Abstract
Periodontal infections increase the risk of atherosclerotic vascular disease via partly unresolved mechanisms. Of the natural IgM Abs that recognize molecular mimicry on bacterial epitopes and modified lipid and protein structures, IgM directed against oxidized low-density lipoprotein (LDL) is associated with atheroprotective properties. Here, the effect of natural immune responses to malondialdehyde-modified LDL (MDA-LDL) in conferring protection against atherosclerosis, which was accelerated by the major periodontopathogen Porphyromonas gingivalis, was investigated. LDL receptor-deficient (LDLR–/–) mice were immunized with mouse MDA-LDL without adjuvant before topical application challenge with live P. gingivalis. Atherosclerosis was analyzed after a high-fat diet, and plasma IgG and IgM Ab levels were measured throughout the study, and the secretion of IL-5, IL-10 and IFN-γ in splenocytes stimulated with MDA-LDL was determined. LDLR–/– mice immunized with MDA-LDL had elevated IgM and IgG levels to MDA-LDL compared with saline-treated controls. MDA-LDL immunization diminished aortic lipid depositions after challenge with P. gingivalis compared with mice receiving only P. gingivalis challenge. Immunization of LDLR–/– mice with homologous MDA-LDL stimulated the production of IL-5, implicating general activation of B-1 cells. Immune responses to MDA-LDL protected from the P. gingivalis-accelerated atherosclerosis. Thus, the linkage between bacterial infectious burden and atherogenesis is suggested to be modulated via natural IgM directed against cross-reactive epitopes on bacteria and modified LDL.
Keywords
Introduction
Periodontal infections have a significant association with the risk of atherosclerotic vascular disease; however, a causative relationship has not been established according to current understanding. 1 Several molecular mechanisms have been suggested to explain the association, for example systemic inflammation, direct bacteremia with vascular infection and molecular mimicry.1,2
Porphyromonas gingivalis (Pg) is recognized as one of the major pathogens in periodontitis, and this bacterium can locally invade periodontal tissue, enter the circulation and evade host defense mechanisms by deregulating the immune and inflammatory responses. Pg is a Gram-negative anaerobe with several potent virulence factors, such as fimbriae, LPS, hemagglutinins and special protease enzymes called gingipains. Infectious challenge with live Pg accelerates the development of atherosclerosis in animal models using homo- and heterozygous apoE-deficient mice,3–6 and rabbits and pigs.7,8 Immunization with killed P. gingivalis confers protection against increased atherosclerosis in apoE–/– mice challenged with live bacteria.9,10 We have shown that immunization with heat-killed Pg diminishes atherosclerosis in low-density lipoprotein (LDL) receptor-deficient (LDLR–/–) mice. 11
Immune responses participate in the process of atherosclerotic lesion development starting from the retention of LDL and increased expression of adhesion molecules in the arterial wall. The progression of lesions is a complex interplay between various immune cells and structural cell types of the vascular wall steered by the actions of pro- and anti-inflammatory mediators. Modification of LDL, for example via oxidation, is required for its uptake by macrophages—a phenomenon that leads to formation of lipid laden foam cells and accumulation of cholesterol inside the advanced atherosclerotic plaques. The presence of bacterial components in the circulation and inside the plaque can also induce atheropromoting effects. For example, Pg induces monocyte chemoattractant protein-1 (MCP-1) secretion by endothelial cells, 12 and increases the uptake of LDL in murine macrophages. 13
The oxidation of LDL generates various immunogenic epitopes on lipid and protein moieties of LDL. Lipid peroxidation reactions of polyunsaturated fatty acids produce reactive aldehydes such as malondialdehyde (MDA) and 4-hydroxynonenal, which efficiently form adducts via amino groups of the phospholipids and lysine residues of proteins. Natural IgM Abs of innate immunity recognize these oxidized lipid and protein adducts in mice and humans.14,15 High-fat feeding of atherosclerosis-prone mice elevates the levels of, for example, IgM to MDA-modified LDL (MDA-LDL), 16 and immunization of LDLR–/– mice with homologous MDA-LDL decreases atherosclerosis via activation of cellular immune responses. 17 Studies with MDA-LDL-immunized heritably hyperlipidemic rabbits, 18 or cholesterol-fed rabbits receiving copper-oxidized LDL as an immunogen,19,20 have also demonstrated reduced progression of atherosclerosis. The mechanisms of this beneficial effect remain unknown. In humans, IgM levels to MDA-LDL are inversely associated with cardiovascular disease.21–23 Therefore, in general, an atheroprotective role for the binding of natural IgM to oxidized LDL and to a variety of other Ags has been proposed.15,24 Corresponding results have been obtained by animal studies using, for example, mice deficient in serum IgM or the depletion of B-1 a lymphocytes.24–26 However, the conclusions have remained controversial and opposing reports also exist. 27
Previously, we characterized a mouse monoclonal IgM to MDA-LDL that recognizes epitopes on hemagglutinin/adhesin domain of Pg gingipain protease. 11 The physiological role and ability of anti-MDA-LDL IgM to dampen the pathogenicity of Pg has not yet been resolved in animal models or humans. The aim of this study was to investigate the effect of MDA-LDL immunization in conferring protection against pathogen-accelerated atherosclerosis. The study design comprised immunization of LDLR–/– mice with MDA-modified mouse LDL (MDA-msLDL) without adjuvant before challenge with live Pg. The extent of atherosclerosis after a high-fat diet (HFD) was analyzed in MDA-LDL-immunized and control animals.
Materials and methods
Lipoprotein isolation and modifications
LDL fraction (density 1.019–1.063 g/ml) was isolated from plasma of LDLR–/– mice on a HFD or from human plasma by sequential density gradient centrifugation. 28 MDA modification to LDL was prepared as described. 11 In brief, 0.5 mol/l solution of 1,1,3,3–tetramethoxypropane malonaldehyde-bis(dimethyl acetal) (#10,838-3; Sigma-Aldrich, St. Louis, MO, USA) in 0.3% hydrochloric acid was incubated at 37℃ for 10 min. pH was adjusted between 6.0 and 7.0 with sodium hydroxide and the final volume was adjusted to 4 ml with sterile water. A total of 900 µl of 0.5 mol/l MDA was added to 6 mg LDL, and the volume was adjusted to 3 ml. The mixture was incubated at 37℃ for 3 h and dialyzed extensively against 0.27 mmol/l EDTA in PBS. The percentage of modified lysine residues was determined with TNBS (2,4,6-trinitrobenzene sulphonic acid trihydrate, #92820; Fluka Chemika, Buchs, Switzerland) testing. 29
Animals, diets and immunization
The animal protocol was approved by the National Animal Experiment Board of Finland (STH190A). Female LDLR–/– mice (C57BL/6J background B6.129S7-Ldlrtm1Her) at the age of 8–15 wk were divided into three groups: (1) imm + Pg + HFD (n = 13); (2) HFD only (n = 12); and (3) Pg + HFD (n = 12). MDA-msLDL immunization (imm), Pg and HFD were performed as presented in Figure 1. During the immunization period, LDLR–/– mice were fed a regular chow diet (4.4% fat and 0.01% cholesterol). Primary immunization with 25 µg MDA-msLDL without adjuvant was injected subcutaneously. Controls received sterile saline. Boosters were given i.p. in 2–3 wk intervals a total of six times containing 10, 10, 10, 10, 25 and 25 µg of MDA-msLDL, respectively. Additionally, immunization was maintained by injecting 25 µg of MDA-LDL i.p. on the day after the end of intensive Pg application period and 4 wk thereafter. Western-type HFD was started on wk 17 of the study. The HFD contained 22% fat and 0.15% cholesterol (TD88137; Harlan Teklad, Madison, WI, USA) and was continued for 12–14 wk. Mice had free access to water and food, and were kept in conventional housing throughout the study.
Experimental scheme and IgG Abs to Pg in LDLR–/– mice. (A, B) Timeline showing the immunizations (circles), period of HFD and Pg applications (boxes). LDLR–/– mice were immunized with MDA-msLDL without adjuvant (group 1). Two control groups received saline (groups 2 and 3). Next, HFD was initiated and, after 2 wks, the oral and anal topical application period, on d 1, 3, 5, 8, 10 and 12, with Pg in carboxymethylcellulose was started. MDA-LDL-immunized mice and one control group received Pg and the other control group received only carboxymethylcellulose. Immunizations and applications were continued once a month until the end of the study. (C) Plasma IgG binding to Pg Ags was analyzed in individual samples with flow cytometry as percentage of events above the background in the gated bacterial population at the end of the study. Plasma IgG binding to (D) Pg LPS at the end of the study, and IgG binding to recombinant domains of gingipain, (E) Rgp44 and (F) Rgp15–27 was analyzed using a chemiluminescence immunoassay. Plasma samples collected before immunization (pre), after immunizations (Imm) and after topical application period (appl) were pooled from three mice (E, F). Samples at the end of the study (end in E and F) and all samples in (D) were analyzed individually, and duplicates for each mouse were measured. *P < 0.05, **P < 0.01.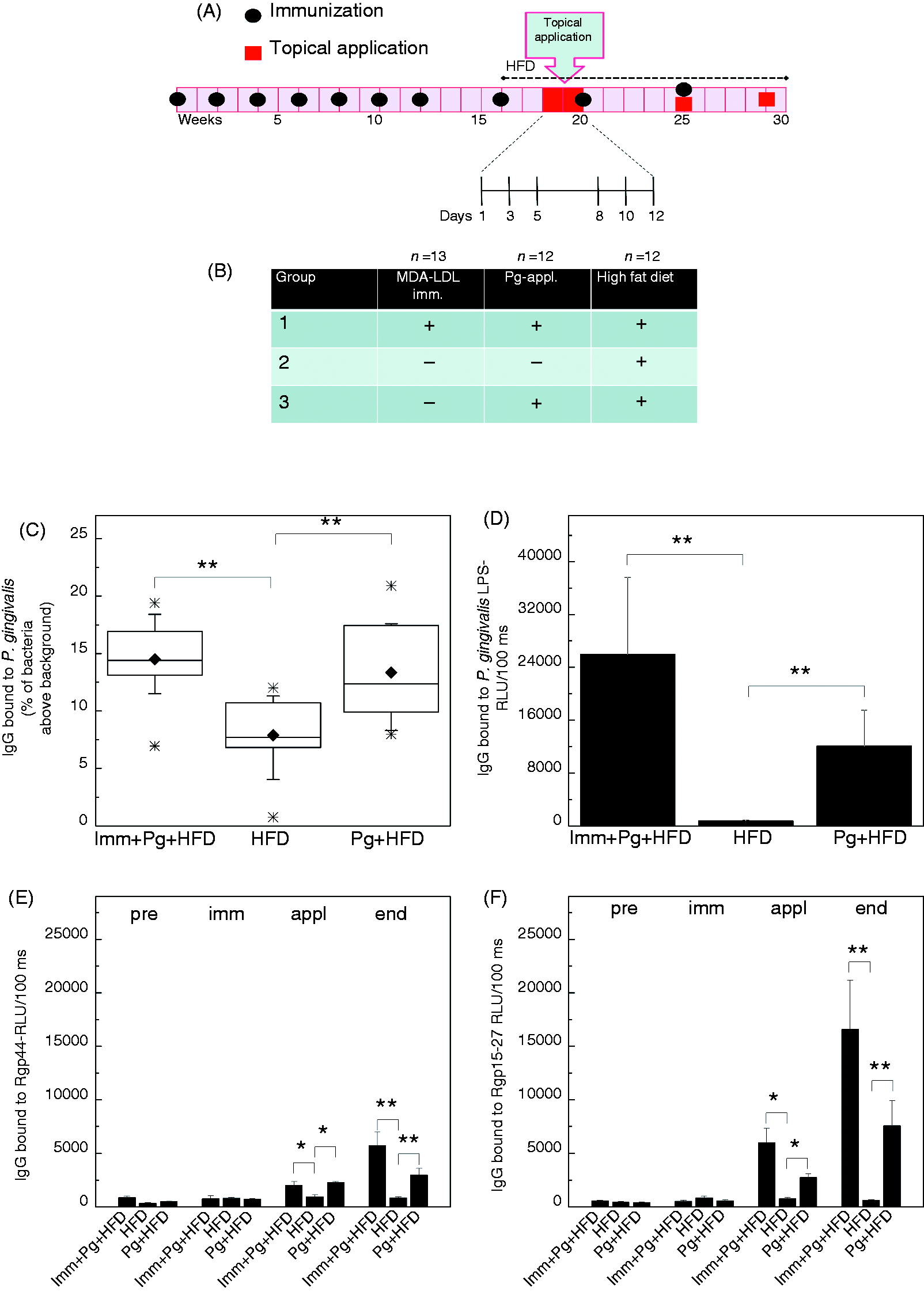
Topical application
LDLR–/– mice were challenged with live Pg (ATCC 33277) grown in anaerobic conditions at 37℃ on Brucella agar plates supplemented with 5% horse blood, 5 µg/ml hemin and 100 µg/ml vitamin K1 for 5 d. Bacteria were suspended in sterile PBS, and the volume was adjusted to give an absorbance of ∼0.75 at 650 nm, corresponding to 1010 CFU/ml. Carboxymethyl cellulose [(CMC) #21902; Fluka Biochemika, Buchs, Switzerland) was added to 3% (w/v). MDA-LDL-immunized mice and one saline immunized group were challenged with bacteria. The other saline immunized control group was treated with 3% CMC in PBS. Topical applications were started 2 wk after introducing HFD and performed on d 1, 3, 5, 8, 10 and 12. After the intensive period the applications were repeated twice at 4-wk intervals (Figure 1). Each mouse received 2.5 × 109 CFU of Pg as oral topical application and 5 × 108 CFU as anal topical application per one treatment. Mice are coprophagic animals, and the anal application route was administered to establish a continuous cycle of oral reinfection with Pg remaining in the feces. Antibiotic prophylaxis has been commonly used before oral bacterial challenge,4,30 but it was omitted in this study.
Plasma Ab binding to Pg cells and LPS
Pg cells were freshly picked from supplemented Brucella-plates, suspended and washed once with PBS. The cell suspension was frozen and thawed for use in flow cytometry analysis. Pg cells were washed with 0.1% BSA in PBS (FACS buffer) using centrifugation at 3200 g for 10 min at 4℃. Mouse plasma samples were diluted in FACS buffer (1:100 for IgG and 1:250 for IgM) and incubated 40 min at 4℃ with bacteria followed by washing with FACS buffer. AlexaFluor 488-conjugated anti-mouse-IgG (#A11001; Invitrogen, Carlsbad, CA, USA) and anti-mouse-IgM (#A21042; Invitrogen) were used as secondary Abs (0.5 µg/ml) and incubated for 35 min at 4℃ followed by washing as described. Pg cells were suspended in FACS buffer for flow cytometer analysis with FACSCalibur (BD Biosciences, San Jose, CA, USA). Data were analyzed with FlowJo 7.6.4. software (TreeStar, Ashland, OR, USA). Plasma Ab binding to Pg LPS was analyzed in plasma samples collected at the end of HFD and diluted in 0.5% fish gelatin-0.27 mmol/l EDTA in PBS (1:125 for IgG and 1:100 for IgM). Pg LPS (5 µg/ml, #tlrl-pglps; InvivoGen, San Diego, CA, USA) was immobilized on microtiter plates and a chemiluminescence immunoassay was performed as described below using alkaline phosphatase-conjugated anti-mouse IgG/IgM and Lumiphos 530 (#P-501; Lumigen, Southfield, MI, USA) as substrate.
Analysis of atherosclerosis and plasma lipids in mice
The extent of atherosclerosis was analyzed using en face lipid staining of the whole aorta and determination of aortic origin cross-sectional plaque areas on hematoxylin-eosin stained sections as described previously.11,31 Necrotic core areas were determined on aortic root paraffin sections stained with modified Movat’s pentachrome.32,33 Plasma total cholesterol (TC), total triglycerides and high-density lipoprotein cholesterol in LDLR–/– mouse plasma were determined after HFD by enzymatic methods using commercial kits from Roche Diagnostics (Mannheim, Germany). LDL cholesterol was estimated using Friedewald's formula.34
Chemiluminescence immunoassays
Chemiluminescence immunoassays using modified lipoprotein Ags, MDA-BSA and recombinant domains of Pg gingipain, Rgp44 and Rgp15–27, were performed as described previously. 11 MDA and phosphocholine (PC) epitopes in plasma apoB-containing particles were determined as described previously. 35 Rgp44 and Rgp15–27 are consecutive sequences at the hemagglutinin/adhesin domain of gingipain. 36 PC-BSA (PC-conjugated BSA) was purchased from Biosearch Technologies (#PC-1011; Novato, CA, USA). Ags (5 µg/ml) were immobilized on microtiter plates and mouse plasma IgG or IgM binding was measured using alkaline phosphatase conjugated secondary Abs (A2429, A9688; Sigma-Aldrich) and Lumiphos 530 as a substrate for the chemiluminescence immunoassay. 11 Results are shown as relative light units (RLU) per 100 ms. Specific binding was analyzed using competitive immunoassay in which the plasma samples were incubated with 100 µg/ml of soluble competitor at 4℃ for 18 h. Immunocomplexes were separated by high-speed centrifugation, and unbound Abs were measured using chemiluminescence immunoassay. For measuring the total IgM and IgG concentration in mouse plasma, 2.5 µg/ml of anti-mouse-IgM (M8644; Sigma-Aldrich) or 3 µg/ml anti-mouse-IgG (M1397; Sigma-Aldrich) in 0.27 mmol/l EDTA in PBS was immobilized to microtiter plates and purified mouse IgM or IgG was used as a standard. 35
Splenocyte assay
Spleen was removed immediately after sacrifice, and single-cell suspensions were prepared using BD Falcon 100 µm cell strainers in DMEM supplemented with 20% FBS, 1% non-essential amino acids, 10 mM HEPES, 100 U/ml penicillin and 100 µg/ml streptomycin (Sigma-Aldrich). Splenocytes of three mice within a group were combined and washed with supplemented DMEM. Erythrocytes were lysed with 0.14 mol/l ammonium chloride-0.02 mol/l Tris buffer (pH 7.2). The washing step was repeated and cells were seeded 106 cells/well to round-bottom 96-well culture plates (BD Biosciences). Ags were added as follows: human MDA-LDL or native LDL (nLDL) 10, 40 or 80 µg/ml and control cultures without Ags. All Ags were added to six replicate wells for 72 h incubation at 37℃ in a humidified atmosphere with 5% CO2. 37 Splenocyte media were collected and stored at –70℃ before analyzing IL-10 (#558300) and IFN-γ (#558296) with Cytometric Bead Assay using Mouse/Rat Soluble Protein Master Buffer kit #558267 (BD Biosciences), and secretion of IL-5 using matched monoclonal Abs (MAB405, BAM705) and recombinant standard (#405-ML) for mouse IL-5 (R&D Systems, Minneapolis, MN, USA) in chemiluminescence based immunoassay.
Gene expression analysis by quantitative real-time PCR
The excised spleen halves and pieces of liver were immersed immediately after blood collection into AllProtect Reagent (Qiagen, Hilden, Germany) and stored at 4℃. RNA was isolated with RNeasy Microarray Tissue Mini kit (Qiagen), including DNase treatment. RNA (0.33 µg) from three samples within a group were combined (total 1 µg of RNA) and cDNA was made with RT 2 First Strand Synthesis Kit (Qiagen). Gene expression in the spleens was analyzed using RT 2 Profiler Array, Mouse Innate & Adaptive Immune Responses Array (PAMM-052A, 84 genes; Qiagen) and Mouse Atherosclerosis Array (PAMM-038A) for analysis of liver samples. All arrays were analyzed in duplicate with an iQ5 Real-time PCR detection system (Bio-Rad, Hercules, CA, USA). Threshold cycle (Ct) values were transformed to fold change values with the RT 2 Profiler PCR Array Data Analysis software provided on the manufacturer’s website. Color-scaled matrices of log2 of fold change in gene expression were constructed using the Matrix2png web interface. 38
Array expression data were confirmed for individual genes using quantitative real-time PCR. Maxima SYBR Green qPCR Master mix (Thermo Scientific, Rockford, IL, USA) and Rotor-Gene 3000 (Qiagen) real-time thermal cycler were used. The PCR protocol consisted of an initial incubation for 15 min at 95℃ followed by PCR cycling using a three-step cycle at 95℃ for 15 s, at 60℃ for 30 s and at 72℃ for 45 s for a total of 40 cycles. The cellular GAPDH mRNA was used as an endogenous control. The primers used were as follows: mouse GAPDH forward 5’-GGTGCTGAGTATGTCGTGGA-3’ and reverse 5’-GTGGTTCA CACCCATCACAA-3’; mouse MCP-1 forward 5’-CAATGAGTAGGCTGGAGAGCTA-3’ and reverse 5’-TCTGGACCCATTCCTTCTTG-3’; mouse IL-5 forward 5’-TGACA AGCAATGAGACGATGA-3’ and reverse 5’-CCCACGGACAGTTTGATT CT-3’. Copy numbers were calculated using the standard curve method. The external standards for GAPDH and MCP-1 were constructed from the cDNA transcripts of mouse liver extracts, and the external standard for IL-5 was constructed from the cDNA transcript of mouse spleen. A dilution series of standards of 101–107 copies per reaction were used for each PCR run.
MDA and PC epitopes in plasma apoB-containing particles
ApoB-containing particles in mouse plasma (1:200) were captured with 5 µg/ml anti-human apoB-100/48 Ab (K45253G; Meridian Life Science, Memphis, TN, USA). 35 Biotinylated mouse monoclonal IgM specific for MDA epitopes (MDmAb; HME-04_07) 11 was used (1.5 µg/ml) for detecting MDA epitopes, and alkaline phosphatase conjugated NeutrAvidin (#31002; Thermo Fischer Scientific) and Lumiphos 530 substrate were used in chemiluminescence detection. PC epitopes in plasma apoB-containing particles were measured using biotinylated mouse monoclonal anti-PC-IgM (EO6) kindly provided by Professor J. Witztum (San Diego, CA, USA). 39
Statistics
A non-parametric Mann–Whitney U-test was used to analyze the differences between the variables in different treatment groups. Data are presented as mean ± SEM. P-Values < 0.05 were considered significant. Differences between paired samples were analyzed using a paired samples t-test. Correlations were analyzed using Pearson’s correlation coefficient.
Results
IgG Ab response to Pg in topically applied LDLR–/– mice
The experimental scheme of the study is described in Figure 1 (A,B). IgG Ab responses induced by the topical application of Pg were verified at the end of the study. Plasma IgG Abs binding to Pg cell Ags, to LPS of Pg and to recombinant hemagglutinin/adhesin domains of gingipain, Rgp44 and Rgp15–27, were analyzed. Both groups receiving live Pg had elevated IgG binding to Pg bacterial Ags (Figure 1C), to Pg LPS (Figure 1D) and to recombinant Rgp44 (Figure 1E) and Rgp15–27 (Figure 1F) compared with the mice receiving vehicle. The IgG levels to Rgp44 and Rgp15–27 remained elevated until the end of the study in groups receiving topical Pg application.
Immunization with MDA-LDL reduces atherosclerosis in Pg-challenged LDLR–/– mice
Aortic lipid deposition was analyzed with an en face method after a 14-wk HFD. Percentage area of aortic lipid depositions was significantly decreased in MDA-LDL-immunized mice after Pg-challenge compared with mice receiving only Pg-challenge (Figure 2A,E). Also, the total aortic lipid deposition area was significantly reduced in MDA-LDL-immunized mice receiving Pg-challenge and in vehicle-treated controls compared with saline-immunized mice challenged with Pg (Figure 2B,E). The extent of atherosclerosis was also analyzed at aortic origin cross-sections. No significant differences were observed in cross-sectional plaque areas (Figure 2C,F) between different treatment groups. Necrotic core areas were determined on Movat-stained aortic origin cross-sections (Figure 2D,G). MDA-LDL-immunized mice receiving Pg challenge and vehicle-treated controls had smaller necrotic core areas compared with the Pg-challenged mice but the differences were not statistically significant.
Quantification of atherosclerosis in Pg-challenged LDLR–/– mice. Atherosclerosis was analyzed with an en face method on the aortas of LDLR–/– mice immunized with MDA-LDL and challenged with Pg (Imm + Pg + HFD), immunized with saline only and challenged with Pg (Pg + HFD) or immunized with saline only and treated only with vehicle (HFD). The aortic lipid deposition area is shown as (A) percentage of aortic area and as (B) total area in mm2. Plaque areas in the (C) aortic origin cross-sections and (D, G) Necrotic core areas (circled in red) on sections stained with Movat’s pentachrome were also measured as absolute area in µm2. Representative images of (E) stained aortas and (F) aortic origin cross-sections in each group are shown. *P < 0.05.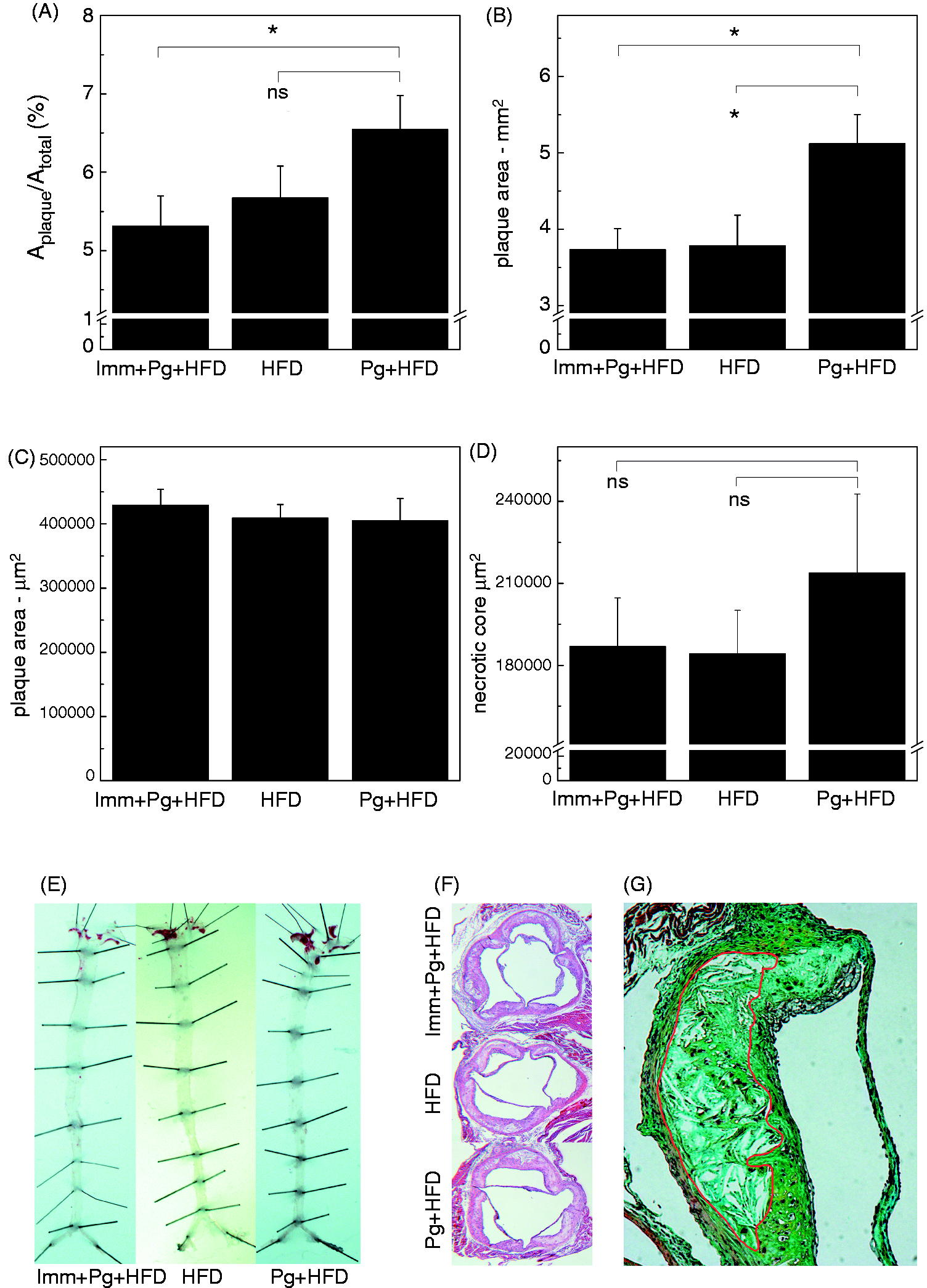
IgM Ab response to MDA and PC epitopes
LDLR–/– mice immunized with homologous MDA-LDL without adjuvant showed increased plasma IgM binding to immobilized MDA-LDL (Figure 3A) after immunizations, after intensive bacterial challenge and at the end of the study when compared with saline-immunized groups. When IgM binding to MDA-LDL was normalized with the total IgM concentration, the relative levels of anti-MDA-LDL IgM remained significantly elevated in the MDA-LDL-immunized mice compared with controls (Figure 3B). MDA-LDL-immunized mice had also elevated IgM binding to nLDL (Figure 3C). Normalized relative levels of IgM to nLDL were also increased in MDA-LDL-immunized mice compared with controls (Figure 3D). Plasma total IgM concentrations increased in all groups throughout the study when compared with the previous measurement point (Figure 3E). MDA-LDL-immunized mice had also elevated IgM binding to MDA-BSA (Figure 3F), and to PC-conjugated BSA (Figure 3G), which resulted, most likely, from a broad spectrum induction of natural IgM production or spontaneous oxidative modification of the native mouse LDL preparation used as Ag.
IgM Ab binding to MDA epitopes, native LDL and PC-BSA. Plasma samples (1:1000) collected before immunizations (pre), after immunizations (imm), after topical application period (appl) and at the end of the study were analyzed for IgM binding to (A) MDA-msLDL with chemiluminescence immunoassay, and (B) IgM binding was normalized with the total IgM concentration. IgM binding to (C) mouse native LDL was analyzed and (D) normalized with the total IgM concentration. (E) Plasma total IgM concentrations were measured at different time points. Plasma samples (1:500) were also analyzed for IgM binding to (F) MDA-BSA and (G) PC-BSA. Samples of three mice were combined for analysis of time points pre, immunized and applicated (A–D, F, G). Samples at the end point and samples for the determination of total IgM levels were measured individually using duplicates. *P < 0.05, **P < 0.01. Differences between paired samples were analyzed using paired samples t-test (§§P < 0.01).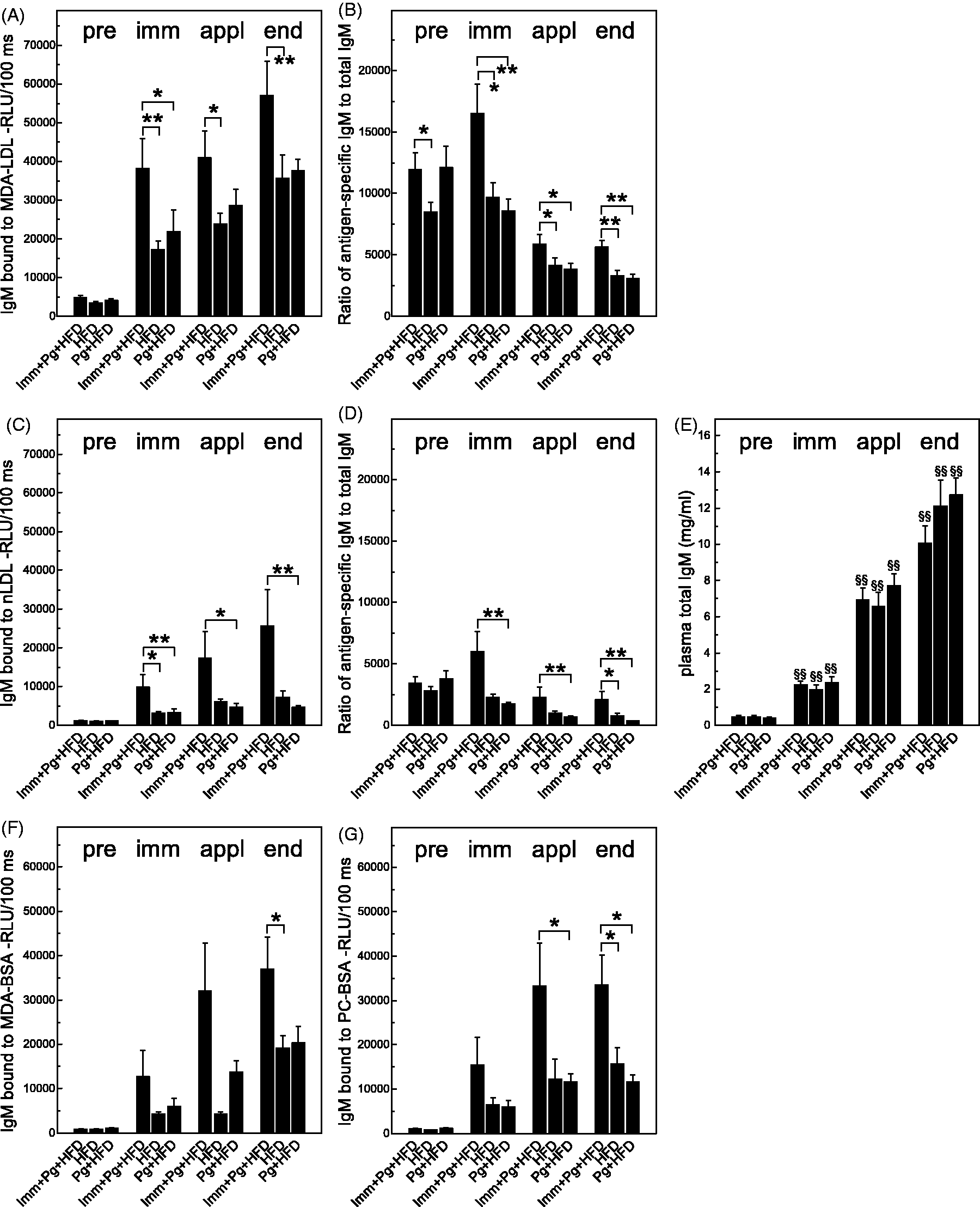
Specificity of IgM response to MDA adducts
Plasma IgM Abs of LDLR–/– mice were analyzed for specific binding to MDA adducts and PC-BSA at different time points of the study using competitive liquid phase immunoassay. IgM binding to MDA-LDL was competed equally (50–60%) by soluble MDA-LDL and MDA-BSA in all groups (Figure 4A–C) after immunizations. No differences were observed either after topical application with Pg or at the end of HFD between the groups (data not shown). nLDL reduced IgM binding to MDA-LDL by, on average, 20–30% (Figure 4 A,B). Specific IgM to PC epitopes were not induced as a result of either MDA-LDL immunization (Figure 4A), HFD (Figure 4B) or Pg application (Figure 4C).
Specific IgM binding to MDA adducts. Competition immunoassays were performed to analyze binding specificity of plasma IgM after the immunization period. Soluble competitors MDA-LDL, MDA-BSA and nLDL (100 µg/ml) were used, and IgM binding to immobilized MDA-LDL was measured. When PC-BSA was used as a competitor, IgM binding to immobilized PC-BSA was measured. (A) Mice receiving MDA-LDL immunization (Imm) prior to Pg-application and HFD, (B) saline-immunized mice prior to HFD only and (C) saline-immunized mice prior to Pg-application and HFD. IgM binding is shown as ratio of binding with and without the competitor (B/B0).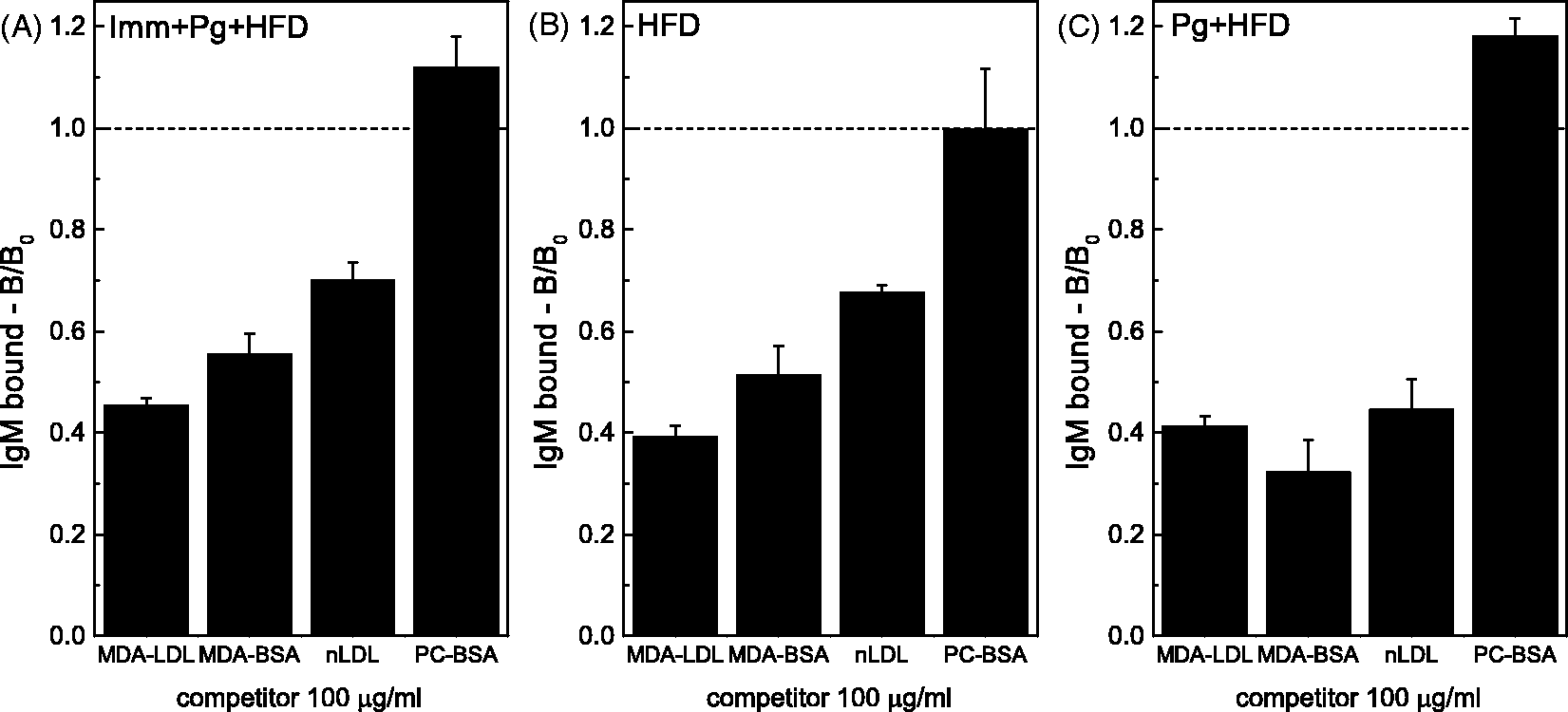
IgM Ab response to Pg-derived Ags
IgM binding to Pg cell Ags was significantly elevated at the end of the study in MDA-immunized mice compared with the vehicle-treated controls. Also, elevated IgM to Pg was detected after intensive application period in MDA-LDL immunized mice compared with the control groups (Figure 5A). MDA-LDL-immunized mice had significantly higher IgM levels to Pg LPS compared with the vehicle-treated group, and also, most notably, compared with the mice receiving the bacterial challenge only (Figure 5B). IgM binding to Rgp44 and Rgp15–27 in MDA-LDL-immunized mice was moderately elevated (Figure 5C,D).
IgM Ab levels to Pg Ags. (A) Plasma (1:250) IgM binding to Pg was analyzed with flow cytometry by pooling four samples within a group before immunizations (pre), after immunizations (imm), and after topical application period (appl). Samples at the end point were measured individually and mean ± SEM is shown. (B) IgM binding to Pg LPS in plasma samples (1:100) at the end of the study was measured with chemiluminescence immunoassay using individual samples measured in triplicate. Mean ± SEM is shown. IgM binding to (C) Rgp44 and (D) Rgp15–27 was analysed with chemiluminescence immunoassay. Samples of three mice were pooled for analysis of time points pre, immunized and applicated, and mean ± SEM is shown for four combined samples within a group. Samples at the end point were measured individually using duplicates (A, C, D). *P < 0.05, **P < 0.01.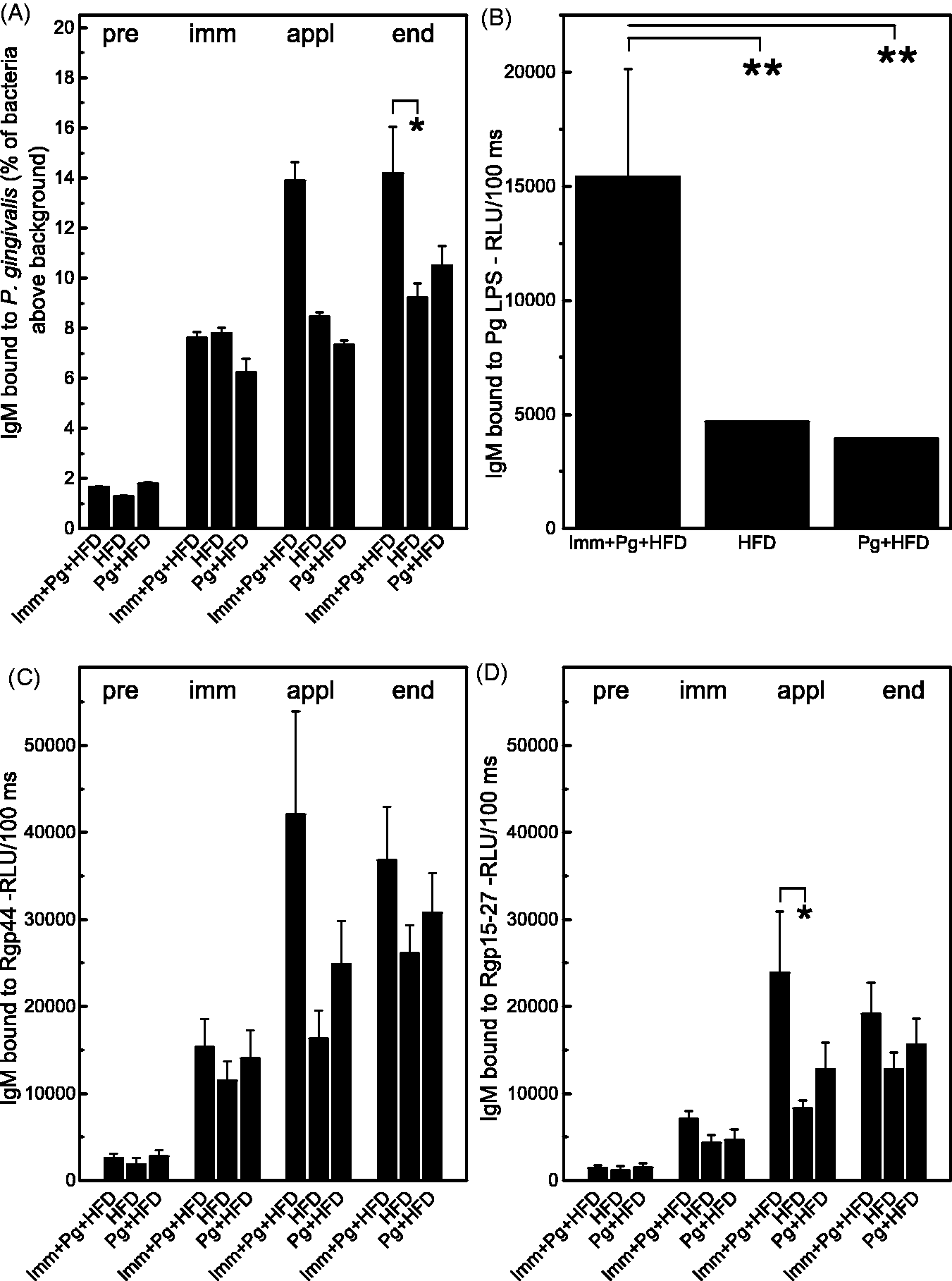
IgG Ab response to MDA-LDL
Low levels of IgG to MDA-LDL were detected in LDLR–/– mice immunized with MDA-LDL, and the IgG response did not increase in the course of the study (Figure 6A). When IgG binding to MDA-LDL was normalized with the total IgG concentration, the relative levels of IgG to MDA-LDL were significantly elevated in the MDA-LDL-immunized mice compared with controls (Figure 6B). Controls without bacterial challenge also had lower relative levels of IgG to MDA-LDL but the difference did not reach statistical significance compared with MDA-LDL-immunized mice (Figure 6B). Significant increases in total IgG concentrations between measurement points were observed in all groups after immunizations and after bacterial topical application (Figure 6C). IgG levels to nLDL were at negligible levels (Figure 6D), and after normalization to total IgG concentration there were no differences between the groups (Figure 6E).
IgG Abs to MDA and native LDL. Plasma samples (1:1000) collected before immunizations (pre), after immunizations (imm), after topical application period (appl) and at the end of the study were analyzed for IgG binding to (A) MDA-msLDL with chemiluminescence immunoassay, and (B) IgG binding was normalized with (C) the total IgG concentration. IgG binding to (D) mouse native LDL was analyzed and (E) normalized with the total IgG concentration. Plasma total IgG concentrations were measured at different time points. Samples of three mice were pooled for analysis of time points pre, immunized and applied (A, B, D, E). Samples at the end point and samples for the determination of total IgG levels were measured individually using duplicates. *P < 0.05. Differences between paired samples were analyzed using paired samples t-test (§§P < 0.01).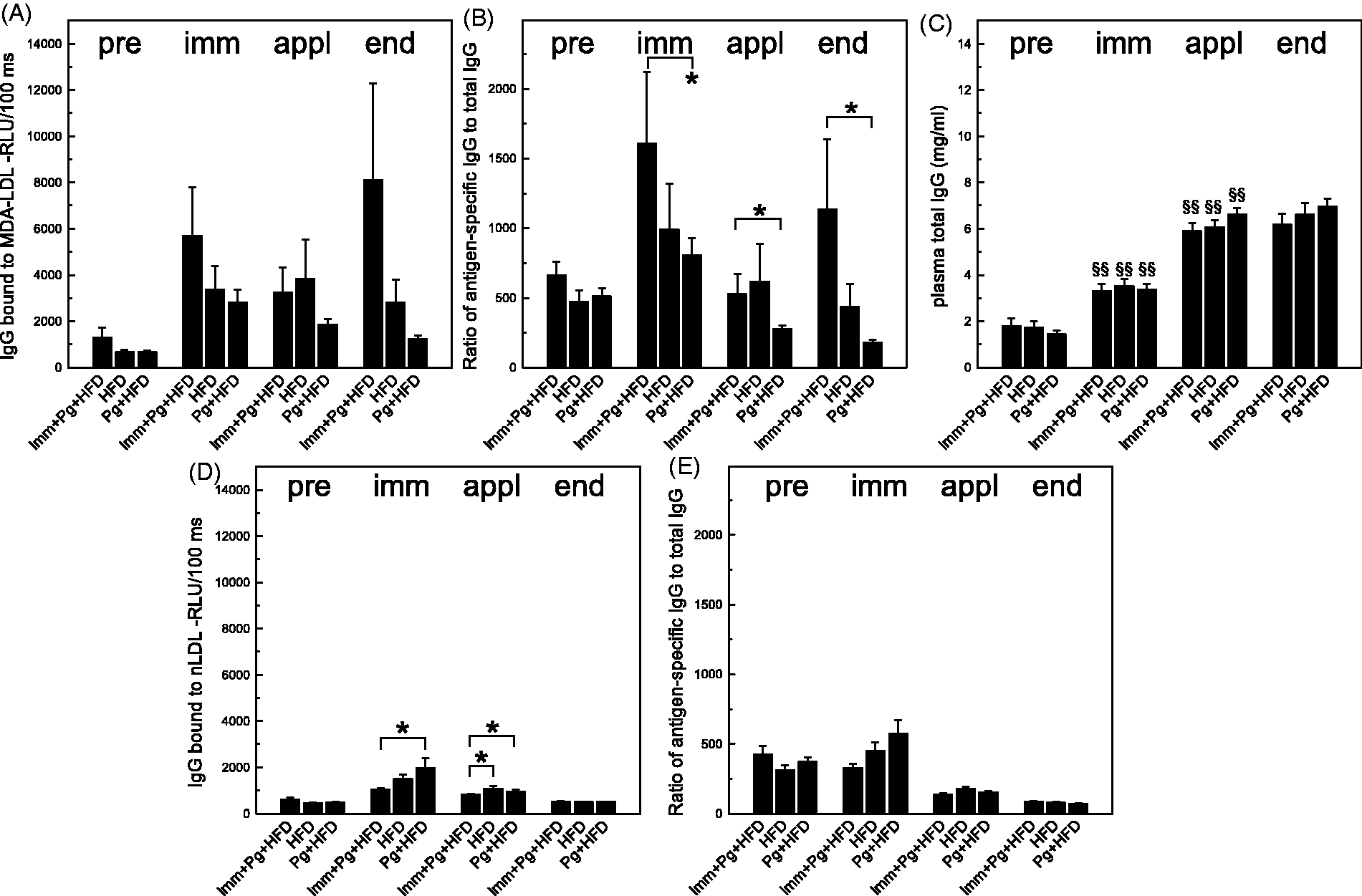
MDA-LDL immunization increases IL-5 secretion by splenocytes stimulated with MDA-LDL
Splenocytes from LDLR–/– mice collected at the end of the study were cultured for 72 h with or without the Ags, MDA-LDL or nLDL. The splenocytes of MDA-LDL-immunized mice secreted elevated levels of IL-5 in a dose-dependent manner upon stimulation with MDA-LDL compared with splenocytes of saline-immunized mice. IL-5 secretion by stimulation with nLDL and without Ags was at equal levels and far less than the MDA-LDL-induced secretion in MDA-LDL-immunized mice (Figure 7A). Splenocytes of mice challenged with Pg demonstrated only minimal increase in IL-5 secretion when stimulated with MDA-LDL compared with the culture without Ags, and IL-5 secretion induced by nLDL was below detection limit (Figure 7A). Splenocytes of mice receiving only HFD showed a very weak dose-dependent IL-5 response to MDA-LDL but not to nLDL (Figure 7A). Gene expression of IL-5 was also analyzed in unstimulated spleen samples collected after sacrifice. The levels of IL-5 cDNA transcripts were extremely low in the spleens of all groups (data not shown). Plasma levels of IL-5 were measured after the fourth and fifth booster immunizations, and MDA-LDL-immunized mice had elevated plasma concentration of IL-5 compared with all saline-immunized mice (Figure 7A, inset). Secretion of IL-10 as a marker of Th2 response and secretion of IFN-γ as a marker of Th1 response were also measured in splenocyte cultures stimulated with MDA-modified or nLDL. There were no significant differences in the secretion of IL-10 between different immunizations groups or Ags in culture (Figure 7B). Th1 responses were also weak in all immunization groups, as demonstrated by low levels of IFN-γ secretion after stimulation with MDA-LDL or nLDL (Figure 7C).
Splenocyte cytokine secretion stimulated by MDA-LDL. Splenocytes from LDLR–/– mice were cultured with the Ags human MDA-LDL and human native LDL (10, 40 and 80 µg/ml) or without Ags. (A) Secretion of IL-5 to splenocyte culture medium (undiluted, pooled samples) was measured after 72 h of Ag stimulation using a chemiluminescence-based immunoassay. Inset shows the plasma IL-5 concentration measured from pooled samples (1:3 dilution) taken after fourth and fifth booster immunization. MDA-LDL-immunized mice are compared with all mice receiving saline injections. Secretion of (B) IL-10 and (C) IFN-γ were measured in splenocyte culture media using Cytometric Bead Assay. #Below detection limit.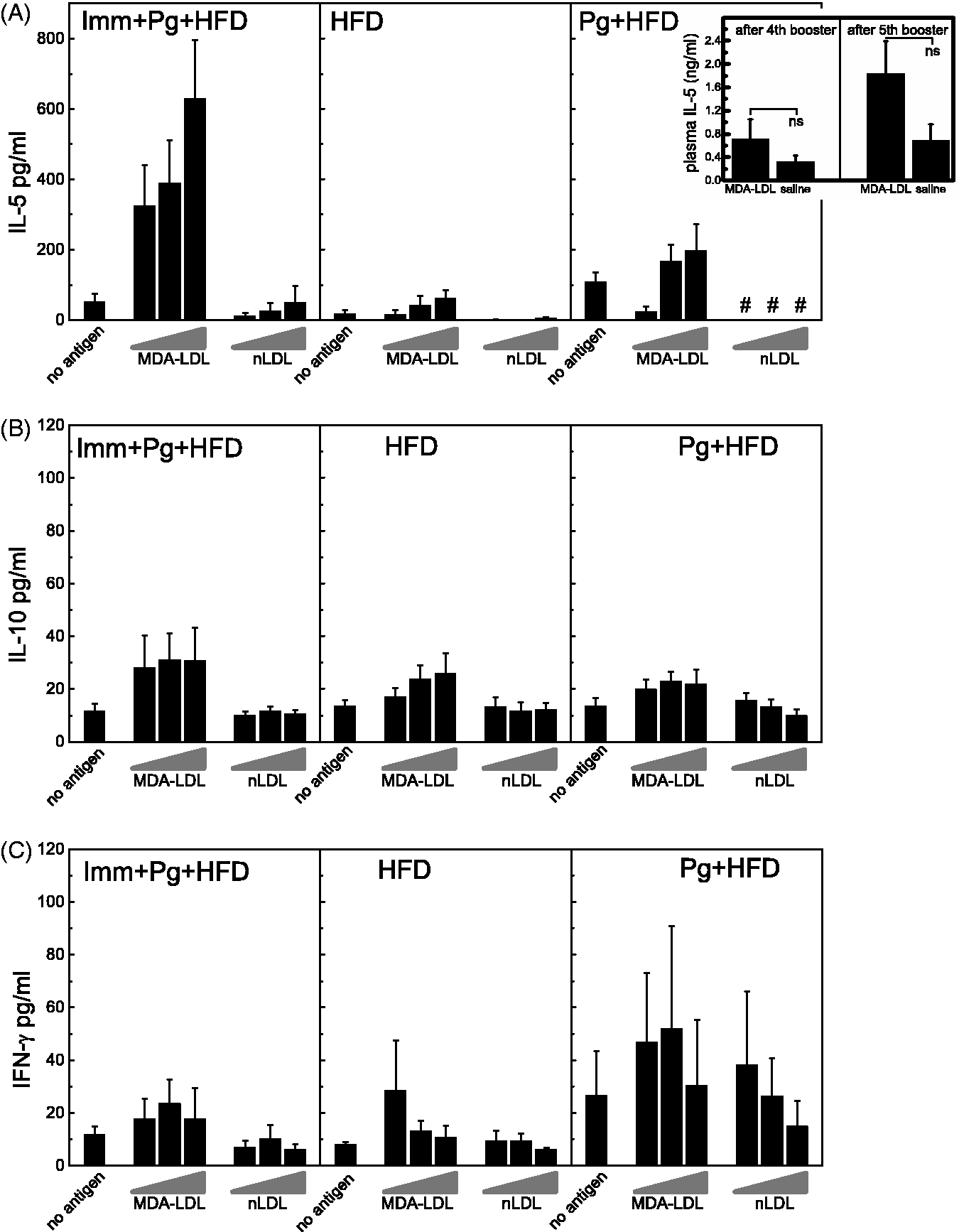
Pg challenge increases MDA epitopes in circulating apoB-100/48-containing particles
Effects of Pg challenge on oxidative stress in the circulation of HFD LDLR–/– mice were investigated by determining the amounts of MDA and PC epitopes in plasma apoB-containing particles. Plasma apoB-100/48-containing particles were captured on microtiter plates, and MDA and PC epitopes were detected with a chemiluminescence immunoassay. MDA epitopes in plasma apoB-containing particles were significantly increased in both groups receiving Pg challenge (Figure 8A). Measurements were made after the intensive application period and compared with the samples taken after immunizations. ApoB-containing particles in plasma contained PC epitopes abundantly, but there were no differences between bacterial challenge and vehicle-treated groups (Figure 8B).
Proatherogenic changes associated with Pg challenge. ApoB-containing particles from plasma were captured and (A) MDA and (B) PC epitopes were measured with specific mouse monoclonal IgMs using chemiluminescence immunoassay. Samples were collected after immunizations (imm) and after intensive topical Pg-application period (appl). At the latter time point, mice had received HFD for 4 wks. Samples were measured individually using triplicates for (A) MDA epitope determination and (B) in the assay with anti-PC Ab three samples were combined and analyzed in triplicate. (C, D) MCP-1 expression in the liver was analyzed as gene copy numbers (C), and the corresponding liver RT2 PCR Array results as fold change are shown in (D). Color-scaled boxes of log2 of fold change in hepatic MCP-1 expression represent comparisons of (1) Imm + HFD + Pg vs. Pg + HFD, (2) Imm + HFD + Pg vs. HFD and (3) Pg + HFD vs. HFD. *P < 0.05, **P < 0.01.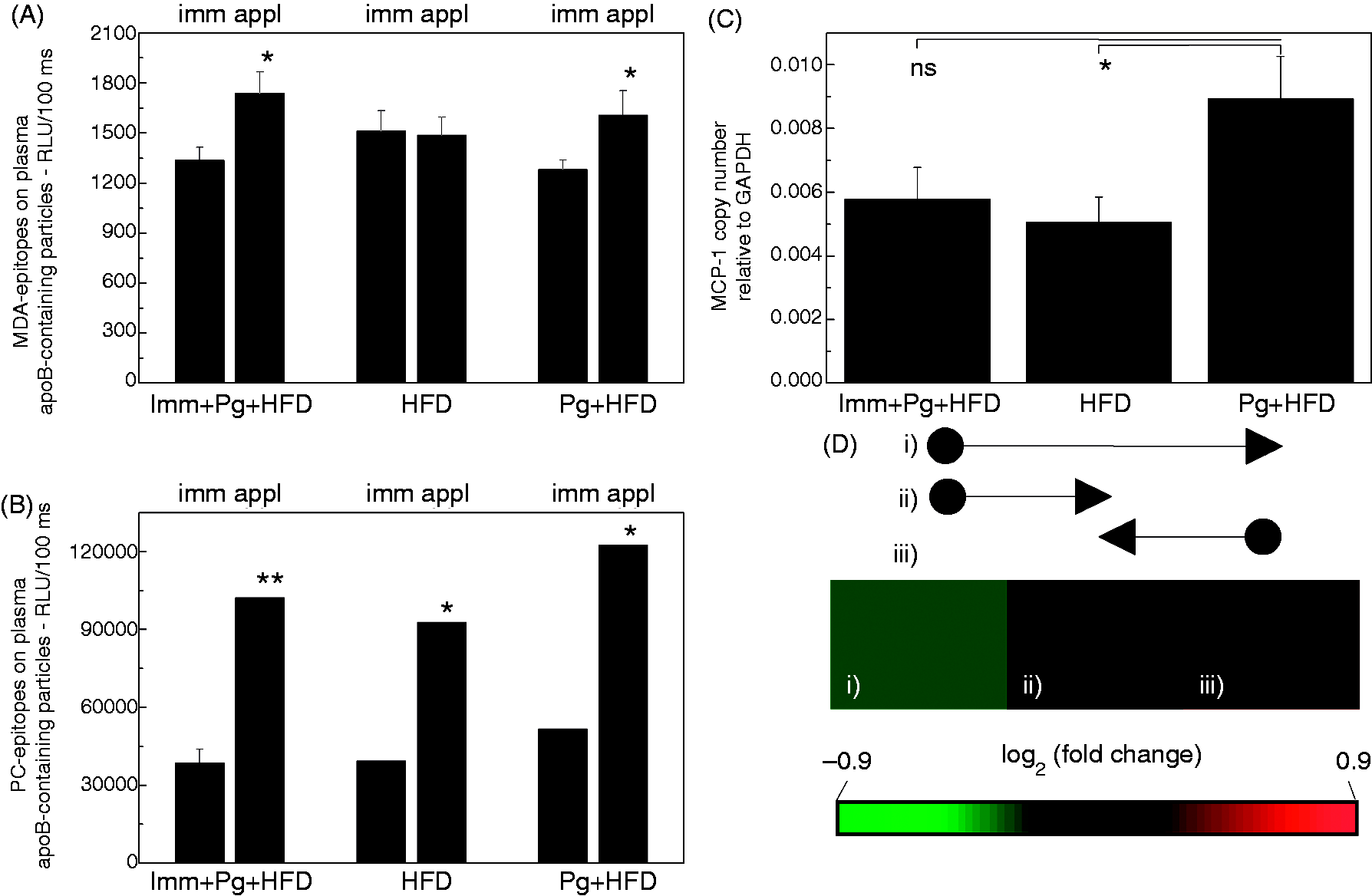
Specific IgM to MDA-LDL associates with reduced cholesterol levels in MDA-LDL-immunized mice
The correlations between results of competitive immunoassay with MDA-LDL as competitor and plasma cholesterol levels in LDLR–/– mice were analyzed at the end of the study. Plasma cholesterol and triglyceride concentrations are presented in the online Supplementary Table. Plasma triglyceride and lipoprotein levels were not different in the experimental groups at any time point. However, MDA-LDL-immunized LDLR-/- mice had a significant inverse association between specific anti-MDA-LDL IgM and plasma TC (online Supplementary Figure S1A) and LDL cholesterol (online Supplementary Figure S1D) after Pg-challenge and HFD. These associations were not observed in the HFD control group without immunization or bacterial challenge (online Supplementary Figure S1B, E). Inverse associations between plasma TC (online Supplementary Figure S1C) or LDL cholesterol (online Supplementary Figure S1F) and specific IgM to MDA-LDL were not seen in Pg-challenged mice but, on the contrary, correlation coefficients were positive in the range of 0.5–0.6 in this group. These findings suggested that MDA-LDL immunization had an effect on the levels and functionality of MDA-specific IgM, which associated with lower plasma LDL cholesterol and TC levels. Plasma LDL cholesterol levels associated positively with the extent of atherosclerosis in the HFD control group without any interventions (online Supplementary Figure S1H). In the MDA-LDL-immunized mice the plasma LDL concentration did no longer associate with the extent of atherosclerosis (online Supplementary Figure S1G). Pg-challenged mice without the MDA-LDL immunization mice did not demonstrate a positive association between LDL cholesterol and atherosclerosis either, suggesting that topical application of Pg drives the accelerated atherosclerosis with mechanisms that are not directly related to LDL cholesterol levels (online Supplementary Figure S1I).
Pg challenge increases hepatic expression of MCP-1
qPCR arrays of atherosclerosis-related genes from the liver samples were used for screening the candidate genes whose expression was altered by Pg challenge and reduced by MDA-LDL immunization. The levels of hepatic MCP-1 gene expression were confirmed in qPCR assays using designed primers (Figure 8C), as the gene expression of MCP-1 was found to be increased in qPCR arrays of the livers of Pg-challenged mice compared to the immunized group or the vehicle-treated controls (Figure 8D). The copy numbers of MCP-1 mRNA were significantly increased in the livers of Pg-challenged mice compared with the vehicle-treated controls (Figure 8C). Also, MCP-1 copy numbers in the livers of MDA-LDL immunized mice receiving Pg-challenge were lower compared with the Pg-challenged group, and the MCP-1 gene copy numbers of the immunized mice were almost equal to those of the vehicle-treated controls (Figure 8C).
Down-regulation of innate and adaptive immune response genes in the spleen by MDA-LDL immunization
Expression of innate and adaptive immune response genes in the spleens was analyzed in an 84-gene array. MDA-LDL-immunized mice receiving Pg challenge mice had down-regulation of splenic MCP-1 expression; additionally, several genes belonging to TNF-α, IFN-γ and NF-κB signaling pathways were down-regulated when compared with Pg-challenged mice (online Supplementary Figure S2A, D). When comparing Pg-challenged mice to controls without bacterial challenge, all these genes were up-regulated (online Supplementary Figure S2B, D). MDA-LDL-immunized mice receiving Pg challenge demonstrated equal expression levels of these genes compared with controls without bacterial challenge (online Supplementary Figure S2C, D). The general tendency in MDA-LDL-immunized mice showed slight suppression of adaptive and innate immune responses despite the atheropromoting burden of Pg challenge and HFD, even though none of the individual genes had statistically significant differences in the expression levels (online Supplementary Figure S2A–G).
Discussion
In this study we have shown that immunization of LDLR–/– mice with MDA-msLDL without adjuvant protected from the accelerated development of atherosclerosis after challenge with the live periodontopathogen Pg. A profound link between periodontal infections and increased risk for atherosclerosis and cardiovascular disease has been established in several studies and meta-analyses.1,40–44 Yet the molecular mechanisms explaining the association have remained unresolved. Infection of the periodontal pocket provides a direct route for bacteria to enter the circulation, and multiple reports describe the contribution of systemic inflammation and bacteremia as a link between periodontitis and atherosclerotic vascular disease. 2 Several components of the innate and adaptive immune system modulate the course of atherogenesis, both at the systemic level and inside each developing plaque. 45 The association of periodontitis and atherosclerosis has also been connected to immune activation as a result of structural resemblance, namely molecular mimicry on bacterial epitopes and Ags derived from the human body. 46 Our previous investigations have revealed intriguing molecular mimicry between oxidatively modified epitopes on atherosclerotic plaque and the oral pathogen Pg. A mouse monoclonal IgM to MDA-LDL binds to hemagglutinin/adhesin domain of Pg gingipain protease, and the presence of similar type of cross-reactive IgM is also demonstrated in human sera. 11
Previously, several studies using apoE-deficient mice have reported the accelerated atherosclerosis after challenge with live Pg.3–6 In this study, we found confirmatory results in LDLR–/– strain using only mucosal topical exposure to Pg. The experimental design of this study was novel compared with previous studies investigating pathogen-accelerated atherosclerosis. We omitted the use of antibiotic prophylaxis before Pg challenge in order to avoid the disturbance of normal microbiota and possible consequences on the development of atherosclerosis.4,30 The presence of normal bacterial flora in mice at the time of Pg challenge is highly important considering the mutual development of periodontal infections and advancing atherosclerosis. Our principal aim was not the establishment of clinically detectable alveolar bone loss by Pg challenge in these mice but to investigate the systemic immune modulation of atherogenesis. Therefore, topical application to mucous membranes was selected as the most suitable means of delivery for Pg as it also closely mimics the infection route from human periodontal pockets. Immune responses to Pg were clearly demonstrated in topically challenged mice as indicated by elevated IgG levels to a mixture of bacterial Ags and LPS of Pg. Additionally, mice challenged with Pg had elevated IgG levels to recombinant domains of Pg gingipain supporting the immunodominant role of gingipain for the virulence of Pg. 11 The ATCC33277 strain of Pg used in this study is non-invasive without an ability to establish chronic infection distant from the actual site of infection.30,47,48
Many studies have shown that immunization of animals with oxidized LDL confers atheroprotection but the mechanisms have remained unresolved. Conclusions may also be difficult to draw because adjuvants have been commonly used in these immunization studies,17–20,37 even though the atheroprotective effects of adjuvants alone have been described. 49 In this study, LDLR–/– mice were immunized with homologous MDA-LDL without adjuvant, and the immunization did not alter the specific IgM response to MDA adducts compared with the other mice on HFD. Increases in plasma total concentration of IgG and IgM throughout the study in all groups were in accordance with the previously published immunization study using another type of modified LDL, homologous carbamylated LDL, without adjuvant as the immunogen. 35 IgG binding to MDA-LDL was elevated in MDA-LDL-immunized mice after normalization with the total IgG concentration, and these findings are comparable with the previously published data demonstrating humoral IgG response after immunization with modified forms of LDL, including MDA-adducted17,37 and carbamylated LDL. 35 Previously, it has not been clear if there is any difference in the specific IgM response against the immunogen used in these studies, 35 and this work provides further evidence that the immunization with MDA-LDL elevates IgM binding to the Ag but the true specificity remains undisclosed.
B-1 cells produce natural IgM Abs that are linked to atheroprotective properties in humans and mice.15,26 Possible suppressive mechanisms of atherogenesis have been investigated in MDA-LDL-immunized mice; for example, IL-5 is suggested to function as a non-cognate stimulator of innate B-1 cells after MDA-LDL immunization and following expansion of MDA-LDL specific Th2 cells. 37 MDA-LDL-immunized mice in this study showed similar dose-dependent induction of splenic IL-5 production by stimulation with MDA-LDL as described earlier. 37 Levels of IL-5 in the plasma were already elevated during the immunization period compared with saline-injected controls before any bacterial challenge or HFD was introduced. Atheroprotective effects of IL-5 have also been broadly described in humans and mice. IL-5 is produced by Th2-lymphocytes after antigenic stimulation, and produced to some extent by mast cells and by endothelial cells as well. 50 IL-5 stimulates B-1 cells to produce natural IgM Abs with a variety of specificities including LPS, PC and complement binding Abs. In humans, plasma IL-5 levels are related to Abs binding to oxidized LDL and remain as an independent determinant for carotid artery bifurcational intima-media thickness even after adjustment for traditional risk factors of atherosclerosis. 51 In this study, IL-10 secretion stimulated by MDA-LDL was not significantly elevated in MDA-LDL -immunized group. According to the recently published data, oxidized LDL is capable of inhibiting the IL-10 production induced by Pg LPS in monocytes. 52 Taken together, both of these modulating and dampening factors, either bacteria or oxidized LDL in the culture containing the mixture of all cell types of the spleen can make the comparison of IL-10 data to the previously published work complicated. 37
Detailed investigation of MDA-specific IgM response in the different treatment groups of this study revealed interesting associations to plasma total and LDL cholesterol levels. MDA-LDL immunization of LDLR–/– mice before Pg challenge mediated inverse association between specific anti-MDA-IgM and plasma total and LDL cholesterol as measured at the end of the study. One circumspect interpretation of these association analyses could be that MDA-LDL immunization had an effect on functions of specific natural anti-MDA-IgM that are linked to maintenance of plasma cholesterol levels. In part, these observations could also explain the beneficial effect seen on the development of atherosclerosis in MDA-LDL-immunized mice despite the Pg challenge. MDA-LDL immunization reduced atherosclerosis in Pg-challenged mice despite the fact that both groups receiving Pg via topical application demonstrated increased levels of MDA epitopes in plasma apoB-100/48-containing particles. It is noteworthy that Pg challenge specifically increased MDA epitopes but not PC epitopes. The levels of PC epitopes in plasma apoB-100/48-containing particles were at equal levels in all treatment groups after intensive topical application period and 1 month of HFD. The ubiquitous oxidative burden of HFD accompanied by aging of these mice presumably explains the increase in PC epitopes. Additionally, the MDA-LDL-immunized mice had suppressed expression levels of innate and adaptive immune response genes in the spleens even after Pg-challenge and HFD. The most important down-regulated genes in MDA-LDL-immunized mice were MCP-1 and members of NF-κB and TNF-α signaling pathways, which can all modulate atheroprogression.53–57 MCP-1 in the vessel wall functions as a chemoattractant regulating the recruitment of monocytes to atherosclerotic plaques. Therefore, MCP-1 is a contributing factor for atherogenesis.54,58 MCP-1 produced, for example, by hepatic stellate cells and Kupffer cells promotes liver inflammation and fibrosis by recruiting macrophages. 59 Thus, the findings of this study support the notion that the atheroprotective effect of MDA-LDL immunization is mediated by means other than the direct induction of specific IgM to MDA-LDL.
In conclusion, we examined the potential of homologous MDA-LDL immunization without adjuvant to alter innate and adaptive immune responses and protect from the pathogen-accelerated atherosclerosis using challenge of LDLR–/– mice with a major periodontitis bacterium, Pg. The findings of the study addressed interesting possibilities for modulating the linkage between bacterial infectious burden and atherogenesis via natural immune responses directed against oxidatively modified epitopes. LDLR–/– mice immunized with homologous MDA-LDL without adjuvant had elevated IgM and IgG levels to MDA-LDL compared with saline-treated controls. We found that immunization with MDA-LDL stimulated production of IL-5, which can be considered as a sign of general activation of B-1 cells. The atheroprotective effects of B-1 cells and IgM secreted by them have been demonstrated in several elegant studies;25,26 therefore, we suggest that the activation of B-1 cells led to the diminished atherosclerosis in MDA-LDL-immunized mice. Interestingly, the activation of innate immunity B-1 cells by MDA-LDL immunization seemed to be potent enough to also protect against the atheropromoting effect of Pg challenge. This finding opens up possibilities for investigating the activation of B-1 cells and their action in the axis of pathogen-accelerated atherosclerosis. Special focus should be directed on further investigating the role of molecular mimicry on MDA adducts and Pg, and on how innate immune recognition of this structural resemblance triggers the mechanisms that suppress atherosclerosis development.
Footnotes
Funding
This work was supported by the Academy of Finland (grant numbers 119012, 1266053, 134763); the Finnish Foundation for Cardiovascular Research; the Sigrid Juselius Foundation; the Sohlberg Foundation; the Finnish Cultural Foundation; the Ida Montini Foundation; the Paulo Foundation; and the Paavo Nurmi Foundation.
Acknowledgement
We thank Ms Sirpa Rannikko for excellent technical assistance.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
