Abstract
Resveratrol, a natural phenolic compound found in red grapes and wine, exists as cis and trans isomers. Recent studies have shown that trans-resveratrol possesses anti-inflammatory, anti-oxidant, anti-carcinogenic, anti-tumor and immunomodulatory properties. However, it remains unclear whether cis-resveratrol may exhibit similar activities. The objective of the present study was to examine the effects of cis- and trans-resveratrol on the production of pro-inflammatory cytokines and mediators in human macrophages. We examined the possibility that cis- and trans-resveratrol may affect cytokine secretion by modulating inflammasomes, intracellular multi-protein complexes, the assembly of which leads to caspase-1 activation and secretion of active IL-1β by macrophages. Our results show that pre-treatment of macrophages with cis-resveratrol not only reduces pro-IL-1β production and IL-1β secretion, but also suppresses ATP-induced transcription and activation of caspase-1 and caspase-4. Notably, cis-resveratrol inhibits the expression of the purinergic receptor, P2X7R, and the endoplasmic reticulum stress marker, Glc-regulated protein 78, but also reduces reactive oxygen species production. Moreover, cis-resveratrol attenuates cyclooxygenase-2 expression and prostaglandin E2 production. cis-Resveratrol also decreases the phosphorylation of p38 MAPK and expression of the c-Jun protein. These results indicate that cis-resveratrol produces anti-inflammatory effects by inhibiting both the canonical and non-canonical inflammasomes, and associated pathways in human macrophages.
Introduction
Resveratrol (3,4’,5-trihydroxystilbene) represents a natural phenolic phytoalexin found in a wide variety of plant species, such as red grapes, peanuts and mulberries.1,2 It exists as two isomers, cis- and trans-resveratrol. The cis isomer is thought to be produced naturally during grape fermentation as a result of isomerization of the trans isomer by yeast isomerases; in addition, cis-resveratrol can be obtained by exposure of the trans isomer to sunlight.3,4 Several studies conducted within the last few years have shown that trans-resveratrol exhibits a wide range of biological activities, including anti-inflammatory, anti-oxidant, anti-carcinogenesis, anti-aging, anti-platelet aggregation, immunomodulatory and chemoprevention effects.5–9 However, only a few studies have been performed to examine the biological effects of the cis isomer. cis-Resveratrol has been shown to possess anti-cancer, antimicrobial and anti-platelet aggregation activities.10–12 Studies are currently under way to determine the properties of cis-resveratrol.
Macrophages are major components of the innate immune system. The activation of macrophages has been shown to play a pivotal role during the initiation and development of inflammatory responses by producing numerous pro-inflammatory cytokines, such as IL-1β, IL-6 and TNF-α, and other pro-inflammatory mediators, such as NO and prostaglandins.13–15 IL-1β and IL-18, which are members of the IL-1 cytokine superfamily, promote a variety of innate immune processes associated with infection, inflammation and autoimmunity.16,17 IL-1β participates in the generation of systemic and local immune responses against various pathogens, and has been implicated in the pathogenesis of inflammatory diseases, such as gout, asthma, inflammatory bowel disease, rheumatoid arthritis and atherosclerosis.18–20 Similarly, IL-18 plays a critical role in the execution of antimicrobial and antiviral immunity, and this cytokine has also been associated with severe inflammatory disorders, such as rheumatoid arthritis, Crohn’s disease, psoriasis, lupus, sarcoidosis and multiple sclerosis.21,22 During inflammation or local infection with pathogens, prostaglandin E2 (PGE2) represents the main prostaglandin derived from arachidonic acid via the action of cyclooxygenase-2 (COX-2). 23 Recent studies have shown that COX-2 and its downstream product PGE2 contribute to the development of many chronic inflammatory diseases, such as cardiovascular disease, rheumatoid arthritis and inflammatory bowel disease.24,25
The pro-inflammatory cytokines IL-1β and IL-18 are synthesized as inactive precursors (i.e. pro-IL-1β and pro-IL-18) and accumulate within the cytosol of monocytes and macrophages exposed to or ‘primed’ with pathogen-associated molecular patterns like the bacterial endotoxin, LPS. 26 However, LPS by itself is usually insufficient to trigger IL-1β and IL-18 secretion from macrophages unless danger-associated molecular patterns provide the second signal responsible for the activation of inflammasome complexes and the protease caspase-1, processing of pro-IL-1β and pro-IL-18, and secretion of the mature cytokines from the cells.27–29 In addition, a recent study demonstrated that inflammasome activation also modulates the production of pro-inflammatory lipid mediators like prostaglandins in macrophages, 30 indicating that the canonical inflammasome pathway is linked to prostaglandin production in these cells.
Extracellular ATP acts as a danger signal released from injured cells during tissue damage and inflammation; it initiates inflammation and further amplifies and sustains cell-mediated immunity through P2 receptor-mediated purinergic signaling. 31 Binding of ATP to the P2X7 receptor (P2X7R) leads to inflammasome activation and secretion of IL-1β and IL-18 by primed macrophages. 32
Activation of caspase-1 requires the assembly and activation of a cytosolic multi-protein complexes known as the inflammasome, which is typically formed by three components consisting of a nucleotide binding and oligomerization domain-like, leucine-rich repeat-containing receptor (NLR), the adaptor protein apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC) and pro-caspase-1. 33 Upon activation, oligomerized NLRs interact with ASC, which, in turn, recruits and activates caspase-1, and leads to cleavage and activation of pro-IL-1β and pro-IL-18. 29 The NLRP1 (NALP1) and NLRP3 (NALP3/cryopyrin) inflammasomes are two of the best-characterized canonical inflammasomes described so far. A large number of stimuli have been shown to trigger activation of the NLRP3 inflammasome, including ATP, monosodium urate crystals, cholesterol crystals, UVB irradiation, pathogen-derived nucleic acids, silica, asbestos and amyloid-β.34–41 In addition, treatment with LPS or muramyl dipeptide (along with ATP) has been reported to induce the NLRP1 inflammasome.42,43
More recently, non-canonical inflammasomes containing murine caspase-11 have also been reported.44,45 Caspase-11 does not exist in humans, but the function of this enzyme is functionally equivalent to human caspase-4 and caspase-5, which also modulate inflammasome activity as revealed in previous studies.46,47
The main objective of the present study was to investigate whether cis- and trans-resveratrol inhibit the production of pro-inflammatory cytokines (IL-1β and IL-18) and the pro-inflammatory mediator (PGE2) in LPS-primed, ATP-stimulated human macrophages. We also aimed to determine whether cis- and trans-resveratrol modulate inflammasome activation in macrophages. We found that cis-resveratrol suppresses IL-1β secretion and PGE2 production by inhibiting the non-canonical inflammasome pathway and COX-2. These findings suggest that cis-resveratrol possesses anti-inflammatory activities that may prove beneficial for the treatment of inflammatory diseases.
Materials and methods
Chemicals and reagents
ATP, LPS and phorbol 12-myristate 13-acetate (PMA) were purchased from Sigma-Aldrich (St. Louis, MO, USA). cis-Resveratrol and trans-resveratrol were purchased from Cayman Chemical Company (Ann Arbor, MI, USA), and a stock solution of 200 mM was prepared in absolute ethanol and kept at –20℃ away from light. Cell culture media (RPMI 1640), FBS, penicillin and streptomycin were purchased from Life Technologies (Grand Island, NY, USA). For Western blot analysis, anti-IL-1β, anti-NF-κB p65, anti-IκB-α, anti-p38, anti-p-p38, anti-ERK1/2, anti-p-ERK1/2, anti-JNK and anti-p-JNK were obtained from Cell Signaling Technology (Danvers, MA, USA); anti-caspase-4 was from MBL (Nagoya, Japan); anti-ASC, anti-COX-2, anti-c-Fos, anti-c-Jun, anti-Glc-regulated protein 78 (GRP78), anti-pro-IL-1β, anti-IL-18 and anti-P2X7R were from Santa Cruz Biotechnology (Santa Cruz, CA, USA); anti-NLRP3 and anti-P2X4R were from Sigma-Aldrich; anti-caspase-1 was from Millipore (Billerica, MA, USA); anti-NLRP1 was from Enzo Life Sciences (Farmingdale, NY, USA); anti-lamin B1 was from Abcam (Cambridge, MA, USA); and anti-β-actin was from Novus Biologicals (Littleton, CO, USA). HRP-conjugated anti-rabbit and anti-mouse secondary Abs were obtained from Santa Cruz Biotechnology.
Cell culture and treatments
Human acute monocytic leukemia THP-1 cells [TIB-202, American Type Culture Collection (ATCC), Manassas, VA, USA] were cultured in RPMI 1640 medium supplemented with 10% (v/v) heat-inactivated FBS, 100 units/ml of penicillin, and 100 µg/ml of streptomycin. Cell lines were cultured at 37℃ in a humidified incubator containing 5% CO2. The THP-1 cells were plated in six-well plates at 2 × 106 cells/well. Cellular differentiation of the suspended cells to adherent macrophages was induced by overnight culture in complete medium supplemented with 500 ng/ml of PMA, followed by culture in fresh complete medium for an additional 2 d. THP-1 macrophages were pre-treated for 20 h with the indicated concentrations of resveratrol or with ethanol as a control, followed by treatment with LPS (0.5 µg/ml) for 3 h and ATP (5 mM) for 1 h. Cell culture supernatants were harvested at 14,000 g for 5 min at 4℃, and the supernatants were collected and stored at –80℃ for cytokine assay. In addition, cell lysates were re-suspended in lysis buffer for RNA extraction and Western blot analysis.
Cell viability assay
Cell viability was determined using a commercial MTT-based in vitro toxicology assay (Sigma-Aldrich), which detects viable cells colorimetrically based on the production of purple formazan by viable cells. THP-1 cells were seeded at a density of 1 × 105 cells/well for 24 h. For macrophage differentiation, cells were treated and incubated with PMA as described above. Cell culture media were replaced by complete media containing the indicated concentrations of resveratrol and then incubated with cells for 24 h. After incubation, 10 µl of MTT (5 mg/ml) were added to each well and the plates were incubated at 37℃ for 4 h. The content of each well was eluted and the precipitate was dissolved with 100 µl of the MTT solubilization solution. Absorbance was read at 570 nm using a VersaMax microplate reader (Molecular Devices, Sunnyvale, CA, USA). Cell viability (%) was calculated as the ratio of surviving cells in the resveratrol-treated group divided by that of the control group. All treated samples and controls were tested in triplicate.
Measurement of human IL-1β, IL-18, caspase-1 and PGE2 by ELISA
THP-1 macrophages were seeded at a density of 2 × 106 cells/well in six-well plates and were pre-incubated with the indicated concentrations of resveratrol in 1 ml of complete medium for 20 h, followed by treatment with LPS (0.5 µg/ml) for 3 h and with ATP (5 mM) for 1 h. Cell culture supernatants were collected and centrifuged at 10,000 g for 5 min at 4℃ to remove insoluble material. Levels of secreted IL-1β, activated caspase-1 and PGE2 in cell culture supernatants were measured using commercial ELISA kits (R&D Systems, Minneapolis, MN, USA) based on the manufacturer’s instructions; IL-18 in cell culture supernatants was measured by using a human IL-18 ELISA kit (MBL).
Reactive oxygen species production
Total reactive oxygen species (ROS) production in THP-1 macrophages was measured using the Total ROS/Superoxide detection kit (Enzo Life Sciences, Farmingdale, NY, USA). Briefly, cells were first seeded at a density of 1 × 105 cells/well in 96-well plates for 24 h. For macrophage differentiation, cells were treated with PMA as described above. Cell culture media were replaced by complete media containing resveratrol at the indicated concentrations and then incubated for 20 h, followed by treatment with LPS (0.5 µg/ml) for 3 h and with ATP (5 mM) for 1 h. In addition, cells were treated with the ROS inducer, pyocyanin (200 µM), at 37℃ for 30 min as a positive control. After treatment, the cells were washed with 200 µl of 1 × washing buffer and loaded with 100 µl of ROS/superoxide detection reagents prior to incubation at 37℃ for 1 h. Absorbance was read at 520 nm using a microplate reader after excitation at 488 nm. The increase in relative fluorescence intensity was used to determine intracellular ROS production.
Protein extraction and Western blot analysis
Total cellular proteins were isolated from cell lines in RIPA lysis buffer (50 mM Tris-HCl, pH 7.4, 150 mM NaCl, 0.25% deoxycholic acid, 1% Nonidet P-40, 1 mM EDTA) (Millipore) containing a protease inhibitor cocktail (Roche, Mannheim, Germany). Cell culture supernatants were harvested as described above. Cytosolic and nuclear proteins were extracted using cytoplasmic and nuclear protein extraction kits (Thermo Fisher Scientific, Rockford, IL, USA). Protein concentration was determined using the Bio-Rad Bradford assay (Bio-Rad, Hercules, CA, USA). Equal amounts of protein were separated by 8–12% SDS-PAGE. After gel electrophoresis, the proteins were transferred to polyvinylidene difluoride membranes (Millipore) and subsequently stained with Ponceau S (Sigma-Aldrich) to confirm protein transfer. The membranes were blocked with 5% non-fat dry milk or BSA in TBS-T (0.1% Tween-20 in 1 × TBS, pH 7.4) for 1 h and probed overnight with the indicated primary Abs. Protein signal was revealed by enhanced chemiluminescence (Millipore) following treatment with HRP-conjugated secondary Ab. Quantification of protein signal obtained by Western blot analysis was performed with the LAS-3000 chemiluminescence detection device (Fujifilm, Valhalla, NY, USA). To adjust for protein loading, the signal of each protein was normalized to β-actin.
Quantitative real-time RT-PCR
Sequences for the quantitative real-time RT-PCR primers.

Statistical analysis
For each experiment, data were obtained in triplicate and presented as means ± SD. Mean comparisons between cis-resveratrol or trans-resveratrol-treated cells and untreated control cells were analyzed using Student’s t-test. P-Values < 0.05 were considered statistically significant.
Results
cis- and trans-Resveratrol produce no toxic effect on human macrophages
To observe whether cis- and trans-resveratrol show cytotoxic effects on human THP-1 macrophages, we treated the cells with either cis- or trans-resveratrol at various concentrations for 24 h, followed by monitoring cell viability using the MTT assay. The resveratrol isomers showed no toxic effect on THP-1 macrophages (Figure 1).
cis-Resveratrol shows no toxicity on human THP-1 macrophages. THP-1 macrophages were treated with various concentrations (1, 10 and 100 µM) of either cis-resveratrol or trans-resveratrol for 24 h, and cell viability was measured by using the MTT assay as described in the ‘Materials and methods’. Data are presented as means ± SD of three experiments preformed in duplicate.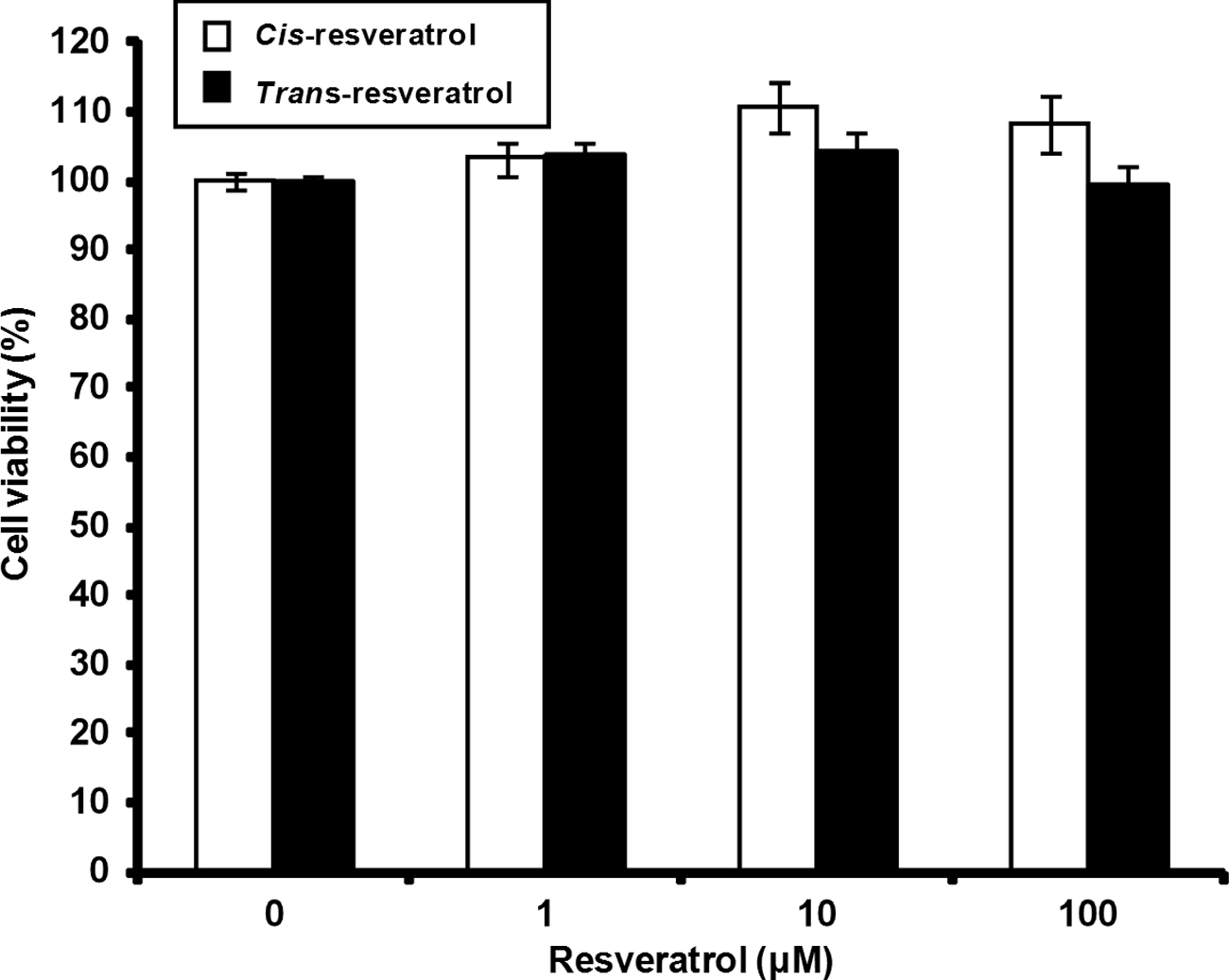
cis-Resveratrol attenuates the production of IL-1β at the transcriptional and translational levels in activated macrophages
To evaluate the effects of cis- and trans-resveratrol on the production of pro-inflammatory cytokines by macrophages, we treated THP-1 cells with either cis- or trans-resveratrol (1, 10 or 100 µM for 20 h), then with LPS (0.5 µg/ml for 3 h) to induce cytokine expression and, finally, with ATP (5 mM for 1 h) to induce secretion of IL-1β and IL-18. ELISA and Western blot analyses showed that cis-resveratrol pre-treatment significantly inhibited IL-1β secretion in the cell culture supernatants (Figure 2a, e) and pro-IL-1β production in whole cell lysates (Figure 2e, f). In contrast, pre-treatment with trans-resveratrol increased IL-1β secretion in a dose-dependent manner (Figure 2a). Neither cis-resveratrol nor trans-resveratrol inhibited IL-18 production in macrophages; actually, cis-resveratrol induced IL-18 secretion under these conditions (Figure 2b, e). Based on these results, we focused our attention in this study on the inhibitory effects of cis-resveratrol on IL-1β secretion, as this anti-inflammatory activity may have potential applications in treating inflammatory diseases in humans.
Effects of cis-resveratrol on the secretion of IL-1β and IL-18 in human THP-1 macrophages. Cells were pre-treated with various concentrations (1, 10 and 100 µM) of cis-resveratrol or 10 µM of trans-resveratrol for 20 h, followed by treatment with LPS (0.5 µg/ml) for 3 h and with ATP (5 mM) for 1 h. Secretion of (a) IL-1β and (b) IL-18 in cell culture supernatants was monitored by ELISA. mRNA expression levels of (c) IL-1β and (d) IL-18 were quantified using real-time RT-PCR. β-Actin gene expression was used for normalization. The results were expressed as fold change of untreated cells. (e) The presence of IL-1β and IL-18 in cell lysates and cell culture supernatants were analyzed by Western blot analysis. β-Actin was used as an internal control. (f) Bands from (e) were analyzed by scanning densitometry and normalized to β-actin. Data were presented as means ± SD of three experiments performed in duplicate. #P < 0.01 versus untreated cells. *P < 0.01 versus resveratrol-untreated (ethanol-treated) control cells. †P < 0.01 versus cis-resveratrol (100 µM) treated cells.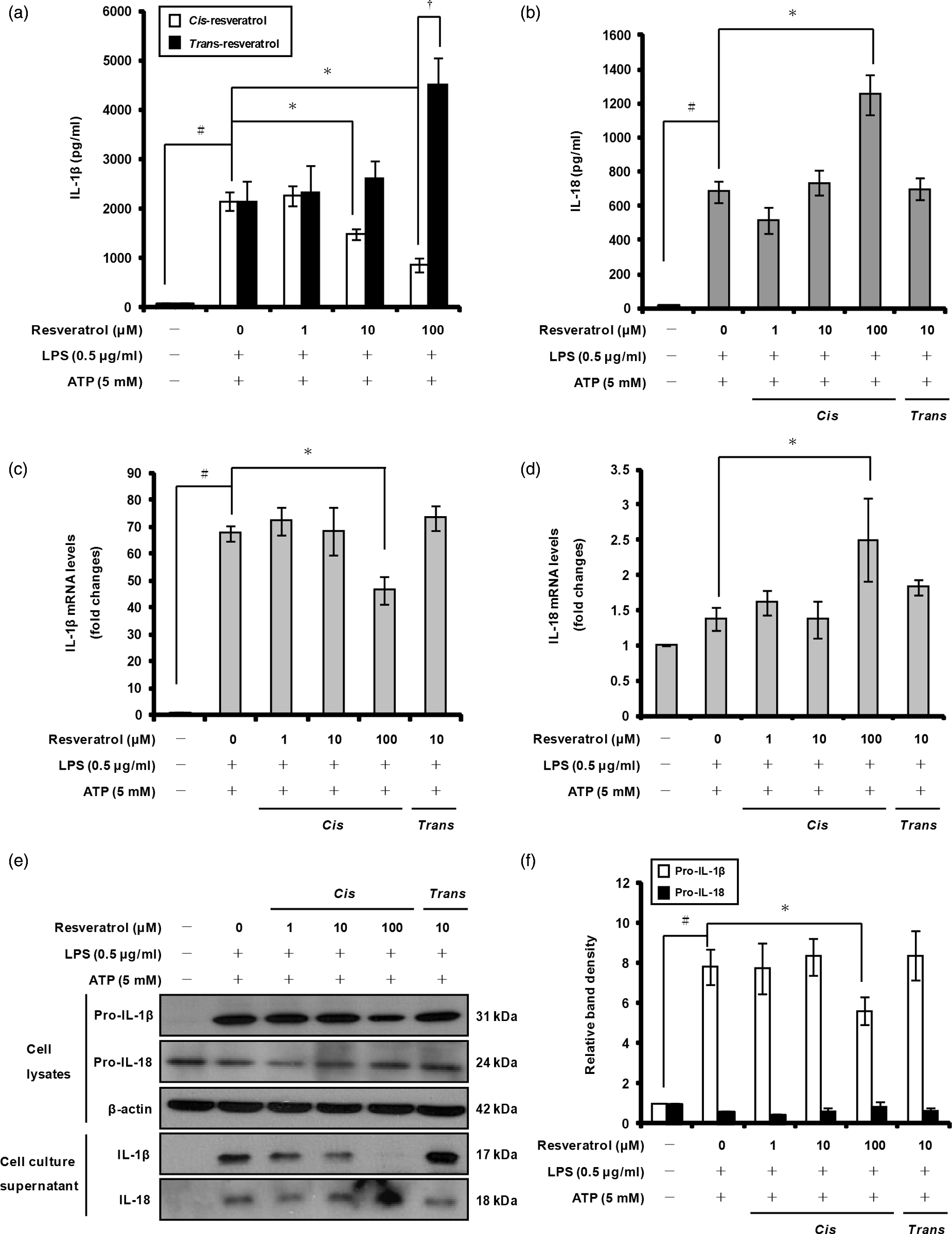
We examined whether the immunomodulatory effects of cis-resveratrol on IL-1β and IL-18 production may be due to changes in gene expression of these cytokines. Quantitative real-time RT-PCR analysis showed that pre-treatment of macrophages with 100 µM cis-resveratrol significantly reduced IL-1β mRNA expression (Figure 2c); in contrast, cis-resveratrol used at the same concentration increased IL-18 mRNA expression (Figure 2d). These findings indicate that cis-resveratrol suppresses IL-1β production by altering the transcription and translation of this cytokine in activated macrophages.
cis-Resveratrol reduces expression and activation of caspase-1 and caspase-4 in macrophages
The cytokine IL-1β is generated as a cytosolic precursor that requires cleavage by the protease caspase-1 in order to generate the biologically active cytokine.
27
To determine whether caspase-1 gene expression and protein activation may be affected by cis-resveratrol, we pre-incubated THP-1 macrophages with this compound for 20 h, prior to LPS and ATP treatments as mentioned above. As shown in Figure 3a, cis-resveratrol reduced caspase-1 mRNA expression under these conditions. cis-Resveratrol used at 100 µM significantly inhibited both caspase-1 secretion in cell culture supernatants and activated caspase-1 (20 ku) in whole cell lysates (Figure 3b–d).
cis-Resveratrol inhibits caspase-1 and caspase-4 gene expression and activation in macrophages. Cells were pre-treated with the indicated concentrations of cis-resveratrol or trans-resveratrol for 20 h, followed by treatment with LPS (0.5 µg/ml) for 3 h and with ATP (5 mM) for 1 h. (a) mRNA expression levels of caspase-1 and caspase-4 were quantified using real-time RT-PCR. β-Actin gene expression was used for normalization. The results were expressed as fold change of untreated cells. (b) Secretion of caspase-1 subunit p20 into the supernatants of THP-1 macrophages was assessed by ELISA. (c) Equal amounts of cell lysates were probed to detect pro-caspase-1 p45 and caspase-1 subunit p20 or pro-caspase-4 p43 and caspase-4 subunit p30. β-Actin was used as an internal control. (d) Bands from (c) were analyzed by scanning densitometry and normalized to β-actin. Data were presented as means ± SD of three experiments preformed in duplicate. #P < 0.01 versus untreated cells. *P < 0.01 versus resveratrol-untreated control (ethanol) cells.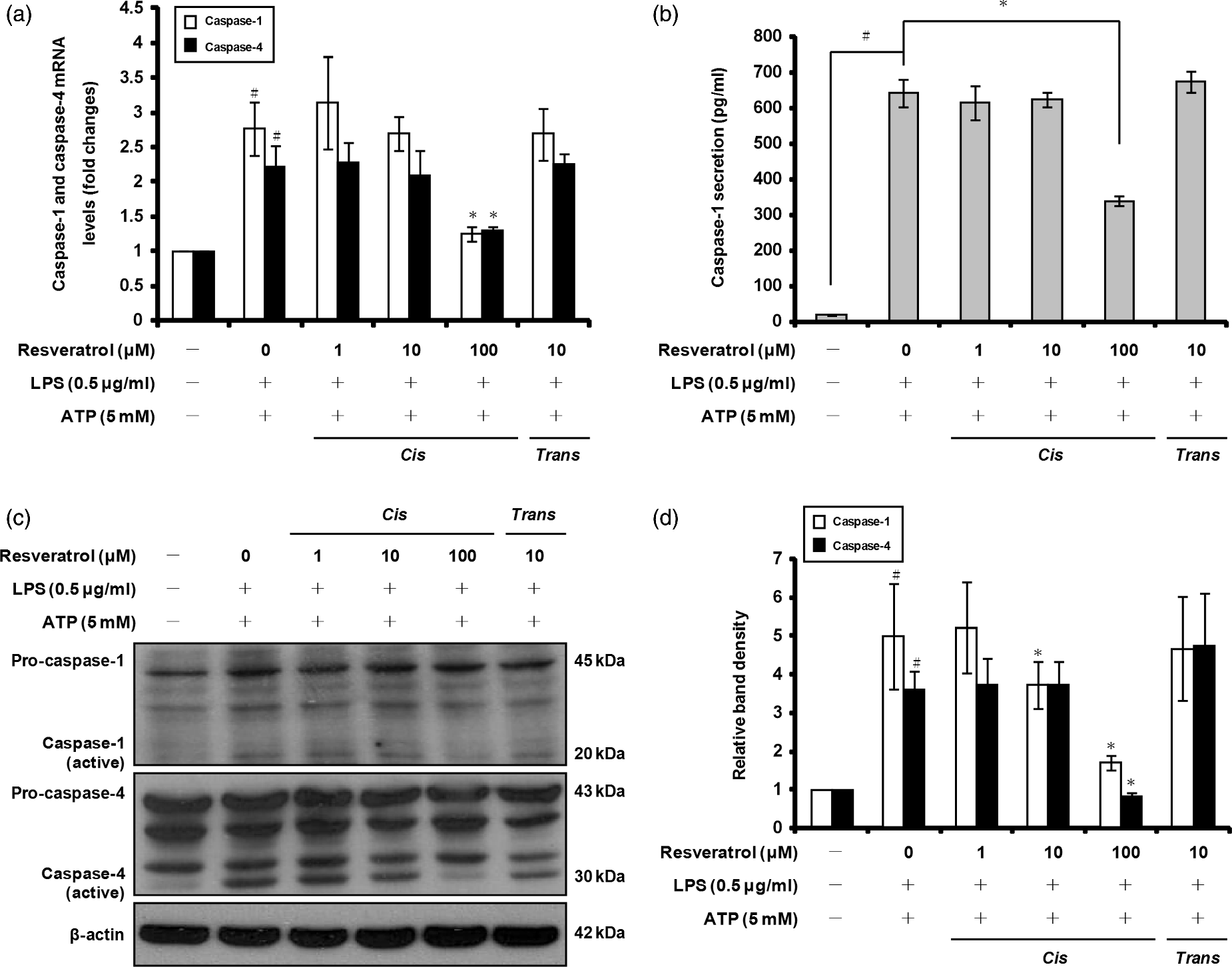
Recent studies have shown that pro-IL-1β is also the substrate of non-canonical inflammasomes containing caspase-4, and that this proteolytic enzyme is able to generate the biologically active form of IL-1β. 46 We examined the effect of cis-resveratrol on caspase-4 gene expression in activated macrophages. As shown in Figure 3a, pre-treatment with cis-resveratrol resulted in a significant decrease of caspase-4 gene expression in macrophages stimulated with LPS and ATP. cis-Resveratrol-treated cells also showed a significant reduction of active caspase-4 protein in cell lysates (Figure 3c, d; 30 ku). Taken together, these results suggest that the reduction in IL-1β secretion induced by cis-resveratrol in macrophages is associated with down-regulation of both caspase-1 and caspase-4 gene expression and protein activation.
cis-Resveratrol does not inhibit the expression of NLRP1, NLRP3, or ASC
Canonical inflammasomes are formed by three components that include an NLR family protein (e.g. NLRP1 or NLRP3), the adaptor protein ASC and pro-caspase-1.
48
To determine whether cis-resveratrol affects the NLRP1 and NLRP3 inflammasomes, we measured the mRNA and protein expression levels of NLRP1, NLRP3 and ASC using quantitative real-time RT-PCR and Western blot analyses. As shown in Figure 4a, c and d, the mRNA and protein levels of ASC, which decreased following treatment with LPS and ATP, appeared to slightly increase in cells pre-treated with cis-resveratrol at 100 µM compared to 1 and 10 µM. Moreover, pre-treatment of macrophages with cis-resveratrol at 100 µM increased the mRNA and protein levels of NLRP1 and NLRP3 compared with cells treated only with LPS and ATP (Figure 4b, c, d). We conclude that the inhibitory effect of cis-resveratrol on IL-1β secretion is not due to down-regulation of NLRP1, NLRP3 or ASC expression.
Effects of cis-resveratrol on the expression of ASC, NLRP1 and NLRP3 in activated macrophages. Cells were pre-treated with the indicated concentrations of cis-resveratrol or trans-resveratrol for 20 h, followed by treatment with LPS (0.5 µg/ml) for 3 h and with ATP (5 mM) for 1 h. mRNA expression levels of (a) ASC, as well as (b) NLRP1 and NLRP3, were quantified using real-time RT-PCR. β-Actin gene expression was used for normalization. The results were expressed as fold change of untreated cells. (c) Cell lysates were analyzed by Western blot analysis using specific anti-ASC, anti-NLRP1 and anti-NLRP3 Abs. (d) Bands from (c) were analyzed by scanning densitometry and normalized to β-actin. Data were presented as means ± SD of three experiments preformed in duplicate. #P < 0.01 versus untreated cells.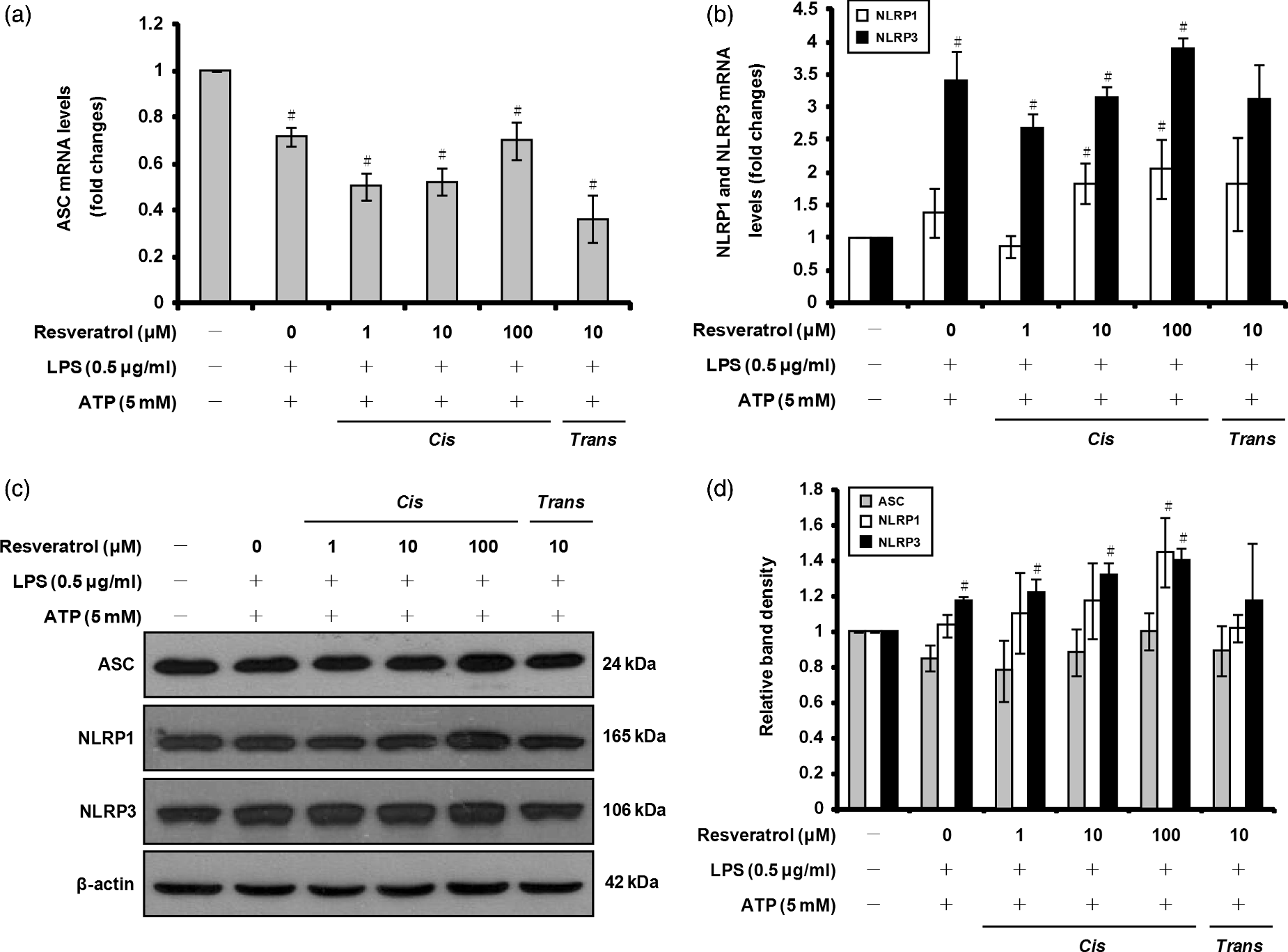
cis-Resveratrol reduces P2X7 receptor expression
Previous studies have shown that activation of the purinergic receptor P2X7R by extracellular ATP activates the inflammasome, which leads to processing and release of both caspase-1 and mature IL-1β into the culture medium of activated macrophages.49,50 To determine whether cis-resveratrol may influence expression of P2X receptors, which represent ATP-gated channels, we pre-treated THP-1 macrophages with cis-resveratrol, and then with LPS and ATP, prior to measuring mRNA and protein expression of P2X7R and P2X4R (receptor for lower concentrations of ATP than P2X7R) using quantitative real-time PCR and Western blot analyses. As shown in Figure 5a and c, the up-regulation of P2X7R expression induced by ATP (and LPS) was significantly suppressed by100 µM cis-resveratrol. In contrast, the mRNA and protein levels of P2X4R increased following treatment with cis-resveratrol (Figure 5a, b). These results suggest that the inhibitory effect of cis-resveratrol on IL-1β secretion may be due, at least in part, to reduced P2X7R expression.
cis-Resveratrol suppresses P2X7R-mediated ROS production in macrophages. Cells were pre-treated with the indicated concentrations of cis-resveratrol or trans-resveratrol for 20 h, followed by treatment with LPS (0.5 µg/ml) for 3 h and with ATP (5 mM) for 1 h. (a) mRNA expression levels of P2X4R and P2X7R were quantified using real-time RT-PCR. β-Actin gene expression was used for normalization. The results were expressed as fold change of untreated cells. (b, c) Cell lysates were analyzed by Western blot analysis used specific anti-P2X4R and anti-P2X7R Abs. The relative protein levels were quantified by scanning densitometry and normalized to β-actin. (d) ROS production was measured with the total ROS detection kit. Pyocyanin (200 µM), a ROS inducer, was used as a positive control for ROS formation. Data were presented as means ± SD of three experiments preformed in duplicate. #P < 0.01 versus untreated cells. *P < 0.01 versus resveratrol-untreated control (ethanol) cells.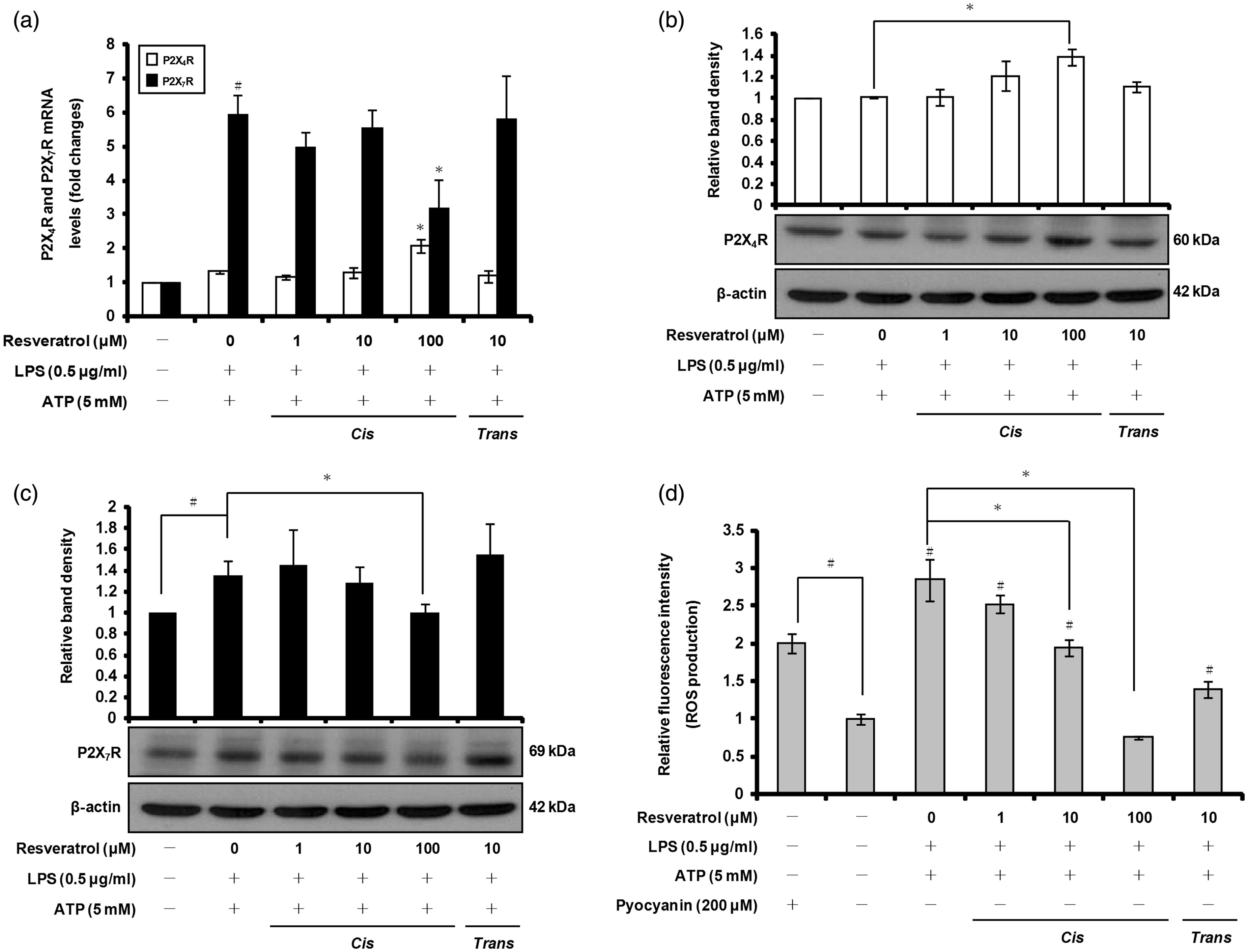
cis-Resveratrol inhibits endoplasmic reticulum stress and ROS production
Previous studies have reported that activation of P2X7R by ATP increases the generation of ROS that are associated with activation of caspase-1 and processing of IL-1β.51,52 We examined whether the ROS production induced by ATP was also affected by cis-resveratrol. We used a commercially available detection kit to measure intracellular ROS production in macrophages pre-treated with cis-resveratrol, prior to LPS priming and ATP activation. ATP treatment in LPS-primed macrophages induced a high amount of ROS compared with untreated cells (Figure 5d; pyocyanin was used as a positive control for ROS formation). Notably, ATP-induced ROS production was suppressed in a concentration-dependent manner by cis-resveratrol pre-treatment (Figure 5d).
Previous studies have shown that ROS can directly induce endoplasmic reticulum (ER) stress.53–55 In addition, caspase-4, a member of the caspase-1 subfamily, has been shown to localize to the ER membrane and to be cleaved and activated in response to ER stress.
56
The prototypical ER stress-responsive gene, GRP78, encodes a molecular chaperone and has been used as a ER stress marker.
57
To assess whether ATP-induced ROS production is involved in ER stress, we evaluated the mRNA and protein levels of GRP78 using quantitative real-time RT-PCR and Western blot analyses. As shown in Figure 6a, GRP78 mRNA expression was down-regulated by pre-treatment with cis-resveratrol compared with cells treated only with LPS and ATP. Further experiments showed that cis-resveratrol produced a similar effect on GRP78 protein levels (Figure 6b). Taken together, these results suggest that cis-resveratrol suppresses caspase-4 activation in activated macrophages, at least in part, by reducing ROS production and GRP78 expression.
cis-Resveratrol suppresses GRP78 and COX-2 expression, and PGE2 production in activated macrophages. Cells were pre-treated with the indicated concentrations of cis-resveratrol or trans-resveratrol for 20 h, followed by treatment with LPS (0.5 µg/ml) for 3 h and with ATP (5 mM) for 1 h. (a) mRNA expression levels of GRP78 were quantified using real-time RT-PCR. β-Actin gene expression was used for normalization. The results were expressed as fold change of untreated cells. (b and c) Cell lysates were analyzed by Western blot analysis used specific anti-GRP78 and anti-COX-2 Abs. The relative protein levels were quantified by scanning densitometry and normalized to β-actin. (d) The concentrations of PGE2 in the cell culture supernatants of THP-1 macrophages were assessed by ELISA. Data were presented as means ± SD of three experiments preformed in duplicate. #P < 0.01 versus untreated cells. *P < 0.01 versus resveratrol-untreated control (ethanol) cells.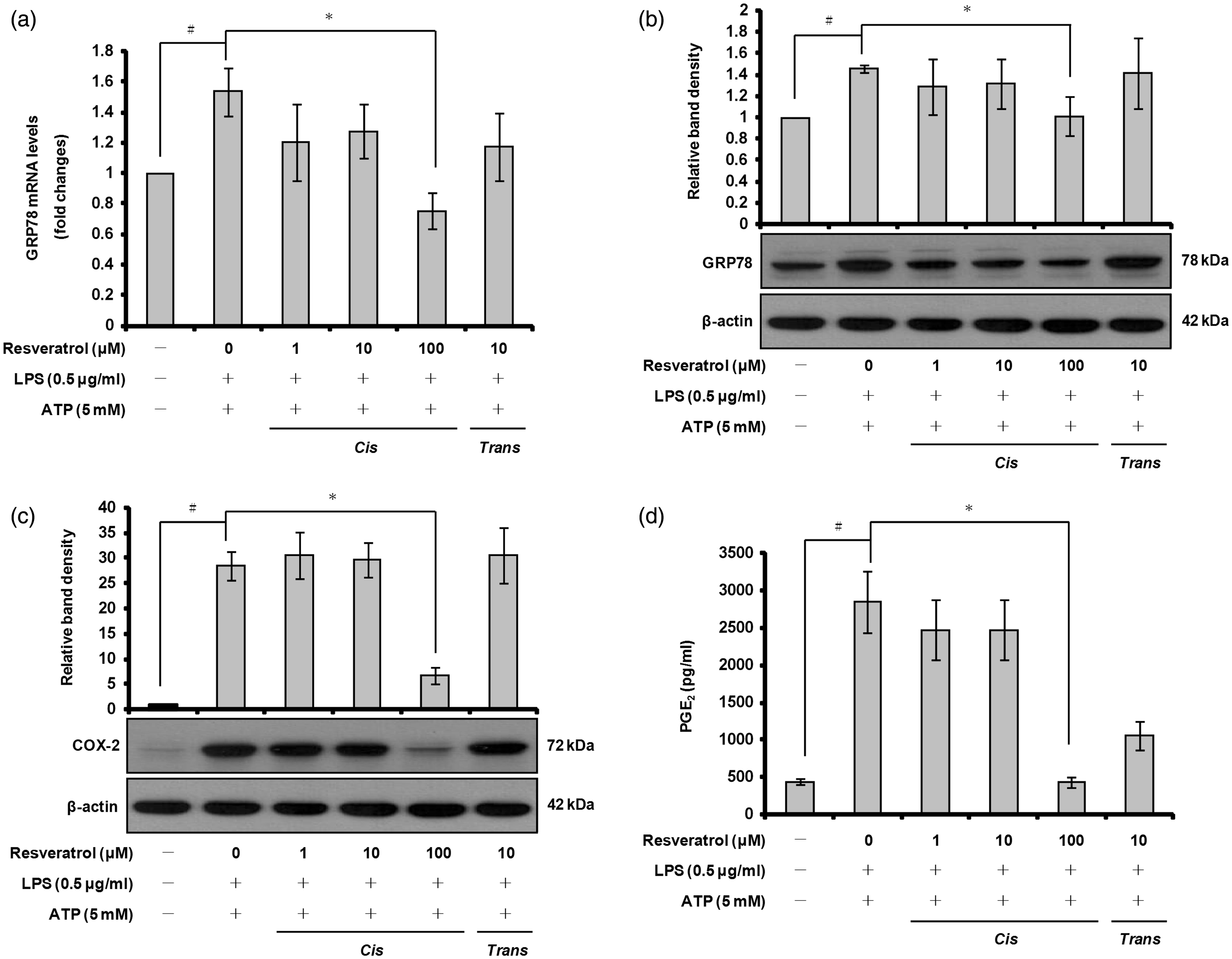
cis-Resveratrol inhibits COX-2 protein expression and PGE2 production
To investigate further the anti-inflammatory properties of cis-resveratrol, we examined the protein level of another important pro-inflammatory enzyme, COX-2, by Western blot analysis. Treatment of THP-1 macrophages with LPS and ATP resulted in a pronounced up-regulation of COX-2 protein level (Figure 6b). Pre-treatment of the cells with cis-resveratrol (100 µM) inhibited COX-2 protein level compared with cells treated only with LPS and ATP (Figure 6c).
PGE2 represents a major active product of the metabolism of arachidonic acid that is produced by the COX pathway in macrophages. 58 To determine whether the inhibitory effect of cis-resveratrol on COX-2 protein level affected the production of PGE2, we performed ELISA to detect PGE2 in cell culture supernatants. As shown in Figure 6d, stimulation of macrophages with LPS and ATP resulted in a marked increase in PGE2 production compared with the untreated control. However, cis-resveratrol decreased the production of PGE2 (Figure 6c; 100 µM). These results suggest that cis-resveratrol may produce anti-inflammatory effects by inhibiting the COX pathway.
cis-Resveratrol suppresses p38 MAPK phosphorylation and c-Jun protein expression
NF-κB is an important transcription factor that regulates gene expression of pro-inflammatory cytokines and mediators in activated macrophages. NF-κB activation occurs via phosphorylation and subsequent degradation of IκB-α, which results in translocation of NF-κB to the nucleus, causing activation of NF-κB-dependent genes.
59
Therefore, we determined whether cis-resveratrol inhibits pro-IL-1β and COX-2 expression by blocking NF-κB activation in THP-1 macrophages. The cytosolic level of IκB-α and nuclear level of NF-κB p65 subunit were analyzed by Western blot analysis. The results showed that pre-treatment with cis-resveratrol at 100 µM led to translocation of NF-κB p65 subunit from the cytosol to the nucleus, suggesting that the NF-κB pathway may not be involved in down-regulation of pro-IL-1β and COX-2 expression by cis-resveratrol (Figure 7a, b).
cis-Resveratrol inhibits p38 MAPK phosphorylation and c-Jun expression in macrophages. Cells were pre-treated with the indicated concentrations of cis-resveratrol or trans-resveratrol for 20 h, followed by treatment with LPS (0.5 µg/ml) for 3 h and with ATP (5 mM) for 1 h. (a, b) After the isolation of cytosolic and nuclear fractions, cytosolic IκB-α and nuclear NF-κB p65 were analyzed by Western blot analysis. β-Actin and lamin B1 were used as internal controls. The relative protein levels were quantified by scanning densitometry. (c) Whole cell lysates were analyzed by Western blotting using various Abs against p38 MAPK, ERK1/2, JNK, phospho-p38 MAPK, phospho-ERK1/2 and phospho-JNK. (d) Bands from (c) were analyzed by scanning densitometry and normalized to total p38 MAPK, ERK1/2 and JNK. (E and F) AP-1 component proteins (c-Jun and c-Fos) were analyzed by Western blot analysis. The relative protein levels were quantified by scanning densitometry and normalized to β-actin. Data were presented as means ± SD of three experiments preformed in duplicate. #P < 0.01 versus untreated cells. *P < 0.01 versus resveratrol-untreated control (ethanol) cells.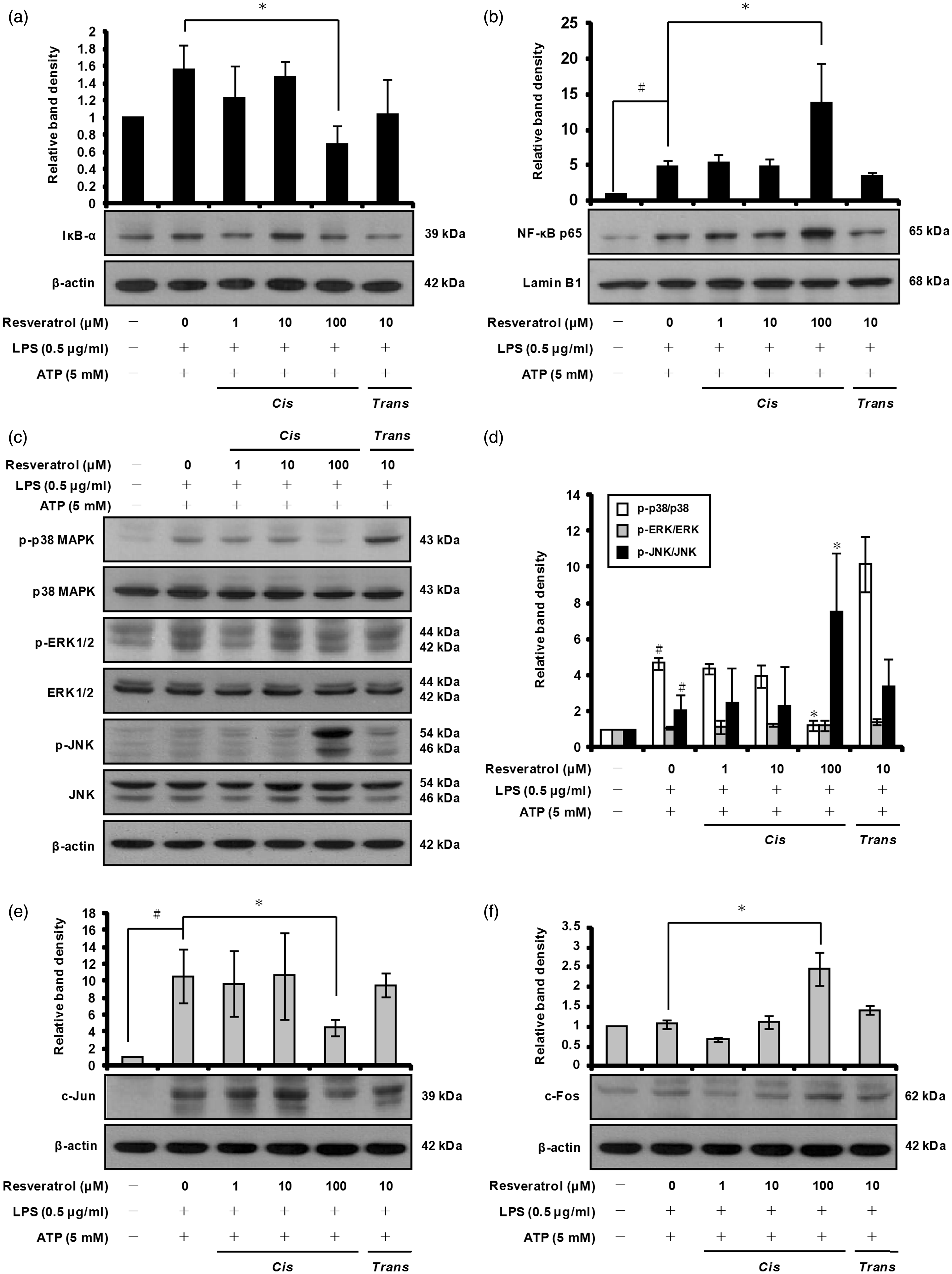
MAPKs represent a group of proline-directed serine/threonine kinases that comprise three major enzymes: p38 MAPK, c-Jun N-terminal kinases (JNK)/stress-activated protein kinases and extracellular signal-regulated kinases 1 and 2 (ERK1/2). MAPKs are thought to be involved in the production of pro-inflammatory cytokines and mediators by activated macrophages in response to various stimuli, including LPS.60,61 For these reasons, we examined the effect of cis-resveratrol on MAPKs in activated macrophages. Western blot analysis was carried out using Abs raised against the phosphorylated and total forms of p38 MAPK, ERK1/2 and JNK. We observed that cis-resveratrol at 100 µM significantly suppressed the phosphorylation of p38 MAPK, but did not suppress the phosphorylation of ERK1/2 or JNK (Figure 7c, d). In addition, cis-resveratrol did not significantly affect the total protein levels of p38 MAPK, ERK1/2 and JNK in activated macrophages (Figure 7c).
Previous work has shown that pro-inflammatory cytokines and mediators are controlled not only by NF-κB but also by activator protein-1 (AP-1). 60 AP-1 is a dimeric transcription factor composed of members of the c-Jun and c-Fos proto-oncogene families. 62 As the activation of AP-1 can be regulated predominately by the MAPK signal transduction pathway, 63 we further examined the effect of cis-resveratrol on the expression of AP-1 component proteins (c-Jun and c-Fos) in activated macrophages. After pre-incubation with cis-resveratrol (100 µM), the induction of c-Jun by LPS and ATP was significantly reduced (Figure 7e); however, the level of c-Fos protein was not suppressed by cis-resveratrol (Figure 7f). Based on these results, it appears that cis-resveratrol may impair pro-IL-1β and COX-2 expression by inhibiting p38 MAPK phosphorylation and c-Jun protein production.
Discussion
The present study demonstrates that cis-resveratrol inhibits IL-1β secretion in activated macrophages by suppressing various pro-inflammatory signaling molecules and pathways. To our knowledge, our report is the first to show a difference between cis-resveratrol and trans-resveratrol in activated macrophages. After identifying that cis-resveratrol inhibits IL-1β secretion, we focused our attention on the modulatory effects of this compound on the inflammasomes and pro-inflammatory mediators.
LPS stimulates the secretion of a large number of inflammatory molecules by macrophages, including IL-1β, IL-6, TNF-α and COX-2.60,64 Our study shows that LPS and ATP significantly induces the expression and production of pro-inflammatory cytokines (IL-1β and IL-18), and enhances expression of COX-2 and production of PGE2 in human THP-1 macrophages. By binding to TLR4, LPS activates several intracellular signaling pathways that include the IκB kinase–NF-κB pathway and MAPK pathways. In turn, these signaling pathways activate a variety of transcription factors that include NF-κB and AP-1. 60 Our study shows that LPS and ATP induces NF-κB activation, phosphorylation of p38 MAPK, ERK1/2 and JNK, and AP-1 component proteins (c-Jun and c-Fos) expression in activated macrophages.
In addition to significantly reducing p38 MAPK phosphorylation and c-Jun protein expression, pre-treatment of macrophages with cis-resveratrol also inhibited the expression and production of pro-IL-1β, caspase-1, caspase-4, COX-2 and GRP78 following activation with LPS and ATP. A previous study has demonstrated that human caspase-1 mRNA is induced through p38 MAPK signaling in cultured human keratinocytes. 65 p38 MAPK has also been shown to up-regulate the expression of the ER chaperone, GRP78, and to activate ER stress. 66 Moreover, p38 MAPK is a key regulator of inflammatory cytokines and mediators—an observation which makes this protein an attractive drug target for treatment. 67 Interestingly, our study shows, for the first time, that cis-resveratrol inhibits the phosphorylation of p38 MAPK. Our findings suggest that the inhibitory effects of cis-resveratrol on p38 MAPK phosphorylation and c-Jun protein expression may involve the reduction of pro-IL-1β, caspase-1, caspase-4, COX-2 and GRP78 expression.
Macrophages were first shown to secrete IL-18 following activation with stimuli such as LPS. 68 The NF-κB pathway has been shown to be involved in the production of IL-18 induced by LPS in THP-1 cells. 69 Cellular signaling leading to IL-18 production also involves MyD88- and IL-1 receptor associated kinase TRAF6-dependent pathways, which, in turn, induce NF-κB. 22 In addition, LPS-induced NLRP3 mRNA expression is reduced by NF-κB inhibitors, indicating that NF-κB may play an important role in regulating NLRP3 gene expression. 70 In the present study, pre-treatment with cis-resveratrol increased NF-κB activation and JNK phosphorylation, which may be responsible for the increased expression and production of IL-18 in activated macrophages. A recent study has shown that blockade of either ERK1/2 or JNK, but not p38 MAPK, inhibits LPS-induced expression of NLRP3 protein in murine macrophages. 71 Consistent with this finding, we found that inhibition of p38 MAPK phosphorylation did not affect the expression of NLRP3, but significantly inhibited IL-1β mRNA and pro-IL-1β protein expression in cis-resveratrol pre-treated macrophages.
It is generally recognized that COX-2 is barely detectable under normal physiological conditions, but is induced mainly at sites of inflammation in response to inflammatory stimuli, including pro-inflammatory mediators and mitogenic stimuli, such as cytokines (IL-1 cytokine family), endotoxins (LPS), growth factors, oncogenes and phorbol esters. 72 Moreover, COX-2 and its products, particularly PGE2, are known to be associated with various tumors and chronic inflammatory diseases.24,25,73–75 Therefore, COX-2 may represent a promising molecular target for the chemoprevention of cancer and inflammatory diseases. Our results demonstrate that cis-resveratrol at 100 µM markedly reduces PGE2 production, as well as COX-2 protein synthesis in activated macrophages, allowing us to conclude that cis-resveratrol has significant modulatory effects on the production of PGE2 by macrophages. These findings also suggest that reduced PGE2 levels may be due to a reduction of COX-2 by cis-resveratrol pre-treatment. Our results confirm the work of Leiro et al. who also demonstrated that cis-resveratrol blocks ROS production by inflammatory murine macrophages and inhibits PGE2 production and COX-2 expression. 76 In addition, our findings are in agreement with previous results showing that canonical inflammasome activation modulates the production of prostaglandins in macrophages. 30
ATP-induced P2X7R activation promotes the rapid production of large amounts of ROS, which, in turn, stimulates activation of the NLRP3 inflammasome. 51 P2X7R stimulation in primary human and murine macrophages is also required for the release of PGE2, and PGE2 release is independent of inflammasome activation. 77 In this study, we demonstrate that pre-treatment of LPS-primed macrophages with cis-resveratrol significantly inhibits ATP-induced P2X7R expression. Moreover, our results show that ATP-induced ROS production is suppressed by cis-resveratrol. In agreement with these findings, previous studies have shown that cis-resveratrol displays typical antioxidant activity, and that this effect may be due to inhibition of NADH/NADPH oxidase enzyme activity.76,78 Our data, which were obtained in human macrophages in vitro, also support previous observations suggesting that a decline in PGE2 production may be due, at least in part, to inhibitory effects on P2X7R expression.
Caspase-4 expression was shown to be required for caspase-1 activation and maturation of pro-IL-1β in keratinocytes and activated THP-1 macrophages, suggesting that caspase-4 may act upstream of caspase-1. 46 ROS accumulation is known to induce ER stress, which further leads to proteolytic cleavage of caspase-4.56,79 Previous studies have also reported that oxidative challenge can lead to ER stress with up-regulation of the protective chaperone protein GRP78, which has been proposed to play a crucial role in preventing oxidative damage to the ER and promoting cell survival.57,80 Pre-treatment of the cells with cis-resveratrol resulted in a significant reduction of caspase-4 expression and activation compared with resveratrol-untreated control cells. We also demonstrated that cis-resveratrol inhibits the expression of the ER stress marker GRP78 and caspase-1 activation, indicating that cis-resveratrol may act upstream of the non-canonical inflammasome and result in down-regulation of GRP78 expression and caspase-4 and caspase-1 activation, thereby leading to reduced IL-1β secretion. However, cis-resveratrol pre-treatment did not impair IL-18 secretion in activated macrophages. In this respect, a recent study has shown that processing and secretion of IL-18 does not require ROS production. 81 Based on these observations, we speculate that the inhibitory effect of cis-resveratrol on ROS production may down-regulate caspase-4 and caspase-1 activation and lead to reduced IL-1β secretion.
Previous studies have reported that trans-resveratrol is quickly absorbed and rapidly metabolized in vivo. 82 Preliminary studies in humans have shown that while trans-resveratrol is quickly absorbed following oral consumption, its bioavailability in the human body is relatively low. 83 Additional studies are needed to examine the absorption, bioavailability and potential beneficial effects of cis-resveratrol in vivo.
In conclusion, this study reveals that cis-resveratrol inhibits IL-1β secretion in activated THP-1 macrophages. The anti-inflammatory effect of cis-resveratrol is associated with reduced P2X7R and GRP78 expression, which leads to suppression of ROS production and reduced caspase-1 and caspase-4 activation (Figure 8). cis-Resveratrol also inhibits COX-2 expression, which may be responsible for the observed decrease of PGE2 production. Furthermore, inhibition of pro-IL-1β and COX-2 expression may occur by down-regulating activation of the p38 MAPK pathway. Therefore, our findings suggest that cis-resveratrol could be used as a potential therapeutic compound for treating inflammatory diseases in the future.
Proposed mechanism underlying the anti-inflammatory effects of cis-resveratrol in macrophages. Pre-treatment of macrophages with cis-resveratrol (c-RSV) suppressed ATP-induced IL-1β secretion and COX-2-produced PGE2 release in LPS-primed human macrophages. cis-Resveratrol also caused down-regulation of P2X7R expression and reduction of ROS production. cis-Resveratrol also reduced GRP78 expression, ER stress-mediated caspase-4 activation and caspase-1 activation. In addition, cis-resveratrol inhibited the phosphorylation and activation of p38 MAPK, which induces the expression of pro-IL-1β. Taken together, our results show that cis-resveratrol inhibits IL-1β secretion in activated macrophages.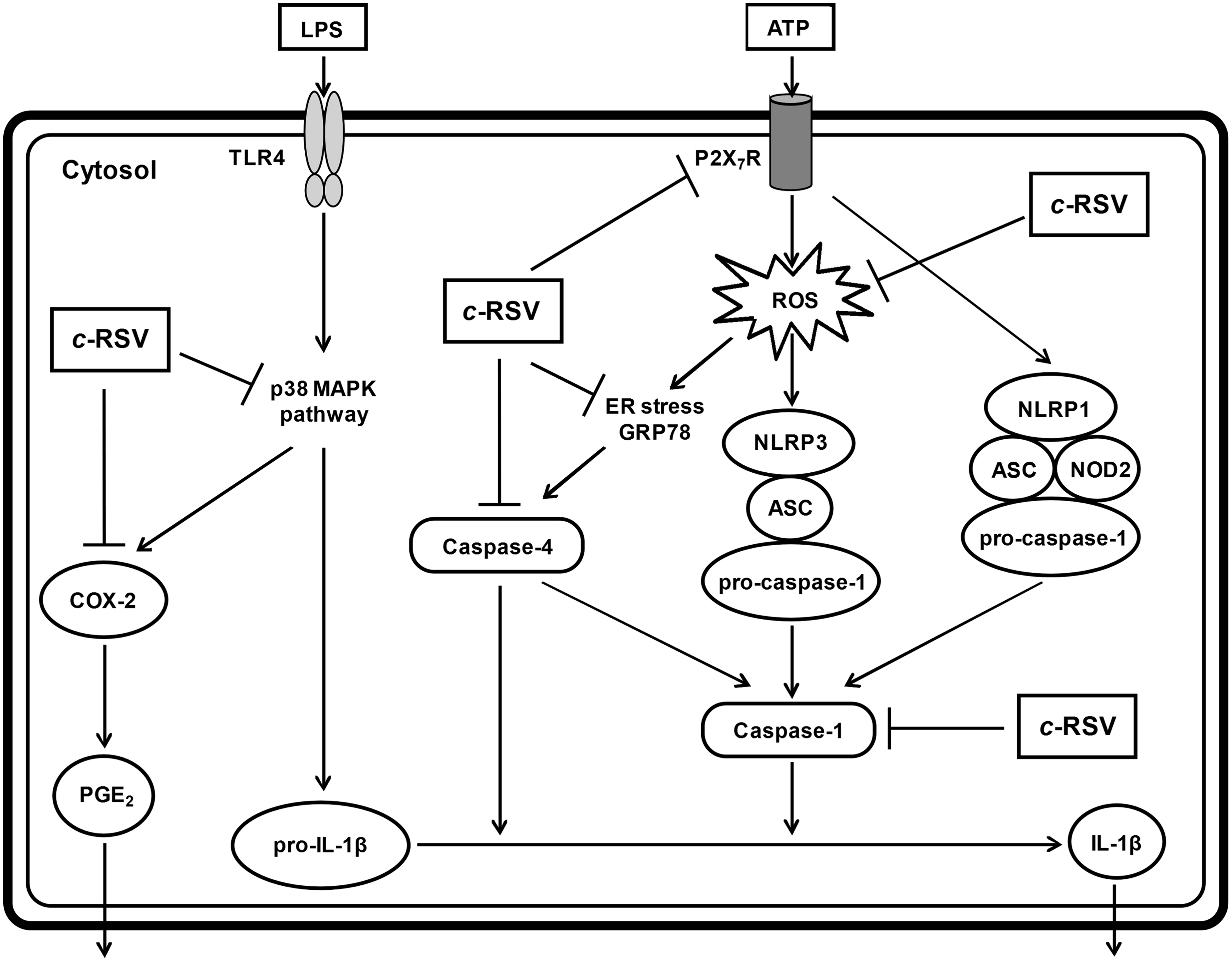
Footnotes
Funding
This work was supported by grants from the National Science Council, Taiwan (NSC 99-2632-B-182-001-MY3 to K.-Y.C., NSC 101-2320-B-182-023-MY3 and NSC 101-2321-B-002-001 to H.-C.L.); Chang Gung Memorial Hospital (CMRPD 180333 and CMRPD 180471 to K.-Y.C., CMRPD190303 and CMRPD190503 to H.-C.L.); and Chang Gung Molecular Medicine Research Center (EMRPD180171 to K.-Y.C.).
Conflict of interest
The do not have any potential conflicts of interest to declare.
Acknowledgements
We thank all colleagues in the laboratory for helpful discussions and technical assistance.
