Abstract
Asymptomatic carriage of Campylobacter jejuni is highly prevalent in chicken flocks. Thus, we investigated whether chronic Campylobacter carriage affects chicken intestinal functions despite the absence of clinical symptoms. An experiment was carried out in which commercial chickens were orally infected with C. jejuni (1 × 108 CFU/bird) at 14 days of life. Changes in ion transport and barrier function were assessed by short-circuit current (Isc) and transepithelial ion conductance (Gt) in Ussing chambers. Gt increased in cecum and colon of Campylobacter-infected chicken 7 d post-infection (DPI), whereas Gt initially decreased in the jejunum at 7 DPI and increased thereafter at 14 DPI. The net charge transfer across the epithelium was reduced or tended to be reduced in all segments, as evidenced by a decreased Isc. Furthermore, the infection induced intestinal histomorphological changes, most prominently including a decrease in villus height, crypt depth and villus surface area in the jejunum at 7 DPI. Furthermore, body mass gain was decreased by Campylobacter carriage. This study demonstrates, for the first time, changes in the intestinal barrier function in Campylobacter-infected chickens and these changes were associated with a decrease in growth performance in otherwise healthy-appearing birds.
Introduction
Campylobacter jejuni is recognized as a leading cause of acute bacterial gastroenteritis in humans worldwide.1,2 Poultry is a major reservoir for C. jejuni and considered as the most common source for human infections as chickens can carry a high load of Campylobacter without clinical signs.3,4 Although C. jejuni is pathogenic to humans, it is believed that it is a commensal in avian hosts. 5 Insights into the mechanisms underlying these differences in infection outcome following Campylobacter exposure are of crucial importance in understanding the interaction of such a microorganism with its host, in assessing the clinical and economic implications of Campylobacter shedding by chicken, and in taking rational preventive measures based on the latter assessments.
The intestinal mucosa acts as a selectively permeable barrier, permitting the absorption of nutrients, electrolytes and water, while it also serves as a barrier of defence against pathogens by stimulating mechanisms that reduce the ability of pathogens and their toxins to invade the mucosa.6–12 The mucosal permeability is adaptable and regulated in response to extracellular stimuli such as bacteria. 10
Man 13 demonstrated in human cell culture models (T84 and Caco-2 cells) that several Campylobacter species have the ability to attach and invade intestinal epithelial cells and macrophages, to compromise the intestinal barrier integrity and physiology, to secrete toxins that kill host cells and to impair host cell functions. He also reported that attachment to the intestinal epithelium by Campylobacter increased the permeability of the intestinal epithelial barrier, decreased transepithelial electrical resistance, and resulted in a loss of tight junction proteins. Moreover, the common adhesive and invasive nature exhibited by several Campylobacter species implies that these mechanisms are one of the primary weapons used by different members of the genus Campylobacter to persist in and colonize the gastrointestinal tract. Nielsen et al. 14 showed that Campylobacter concisus induced a time- and dose-dependent decrease in tissue resistance, and increased permeability and apoptosis together with moderate changes in tight junction protein expression (claudin-5) in HT29/B6 colonic epithelial cells, and this observation coincided with the ability of the bacterium to compromise barrier functions.
In addition to affecting barrier function, enteric bacterial pathogens may also induce fluid and electrolyte secretion, and initiate inflammatory responses. 15 We recently demonstrated that these pathological consequences of infection differ greatly between the intestinal epithelium of chickens compared to what is known for mammals and humans. In our previous study, exposure of jejunal and cecal epithelia from healthy chickens to living Salmonella Enteritidis or its endotoxin acutely decreased the intestinal ion permeability and induced (de novo) sensitivity to the inflammatory and neuronal mediator histamine. 16 This indicated marked differences in the innate pathogen recognition processes between chickens and mammals, and may explain why chickens react to Salmonella and other invasive Gram-negative bacteria (e.g. Campylobacter) with subclinical carriage rather than overt disease. Whether and how the intestinal function is altered during the subsequent chronic carrier state, however, has not been investigated so far. Therefore, the aim of this study was to investigate the changes in passive ion permeability and histamine responsiveness in the chronic carrier state of C. jejuni infections. As such, this study can be considered as the first approach to obtain insight into the functional consequences of chronic Campylobacter colonization, using an integrative combination of in vivo and in vitro experiments.
Materials and methods
Ethics statement
The animal experiment was discussed and approved by the institutional ethics committee of the University of Veterinary Medicine and Austrian Federal Ministry for Science and Research under the license number GZ 68.205/0227-II/3b/2011. All husbandry practices and euthanasia were performed with full consideration of animal welfare.
Birds and feeding
Forty-eight 1-d-old broiler chickens (males and females) were obtained from a commercial hatchery (Ross-308, Geflügelhof Schulz, Graz, Austria) and randomly divided into two groups (24 birds/group). The birds were housed on wood shavings and were provided with food and water ad libitum. The broilers were fed diets based on wheat, maize, barley, soybean meal, soybean oil, sunflower oil and a premix with vitamins, minerals, amino acids, salt and mono-calcium phosphate. The diet contained 22% crude protein, 8.5% fat, 3.3% crude fiber and 1.4% lysine. At 1 and 14 d of age, cloacal swabs were taken from all birds and directly plated on modified charcoal–cefaperazone–deoxycholate agar (CM0739; OXOID, Hampshire, UK) for Campylobacter determination (at 42℃ for 48 h) to ensure absence of C. jejuni.
One group was kept as infected and the other one as the non-infected control group. At 14 d of age each bird in the infected group was inoculated orally by a feeding tube (gavage) with 1 × 108 CFU/bird of the Campylobacter reference strain NCTC 12744 as previously described.17,18
Body mass of birds
Body mass (BM) was determined at different time points and the body mass gain (BMG) was calculated as the difference between the final and initial bird mass during each of the weighing periods. Furthermore, feed intake over the course of the experiment was measured for control and infected birds and, consequently, feed conversion ratio (FCR) was calculated. In this experiment, six birds from each group were killed at each time point 7, 14 and 21 d post-infection (DPI) by bleeding of the jugular vein under anesthesia with a single dose of thiopental (1 mg/kg) injection into the wing vein.
Histomorphological examination of the small intestine
For histomorphological examination (crypt-villus measurements), tissue samples were taken from jejunum close to the junction of Meckel’s diverticulum. The samples were fixed in 4% buffered formalin for 48 h. The processing consisted of serial dehydration, clearing and impregnation with wax. Tissue cross-sections (three per bird, 5 µm thick) from each of the six birds per treatment were cut by a microtome and fixed on slides. Afterwards, deparaffination was performed in xylene (twice, 5 min each time) followed by rehydration in alcohol 100% (5 min), 96% (5 min) and 70% (5 min). A routine staining procedure was carried out using hematoxylin and eosin. 19 The slides were examined with an Olympus BX43F light microscope (Olympus, Tokyo, Japan) fitted with a digital video camera (Olympus DP-26; Olympus). The images were analyzed using Olympus Stream image analysis software v. 1.7 (Olympus). The total of the intact well-oriented, crypt-villus units were selected in triplicate for each intestinal cross-section per sample. The criterion for villus selection was based on the presence of an intact lamina propria. Villus height was measured from the tip of the villus to the villus-crypt junction, while crypt depth was defined as the depth of the invagination between adjacent villi. The villus width was measured at the basal and at the apical transverse of the villus. Villus surface area was calculated from villus height and width at half height, and the villus height to crypt depth (H:D) ratio was also calculated as previously described.20,21
Bacteriological investigation
Concurrent with the killing of birds, contents of jejunum and cecum (1 g) were collected for C. jejuni enumeration. Samples were diluted 1:10 (mass:vol) in PBS (BR0014G; OXOID) and the mixture homogenized using an Ultra-Turrax (IKA, Staufen, Germany). Afterwards, 10-fold dilutions were made from the stock suspension, and each dilution was direct-plated on Campylosel agar (BioMerieux, Vienna, Austria). The plates were incubated microaerophilically at 42℃ for 48 h. After incubation, typical Campylobacter spp. colonies were counted as CFU/g.
Ussing chamber analysis
The intestinal segments were taken from the mid-jejunum, cecum and colon (two replicates of each segment/bird) immediately after killing of birds (six birds/group). The preparation of epithelia and mounting in Ussing chambers was done as previously described.
22
The intestinal segments were harvested from the birds and placed into ice-cold buffer solution (contained in mmol/l: NaCl, 115; KCl, 5; CaCl2, 1.5; MgCl2, 1.2; NaH2PO4, 0.6; Na2HPO4, 2.4;
Statistical analysis
All data are presented as means and SEM. Following tests for normality (Kolmogorov–Smirnov’s test), Student’s t-test was used to detect significant differences between infected and control groups for performance, intestinal histology and electrophysiological responses in Ussing chambers. The mean of each bird was the experimental unit for histology data. Differences were considered significant at a level of P ≤ 0.05. All tests were performed using appropriate software (PASW statistics 20; SPSS, Chicago, IL, USA).
Results
Campylobacter-associated change in BM and bacterial colonization during the entire trial
All birds appeared clinically normal during entire trial. There were no signs of diarrhea, and fecal droppings looked normal in both control and infected birds. No mortality occurred over the course of the experiment. In addition, no differences were found between the BM of birds in the different groups prior to infection.
Growth performance of infected broiler chickens, in terms of the average BM, numerically decreased (P < 0.1) at 7 and 14 DPI (510 ± 61 and 1024 ± 65 g, respectively) compared with the controls (620 ± 26 and 1101 ± 67 g, respectively). Later on, at 21 DPI, BM (1398 ± 117 g) was significantly depressed by C. jejuni infection compared with the controls (1768 ± 109 g; P < 0.05; Figure 1). In addition, no differences were observed in the average daily feed intake between control birds (106 g/bird/d) and infected birds (104 g/bird/d). However, the FCR of infected birds (1.81) was numerically higher than control birds (1.41), which may indicate that C. jejuni infection can affect feed efficiency. These results suggest that the lower slaughtering mass after C. jejuni infection may be due to the reduction in the feed efficiency of infected birds.
Effect of a Campylobacter infection at 14 d of age on BM and BMG of broilers. (a) Mean BM and (b) mean BMG from non-infected control chickens and Campylobacter-infected chickens receiving 108 C. jejuni (n = 10). Data are means + SEM. Asterisks mark significant differences: *P < 0.05.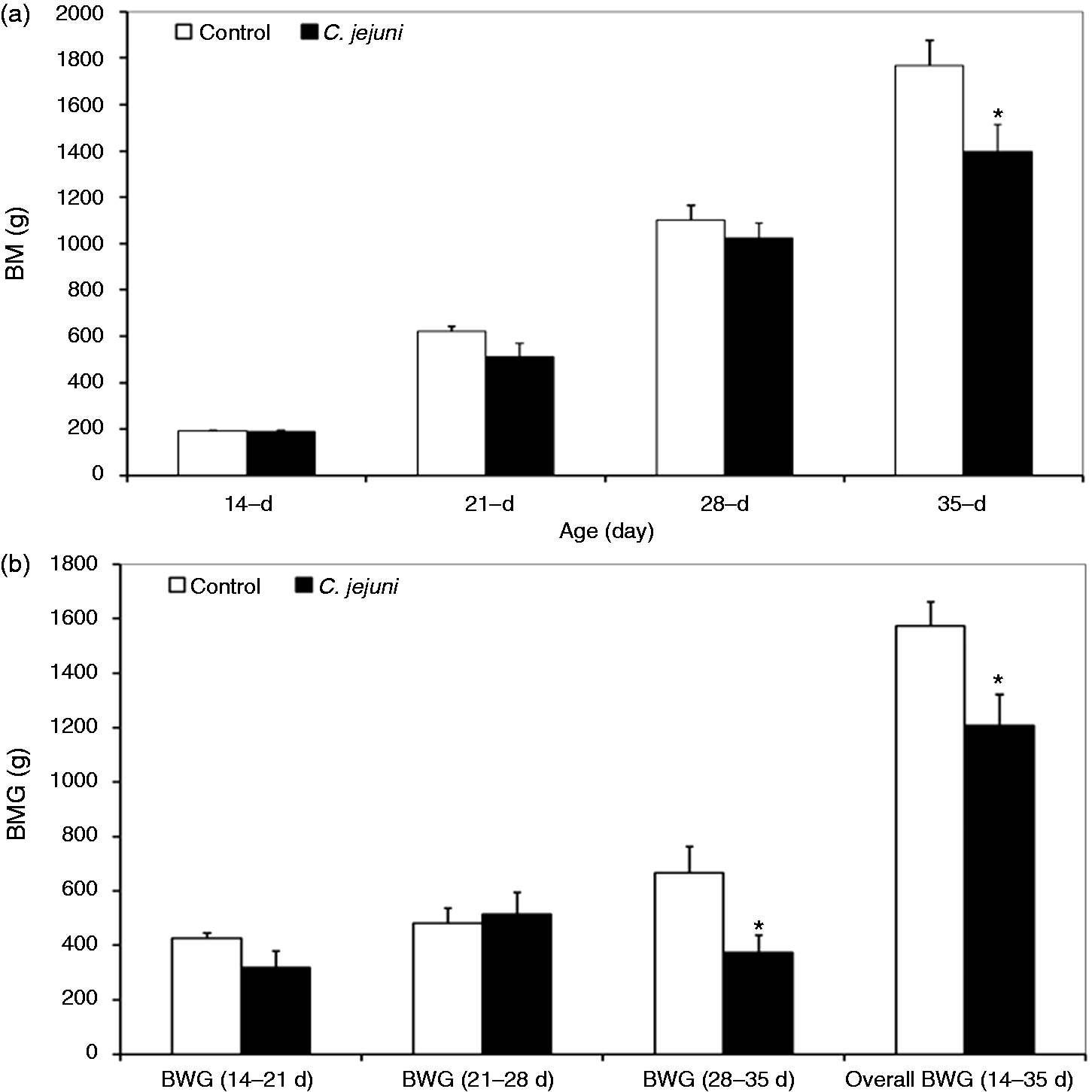
In the infected birds, at 7 DPI, C. jejuni colonization in jejunum and cecum were 0.69 × 103 ± 3.51 and 0.11 × 106 ± 0.69 CFU/g of contents, respectively. Later on, at 14 DPI, values of 0.26 × 105 ± 1.42 and 0.67 × 108 ± 4.74 CFU/g were recorded in jejunum and cecum, respectively. Furthermore, C. jejuni colonization in jejunum (0.66 × 105 ± 5.50 CFU/g) and cecum (0.46 × 108 ± 4.12 CFU/g) at 21 DPI was similar to 14 DPI. Campylobacter colonization was higher (P < 0.01) in the cecum and jejunum (P < 0.1) at 21 DPI than the colonization at 7 DPI. In contrast, C. jejuni was not detected in any of the control animals.
Influence of Campylobacter infection on intestinal histomorphology
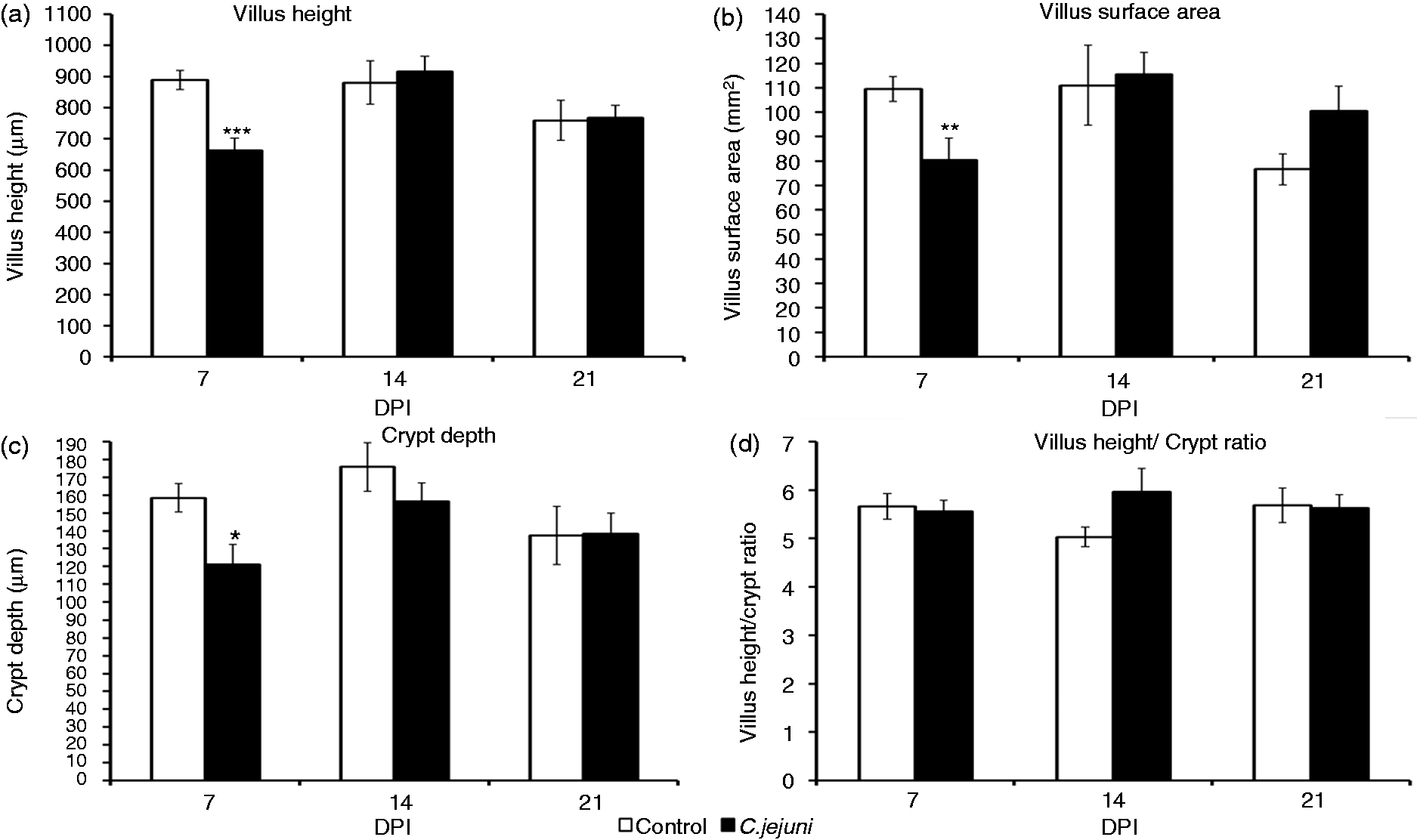
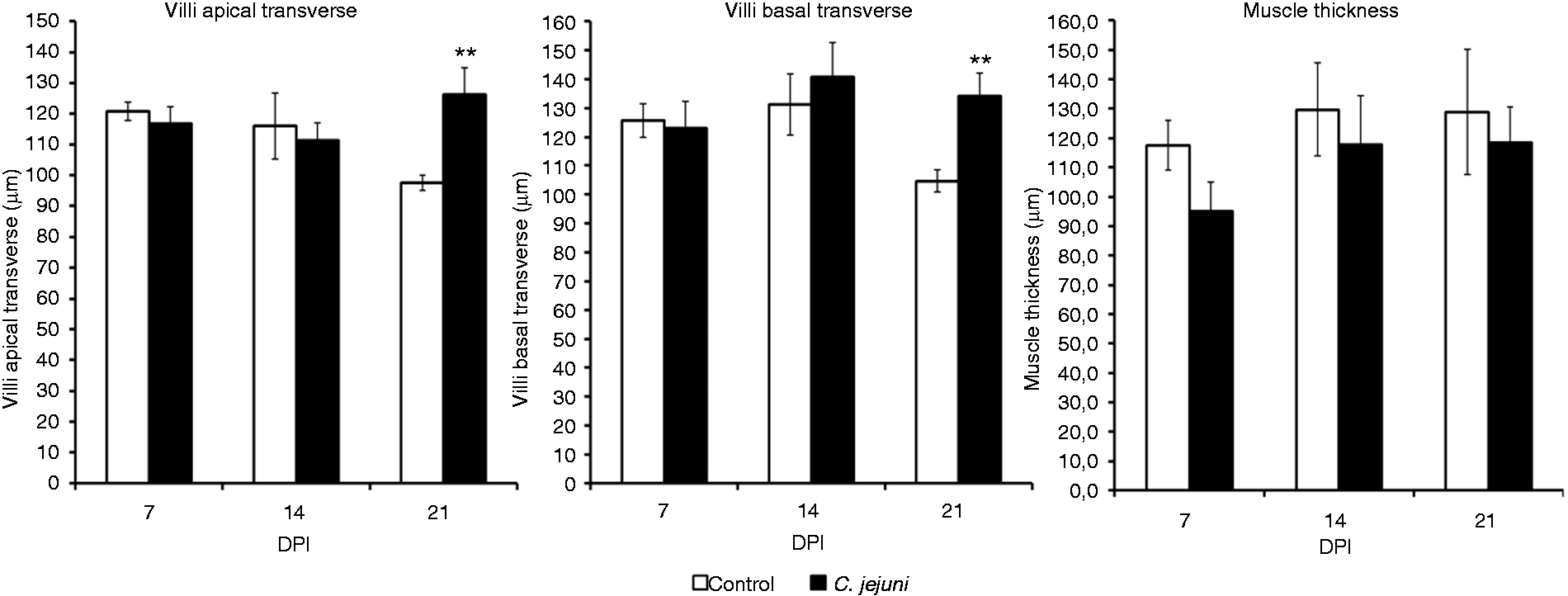
Intestinal histomorphometric analysis for the jejunum revealed that the C. jejuni colonization altered the small intestinal architecture (Figures 2–4). At 7 DPI, the villi in the jejunum were significantly (P < 0.001) shorter (662 µm) in the infected group than in the controls (889 µm) (Figure 3). Campylobacter infection also resulted in a decreased (P < 0.05) crypt depth and villus surface area (121 µm versus 80 mm2) compared with controls (159 µm versus 110 mm2). These changes resulted in a significantly decreased apparent villus surface area (80 versus 110 mm2, P <0.01) in the infected group. However, no effect of the infection was apparent for villus width, muscle thickness (Figure 4) and villus H:D ratio in the jejunal mucosa at 7 DPI. Interestingly, at 14 DPI, there was a trend for a greater H:D ratio in the Campylobacter-infected group, indicating a rapid cellular migration to permit renewal of the villus. Towards 21 DPI, differences in villus height and crypt depth disappeared, but the villi in the Campylobacter-infected group became wider than those in the control group, resulting in a trend for a larger apparent villus surface area in the infected group at 21 DPI (P = 0.071; Figure 3).
Histomorophology of jejunal villi of control (a) and Campylobacter jejuni-infected birds (b). Sections were stained with hematoxylin and eosin. Effect of Campylobacter infection on the jejunal villus height, villus surface area, crypt depth and villus H:D ratio. The data are represented as mean ± SEM (n = 6). Asterisks mark significant differences: ***P < 0.001, **P < 0.01, *P < 0.05. Effect of Campylobacter infection on the apical and basal transverse of jejunal villi, and muscle thickness of jejunal villi. The data are represented as mean ± SEM (n = 6). Asterisks mark significant differences: **P < 0.01.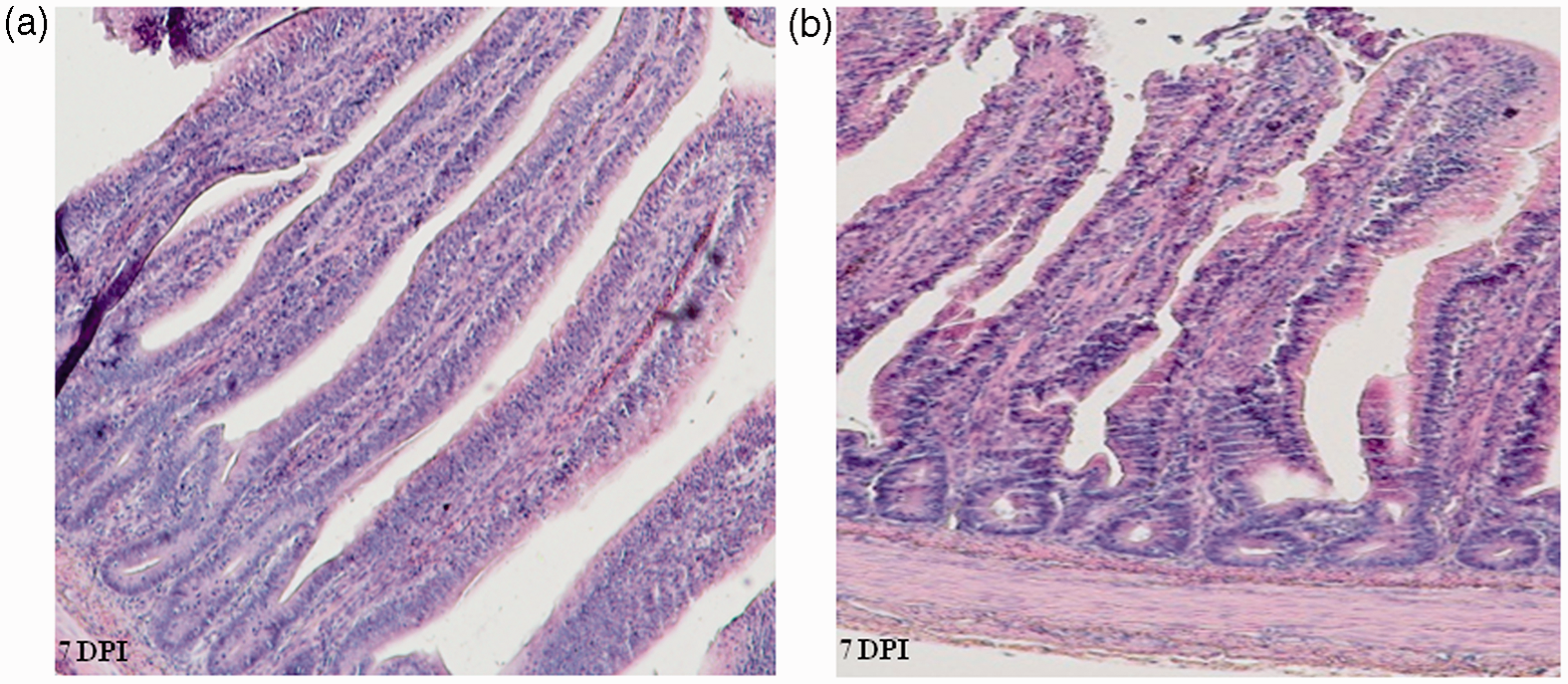
Campylobacter-associated changes in intestinal permeability and ion transport function
To characterize if infection with Campylobacter affected the electrophysiological properties of the gut, jejunal, cecal and colon sections were assayed for changes in ion transport and ion permeability, as reflected by short-circuit current (Isc) and trans-epithelial ion conductance (Gt).
Campylobacter infection decreased the basal Gt in the jejunum (P < 0.01) at 7 DPI. However, with the progression of infection, at 14 DPI the basal Gt recorded for the jejunum increased compared with control birds (P < 0.001) and tended to be higher in infected birds at 21 DPI (P < 0.10; Figure 5). However, active charge transfer across the jejunal epithelium was reduced at 14 DPI (P < 0.05) and as a trend also at 7 DPI (P < 0.10), as evidenced by decreased basal Isc (Figure 5).
Effect of a Campylobacter infection at 14 d of age on intestinal epithelial barrier function of broilers. (a) Tissue conductance (Gt; mS/cm2) and (b) short-circuit current (Isc; µA/cm2) from non-infected control chickens and Campylobacter-infected chickens receiving 108 C. jejuni (n = 6). Data are means + SEM. Asterisks mark significant differences: ***P < 0.001, **P < 0.01, *P < 0.05.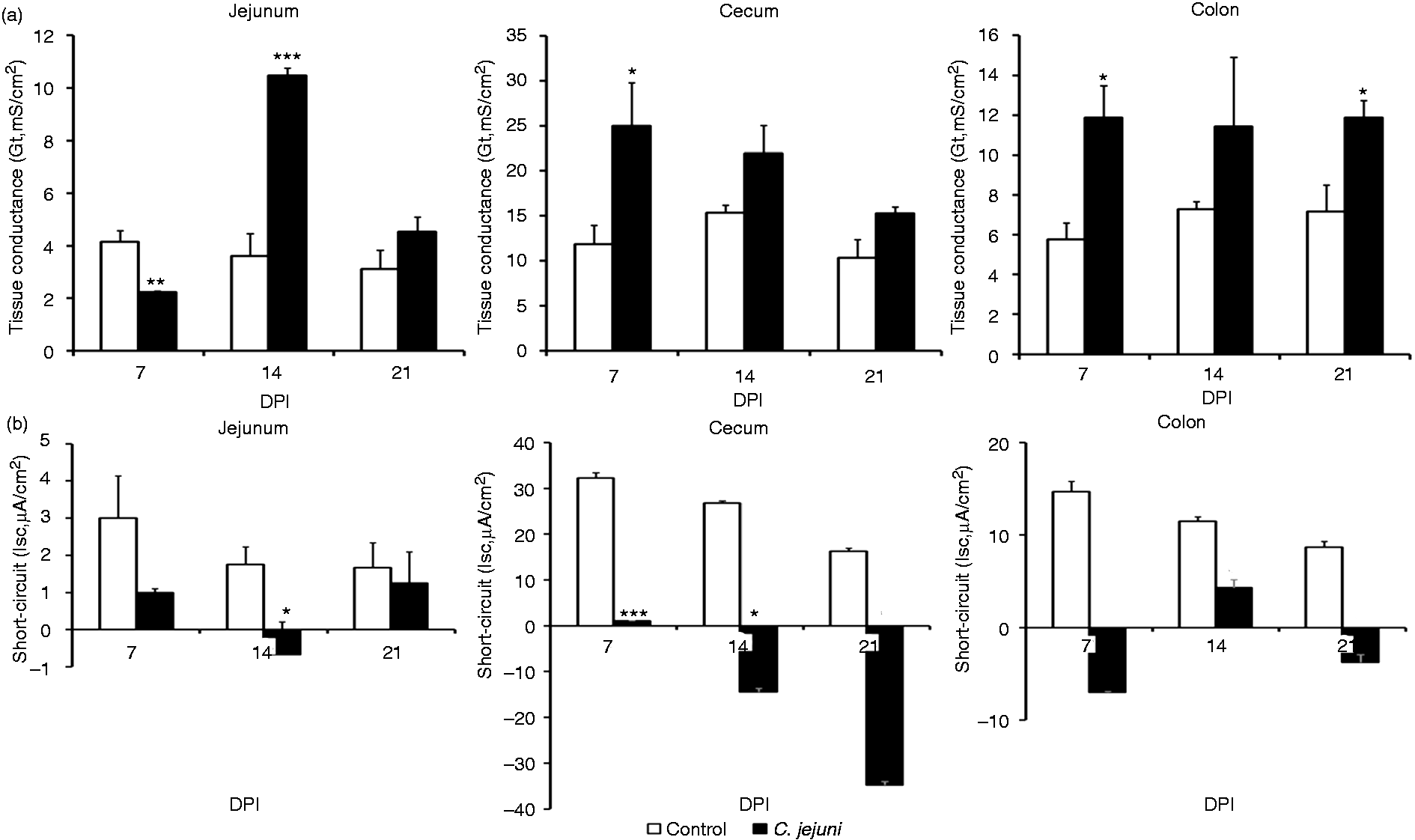
In cecum and colon, the Campylobacter infection increased or tended to increase the basal Gt at all sampling points (7, 14 and 21 DPI); the most prominent and statistically significant increase being observed at 7 DPI (P < 0.05; Figure 5). The increase in basal Gt was accompanied by a decrease in basal Isc that was significant in the cecum at 7 DPI (P < 0.001) and 14 DPI (P < 0.05), and observed as a trend in the cecum at 21 DPI and in the colon at 7 and 21 DPI (P < 0.10; Figure 5).
The changes of transmural conductivity (Gt) and short-circuit current (Isc) of broiler chickens intestine in response to histamine.
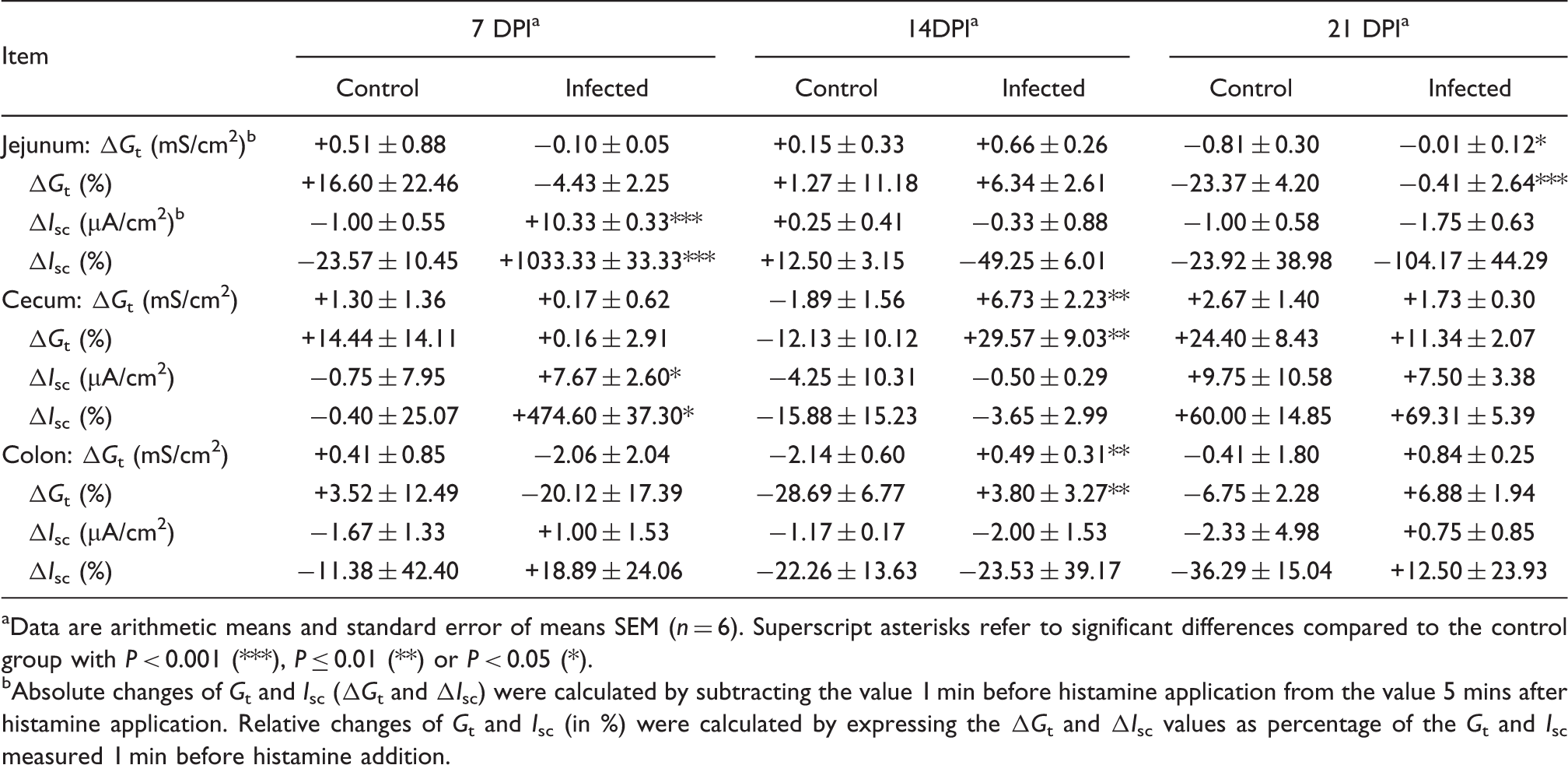
Data are arithmetic means and standard error of means SEM (n = 6). Superscript asterisks refer to significant differences compared to the control group with P < 0.001 (***), P ≤ 0.01 (**) or P < 0.05 (*).
Absolute changes of Gt and Isc (ΔGt and ΔIsc) were calculated by subtracting the value 1 min before histamine application from the value 5 mins after histamine application. Relative changes of Gt and Isc (in %) were calculated by expressing the ΔGt and ΔIsc values as percentage of the Gt and Isc measured 1 min before histamine addition.
Discussion
Previous functional studies in Campylobacter research have largely focused on the pathomechanisms that lead to diarrhea in humans, including, for example, invasion mechanisms and immunologic host reactions.23,24 Campylobacter research in poultry has largely been centered on the factors that determine the shedding and introduction of this bacterium into the human food chain.25,26 Furthermore, it was shown that the chick immune system is inefficiently activated and expression of several antimicrobial peptide genes is reduced, which both contribute to the persistent high level of Campylobacter colonization in chicken gut.27–29 Byrne et al. 30 and Van Deun et al. 17 showed that C. jejuni could adhere to and invade chicken primary intestinal cells. However, there is a lack of knowledge about the effects of Campylobacter infection on intestinal barrier function, except for a few reports that were mostly performed in human cell culture models.13,31
Nonetheless, the potential effects of Campylobacter challenges on the intestinal function of chickens are of great interest given the high prevalence of this bacterium in chicken flocks and the potential consequences for animal performance. With this study, we provide novel information about such effects of Campylobacter infection in chickens, as evidenced by alterations in intestinal morphology and electrophysiology.
Earlier experimental studies have already demonstrated that a high incidence of C. jejuni can influence BMG of chickens.32,33 The results of this study confirm these observations. Oral infection of chickens with a dose of 108 CFU/ml resulted in a reduction in mass gain compared with controls during the 3-wk post-infection period, thereby underlining the deleterious effect of C. jejuni on chicken growth. However, all birds appeared clinically healthy and C. jejuni produced no gross lesions in chicken organs.
Gharib-Naseri et al. 34 have also demonstrated that the BMG of C. jejuni-infected broiler chickens was reduced 4 wk after infection. The authors hypothesized that the reduction of the BM may be due to a compromised intestinal absorption of nutrients, which is supported by our results. Furthermore, the effect on BM in this study might be strain-specific, as it had been shown that C. jejuni isolates can have different colonization potential. 35
For the efficiency of nutrient absorption, the size of the epithelial surface area is one important criterion. 36 The small intestinal surface area is characterized by the presence of villi and crypts. In this study, C. jejuni infection induced a decrease in the jejunal villus height and crypt depth, as well as a reduction of the villus absorptive area at 7 DPI. These changes point either to a decreased life span of enterocytes or to a compromised enterocyte renewal and, thereby, indirectly show interaction of luminal Campylobacter with the intestinal epithelium. Furthermore, the observed villus atrophy in infected birds may also be explained by production of cytolethal distending toxin by C. jejuni, which might be important in suppressing immune responses, thus favoring persistent (asymptomatic) colonization in chickens. 2 Interestingly, at 21 DPI, histomorphometrical examination of the jejunum revealed wider villi and a trend for a greater villus surface area in Campylobacter-infected birds. The latter histomorphometrical changes likely indicate an early onset of epithelial repair mechanisms, which are targeted at the rapid restoration of the villus epithelial monolayer in order to prevent absorption of bacterial toxins. 37 In another recent study, however, villus height was still smaller in the duodenum and jejunum 4 wk after infection with C. jejuni. 34 Possible explanations for a longer-lasting decrease of epithelial surface area may be the higher dose (109 CFU/bird) and different strain (ATCC 33291) of C. jejuni used in that study.
Apart from confirming morphological changes in the intestine of Campylobacter-infected chickens, this study revealed, for the first time, that Campylobacter altered electrolyte absorption by decreasing baseline Isc. Because Na+ absorption contributes significantly to baseline Isc in many intestinal segments,38–40 this could suggest that infection reduced Na+ absorption and subsequently decreased the activity of transport mechanisms for nutrients such as amino acids and glucose, which are often co-transported with Na+.41–43 Our results further demonstrate that Campylobacter induced a decrease in the transmural conductance (Gt) in the jejunum at 7 DPI. Part of this decrease can be attributed to the decrease in the conducting surface area, as evidenced by histomorphometry. However, the decrease in conductance (by 47%) was larger than the decrease in epithelial surface area (by 28%). Therefore, it can be assumed that a certain part of the conductance decrease was attributable to qualitative changes of epithelial barrier properties. In this context, we recently observed a rapid decrease in Gt when acutely exposing chicken jejunum or cecum to Salmonella Enteritidis or its endotoxin in vitro. 16 Such acute effects could well explain the remaining part of jejunal conductance decrease at 7 DPI in this study. Taking into account a concurrently decreased Isc, we previously proposed that this type of response would counteract ion and fluid secretion and may partly explain why chickens do not develop overt diarrhea upon exposure to certain pathogens. 16
At later stages of infection, however, Campylobacter increased the Gt in the jejunum. In the cecum and colon, Gt was increased even as early as 7 DPI. The earlier increase in Gt in the large intestine compared with the small intestine may be related to the much higher bacterial load in the large intestine throughout the infection. Furthermore, the results indicate that C. jejuni cannot pass the epithelial barrier at early stages of infection and the increase of Gt thereafter indicates that C. jejuni-induced intestinal epithelial barrier disruption may be due to alteration of tight junction proteins and opening of tight junctions at later stages of infection. However, the effects of Campylobacter infection on the expression of tight junction proteins were not verified in this study. This conclusion is compatible with results of Connell et al., 44 who demonstrated the involvement of cadherins and calmodulin in C. jejuni intracellular invasion and colonization of chicken intestinal epithelial cells in vivo, leading to a loss of the adherens junction and cell-to-cell adhesion, thus disrupting the epithelial barrier. The results of this study indicate that C. jejuni infection disrupts the intestinal barrier, as revealed by higher intestinal permeability. Furthermore, this disruption was associated with higher intestinal colonization with C. jejuni, suggesting that alteration of intestinal barrier function is a significant mechanism by which C. jejuni colonizes the intestines of chickens.
Gut infections in mammals are associated with an increased release of histamine from mast cells, which contributes to the development of diarrhea. 45 In all intestinal tissues studied so far, the main effect of histamine was an increase in chloride secretion, which can be seen in Ussing chambers by an increased Isc.46,47 In this study, histamine increased the Isc only in the jejunum at 7 DPI, and no effect could be observed in the other tested tissues. The pathophysiological significance of this response remains to be determined. It could be due to an altered expression of histamine receptors or signaling pathways. Alternatively, it was shown that the Cl secretory function may increase in the damaged mucosa owing to a higher expression of the chloride channel ClC-2. 48
Overall, this study is the first to address the electrophysiological changes associated with a Campylobacter infection in vivo. The results indicate that Campylobacter can, indeed, alter absorptive surface area with indirect negative consequences for production efficiency. We also found that C. jejuni initially decreased the ion conductance across the jejunal epithelium and the secretory Isc responses after histamine application were restricted to 7 DPI, which may partly explain the absence of diarrhea after Campylobacter infection in chickens. However, intestinal permeability was increased very early in the large intestine and also later in the small intestine of Campylobacter-infected chicken, which may be important for the persistent colonization of the gastrointestinal tract and for the spread of Campylobacter to internal organs. Furthermore, these effects on intestinal epithelial cells are sufficient to promote the translocation of other enteric microorganisms. Therefore, such effects may be of great clinical significance to gastrointestinal health and disease. For validation and extension of those findings, further future studies will investigate the specific interactions between Campylobacter, intestinal tight junction protein expression, and inflammatory response and their association with the bird performance, as well as the disruption of absorption of ions and other solutes.
Footnotes
Funding
This study was financed by a grant from the CEPO (Centre of Excellence for Poultry) project, which is funded by the European Regional Development Fund, Cross-border Cooperation Programme Austria–Hungary 2007–2013. Grant No: L00112.
Acknowledgements
We would like to thank all staff who assisted with the animal infection experiments at the Clinic for Avian, Reptile and Fish Medicine.
