Abstract
Dysregulation of the cytokine network in severe malaria owing to variations in factors like parasite load, strains and host factors is well documented but the key cytokines that are dysregulated remain poorly elucidated. Longitudinal changes in cytokine levels in an individual with parasitemia and disease resolution is likely to identify the key cytokines. We have analyzed the mRNA expression of cytokines over a 7-d period in severe (SM) and uncomplicated (UM) Plasmodium falciparum malaria. We found up-regulated expression of TNF-α, IL-1β, IFN-γ and TGF-β in SM, with decreased expression of IL-10 on d 0. Further, we observed a negative correlation of IL-10 expression with parasitemia and pro-inflammatory cytokines, suggesting IL-10 to be the key cytokine in tilting the balance to an inflammatory response. Longitudinal analysis revealed that the key cytokines associated with disease were TNF-α, IL-1β, IFN-γ, IL-12α, RANTES and TGF-β, while TNF-α, IL-10 and TGF-β discriminated between SM and UM. A higher neutrophil count in SM and its positive association with parasite density and IL-1β and IL-8 provides support for neutrophils in inflammation in malaria. Our findings suggest subversion of anti-inflammatory response in SM by parasite factors towards an exaggerated pro-inflammatory response with involvement of neutrophils, the classical inflammatory cells.
Introduction
The ability to mount an effective innate immune response is crucial in limiting the initial phase of malaria infection. Notably, in addition to the involvement of parasite factors, geoclimatic and social factors, the host’s immunological response has also been suggested to play a pivotal role in malaria pathogenesis. 1 A dysregulation in the cytokine balance, with overproduction of pro-inflammatory cytokines, is well documented in malaria severity. Of the pro-inflammatory cytokines, TNF-α, IL-1β and IFN-γ have been suggested to be the major players associated with malaria complications. 2 However, other studies have also linked severe malaria with high levels of IL-6 and IL-18.3,4 Again, there is a lack of consensus in the reports elucidating the role of anti-inflammmatory cytokines, with some studies suggesting the involvement of elevated IL-10 and IL-13, while others have observed low levels of IL-10 to be associated with severe malaria.5,6 The association of different cytokines with severe malaria by different studies can be attributed to variable factors like parasite load and strain, as well as host factors, and longitudinal studies of immune responses have been suggested to normalise these variables.7–9 Thus, the changes in cytokine expression profile with disease resolution in a patient would yield more precise information on association of cytokines with disease.
We analyzed the cytokine profile in a cohort of severe (SM) and uncomplicated (UM) malaria individuals over a 7-d period in populations residing in malaria endemic region of Assam. To the best of our knowledge, this is the first study to analyse the longitudinal changes in cytokine profile from disease diagnosis until clearance of parasitemia and resolution of clinical symptoms in the same individual. We observed a dysregulated cytokine response in SM with decreased IL-10 expression and enhanced pro-inflammatory cytokine expression, as well as the possible role of neutrophils in disease pathology suggested by an enhanced neutrophil count and its positive association with parasitemia and pro-inflammatory cytokines.
Materials and methods
Study site, study design and subjects
The study was conducted in Manja region of Karbi Anglong district, Northeast India, where Plasmodium falciparum (Pf) malaria transmission is meso-endemic, with intense transmission during the rainy season, which usually extends from May to July. The study area and the study population have been described in detail elsewhere. 10 Individuals who were diagnosed with Pf positivity by Plasmodium lactate dehydrogenase-based rapid diagnostic test kits and confirmed by microscopy were recruited. In addition to microscopy, a real-time PCR approach for quantifying parasite density was adopted. Pf 18S rRNA specific primer and TaqMan probe was used to quantify parasitemia in the clinical samples following the method of Malhotra et al. 11 Patients admitted in hospital with symptoms of malaria complications as defined by World Health Organization guidelines were grouped under SM. Patients were considered to have UM if they had signs and symptoms of malaria without evidence of complications. The healthy status of control participants at the time of recruitment was based on a medical history, as well as a physical examination.
Peripheral blood in EDTA and RNAlater (Ambion, Austin, TX, USA) was obtained from SM (n = 51) and UM (n = 64) patients by venipuncture on d 0 before treatment. The SM and UM patients were followed up on d 3 and d 7 post-treatment. For the control group (n = 20), only one blood sample was collected. Written, informed consent was obtained from the study participants. The study was approved by the Tezpur University Ethical Committee (DoRD/TUEC/10-14/453 dated 23/09/10).
Gene expression analysis
Blood samples stored in RNA later were used for total RNA isolation using RiboPure Blood kit (Ambion) and reverse transcribed into cDNA by a High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA). Gene expression analysis was performed using TaqMan based assays (Applied Biosystems) for IL-2, IL-4, TNF-α, IL-1β, IFN-γ, IL-12α, IL-17A, IL-18, IL-8, RANTES, IL-10, IL-13, TGF-β and Foxp3 on a StepOnePlus Real-Time PCR System (Applied Biosystems). GAPDH was used as the endogenous control for normalization of expression levels, while controls were used as the calibrator for mRNA quantification by comparative CT method.
Multiplex cytokine assay
PBMCs isolated using Histopaque (Sigma-Aldrich, St. Louis, MO, USA) were adjusted to a final concentration of 105 cells/ml in complete RPMI 1640 culture medium and stimulated with 40 µg/ml of crude Ag isolated from the local Pf strain following the Percoll method of Troye-Blomberg et al. and non-specific mitogen BCG. 12 After 24 h of incubation, cell-free supernatants and remaining cell pellets were analysed for cytokine protein levels using the MILLIPLEX MAP Human Cytokine/Chemokine Magnetic Bead Panel (Millipore, Bedford, MA, USA) on Millipore’s MAGPIX instrumentation platform for selected cytokines and mRNA expression levels, respectively.
Statistical analysis
Participants with incomplete data either due to loss of follow-up or incomplete investigations were excluded from analysis. Statistical analysis of the data was performed using XLSTAT 2013 version. Comparison of cytokine expression between the groups and within the same group was performed using the unpaired and paired Student’s t-test respectively. Correlations between parameters were evaluated by Pearson’s correlation coefficient test. Relationships between cytokine gene expression levels and disease status were predicted using logistic regression models. A P-value < 0.05 was considered statistically significant.
Results
Clinical and demographic characteristics of malaria patients
Demographic and clinical characteristics of patients with SM and UM.
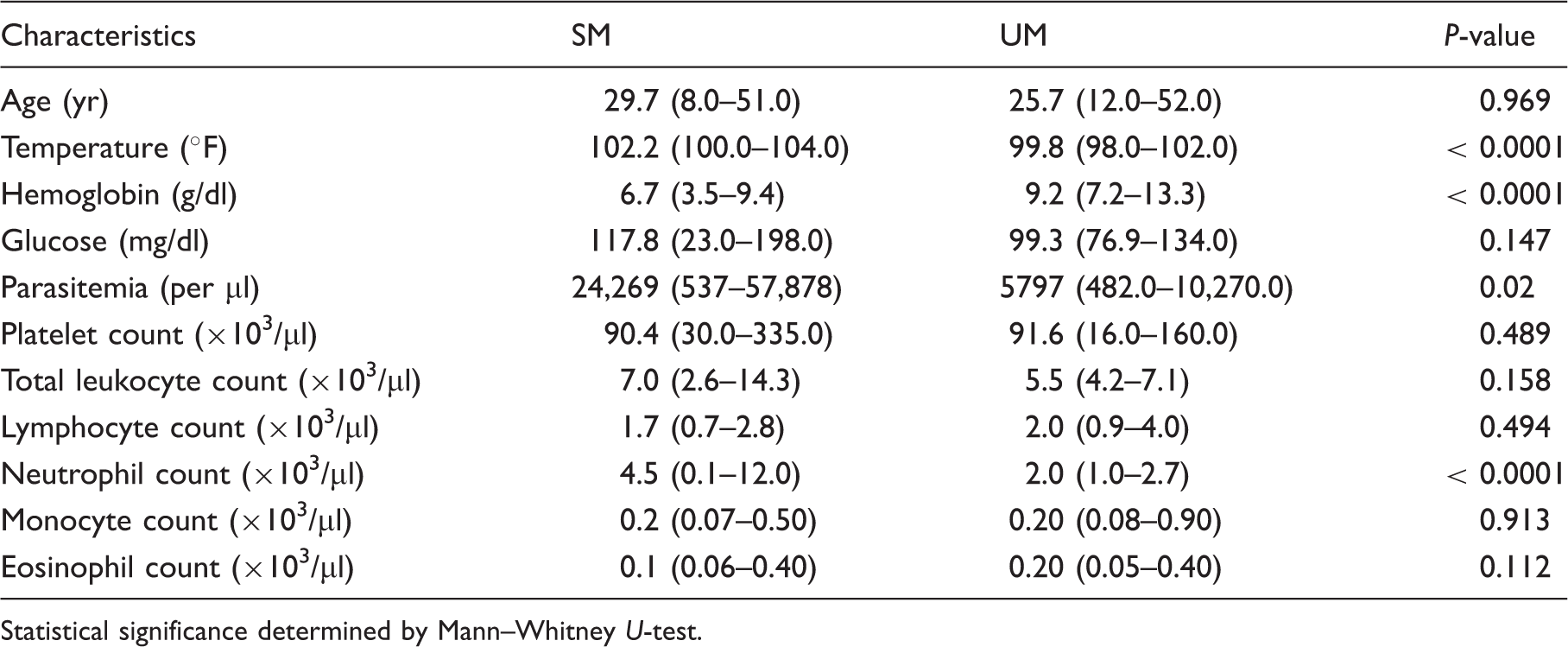
Statistical significance determined by Mann–Whitney U-test.
Cytokine expression differs in SM and UM on d 0
The expression of TNF-α (P < 0.0001), IFN-γ (P < 0.0001), IL-1β (P < 0.0001) and TGF-β (P = 0.04) was up-regulated in SM compared with UM on d 0 prior to treatment (Figure 1). Interestingly, the comparison showed down-regulation in the expression of IL-10 in SM (P = 0.02), suggesting dysregulated balance of the host immune response. However, the CT values of IL-2, IL-4 and IL-13 were beyond the detection limit, and these cytokines were not included for further analysis. Modelling of data by logistic regression taking SM and UM as binary variables showed that IL-1β, TNF-α and the combination of IFN-γ*IL-18 were predictive for SM, with the area under the receiver–operator curve of 0.986 indicating the strength of the model.
mRNA expression profile of cytokines/chemokines and Foxp3 with disease resolution in (A) SM and (B) UM. The mean relative expression was calculated with 2−ΔΔCt formula using healthy control as the calibrator. Statistical comparisons were made between d 0–3, d 3–7 and d 0–7 expression levels by paired t-test. Single and double asterisks indicate significant changes in d 3 expression of P < 0.05 and P < 0.0001, respectively.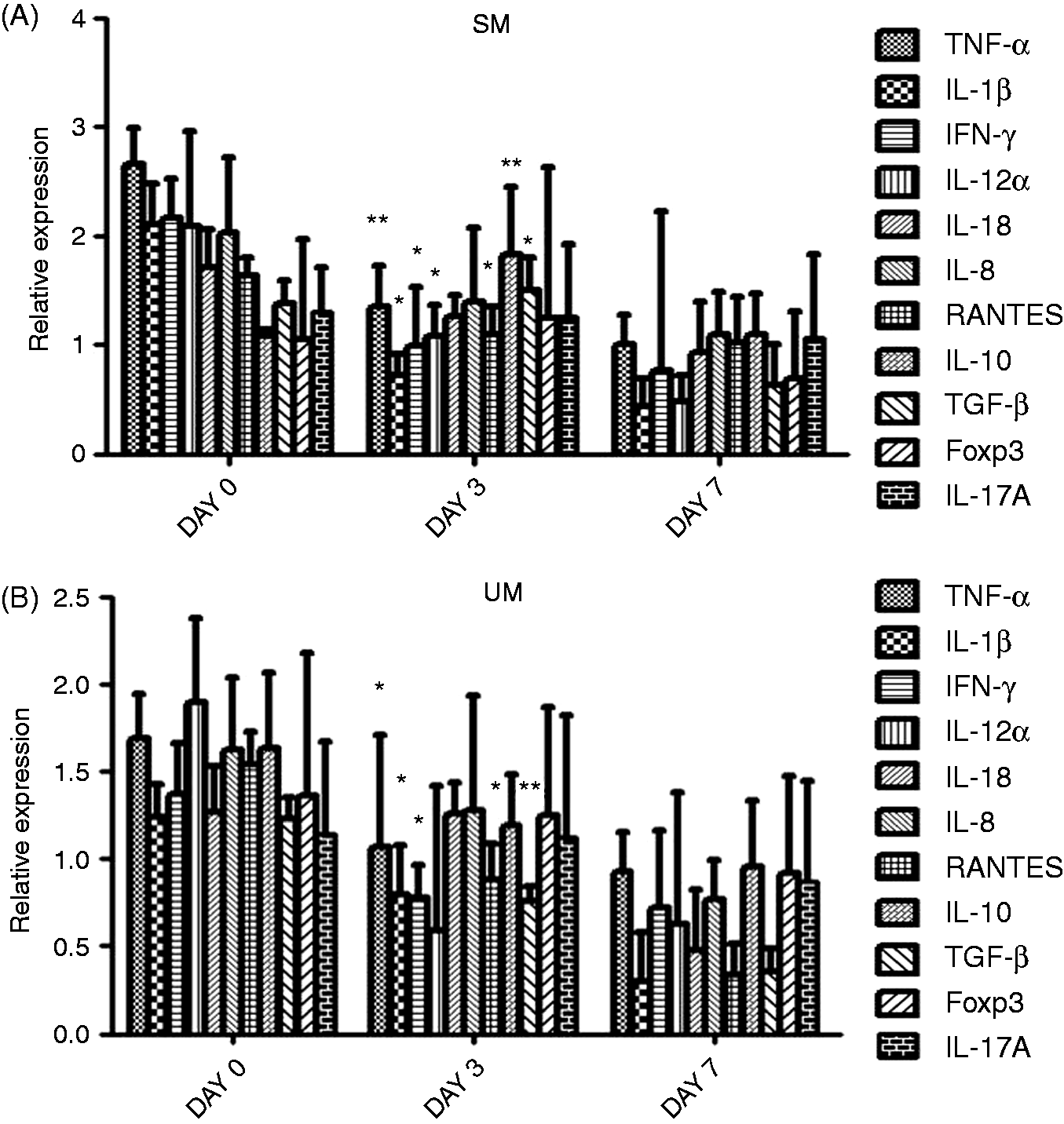
Changes in cytokine and Foxp3 expression with disease resolution in SM and UM
In order to gain insight into the temporal changes in cytokine expression, statistical comparisons were made between d 0–3, d 3–7 and d 0–7 expression levels. The longitudinal analysis revealed that the studied pro-inflammatory cytokines were up-regulated in active infection and tended to normal levels by d 7, suggesting the involvement of multiple cytokines in disease (Figure 1). However, the key cytokines associated with disease characterised by significant changes in expression by d 3 were observed to be TNF-α, IL-1β, IFN-γ, IL-12α, RANTES and TGF-β. In addition, longitudinal changes in the cytokine levels also provided evidence for the role of TNF-α, IL-10 and TGF-β as the discriminating cytokines between SM and UM. Significantly, in SM, the expression of Foxp3 was up-regulated on d 3, which then attained normal levels by d 7, suggesting suppression of regulatory T cell (Treg) activation during malaria severity.
Cytokine protein levels in UM
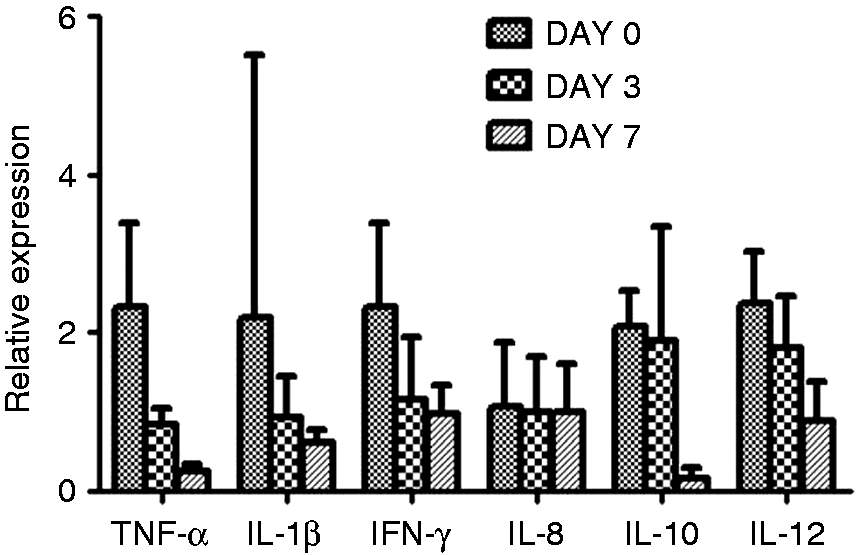
Cytokine protein levels showed a decreasing trend with disease resolution in supernatants of PBMC culture stimulated with crude Pf Ag except for increased levels of IL-1β on d 3 (Figure 2). Further, BCG stimulation elicited increased levels of the cytokines compared to Pf-stimulated culture except for TNF-α. Cytokine gene expression in these Pf-stimulated PBMCs was similar as that at the protein level except IL-1β, which differed in d 3 data (Figure 3). Thus, the longitudinal changes in cytokine mRNA expression in Pf-stimulated PBMCs was similar to blood mRNA levels.
Cytokine/chemokine protein levels with disease resolution in cell culture supernatants stimulated with 40 µg/ml of crude Pf Ag and BCG. Asterisks indicate significant changes in d 3 expression compared with d 0 (P < 0.05). mRNA expression profile of cytokines/chemokines with disease resolution in cultured PBMCs stimulated with crude Pf Ag.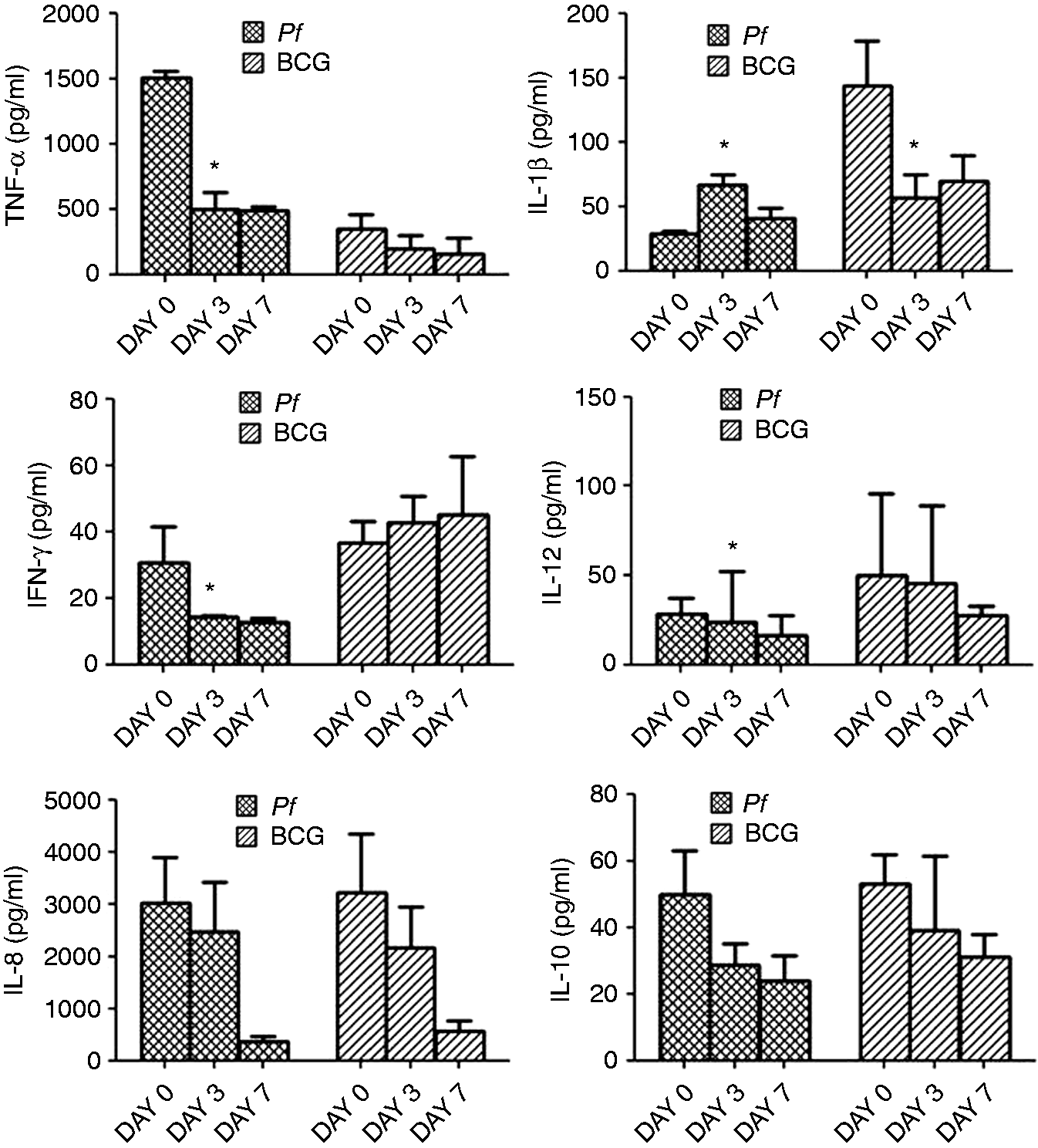
Correlation between cytokine expression, clinical and parasitological parameters in SM and UM during active infection
In SM, neutrophil counts positively correlated with parasite density and mRNA levels of IL-1β and IL-8, suggesting the role of neutrophils in disease pathology (Figure 4A, B). Further, a positive association of parasitemia with TNF-α, IL-1β and IFN-γ was observed (Figure 4C). Interestingly, parasitemia inversely correlated with IL-10 mRNA levels (Figure 4D). Correlation analysis among the cytokines revealed positive association between IFN-γ and TNF-α expression, while IL-10 levels were negatively associated with TNF-α, IL-1β, IFN-γ and IL-8, indicating the overproduction of pro-inflammatory cytokines in presence of low IL-10 levels (Figure 4E, F). In UM, positive association of parasite count was seen with TNF-α, IL-1β and IL-8 (Figure 4G). Further, IL-1β positively correlated with TNF-α and IFN-γ. In addition, TGF-β was associated with TNF-α, IL-1β and IFN-γ, while IL-10 with TNF-α, IL-1β and TGF-β, suggesting a balanced inflammatory response in UM (Figure 4I, J).
Correlation between neutrophils, parasitemia, expression of cytokines in (A–F) SM and (G–J) between parasitemia and cytokines in UM. [(A) r = 0.491, P = 0.003; (B) r = 0.412, 0.423, P = 0.01, 0.01; (C) r = 0.522, 0.394, 0.543, P = 0.002, 0.02, 0.001; (D) r = –0.546, P = 0.001; (E) r = 0.359, P = 0.03; (F) r = –0.408, –0.394, –0.368, –0.354, P = 0.01, 0.02, 0.03, 0.04; (G) r = 0.560, 0.363, 0.498, P < 0.0001, 0.01, <0.001; (H) r = 0.426, 0.329, P = 0.003, 0.02; (I) r = 0.380, 0.386, 0.439, P = 0.008, 0.007, 0.002; (J) r = 0.545, 0.486, 0.362, P < 0.0001, 0.001, 0.01].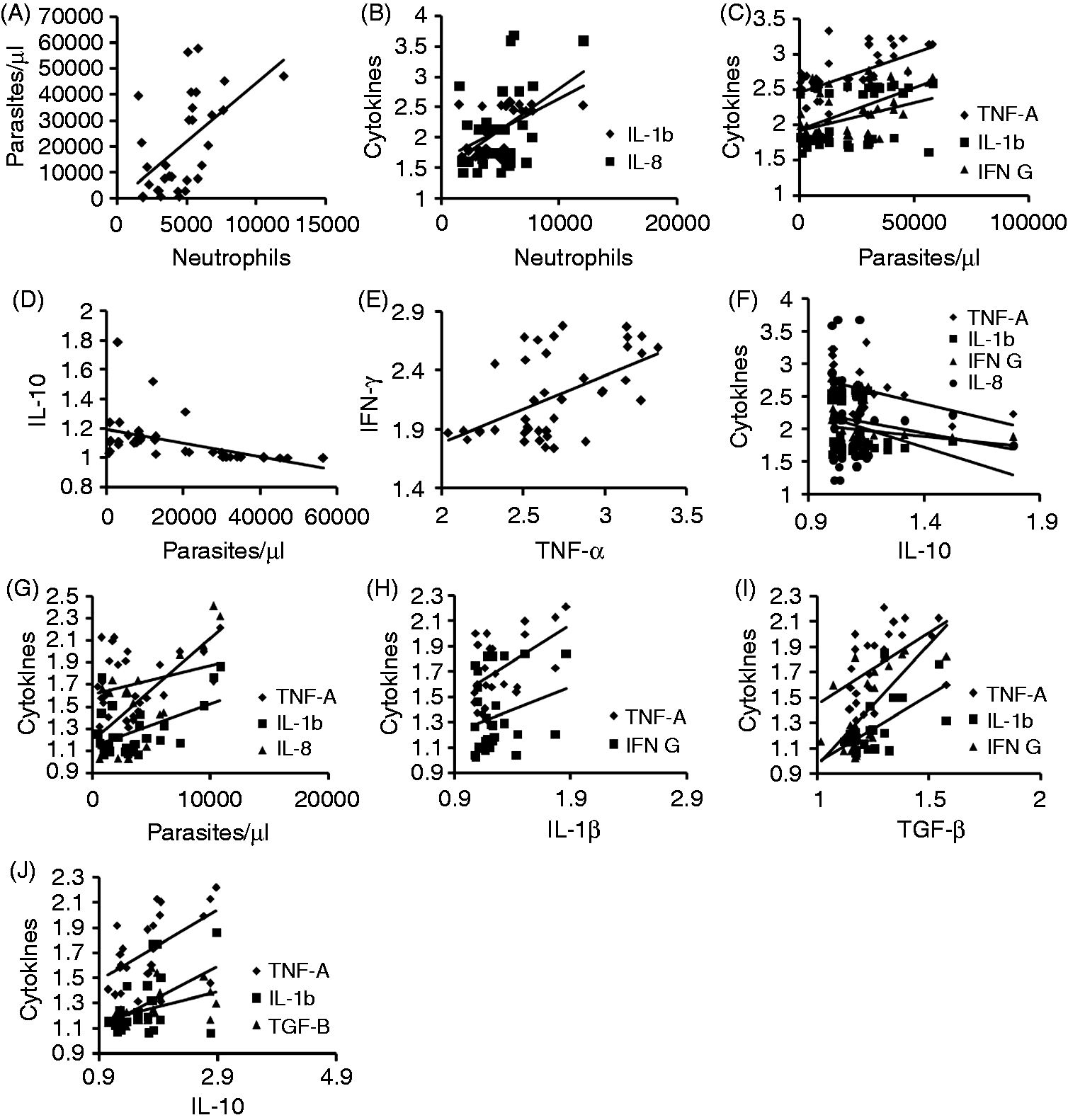
Discussion
Cytokines have been implicated in the contribution to the protection, as well as the pathophysiology of malaria, and it has been argued that clinical outcome of an infection may depend on appropriate induction and counter-regulation of both pro- and anti-inflammatory cytokine secretion.13,14 Pro-inflammatory cytokine response was seen in both SM and UM, with a higher magnitude of response in SM, as reported in earlier studies. 2 The difference appeared to lie in the level of anti-inflammatory cytokines, particularly of IL-10, which was down-regulated in SM but not in UM, suggesting the failure to counterbalance the inflammatory response in SM. The fact that controlled inflammation, i.e. balanced cytokine response is being maintained in UM, is reflected in our findings of positive association of IL-10 with TNF-α, IL-1β and TGF-β, and of TGF-β with TNF-α, IL-1β and IFN-γ, which is consistent with previous reports from our laboratory and other investigators.15,16 Interestingly, in SM, IL-10 levels were negatively correlated with parasitemia and the pro-inflammatory cytokines TNF-α, IL-1β, IFN-γ and IL-8. Notably, a study of cytokine networks in Brazilian Amazon also revealed negative correlation of IL-10 with parasitemia in severe Plasmodium vivax malaria, thus suggesting the association to be malaria specific. 17 At this point, it is interesting to ask if lower IL-10 levels are parasite driven; but then how does the increased inflammatory response benefit the parasite? The answer appears to lie in the inflammatory cytokine and TGF-β-dependent up-regulation of cell adhesion molecules favoring sequestration and parasite survival. 18 The observed relatively higher levels of TGF-β in SM tend to support a role for the cytokine in parasite immune evasion. IL-10 and TGF-β are known to be secreted by different cells in response to pathophysiological stimuli.19,20 Thus, it will be important to understand the cellular sources of these two anti-inflammatory cytokines, as well as elucidating their regulatory mechanisms in malaria. IL-2 is crucial for induction of Tregs. 21 Thus, in our study, where we had undetected levels of IL-2, induction of Tregs is not expected, which is also supported by low Foxp3 in SM. Hence, it is likely that there are cells other than IL-2-induced suppressor cells that could be the source of TGF-β.
Further, our data on longitudinal analysis of changes in cytokine expression revealed that TNF-α, IL-1β, IFN-γ, IL-12α, RANTES and TGF-β were the key cytokines associated with malaria, with up-regulated levels in malaria. Longitudinal changes in the cytokine levels also provided evidence for the role of TNF-α, IL-10 and TGF-β as the discriminating cytokines between SM and UM.
Another important observation was a significantly higher neutrophil count in SM compared with UM. In addition, a strong positive association of neutrophil count with parasite density was observed in SM. The role of reactive oxygen intermediates secreted by neutrophils were reported to be correlated with severe malarial anemia in an earlier study. 22 Importantly, a recent study, which emphasized the role of intracellular reactive oxygen species in cerebral malaria pathogenesis, has reported the use of Tempol as an antioxidant that increased survival in mice with cerebral malaria. 23 Further, it has become evident that neutrophils express an expanding repertoire of cytokines, thus contributing to the inflammatory response.24,25 We also observed a moderate positive correlation of neutrophil count with expression levels of IL-1β and IL-8 in SM. Neutrophil-derived IL-8 plays an important role in the recruitment of other neutrophils, while IL-1β induces other cells to produce neutrophil chemoattractants, thus leading to exaggerated neutrophil function and may explain the increased neutrophil count obtained in our study.26,27 As IFN-γ is known to prolong neutrophil lifespan so the increased expression of IFN-γ in our SM study group could be suggested to influence indirectly the serious, detrimental effects of neutrophils in disease severity. 28 Based on the observations of up-regulated neutrophil characteristic cytokines IL-1β and IL-8, increased neutrophil count and correlation with disease severity, the question that may be asked if neutrophils were the source of TGF-β in malaria? Importantly, studies have reported the involvement of neutrophils in TGF-β secretion in response to different signals.24,29
In conclusion, our data suggests failure of mechanisms that regulate the cytokine balance, as indicated by decreased IL-10 expression, which resulted in exaggerated inflammatory response in SM. Longitudinal changes in the cytokine levels also provided evidence for the role of TNF-α, IL-10 and TGF-β as the discriminating cytokines between SM and UM. Further, we hypothesize that immunosuppression of T helper arm and manipulation of innate cells and pathways by parasite factors appear to be the major factors contributing to heightened inflammation and disease severity in Pf malaria. In addition, a significant role of neutrophils in disease severity was observed, which necessitates the importance to understand the host–pathogen interactions that would help design therapeutic interventions to provide optimal pathogen killing with minimal host damage.
Footnotes
Funding
This study was funded by Department of Biotechnology, India (Grant no: BT/CP/11/NE/TBP/2010) and DBT Institutional Biotech Hub (Grant no: BT/22/NB/2011).
Acknowledgements
We would like to thank all the participants in the study and the clinical staff who helped us in sample collection.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
