Abstract
Intestinal macrophages originate from inflammatory blood monocytes which migrate to the intestine, where they differentiate into anti-inflammatory macrophages through a number of transitional stages. These macrophages typically remain hypo-responsive to commensal bacteria and food Ags in the intestine, yet also retain the ability to react to invading pathogens. In this study we examined the role of epithelial cells in inducing this intestinal macrophage phenotype. Using an in vitro system we showed that, in two-dimensional culture, epithelial cell-derived factors from a murine cell line, CMT-93, are sufficient to induce phenotypic changes in macrophages. Exposure of monocyte-derived macrophages, J774A.1, to soluble factors derived from epithelial cells, induced an altered phenotype similar to that of intestinal macrophages with decreased production of IL-12p40, IL-6 and IL-23 and expression of MHC ІІ and CD80 following TLR ligation. Furthermore, these conditioned macrophages showed enhanced phagocytic activity in parallel with low respiratory burst and NO production, similar to the response seen in intestinal macrophages. Our findings suggest a role for colonic epithelial cells in modulation of macrophage phenotype for maintenance of gut homeostasis. Further understanding of the cell interactions that maintain homeostasis in the gut could reveal novel therapeutic strategies to restore the balance in disease.
Introduction
The immune system in the intestine represents a unique environment that can quickly respond to harmful pathogens, but it remains tolerant to Ags from food and commensal bacteria. This balance between protective immunity and tolerance is largely dependent on the mononuclear phagocytes in the intestine, such as macrophages. 1 Macrophages are the most abundant mononuclear phagocytes in the gut, especially in the large intestine. 2 They are critical for the maintenance of gut homeostasis, as depletion of intestinal macrophages leads to increased susceptibility to intestinal inflammation. 3 They also regulate epithelial cell renewal and integrity, 4 and support the induction and survival of Treg cells in the mucosa.5,6 Unlike other tissue macrophages, intestinal macrophages are in a state of hypo-responsiveness. They do not produce pro-inflammatory cytokines, chemokines or NO in response to stimuli, neither do they up-regulate co-stimulatory molecules or Ag-presenting receptors.7,8 Their respiratory burst capacity is also down-regulated, 9 even though they are highly phagocytic and bactericidal.6,8,10 They do, however, produce the anti-inflammatory cytokine IL-10 both in a steady state and in response to stimulation.5,6,11
Recently, it has been shown that intestinal macrophages originate from inflammatory blood monocytes that migrate to the intestine, where they differentiate into anti-inflammatory macrophages through a number of transitional stages. 12 As this plasticity happens only in the intestine, it is probable that the intestinal environment is responsible for altering the phenotype of inflammatory monocytes upon their arrival in the gut. Indeed, increasing evidence has shown that the intestinal epithelium has an immunomodulatory role, influencing immune cells and shaping their function. For example, culture of human monocyte-derived dendritic cells (DC) with epithelial cell supernatants resulted in the induction of non-inflammatory DCs that favoured the generation of a Th2 response. 13 Furthermore, co-culture of monocyte-derived DCs with Caco-2 intestinal epithelial monolayer resulted in the induction of DCs with reduced expression of MHC class II, CD86 and CD80, and reduced levels of inflammatory cytokine production. 14
In order to investigate whether the intestinal environment can modulate macrophage phenotype, we established an in vitro system where we cultured J774A.1 murine monocyte-derived macrophages with CMT-93 colonic epithelial cell-conditioned media. Conditioned macrophages showed an altered phenotype and an impaired ability to respond to bacterial ligand, similar to that of intestinal macrophages. Our data therefore indicate a role for epithelial cells in the alteration of macrophage phenotype for the maintenance of gut homeostasis.
Materials and methods
Cell culture and conditioning
CMT-93 mouse colonic epithelial cells and J774A.1 mouse macrophages were obtained from the European Collection of Cell Cultures and maintained in DMEM (Sigma-Aldrich, St. Louis, MO, USA) supplemented with 10% FCS (Gibco) and 2% penicillin-streptomycin (Gibco, Carlsbad, CA, USA). Cells were incubated at 37℃ in a humidified atmosphere of 5% CO2.
CMT-93 cells were grown until they were approximately 80% confluent after which fresh media was added, left on the cells for 24 h and then collected, centrifuged and stored at −20℃ for conditioning experiments.
J774A.1 macrophages were incubated for 6 h or 24 h with medium alone (unconditioned) or CMT-93-conditioned medium. After incubation, cells and supernatants were collected for further analysis. To determine the macrophage response to stimuli, macrophages were conditioned for 6 h or 24 h in the presence or absence of CMT-93-conditioned medium before being stimulated with LPS (100 ng/ml; Enzo Life Sciences, Exeter, UK) for 24 h.
Cytokine and chemokine analysis and flow cytometry
Supernatants from unconditioned and CMT-93-conditioned macrophages were collected and spun to remove cell debris. Cytokines (IL-12p40, TNF-α, IL-6, IL-23 and IL-10) and chemokines (MIP-1α, MIP-2 and MCP-1) were measured in supernatants using a DuoSet ELISA kit (R&D Systems, Minneapolis, MN, USA) according to manufacturer’s instructions.
For flow cytometry, macrophages were harvested and stained with appropriate fluorescently labelled Abs—anti-CD86, anti-CD80, anti-CD40 (BD Biosciences), anti-MHCII, anti-TLR2 and anti-TLR4 (eBioscience)—for 30 min at 4℃ in the dark. Cells were then analysed using FACSAria I flow cytometer (BD Biosciences, Franklin Lakes, NJ, USA) and FlowJo software (Treestar, Ashland, OR, USA). In all experiments, forward and side scatter were adjusted to exclude debris and cell doublets. Dead cell exclusion was performed using propidium iodide staining (Miltenyi Biotec, Bergisch Gladbach, Germany) at a final concentration of 1 µg/ml.
Phagocytosis
Unconditioned and conditioned J774A.1 macrophages were incubated with 1-µm fluorescent latex beads (Sigma-Aldrich), at a concentration of 20 beads per cell, for 2 h at 37℃. Cells were then washed with PBS/2% FCS and the uptake of beads (λex ∼ 470 nm; λem ∼ 505 nm) was measured by flow cytometry.
Nitrite analysis and the measurement of reactive oxygen species
The concentration of nitrite in the supernatants of unconditioned and conditioned macrophages was determined spectrophotometrically using Griess reagent (Promega, Madison, WI, USA) according to manufacturer’s instructions. Absorbance was measured at 543 nm using a VersaMax microplate reader (Molecular Devices, Sunnyvale, CA, USA). The nitrite concentration was determined using sodium nitrite as a standard. To measure the intracellular production of reactive oxygen species (ROS), unconditioned and conditioned cells were incubated with 20 µM of the cell permeant reagent 2′,7′-dichlorofluoresceindiacetate (DCFDA; Abcam, Cambridge, UK) for 30 min at 37℃ in dark, according to the manufacturer’s instructions. Fluorescence intensity of 2′,7′-dichlorofluorescin (DCF; λex ∼ 488 nm, λem ∼ 535 nm) was then measured by flow cytometry.
TGF-β and thymic stromal lymphopoietin neutralisation experiments
Neutralising Abs against TGF-β (10 µg/ml; R&D) or thymic stromal lymphopoietin (TSLP;) (10 µg/ml; R&D systems) or appropriate isotype controls [rabbit IgG (10 µg/ml) and rat IgG2A (10 µg/ml), both from R&D Systems) were added to conditioned media for 2 h at 4℃. Conditioned media with neutralised TSLP or TGF-β was then added to J774A.1 macrophages and left for 24 h. After 24 h, macrophages were collected and the production of cytokines (TNF-α, IL-6) was measured by ELISA (R&D Systems). In parallel, as a control, macrophages were grown in conditioned or unconditioned media without neutralising Abs.
Statistics
All data were analysed using Prism version 5 (GraphPad, La Jolla, CA, USA). For comparison between groups, the unpaired t-test was used and data were considered statistically significant if P < 0.05.
Results
Conditioning with colonic epithelial cell media modulates cytokine and chemokine secretion from steady-state macrophages
The development of resident colonic macrophages seems to be driven by the intestinal environment. 12 As colonic epithelial cells provide a first line of defence against commensal and pathogenic microorganisms, we hypothesised that these cells could be an important factor in driving the colonic macrophage phenotype. In order to explore this idea, we incubated J774A.1 macrophages with the supernatants from the mouse colonic epithelial cell line CMT-93. We first tested whether CMT-93 supernatant can affect the cytokine and chemokine secretion from unstimulated (steady-state) macrophages. We also investigated whether the length of time of conditioning further affects these parameters.
Conditioning of macrophages with CMT-93 supernatants modulated cytokine and chemokine secretion. Both 6-h and 24-h conditioning induced up-regulation of TNF-α and down-regulation of MIP-2 from conditioned macrophages when compared with unconditioned control macrophages (Figure 1A, B). MIP-1α was significantly up-regulated only after 24-h conditioning (Figure 1B), while there was no change observed in the production of IL-10 and MCP-1 from conditioned macrophages (Figure 1A, B). The presence of IL-12p40, IL-6 and IL-23 could not be detected using a commercial ELISA (R&D Systems). The cytokine and chemokine content was also measured in the conditioned media itself, prior to addition to macrophages. The levels of TNF-α, IL-10 and MIP-1α were undetectable, while the levels of MIP-2 and MCP-1 were <150 pg/ml (data not shown). These results suggest that the soluble factors present in the epithelial cell supernatant have the ability to change selectively the basal levels of cytokine and chemokine production from macrophages.
Conditioning with colonic epithelial cell media modulates cytokine and chemokine secretion from steady-state macrophages. J774A.1 macrophages were incubated with unconditioned medium as a control or with CMT-93 supernatants (conditioned) for 6 h or 24 h. (A) Cytokine and (B) chemokine release was measured by ELISA. Data are presented as mean ± SEM of three replicates and are representative of three independent experiments. *P < 0.05, **P < 0.01,*** P < 0.001 determined by unpaired t-test. ns: not significant.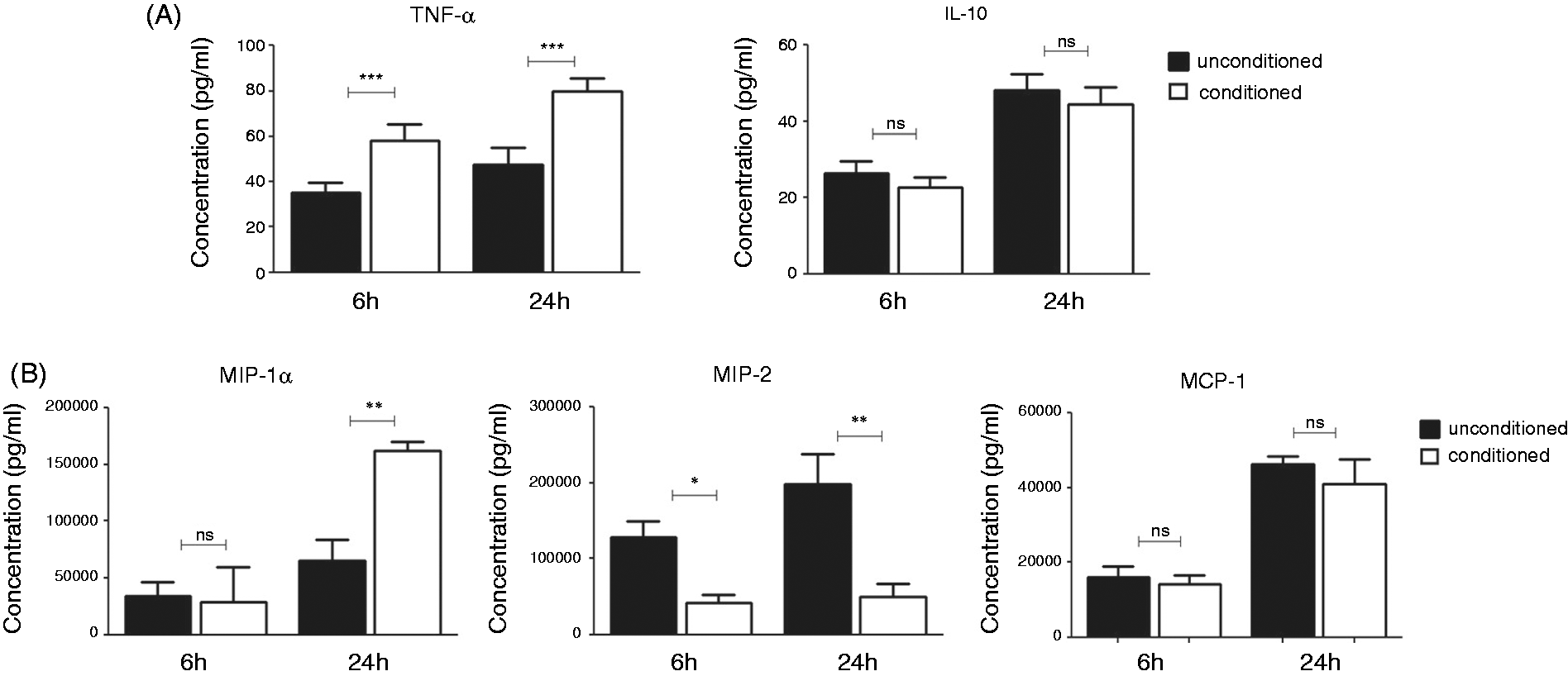
Surface phenotype of steady-state macrophages is modulated after conditioning with colonic epithelial cell media
In order to compare the phenotype of conditioned and unconditioned macrophages, we investigated the expression of the most important innate surface markers following 6-h and 24-h conditioning with CMT-93 supernatant.
Expression of CD40 was down-regulated on conditioned macrophages after both 6-h (MFIunconditioned = 1387 versus MFIconditioned = 914) and 24-h conditioning (MFIunconditioned = 1272 versus MFIconditioned = 572) (Figure 2). CD80, MHC class II and TLR4 were down-regulated after 24-h conditioning (MFIunconditioned = 817 versus MFIconditioned = 486; MFIunconditioned = 1976 versus MFIconditioned = 631; MFIunconditioned = 4308 versus MFIconditioned = 2151 respectively) compared with unconditioned control, while there was no change observed in the expression of CD86 and TLR2 at 24 h (MFIunconditioned = 2198 versus MFIconditioned = 2204; MFIunconditioned = 1495 versus MFIconditioned = 1482 at 6 h respectively and MFIunconditioned = 1533 versus MFIconditioned = 1490; MFIunconditioned = 1299 versus MFIconditioned = 1278 respectively) (Figure 2). Statistical analysis of flow cytometry data is included in Supplementary Figure S1 and shows the MFI percentage change from three independent experiments. Thus, soluble factors from epithelial cell supernatants also modulate macrophage phenotype.
Surface phenotype of steady-state macrophages is modulated following conditioning with colonic epithelial cell media. J774A.1 macrophages were incubated for 6 h or 24 h with unconditioned media as a control or CMT-93 supernatants (conditioned) and analysed for the expression of surface markers by flow cytometry. Histograms show surface marker expression on unconditioned cells (black line) and conditioned cells (dashed line). Filled histograms represent fluorescence of unstained cells. Data are representative of three independent experiments.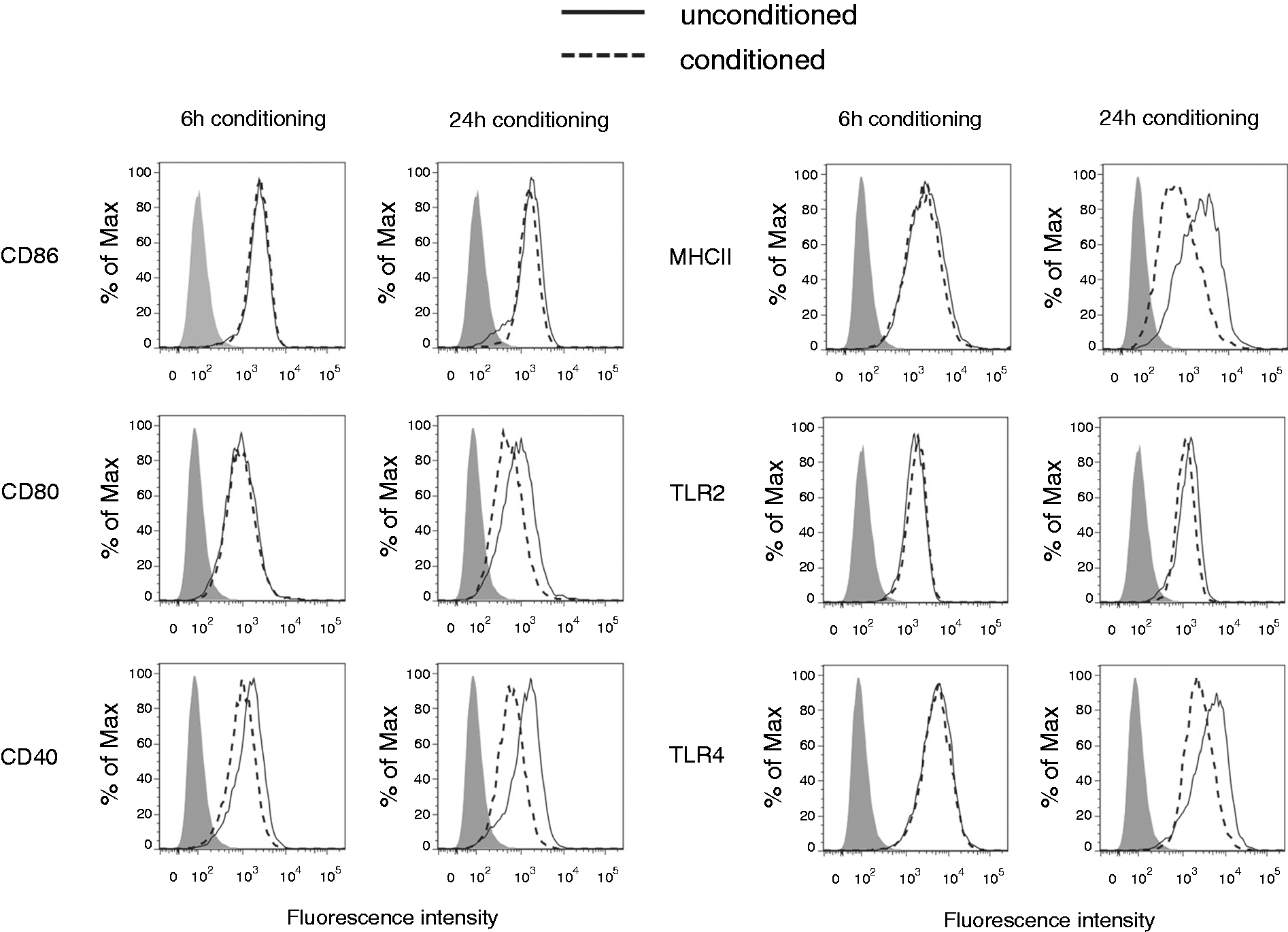
Conditioned macrophages show distinct cytokine and chemokine secretion in response to LPS stimulation
Macrophages conditioned with epithelial cell media were next analysed for their ability to respond to TLR4 stimulation. In order to assess this we pre-conditioned macrophages with CMT-93 supernatants for 6 h or 24 h before stimulation with LPS for 24 h.
Conditioned macrophages showed significantly decreased production of IL-12p40, IL-6, IL-23 and IL-10 in response to LPS compared with unconditioned LPS-stimulated macrophages, both after 6 h and 24 h of pre-conditioning (Figure 3A). However, they produced significantly higher levels of TNF-α when conditioned (Figure 3A). Conditioning did not have a strong effect on the production of chemokines, except for MCP-1 which was slightly up-regulated on LPS-stimulated macrophages following conditioning for 24 h (Figure 3B). These results suggest that exposure to epithelial cell media impairs macrophage response to inflammatory stimuli.
Conditioned macrophages show altered cytokine and chemokine secretion in response to LPS stimulation. J774A.1 macrophages were pre-conditioned with colonic epithelial cell media for 6 h or 24 h before being stimulated with LPS (100 ng/ml) for 24 h. Unconditioned, unstimulated cells were used as a control. (A) Cytokine and (B) chemokine levels were measured by ELISA. Data are presented as mean ± SEM of three replicates and are representative of three independent experiments. *P < 0.05, **P < 0.01, ***P < 0.001 determined by unpaired t-test. ns, not significant.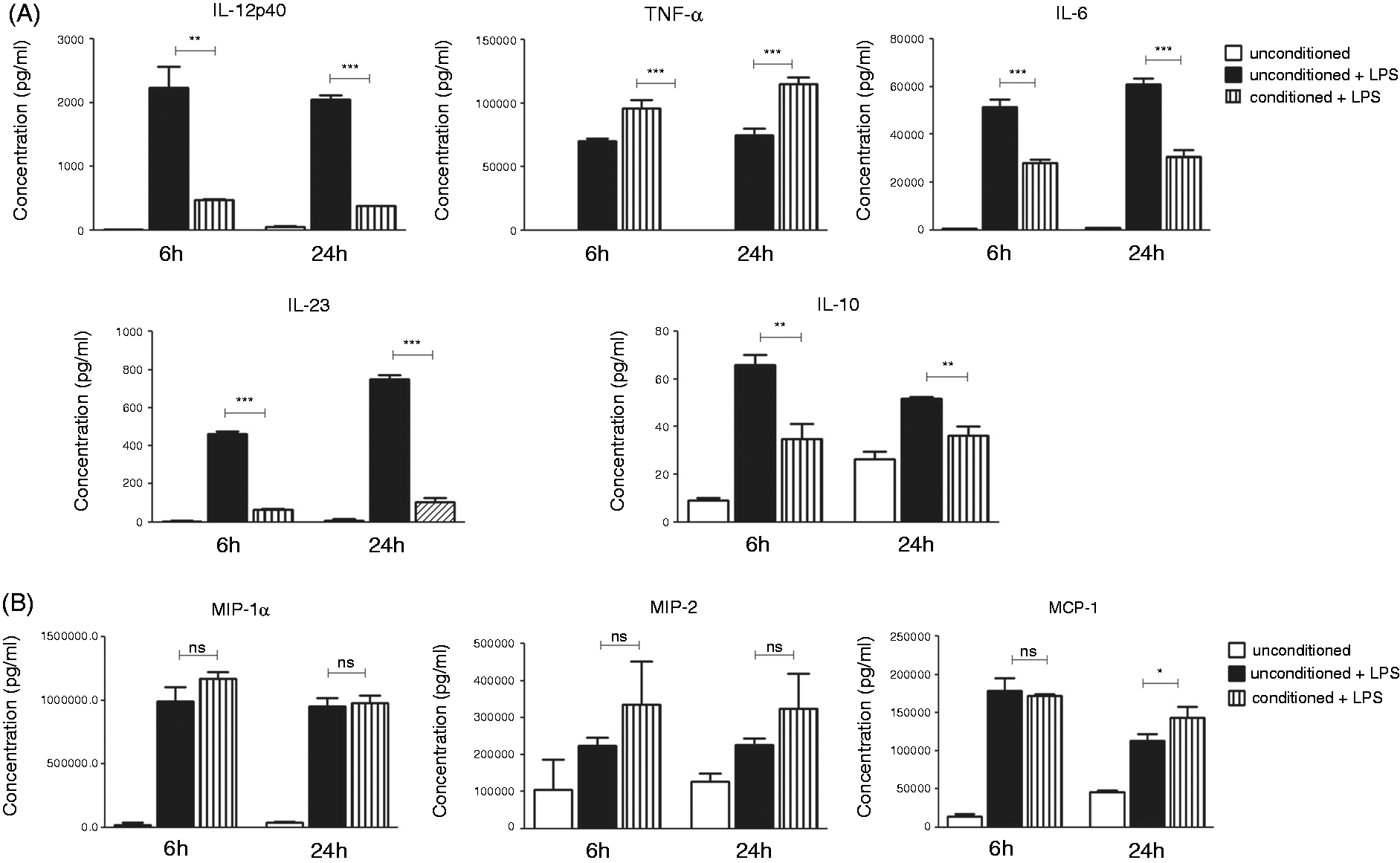
Conditioned macrophages have decreased expression of CD80, MHC class II and TLR4 in response to LPS stimulation
After demonstrating that conditioning macrophages modulate their cytokine and chemokine profile in response to inflammatory stimuli, we also wanted to investigate the cell surface marker expression on LPS-stimulated macrophage following conditioning.
Both 6-h and 24-h pre-conditioned macrophages showed decreased expression of CD80 and MHC class II in response to LPS (MFIuncond+LPS = 1768 versus MFIcond+LPS = 1161; MFIuncond+LPS = 8242 versus MFIcond+LPS = 2793 at 6 h, respectively, and MFIuncond+LPS = 2122 versus MFIcond+LPS = 1168; MFIuncond+LPS = 6533 versus MFIcond+LPS = 2467 at 24 h respectively) compared with unconditioned control macrophages (Figure 4). TLR4 expression was also decreased (MFIuncond+LPS = 10566 versus MFIcond+LPS = 5539 at 6 h and MFIuncond+LPS = 9433 versus MFIcond+LPS = 5832 at 24 h) (Figure 4). Pre-conditioning did not appear to have any effect on the expression of CD86, CD40 or TLR2 (MFIuncond+LPS = 3061 versus MFIcond+LPS = 2745; MFIuncond+LPS = 5895 versus MFIcond+LPS = 6259; MFIuncond+LPS = 2932 versus MFIcond+LPS = 2907 at 6 h, respectively, and MFIuncond+LPS = 3369 versus MFIcond+LPS = 3257; MFIuncond+LPS = 4390 versus MFIcond+LPS = 4329; MFIuncond+LPS = 3062 versus MFIcond+LPS = 2874 at 24 h respectively). Statistical analysis of flow cytometry data is included in Supplementary Figure S1 and shows the MFI percentage change from three independent experiments.
CD80, MHC class II and TLR4 expression are decreased on conditioned macrophages in response to LPS stimulation. J774A.1 macrophages were incubated for 6 h or 24 h with unconditioned media or CMT-93 supernatants (conditioned) before being stimulated with LPS (100 ng/ml) for 24 h. Expression of surface markers was analysed by flow cytometry. Histograms show surface marker expression on unconditioned cells (black line) and conditioned cells (dashed line) following LPS stimulation. Grey line represents the expression on unconditioned, unstimulated controls. Filled histograms represent fluorescence of unstained cells. Data are representative of three independent experiments.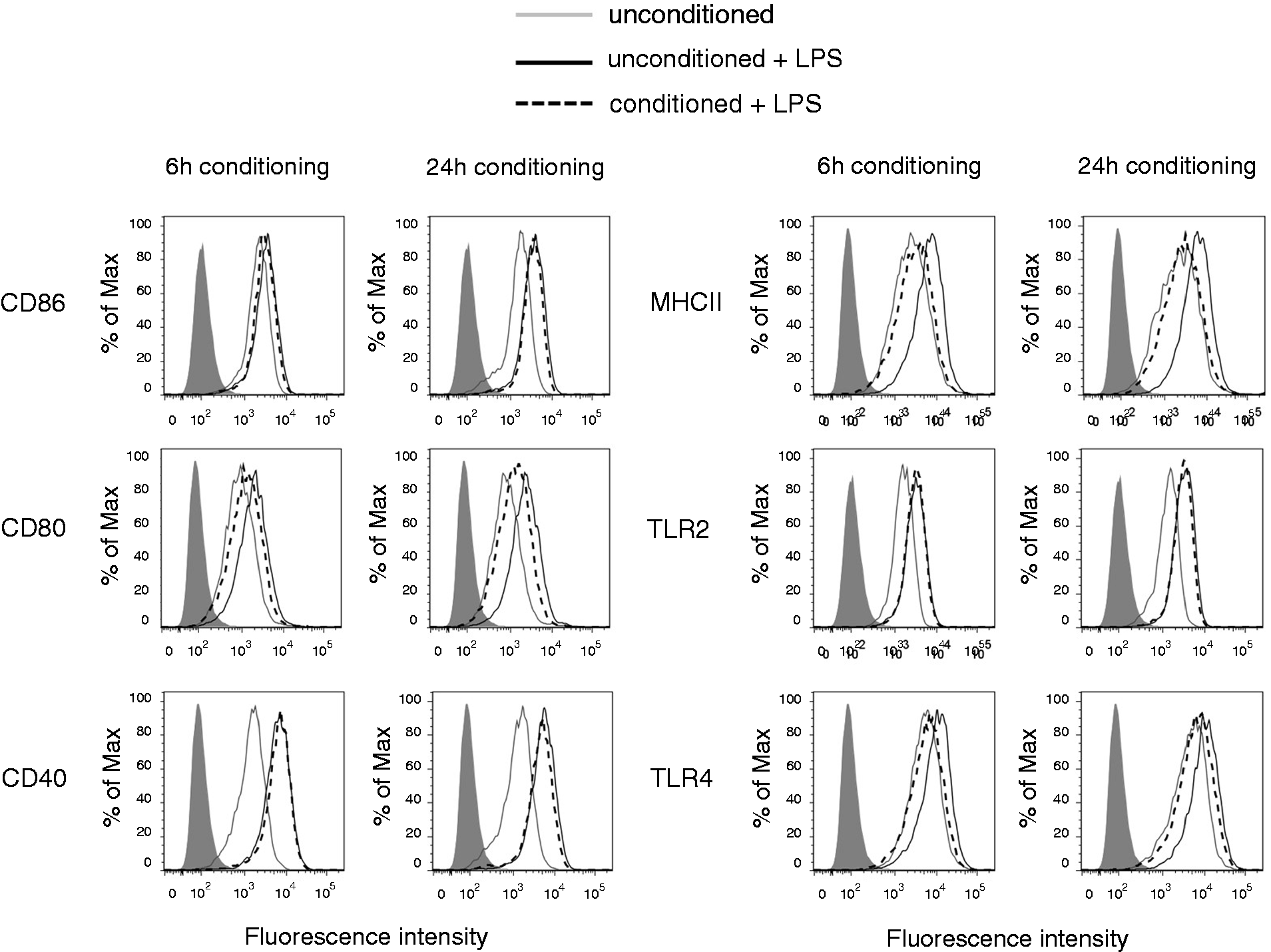
This data show that the epithelial cell environment selectively modulates macrophage phenotype in response to a TLR agonist.
Conditioned macrophages produce lower amounts of nitrite and ROS
Classically activated macrophages produce large quantities of inflammatory mediators such as NO and ROS in response to bacterial endotoxins. However, colonic macrophages do not generate a significant respiratory burst and NO production under the same conditions.7,9 We measured the levels of NO and ROS on conditioned macrophages to investigate whether epithelial cell soluble factor also affected these parameters. NO levels were detected by measuring the production of nitrite (NO2–) which is a stable breakdown product of NO. Levels of nitrite from unstimulated (steady state) macrophages were undetectable, but following stimulation with LPS, macrophages produced high amounts of nitrite (Figure 5). Conditioned macrophages produced significantly lower levels of nitrite in response to LPS, both after 6-h and 24-h pre-conditioning (Figure 5).
Conditioned macrophages produce lower amounts of nitrite in response to LPS stimulation. J774A.1 macrophages were pre-conditioned with colonic epithelial cell media for 6 h or 24 h before being stimulated with LPS (100 ng/ml) for 24 h. The concentration of nitrite in supernatants, after LPS stimulation, was determined spectrophotometrically using Griess reagent. The basal levels of nitrite could not be detected. Data are presented as mean ± SEM of three replicates and are representative of three independent experiments. *P < 0.05, **P < 0.01 determined by unpaired t-test.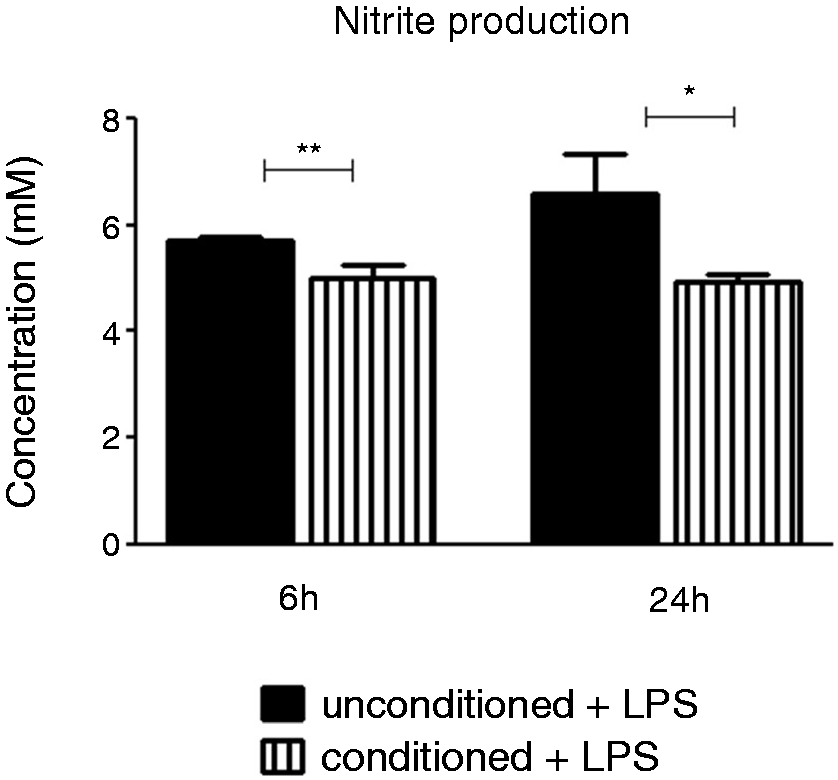
ROS levels were measured using the cell permeant reagent DCFDA, a fluorogenic dye that measures hydroxyl, peroxyl and other ROS activity within the cell. Conditioning decreased the basal levels of ROS in unstimulated macrophages (Figure 6A), as well as in response to LPS (Figure 6B). While 6-h conditioning was enough to decrease the ROS production from unstimulated macrophages, 24-h pre-conditioning was necessary to detect the decrease of ROS in LPS-stimulated conditioned macrophages (Figure 6B).
Conditioned macrophages produce lower levels of ROS in a steady state and in response to LPS stimulation. (A) J774A.1 macrophages were incubated with unconditioned media or CMT-93 supernatants for 6 h or 24 h. (B) After pre-conditioning, cells were stimulated with LPS (100 ng/ml) for 24 h. To measure the production of ROS, cell-permeant reagent DCFDA was added to cells for 30 min at 37℃, according to manufacturer’s instruction. After 30 min, fluorescence was analysed by flow cytometry. Histograms show fluorescence intensity of DCF in unconditioned cells (black line) and conditioned cells (dashed line). Data on graphs presents mean ± SEM of two replicates and are representative of three independent experiments. *P < 0.05, ***P < 0.001 determined by unpaired t-test. MFI percentage change was calculated relative to unconditioned, unstimulated cells.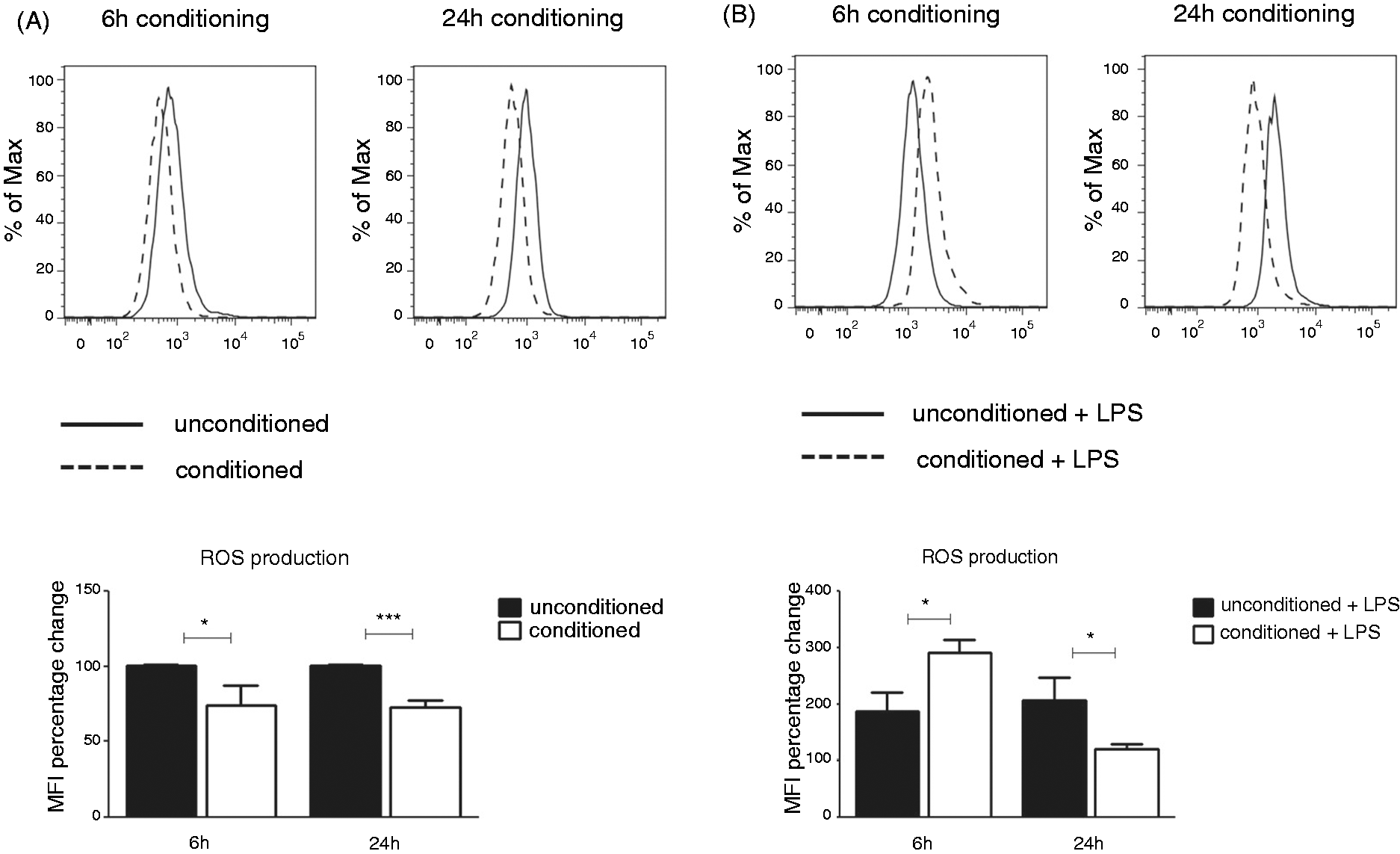
Conditioning macrophages with epithelial cell media enhances their phagocytic ability
Phagocytosis and clearance of pathogens is a hallmark of macrophage function and while colonic macrophages are hypo-responsive, they maintain a strong phagocytic capacity.
15
Therefore, we investigated the effect of CMT-93 conditioning on macrophages’ ability to phagocytose. Phagocytosis was measured by the percentage of cells that contained fluorescent beads after 2 h of incubation. Both unstimulated and LPS-stimulated conditioned macrophages showed increased uptake of fluorescent beads when compared with unconditioned controls (Figure 7). We repeated this experiment using CMT-93 cells as a negative control as they do not phagocytose. As expected there was no uptake of beads (data not shown). Thus, epithelial cell conditioning enhanced macrophage phagocytic ability.
Conditioned macrophages display enhanced phagocytic ability. (A) J774A.1 macrophages were incubated with unconditioned media or colonic epithelial cell supernatants for 6 h or 24 h. (B) After pre-conditioning, cells were stimulated with LPS (100 ng/ml) for 24 h. To assess phagocytic ability of cells, 1-µm fluorescent latex beads (Sigma-Aldrich) were added to the culture and macrophages were left to phagocytose for 2 h. Cells were then washed and analysed by flow cytometry for the uptake of beads. Histograms show the percentages of cells that contain (phagocytes) or do not contain (non-phagocytes) beads. Data on graphs presents mean ± SEM of two replicates and are representative of three independent experiments. *P < 0.05, **P < 0.01 determined by unpaired t-test.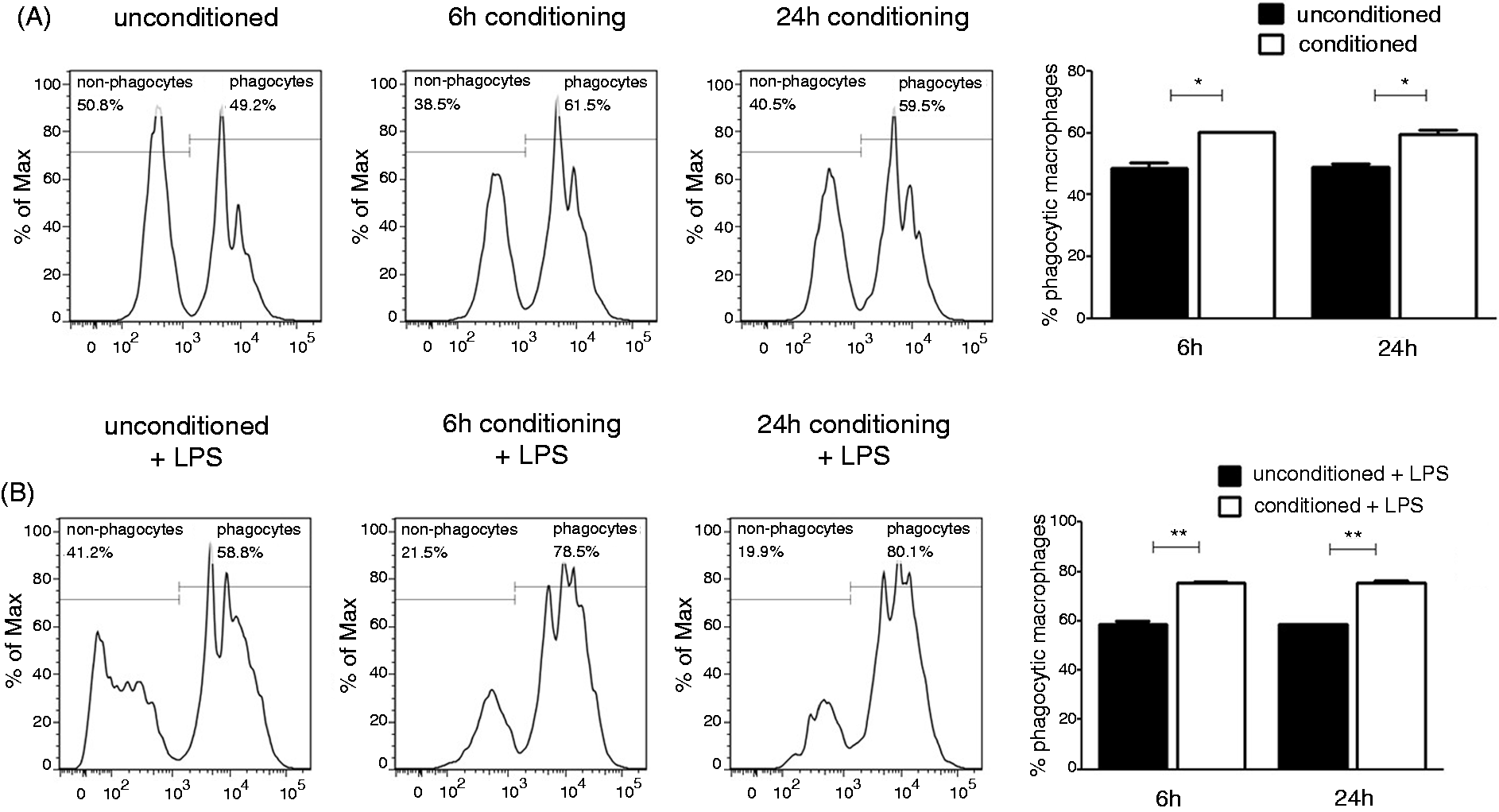
TGF-β and TSLP do not affect macrophage phenotype
TGF-β and TSLP, secreted by intestinal epithelial cells, have been shown to induce a tolerogenic phenotype in monocyte-derived DCs in vitro.13,16 In order to explore whether TGF-β and TSLP can also influence macrophage phenotype, we neutralised TGF-β or TSLP in CMT-93 media and investigated if the lack of these factors in the media affected the change in macrophage phenotype. Neutralisation of TGF-β or TSLP did not have any effect on TNF-α and IL-6 production from conditioned macrophage, both in a steady state and after LPS stimulation (Figure 8A, B). We also did not observe any effect on IL-12p40, IL-10 and IL-23 production, as well as the expression of CD86, CD80, CD40, MHCII, TLR2 and TLR4 cell surface markers (data not shown). The effectiveness of neutralising Abs is shown in Supplementary Figure S2.
TGF-β and TSLP do not affect macrophage phenotype. Neutralising Abs against TGF-β (10 µg/ml) or TSLP (10 µg/ml) or appropriate istoype controls were added to CMT-93 media for 2 h at 4℃. Conditioned media with neutralised TSLP or TGF-β was then added to J774A.1 macrophages and left for 24 h. After 24 h, macrophages were collected and the effect of (A) anti-TGF-β and (B) anti-TSLP on TNF-α and IL-6 production was measured by ELISA (R&D Systems). Data are presented as mean ± SEM of three replicates and are representative of three independent experiments. Statistical significance was determined by unpaired t-test. ns, not significant.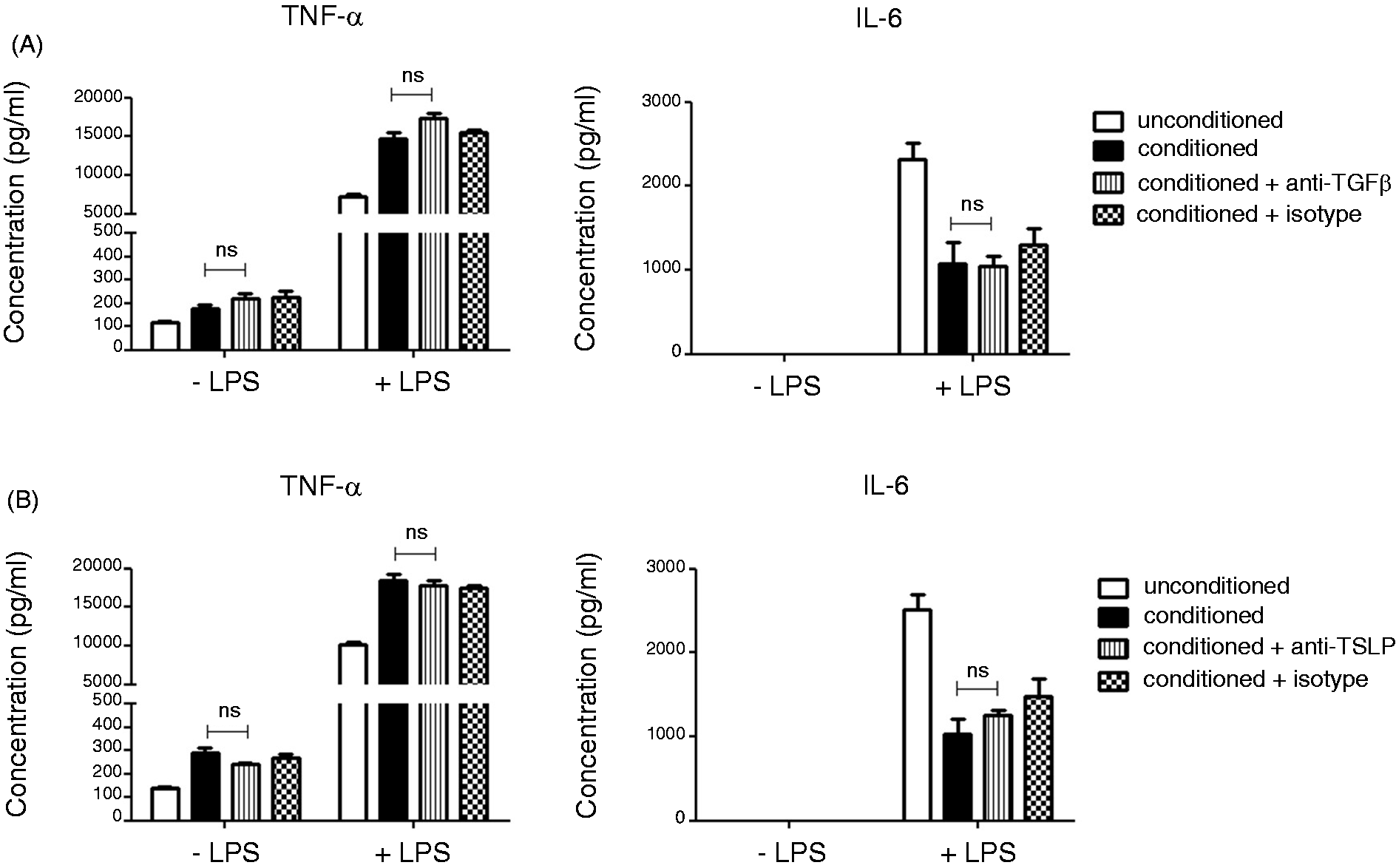
Discussion
In order to preserve homeostasis in the gut, which is constantly exposed to commensal bacteria and food Ags, intestinal macrophages adopt a state of hypo-responsiveness, while still retaining the ability to react to invading pathogens. Although these adaptations do not come as a surprise, little is actually known about what is driving them. One of the hypotheses is that the local intestinal milieu can influence immune cells and modulate their phenotype. Here we show that the soluble factors secreted by colonic epithelial cells can induce differentiation of normal macrophage into a tolerogenic phenotype resembling intestinal macrophages.
Macrophages conditioned with epithelial cell media showed altered expression of cell surface receptors involved in the immune response. We observed decreased expression of CD40, CD80, MHCII and TLR4 following conditioning of steady-state macrophages. Colonic macrophages have also been shown to have low levels of expression of co-stimulatory molecules and TLRs.8,17 Conditioned macrophages also showed impaired sensitivity to LPS by down-regulating the expression of TLR4 in response to this ligand. Furthermore, the LPS-induced up-regulation of CD80, MHCII and TLR4 was decreased on conditioned macrophages, indicating a development of a more hypo-responsive phenotype. Development of this hypo-responsive phenotype following conditioning was also observed when we examined the production of cytokines from macrophages following LPS stimulation. While unconditioned macrophages produced high amounts of IL-12p40, IL-6 and IL-23 in response to LPS, production of these pro-inflammatory cytokines was significantly suppressed from conditioned macrophages. It has been previously shown that colonic macrophages do not produce pro-inflammatory cytokines following stimulation with TLR ligands.5,10 Reduced production of IL-12p40 was also observed by other groups after they exposed monocyte-derived DCs to human epithelial cell line, Caco-2, supernatants.13,14 The same groups reported down-regulation of co-stimulatory molecules on DCs following conditioning.
While other groups concentrated only on the mentioned parameters, we also investigated the effects of epithelial cells on chemokine production and macrophage function, such as phagocytosis. Conditioning with epithelial cell media significantly increased the production of MIP-1α chemokine from unstimulated macrophages and MCP-1 from LPS-stimulated macrophages. Intestinal macrophages also produce MCP-1, as has been shown by Takada et al. 18 The same study showed that intestinal macrophages from MCP–/–mice produce lower amounts of IL-10 compared with WT mice, 18 which supports the homeostatic role of this chemoattractant in the intestinal environment. MIP-1α is important for the recruitment of eosinophils and neutrophils.19,20 Contrary to our previous understanding that eosinophils appear in the intestine only in a rare parasitic infections, recent studies show unexpectedly large amounts of eosinophils in preparations from healthy small intestine and colon. 21 It is speculated that they may contribute to epithelial renewal and barrier integrity in the gut, or may even be a source of the conditioning factors that maintain local macrophage hypo-responsiveness.21,22 If that is the case, the up-regulated production of eosinophil-recruiting chemokines that we report after epithelial cell conditioning may suggest a mechanism that supports intestinal homeostasis.
Phagocytosis is the hallmark of macrophage function. It is vital for the clearance of infectious pathogens, as well as apoptotic cells. 23 Colonic macrophages have been shown to be highly phagocytic even though they do not activate an immune response following phagocytosis. 15 Here we show that the phagocytic ability of macrophages is enhanced following conditioning with epithelial cell supernatant. Macrophages cultured in the presence of epithelial cell media increased their intake of fluorescent beads, both in a steady state and after LPS stimulation. However, although our conditioned macrophages became highly phagocytic they did not demonstrate any increase in their respiratory burst capacity and NO production, which would normally accompany phagocytic activity. This further suggests that conditioning the macrophages with epithelial cell media induces a similar phenotype to that of a colonic macrophage, as intestinal macrophages isolated from normal, non-inflamed human colonic mucosa do not show significant respiratory burst and neither do they express inducible nitric-oxide synthase.7,9 Overproduction of these inflammatory mediators can lead to significant tissue damage and cell death. 24 Therefore, by keeping the levels of ROS and NO low, intestinal macrophages can phagocytose microbes without inducing an inflammatory cascade.
Interestingly, although some changes occurred even after a short 6-h exposure to epithelial cell supernatants, longer conditioning had a more potent modulatory effect and macrophages developed a more pronounced anti-inflammatory phenotype when exposed to the epithelial cell media for longer. This gradual transition into an anti-inflammatory macrophage is also observed in vivo. Bain et al. showed that inflammatory monocytes, 12 once they arrive in the gut, differentiate into resident macrophages through a number of transitional stages, which they named P1–P4. The P1 population, which is similar to inflammatory monocytes, gradually acquires tolerogenic properties, and evolves through P2 and P3 stage into the P4 anti-inflammatory resident macrophages. 12 Our 24-h conditioned macrophages resemble P4 population with a decreased production of IL-6, IL-12p40, NO and ROS. Furthermore, they are hypo-responsive to TLR stimulation, but have an increased phagocytic ability, correlating with P4 population. 12 Weber et al. published similar observations. 25 They characterised two distinct macrophage populations in the colon, CX3CR1lo and CX3CR1hi, which correlate to P1 and P4, respectively, and showed that CX3CR1hi macrophages secrete significantly lower levels of IL-6 and IL-12, similar to our conditioned macrophages. 25
Considering that conditioned macrophages exhibited primarily anti-inflammatory properties, we were surprised by the finding that both unstimulated and LPS-stimulated conditioned macrophages significantly up-regulated their production of TNF-α. TNF-α is usually seen as a potent pro-inflammatory cytokine implicated in various autoimmune and inflammatory diseases, such as rheumatoid arthritis, psoriasis, Crohn’s disease, sepsis, diabetes and obesity.26–28 Although it was previously thought that colonic macrophages do not produce TNF-α,29,30 recent, more in-depth studies show that there is a constitutive production of TNF-α by resident colonic macrophages. 12 Indeed, TNF-α mRNA levels between P1 and P4, as well as CX3CR1lo and CX3CR1hi, are comparable.12,25 We believe that, although soluble factors secreted by epithelial cells media down-regulate other pro-inflammatory mediators, TNF-α production is left intact because TNF-α is not just a powerful pro-inflammatory agent that drives inflammatory disease, it is equally essential in maintaining normal homeostasis as it regulates a broad range of biological activities. Autocrine TNF-α signalling is important for the long-term survival of macrophages, 31 as well as their proliferation and differentiation.32,33 It also influences growth and differentiation of non-immune cells; it is shown to be important for gastric epithelial cell proliferation during ulcer repair, 34 and it is involved in the organogenesis of secondary lymphoid tissue of the intestine. 35 We do, however, see an increase in TNF-α production following conditioning, unlike Bain et al. and Weber et al.12,25 This could be due to a lack of IL-10 in our model, as well as the lack of soluble factors produced by other intestinal cell types. IL-10 and other factors might limit and regulate TNF-α production in the intestine, which is supported by the fact that most of the TNF-α producing P4 macrophages also produce IL-10, and these dual TNF-α+IL-10+ cells increase from P1 to P4. 12
It has been shown in several studies that intestinal macrophages produce anti-inflammatory IL-10.5,6 As mentioned before, we failed to observe induction of IL-10 following conditioning. Increased IL-10 production was also not observed in the study by Rimoldi et al. in which they conditioned DCs with Caco-2 supernatants; 13 therefore, it is possible that IL-10 production depends on different factors and it is not induced by epithelial cell priming. A study by Ueda et al. showed that IL-10 production is dependent on the presence of commensal microbiota and that colonic macrophages isolated from germ-free mice produce lower levels of IL-10. 36 Zeuthen et al. also investigated the effects of Gram-positive (G+) and Gram-negative (G−) commensals on Caco-2-conditioned DCs and showed that IL-10 is increased following encounter will G+ bacteria, but decreased upon encountering G- bacteria. 16 Use of LPS in our study, which is a major component of the outer membrane of G- bacteria, may therefore explain a decrease of IL-10 following conditioning. This is supported by our finding that epithelial cell conditioning did not alter IL-10 expression from unstimulated macrophages.
Another unexpected finding was the decrease in expression of MHC class II following conditioning. It is well documented that colonic macrophages constitutively express MHC class II molecule although it is not up-regulated following stimulation.10,37 It is possible that epithelial cell factors are not essential for the maintenance of MHC II expression as the same observation was recorded after co-culturing DCs with Caco-2 monolayers. 14 We also have to keep in mind that in our in vitro system we only explored the effects of intestinal epithelial cells on macrophages, and did not take into account the influence of other cell types present in the gut. Also, we have shown that in some cases the time of conditioning has an effect on the phenotypic change, with longer conditioning showing a more potent modulatory effect. Therefore, it is possible that longer conditioning is needed for some of the phenotypic changes to take place. These time-dependent phenotypic changes, as well as the existence of an intermediate macrophage phenotype in the gut (P2, P3) might explain why we see an increased ROS production in response to LPS after 6-h pre-conditioning but not 24-h pre-conditioning. Six hours of exposure to epithelial cell media might not be enough to induce changes in the ROS-induced signalling pathway, leaving macrophages still responsive to stimulation. However, this needs to be further investigated.
Intestinal epithelial cells constitutively secrete a broad range of antimicrobial peptides that are needed for the neutralisation of luminal bacteria. 38 They also secrete different chemokines and cytokines that have an effect on APCs and lymphocytes in the mucosa. 38 Two of these cytokines have been implicated in the induction of a tolerogenic phenotype of intestinal immune cells; TSLP and TGF-β. With this in mind, we anticipated that either TSLP or TGF-β are responsible for the induction of the anti-inflammatory phenotype following conditioning. However, we failed to observe any difference in macrophage phenotype after neutralisation of these factors in epithelial cell media. Still, we have only investigated the lack of TGF-β and TSLP on cytokine production and the expression of cell surface marker; therefore, we cannot rule out the importance of these factors in contributing to some other intestinal macrophage properties. The effect of other immunoregulatory molecules, such as protaglandin E2, which can regulate multiple innate and adaptive immune cell functions, 38 should also be investigated.
Although some authors reported that the differentiation of macrophages into the intestinal phenotype in vitro is only possible in the complex three-dimensional co-culture model, 39 here we show, for the first time, that epithelial cell-derived factors are enough to induce phenotypic changes. Taken together, our data show that exposure to epithelial cell-derived factors results in the development of an anti-inflammatory macrophage that resembles the intestinal macrophage phenotype. Conditioned macrophages became highly phagocytic but with a dampened production of pro-inflammatory mediators, which are key features of intestinal macrophages. We therefore suggest that colonic epithelial cell-derived factors play a key role in monocyte differentiation into intestinal macrophages. This complex crosstalk might be disrupted in disease states such as IBD where the epithelial cell barrier is compromised. Therefore, this may lead to incomplete differentiation of infiltrating monocytes that do not become tolerogenic to their environment, but remain pro-inflammatory. A better understanding of the mechanisms behind the cell interactions that maintain homeostasis in the gut could reveal novel therapeutic strategies to restore the balance in the disease and attenuate acute and chronic intestinal inflammation.
Footnotes
Funding
We acknowledge funding provided under the Programme for Research in Third Level Institutions (PRTLI) Cycle 4. The PRTLI is co-funded through the European Regional Development Fund (ERDF), part of the European Union Structural Funds Programme 2007–2013.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Acknowledgements
We would like to thank Claire O’Connell for constructive review of the manuscript.
