Abstract
Prior exposure to endotoxins renders the host temporarily refractory to subsequent endotoxin challenge (endotoxin tolerance). Clinically, this state has also been pointed out as the initial cause of the non-specific humoral and cellular immunosuppression described in these patients. We recently demonstrated the restoration of immune response with mifepristone (RU486), a receptor antagonist of glucocorticoids. Here we report the treatment with other modulators of glucocorticoids, i.e. dehydroepiandrosterone (DHEA), a hormone with anti-glucocorticoid properties, or metyrapone (MET) an inhibitor of corticosterone synthesis. These drugs were able to partially, but significantly, restore the humoral immune response in immunosuppressed mice. A significant recovery of proliferative responsiveness was also observed when splenocytes were obtained from DHEA- or MET-treated immunosuppressed mice. In addition, these treatments restored the hypersensitivity response in immunosuppressed mice. Finally, although neither DHEA nor MET improved the reduced CD4 lymphocyte count in spleen from immunosuppressed mice, both treatments promoted spleen architecture reorganization, partially restoring the distinct cellular components and their localization in the spleen. The results from this study indicate that DHEA and MET could play an important role in the restoration of both adaptive humoral and cellular immune response in LPS-immunosuppressed mice, reinforcing the concept of a central involvement of endogenous glucocorticoids on this phenomenon.
Introduction
Septic processes constitute one of the major causes of death in intensive care units, and epidemiological studies indicate that mortality reaches around 30% of patients both in Europe and the USA. 1
Over the last 30 yrs, most therapies for sepsis have focused on attenuating the initial inflammatory response, ignoring the development of immunosuppression during the late phase.2,3 However, although these approaches have demonstrated modest benefits in some groups of patients, the majority of deaths occur in immunosuppressed septic patients.4–7 The failure of these treatments has been, in part, because of a lack of understanding of the pathogenic mechanisms driving sepsis. 8
In fact, although both pro- and anti-inflammatory responses begin simultaneously after sepsis initiation, in general and at clinical level, there is a predominance of an initial hyper-inflammatory phase, which, depending on host genetic factors, age, pathogen virulence, bacterial load, etc., can end in multiple organ failure.8,9 However, days or weeks after the beginning of sepsis, the clinical evidence indicates an altered immune status, which frequently leads to a progressive immunosuppression, the main cause of the inability of patients to overcome the original or, eventually, secondary infections.2,8–10
In sepsis caused by Gram-negative bacteria, endotoxins, a normal constituent of the bacterial outer membrane also known as LPS, have been described as one of the main agents causing the undesirable effects in this critical illness. 11
Thus, the response to LPS involves a rapid secretion of pro-inflammatory cytokines, such as TNF-α, IFN-γ, IL-1, IL-6 and IL-8, as well as anti-inflammatory mediators, such as IL-10 and TGF-β, or glucocorticoids (GC).12–17 Repeated exposure to LPS renders the host temporarily refractory to subsequent lethal doses of LPS challenge in a process known as LPS or endotoxin tolerance.18–21
Although it is recognized as a mechanism to prevent systemic inflammation, endotoxin tolerance has also been suggested as the main cause of inducing non-specific humoral and cellular immunosuppression described in septic patients.20,22–24
Taking this into account, and in agreement with Hotchkiss et al., we consider that one of the key aspects of late sepsis is immunosuppression. 8 Whereas some patients succumb during early stages, probably owing to the massive inflammatory response that usually involves multiple organic failure, we have to take into account that more than 70% of deaths in sepsis occur after the first 3 d of the initiation of the disorder, with many of them occurring weeks later—a period in which the immunosuppression is established.9,10 Therefore, the recovery or preservation of the host immune functions would be one of the principal aims in the management of sepsis.
Using a mouse model of LPS-induced tolerance/immunosuppression, we recently demonstrated the recovery of humoral and cellular immune response with mifepristone (RU486), a glucocorticoid receptor antagonist.25,26 In addition, we also showed that immature myeloid-derived suppressor (GR-1+CD11b+) and regulatory T cells [CD4+CD25+FoxP3+], two types of cells claimed to be involved in different mechanisms of immunosuppression, were not relevant in the maintenance of endotoxin tolerance, the initial phase of immunosuppression. Moreover, as these suppressor and regulatory cells can be induced by GC, these findings suggest that the presence of these cells is more an epiphenomenon induced by glucocorticoid action than as a result of endotoxin tolerance. 26 Thus, in our model, GC seem to be the central molecules in the management of cellular and humoral immunosuppression induced by LPS.
However, to gain insight into this effect and in order to discard the possibility that any undesirable or secondary RU486 effect could be responsible for re-installing the immune response, other modulators of GC were evaluated in this work. Thus, dehydroepiandrosterone (DHEA), a steroid hormone precursor that exhibits both immune stimulatory and anti-glucocorticoid effects,27,28 and metyrapone (MET), a recognized inhibitor of corticosterone synthesis,29,30 were assessed in their ability to restore the immune response in the LPS-induced immunosuppression model.
In brief, our results indicated that, similar to RU486, DHEA and MET could play an important role in the restoration of both adaptive humoral and cellular immune response in LPS immunosuppressed mice, highlighting the central involvement of endogenous GC in this phenomenon.
Materials and methods
Reagents
DHEA, MET, FITC, ConA, phorbol 12-myristate 13-acetate (PMA), propyleneglycol (1,2-propanediol) and LPS from Escherichia coli O111:B4 purified by phenol extraction were obtained from Sigma-Aldrich (St Louis, MO, USA). Dihydrorhodamine123 (DHR123) was obtained from Invitrogen (Carlsbad CA, USA). SRBC were obtained from Alfredo Gutierrez (Buenos Aires, Argentina).
Mice
BALB/c mice were bred in the animal facility of the Academia Nacional de Medicina, Buenos Aires. Female mice aged 12–16 wks and weighing 20–25 g were used throughout the experiments. They were maintained under a 12-h light–dark cycle at 22 ± 2℃ and fed with a standard diet plus water ad libitum. The experiments performed herein were conducted according to the principles set forth in the Guide for the Care and Use of Laboratory Animals. 31
LPS-induced immunosuppression model
The induction of a strong immunosuppression state was obtained using the model of LPS-induced immunosuppression as previously described. 26 Briefly, mice were treated with different doses of LPS for 12 consecutive days. The inoculation regimen began with 5 µg/mouse i.p. for the first 4 d, followed by 100 µg/mouse i.p. for the following 8 d.
Drug administration
The effective dose of DHEA for models of systemic inflammation or hemorrhagic shock goes from 4 to 100 mg/kg every 24 h.32,33 Thus, a dose of 40 mg/kg was used in our experiments. DHEA was dissolved in propylene-glycol and administered subcutaneously. MET was dissolved in deionized water and used at a dose of 80 mg/kg and injected i.p.
Immunization and drug treatment
DHEA administration
Twenty-four h after the last dose of LPS, immunosuppressed mice were inoculated with DHEA 4 and 2 h prior to the immunization with SRBC in PBS (5 × 108/mouse; 0.1 ml i.p.). Then, mice were treated again with one dose of DHEA 3 h after immunization. The following day, three doses of DHEA were administered at 3-h intervals each and, finally, the schedule continued with a daily dose during the following 4 d.
MET administration
Twenty-four h after the last dose of LPS, immunosuppressed mice were inoculated with MET. Three h after first MET administration, mice were immunized with SRBC (5 × 108/mouse; 0.1 ml i.p.) and two additional doses were administered at 2 and 4 h after immunization. On the second d, three doses of MET were administered at 3-h intervals each. The control mice (non-immunosuppressed) were also immunized with SRBC using the same schedule. Seven d after immunization, all mice were sacrificed; serum samples collected and the spleens were removed under sterile conditions. The spleens were placed in a 10% phosphate-buffered formalin solution until processing for histological studies. Immunosuppressed mice treated with vehicles used to dissolve DHEA and MET (propylene-glycol and deionized water) did not modify the level of immunosuppression induced by LPS (data not shown).
Assessment of the hypersensitivity reaction
Type IV delayed hypersensitivity response
The immunosuppressed mice were sensitized with SRBC (2 × 108 SRBC/mouse; 0.1 ml, i.p.) 24 h after the last LPS inoculation (d 1). Control mice (non-immunosuppressed) were sensitized using the same schedule. Afterwards, animals were treated with a daily injection of DHEA (40 mg/kg, s.c.) or MET (80 mg/kg, i.p.) during the following 4 or 2 d respectively. A group was left untreated after the sensitization with SRBC. Then, all mice were challenged on d 6 by injecting 1 × 108 SRBC in 30 µl PBS s.c. into the left hind footpad. Footpad thickness was measured by micrometer before and 24 h after the challenge. The degree of footpad swelling was calculated as the ratio of the footpad thickness after Ag challenge/footpad thickness before Ag challenge. A mice group was sensitized with PBS (vehicle for SRBC) and subsequently challenged.
Contact hypersensitivity response
Twenty-four h after the last LPS inoculation, the immunosuppressed mice were sensitized by painting with 50 µl of 1% FITC in 1:1 acetone/dibutyl phthalate onto the previously shaved ventro-lateral region (d 1). Control mice (non-immunosuppressed) were sensitized using the same schedule. Then, mice were treated with DHEA (40 mg/kg, s.c.) or MET (80 mg/kg, i.p.) administered daily during the following 4 or 2 d, respectively. A group was left untreated after the sensitization with FITC. At d 6, all animals were challenged with 10 µl 1% FITC onto both sides of one ear. Ear-swelling response was determined by ear thickness, measured in micrometers, before and 24 h after the second challenge. The degree of ear-swelling was calculated as the ratio of the ear thickness after Ag challenge/ear thickness before Ag challenge. A group was sensitized with acetone/dibutyl phthalate (vehicle for FITC) and subsequently challenged as the other groups.
Isolation of splenocytes
Spleens were removed and passed through a sterile mesh with ice-cooled PBS supplemented with 10% FCS, 1% penicillin and 1% streptomycin to obtain a single cell suspension. Erythrocytes were eliminated with red blood cell lysis buffer (ClNH4 0.15 M; NaHCO3 10 mM; EDTA 0.1 mM; pH 7.3), and cells were washed and counted.
Hemagglutination assay
The Ab response to SRBC was evaluated through a hemagglutination assay, as described previously. 26 Briefly, serum samples were initially diluted 1:9 and later serially diluted (1:0.33). The titer was considered as the reciprocal of the last positive dilution.
Splenocyte proliferation assay
Splenocytes (4 × 105cells/well) were incubated in 200 µl of complete medium (RPMI1640-10% FCS, 1% penicillin–streptomycin) in 96-well microtiter plates (Costar, Cambridge, MA, USA) in the presence or absence of Con A (2 µg/ml). Cells were incubated for 72 h at 37℃, 5% CO2 and 90% humidity, and pulsed with 1 µCi [3H] thymidine 18 h before harvesting the cells. The proliferative activity was calculated by measuring the 3H-thymidine incorporation. Results were expressed as the mean of (counts per minute) c.p.m. of triplicate wells minus the basal c.p.m. of wells containing pulsed, but unstimulated, cells (Δ c.p.m.).
Peritoneal macrophages
Mice were sacrificed on the seventh day and macrophages were harvested by peritoneal lavage with cold PBS supplemented with 3% FCS as described previously. 25
Determination of TNF-α production
Peritoneal macrophages (1 × 105/well) or splenocytes (4 × 105 cells/well) were incubated in complete RPMI medium in 96-well microtiter plates (Costar) for 6 or 20 h, respectively, in the presence or absence of LPS (100 ng/ml). Then, supernatants were removed and stored at −20℃ until assayed. TNF-α was evaluated by specific ELISA performed on flat-bottomed polystyrene microtiter plates (ELISA Ready-SET-Go; eBioscience, San Diego, CA, USA), according to the manufacturer’s instructions.
Flow cytometry
Splenocytes (1 × 106 cells) were immunostained with a combination of the following mAbs: (FITC)-CD4, (Cy5PE)-F4/80 (Invitrogen); [phycoerythrin (PE)]-CD8, (Cy5PE)-CD45R/B220, (FITC)-CD11b, (PE)-MHC-II (BD Pharmingen, San Diego, CA, USA). Cells were suspended in 10 µl of PBS with 1% of normal mouse serum (15 min) and incubated with the corresponding Abs for 30 min at 4℃. Then, cells were washed, fixed in PBS/0.5% paraformaldehyde and finally evaluated in a Becton Dickinson fluorescence activated cell sorter (FACScan) using FSC Express software (De Novo Software, Los Angeles, CA, USA).
Histology
Following fixation in 10% phosphate-buffered formalin, splenic tissue blocks were dehydrated in graded alcohol using standard histological techniques and embedded in paraffin. Semi-serial sections were performed using a microtome and stained with hematoxylin and eosin, or analyzed by immunohistochemistry.
Immunohistochemistry
The immunohistochemistry analysis was performed using a panel of Abs defining common leukocyte subgroups normally found in the spleen. These Abs included anti-CD20 (StaCruz, CA, USA; goat pc, 1:250) defining B cells, anti-CD3 (StaCruz mouse mc, 1:400) defining T cells. For the recognition of T cell subtype, we used anti-CD4 (Leica-Biosystems, Newcastle, Ltd, UK; CD4, pre-diluted) and anti-CD8 (StaCruz mouse mc, 1:400) Abs.
Incubations with primary Abs were performed overnight (18 h) in a moist chamber at 25℃ (room temperature). The procedure was completed using a streptavidin–biotin complex method (ABC-Kit; Vector, Burlingame, CA, USA) with 3,3’ diaminobenzidine as the chromogen. Slides were counterstained with Mayer's hematoxylin and analyzed by standard light microscopy. Negative control slides were prepared by substituting primary antiserum with PBS.
Oxidative burst
Peritoneal macrophages (4 × 105/200 µl) were incubated with 20 µl of DHR123 (5 nM) for 30 min at 37℃. Then, 40 µl of either PMA (16 µM) or medium was added and incubated for 20 min at 37℃. Cells were washed in ice-cold PBS, re-suspended in 10 µl of PBS-1% normal mouse serum for 10 min and then labeled with anti-F4/80-Cy5PE by incubation on ice for 15 min. Finally, after washing, the cells were re-suspended in IsoFlow and evaluated by flow cytometry. Data were reported as median fluorescence intensity (MFI) from rhodamine123 in the F4/80+ cell gate.
Phagocytosis assay
Peritoneal macrophages were cultured in 12-well tissue culture plates (Costar) containing coverslips at a concentration of 8 × 105 macrophages/well in complete RPMI medium. After overnight incubation at 37℃ in a humidified atmosphere of 5% CO2 (and 95% air), cells were washed twice with PBS to remove non-adherent cells. For phagocytosis, previously IgG-opsonized SRBC were added at a ratio of 30:1. Culture medium was supplemented with pooled normal mouse serum as a source of complement (1/10). Afterwards, the plates were centrifuged to promote contact between the added cells and macrophages, and incubated for 30 min at 37℃. To stop phagocytosis, plates were transferred onto ice and the cells were washed with cold PBS. Non-ingested SRBC were lysed with lysis buffer and coverslips were then washed and stained. Finally, cells were stained with May–Grunwald–Giemsa. Coverslips were mounted for examination under 100 × oil immersion objective. The percentage of phagocytosis was determined by expressing the number of macrophages capable of engulfing erythrocytes from a total number of 200 macrophages counted.
Statistical analysis
Values are expressed as the mean ± SEM of n observations. A one-way ANOVA method, followed by the Student–Newman–Keuls test as a post hoc test for multiple comparisons, was used to determine the significance of differences between experimental means; a normality test was previously evaluated. All statistical tests were interpreted in a two-tailed fashion and a P-value < 0.05 was considered statistically significant.
Results
DHEA and MET restore the primary humoral immune response in LPS-induced immunosuppressed mice
To evaluate the effect of treatment with DHEA or MET at the level of immune response, LPS-induced immunosuppressed mice were treated with each one of the drugs and immunized with SRBC. Seven d after immunization the animals were bled and the humoral immune response was evaluated.
Figure 1 shows a profound decreased in anti-SRBC Ab titre from the immunosuppressed mice compared with the control mice. The administration of DHEA or MET enabled a partial, although significant, restoration of the humoral immune response.
DHEA and MET restore the humoral immune response in LPS immunosuppressed mice. LPS-induced immunosuppressed mice treated or not with DHEA or MET, and the control mice (non-immunosuppressed) were immunized with SRBC (5 × 108/mouse i.p.) (see ‘Materials and methods’). Seven d after immunization the animals were bled and serum Abs were evaluated through a hemagglutination assay. Results are expressed as mean ± SEM (hemagglutination titre). Each point represents an individual mouse. The data represent three independent experiments with 4 to 5 mice per group. IS: LPS-immunosuppressed mice; IS-DHEA: DHEA-treated LPS-immunosuppressed mice; IS-MET: MET-treated LPS-immunosuppressed mice; C: control non-immunosuppressed mice; *P < 0·05 versus C; #P < 0·05 versus IS.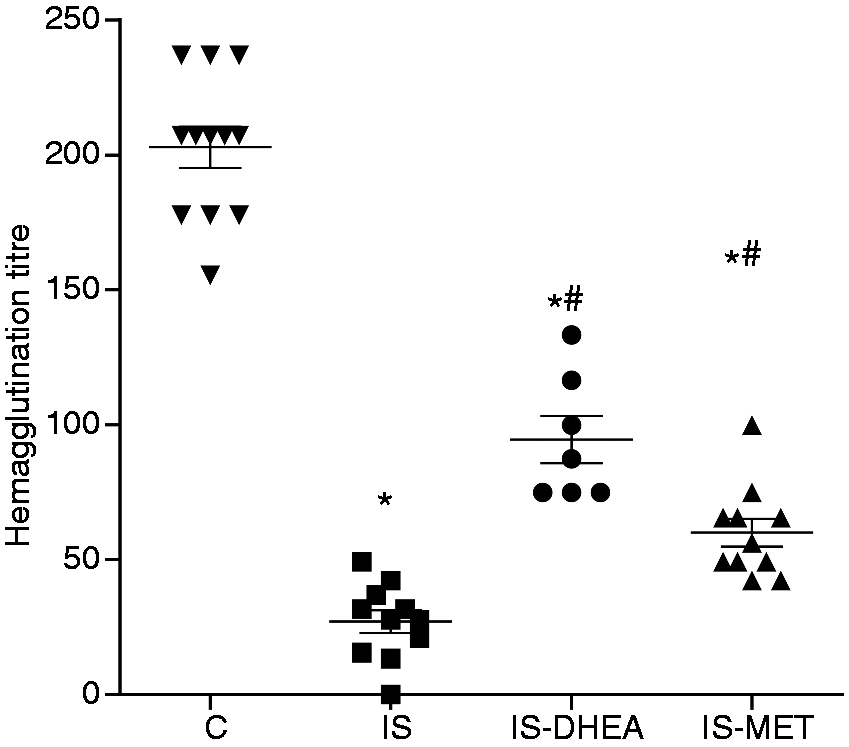
DHEA and MET recover the cellular immune response in LPS-immunosuppressed mice
In order to determine if DHEA and MET were also capable of overcoming the LPS-induced immunosuppression at the cellular proliferation level, ex vivo experiments were performed. LPS-immunosuppressed mice were treated with DHEA or MET in different protocols, as described in the ‘Materials and methods’ section. After 7 d, spleens were removed and cell proliferation was assayed through the non-specific proliferation of splenocytes stimulated with Con A. As shown in Figure 2, proliferation of splenocytes from immunosuppressed mice was significantly lower than those of the control mice. However, the splenocytes from mice of the DHEA- and MET-treated immunosuppressed groups showed a partial recovery of the proliferative response.
DHEA and MET recover the proliferative response of splenocytes to Con A in LPS-immunosuppressed mice. Splenocytes from LPS-immunosuppressed mice treated or not with DHEA or MET, and subsequently immunized with SRBC were obtained on d 7 post-immunization and then evaluated for proliferation assay with Con A (see ‘Materials and methods’). Spleens from control mice previously immunized were also processed. Results are expressed as mean ± SEM (Δ c.p.m.) of 6–8 mice per group. The experiment was repeated twice. IS: LPS- immunosuppressed mice; IS-DHEA: DHEA-treated LPS-immunosuppressed mice; IS-MET: MET-treated LPS-immunosuppressed mice; C: control non-immunosuppressed mice; *P < 0.05 versus C; #P < 0.05 versus IS.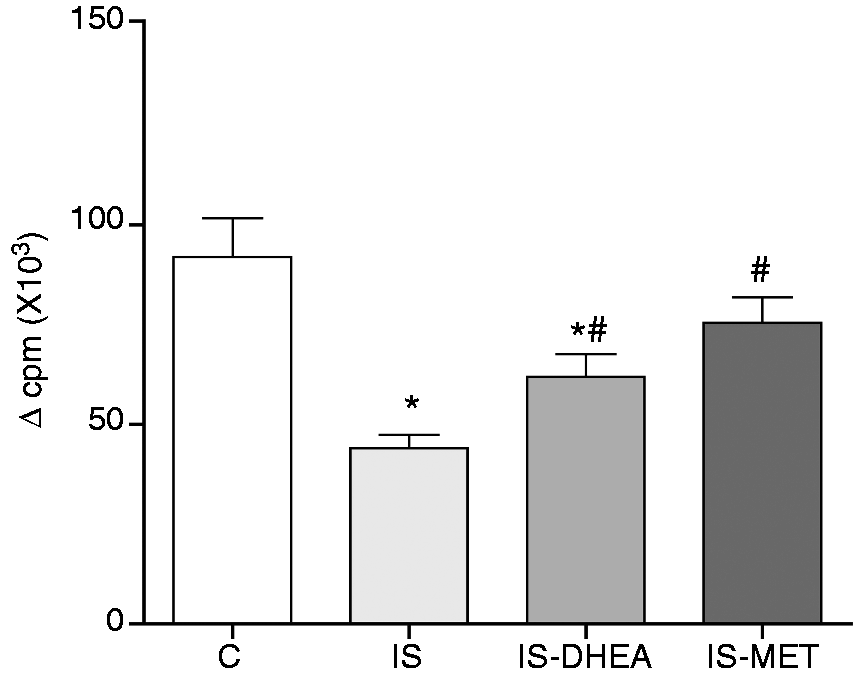
Two additional experiments were performed to evaluate the cellular response. For in vivo experiments, we used two readily measurable inflammatory immune responses, the delayed-type hypersensitivity response (DTH) to SRBC and the contact hypersensitivity (CHS) response to FITC. As shown in Figure 3, DTH and CHS responses were significantly lower in immunosuppressed mice compared with the control mice.
DHEA and MET recover the DTH and CHS response in LPS-immunosuppressed mice. (A, C) LPS-immunosuppressed mice were sensitized with an i.p. injection of SRBC or (B, D) with FITC onto their ventro-lateral region on d 1.Twenty-four h later the animals were treated or not with DHEA or MET. These drugs were administrated daily during the following 4 or 2 d, respectively. On the sixth d the animals were challenged either in the footpad with SRBC or on both sides of one ear with FITC. Footpad and ear thickness were measured prior to challenge and 24 h post-challenge. The control mice were exposed to the same schedule for each Ag. The challenged mice (chall) were challenged without a previous sensitization with Ag. Results are expressed as mean ± SEM of the thickness ratio (thickness after antigen challenge/thickness before challenge). Each point represents an individual mouse. Data represent two independent experiments with 3–5 mice per group. IS: LPS-immunosuppressed mice; IS-DHEA: DHEA-treated LPS-immunosuppressed mice; IS-MET: MET-treated LPS-immunosuppressed mice; C: control non-immunosuppressed mice. *P < 0.05 versus C; #P < 0.05 versus IS.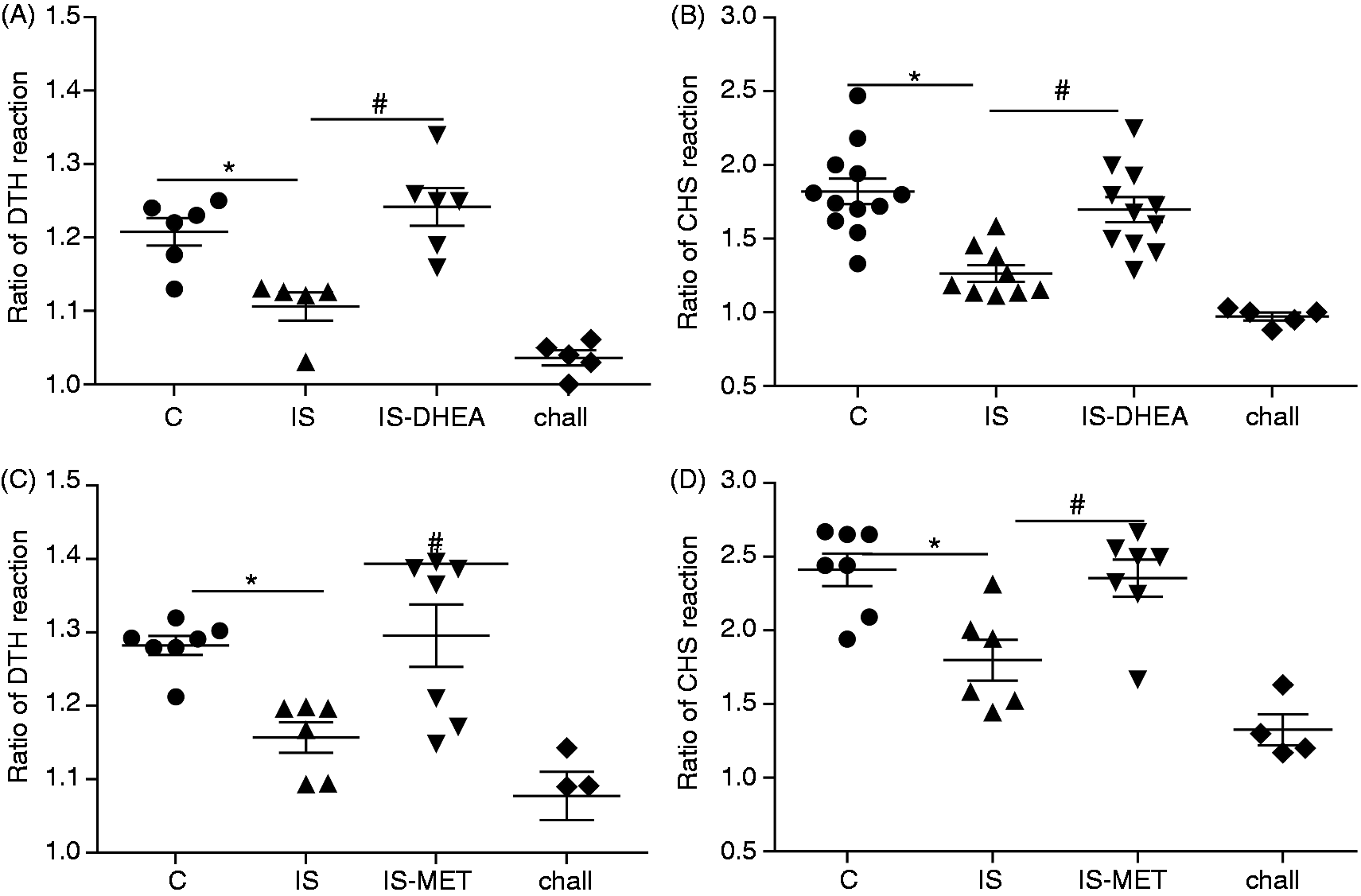
However, the administration of DHEA to immunosuppressed mice led to a complete restoration of both DTH and CHS reactions (Figure 3A, B). MET treatment also induced a restitution of the cellular immunity for both reactions in immunosuppressed mice (Figure 3 C, D). DHEA or MET administration did not affect both reactions in control mice (control-DHEA 1.19 and control-MET 1.28 versus control 1.24, DTH reaction; control-DHEA 1.82 and control-MET 2.21 versus control 2.11, CHS reaction)
DHEA and MET do not restore the number of CD4 cells in spleen but partially rearrange the splenic architecture and the distribution of B and T lymphocytes in immunosuppressed mice
Spleens from immunosuppressed mice show a decreased number and percentage of CD4 lymphocytes and a reduced expression of MHC-II on macrophages.
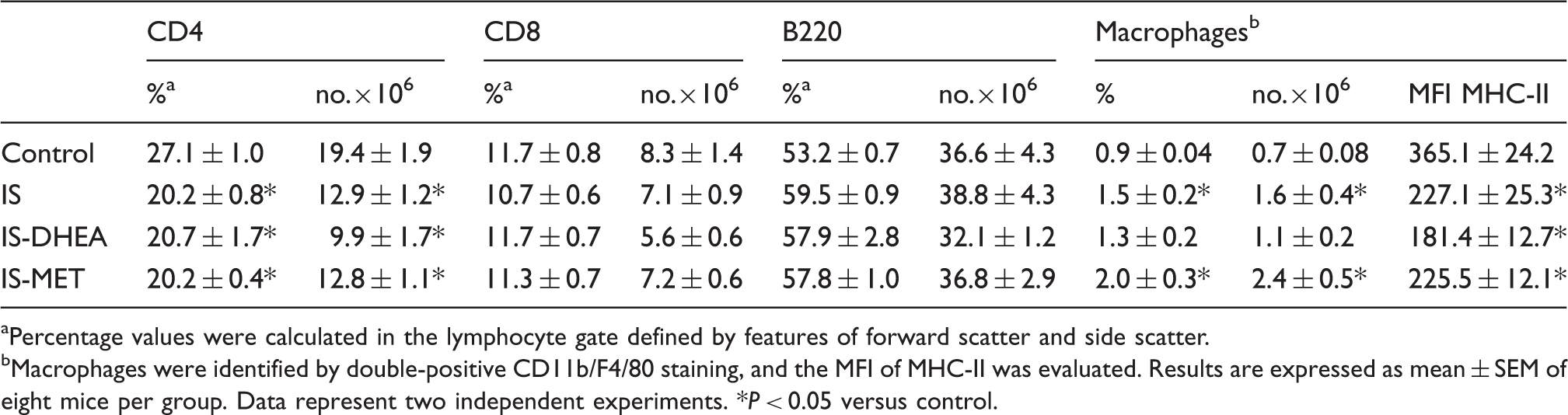
Percentage values were calculated in the lymphocyte gate defined by features of forward scatter and side scatter.
Macrophages were identified by double-positive CD11b/F4/80 staining, and the MFI of MHC-II was evaluated. Results are expressed as mean ± SEM of eight mice per group. Data represent two independent experiments. *P < 0.05 versus control.
The histological analysis of immunosuppressed animals showed a relative enlargement of the red pulp due to a decrease of cellular components of the white pulp. In addition, the white pulp nodules composed of mature lymphocytes with germinal formations were smaller than those of the controls and no apparent marginal zones were detected. Interestingly, treatment with DHEA and MET partially restored the histology pattern in immunosuppressed mice. Thus, in some animals the white pulp structures reassumed their size and shape in an irregular fashion, showing complete and normal cell populations. However, other animals showed an incomplete cell population lacking, mainly, the marginal zone. However, the relationship between the red and white pulp, together with the cell populations, seemed to be completely restored with both drugs (Figure 4A).
DHEA and MET promote a partial rearrangement of the splenic architecture in LPS-immunosuppressed mice. (A) Hematoxylin and eosin, (original magnification 250 ×). Control spleen with follicles in the white pulp, which are regular in shape and size along with proportional red pulp; spleen of immunosuppressed animals with small and irregular follicles, without recognizable marginal zone and relatively increased red pulp (IS); spleen of immunosuppressed mice treated with DHEA with partial restoration of architecture and irregular coalescing follicles with a slight predominance of the red pulp (IS-DHEA); spleen from immunosuppressed mice treated with MET with regular follicles (in shape and size) partially recovered along with white/red pulp relationship recovered (IS-MET). (B) Splenic tissue immunostained with anti-CD20. Control spleen with lymphocyte B arranged in the peripheral region of the follicle; in spleen IS few B lymphocytes were arranged in the periphery of the follicle; spleen from IS-DHEA or IS-MET were found with abundant B lymphocytes in the peripheral zone of the follicle. (Original magnification 250×, except IS-MET which had an original magnification of 400×). (C) Splenic tissue immunostained with anti-CD3. Control spleen showed CD3 T lymphocytes in the mantle zone and in the red pulp; in IS, few CD3 T lymphocytes were observed in the red pulp; IS-DHEA and IS-MET showed T lymphocytes in the mantle zone and red pulp. (Original magnification: 250×; IS: 250×; IS-DHEA: 400×; IS-MET: 100×). (D) Splenic tissue immunostained with anti-CD4 and (E) immunostained with anti-CD8. Control spleen showed CD4 and CD8 T lymphocytes dispersed in the inter-follicular areas; spleen from IS displayed a few CD4 T cells in the periphery of follicle and CD8 cells scattered in the inter-follicular areas; spleen from IS-DHEA showed CD4 T lymphocytes in the periphery of the follicle and the PALS, as well as CD8 T lymphocytes in the inter-follicular areas; IS-MET revealed CD4 T lymphocytes in the PALS and CD8 lymphocytes dispersed in the red pulp. (Original magnification 400× except for anti-CD4 in IS-MET, which was 250×).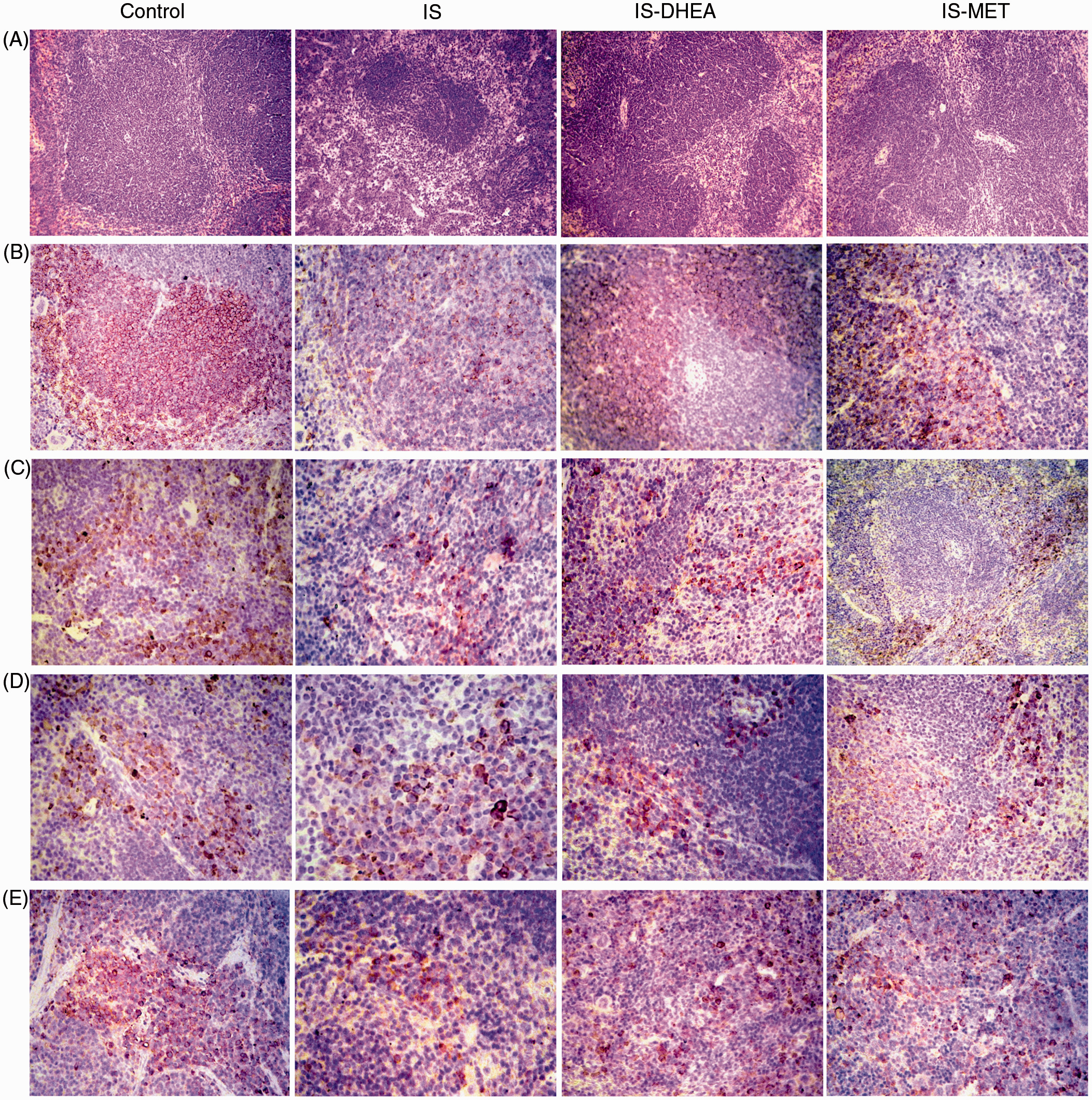
The immunohistochemical analysis of spleens demonstrated a lower presence of CD20 B cells in the white pulp nodules and in the red pulp of immunosuppressed animals (Figure 4B). Although CD4 and CD8 T lymphocytes displayed a normal histological location, they appear to be diminished in quantity. A similar rearrangement of the lymphocytes was observed using Abs against CD3 (Figure 4 C–E). In contrast, the spleen from immunosuppressed mice treated either with DHEA or MET showed an increased intensity for CD20 immunostaining within the follicles (Figure 4B). Furthermore, a reorganization of inner PALS CD4 T lymphocytes and inter-follicular CD8 T lymphocytes was observed in both groups (Figure 4B). A similar rearrangement of T lymphocytes was observed with Abs against CD3 when treated with any of the drugs (Figure 4 C–E).
TNF-α production
As TNF-α is a pro-inflammatory agent usually involved in the pathophysiology of inflammation, we analyzed the effect of DHEA and MET on the regulation of this cytokine in splenocytes and peritoneal macrophages.
Splenocytes from immunosuppressed mice exhibited a decreased production of TNF-α upon stimulation with LPS, whereas the corresponding responses were almost restored to normal values in splenocytes from immunosuppressed mice treated with DHEA by 6 d. Conversely, treatment with MET did not induce an increase in the TNF-α levels on LPS-stimulated splenocytes from immunosuppressed mice (Figure 5A).
DHEA reverses the decreased release of TNF-α in splenocytes and peritoneal macrophages from LPS-immunosuppressed mice. (A) Splenocytes and (B) peritoneal macrophages isolated from control and immunosuppressed mice inoculated or not with DHEA or MET were stimulated with LPS (100 ng/ml) in culture. The stimulation of splenocytes and peritoneal macrophages were performed for 20 and 6 h, respectively. The levels of TNF-α were determined by specific ELISA. Results are expressed as the mean ± SEM (% TNF-α production from to control) of 4–5 animals per group. This experiment was repeated twice. IS: LPS-immunosuppressed mice; IS-DHEA: DHEA-treated LPS-immunosuppressed mice; IS-MET: MET-treated LPS-immunosuppressed mice; C: control mice. *P < 0.05 IS versus control; #P < 0.05 IS versus IS-DHEA.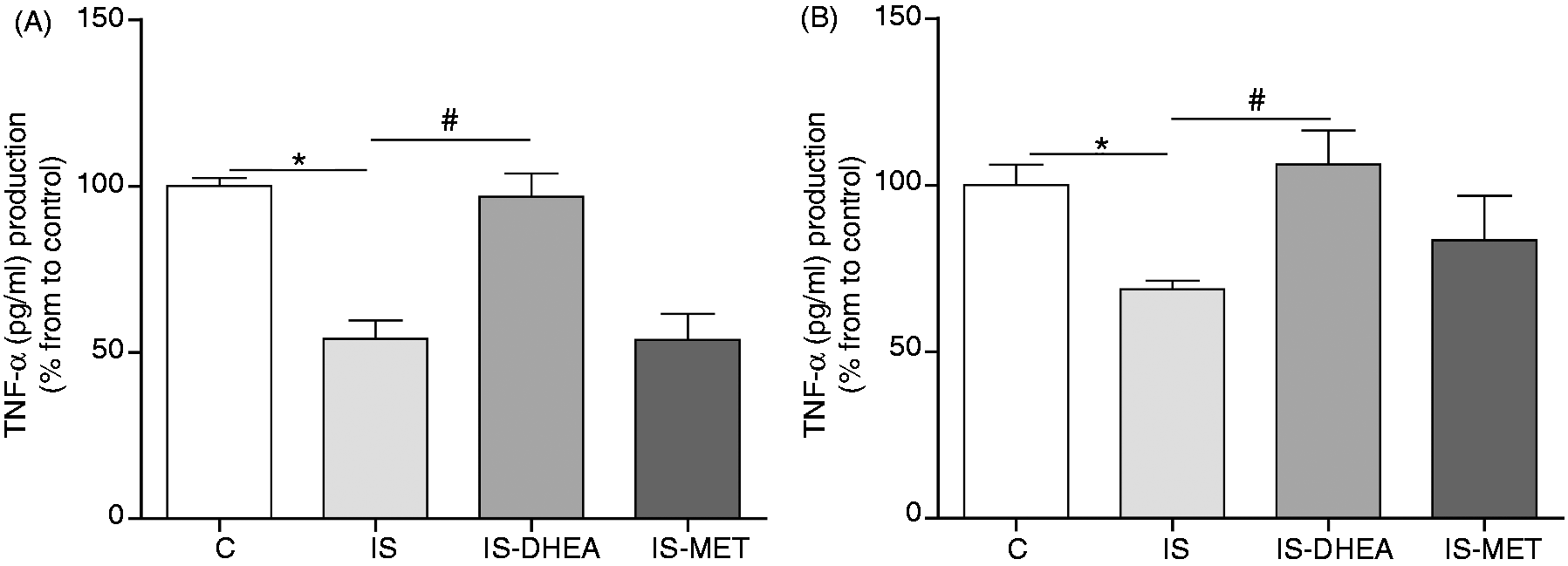
Regarding peritoneal macrophages from immunosuppressed mice, these cells displayed a reduced production of TNF-α in response to LPS stimulation and the in vivo treatment with DHEA clearly restored cytokine levels. MET treatment induced a low increase of TNF-α production; this increment, however, was not statistically significant (Figure 5B).
Phagocytic capacity and oxidative burst
As treatment with DHEA or MET improved both the adaptive immune response, as well as TNF-α production, we proceeded to evaluate the effect of these treatments on innate immune effector mechanisms by assessing phagocytic capacity and reactive oxygen species (ROS) production in peritoneal macrophages. For this experiment, the immunosuppressed mice were treated or not treated with DHEA or MET for 6 or 2 d, respectively. Then, peritoneal macrophages were incubated with Ab-sensitized SRBC for 30 min for the phagocytosis assay. The results showed an increased phagocytic capacity in macrophages from immunosuppressed mice measured as the percentage of cells that engulfed SRBC compared with the control group (Figure 6A). Furthermore, the analysis of frequency distribution median of the number of SRBC per phagocyte indicated a greater number of ingested SRBC per cell in the same group (Figure 6B). However, in vivo DHEA treatment did not modify the phagocytic capacity observed in the macrophages from immunosuppressed mice (Figure 6A). MET treatment showed similar results to DHEA (data not shown).
Peritoneal macrophages from immunosuppressed mice show exacerbated phagocytic capacity (A) Peritoneal macrophages isolated from control and immunosuppressed mice treated or not with DHEA for 6 d were incubated with previously opsonized SRBC (30:1 ratio) for 30 min. Phagocytosis was calculated as the percentage of macrophages containing phagocytosed SRBC determined by counts with May–Grunwald–Giemsa staining. Results are expressed as mean ± SEM (% macrophages with SRBC) of 4–6 mice per group. This experiment was repeated three times. (B) Data show a representative experiment of three of distribution relative frequencies of SRBC number per macrophages from immunosuppressed and control mice. IS: LPS-immunosuppressed mice; IS-DHEA: DHEA-treated LPS-immunosuppressed mice; C: control mice. *P < 0.05 versus control.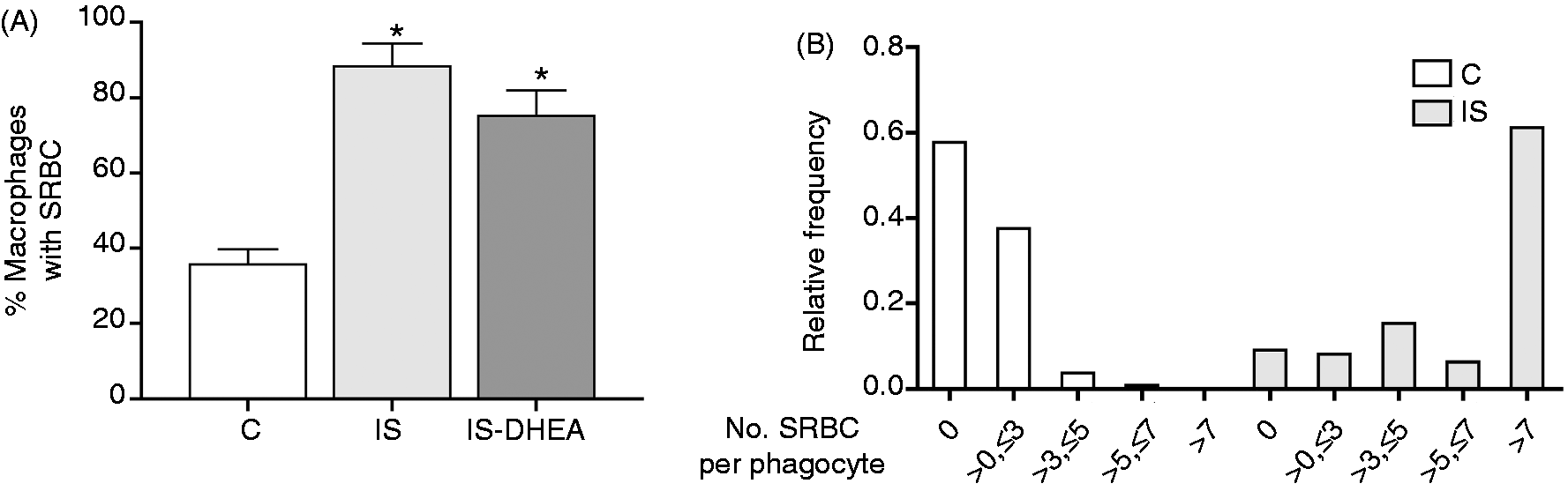
To asses ROS production, the peritoneal macrophages were isolated from all groups as described above and analyzed for their oxidative burst activity using a flow cytometric-based assay. A significant decrease in the baseline of the oxidative burst activity of peritoneal macrophage was observed in immunosuppressed mice compared with the control in the absence of PMA stimulation. Although this activity maintained a similar magnitude compared with the baseline in presence of PMA stimulation, the macrophages from immunosuppressed mice showed a lower oxidative burst activity than the control. Nevertheless, no recovery of the oxidative response was detected for any of the drugs in the macrophages from immunosuppressed mice (Figure 7).
Peritoneal macrophages from immunosuppressed mice show low ROS production. Peritoneal macrophages isolated from control and immunosuppressed mice treated or not with DHEA or MET for 6 and 2 d, were stained with DHR123 and then treated with (+) or without (-) PMA for 20 min. Flow cytometry was used to measure oxidative burst from macrophages identified by positive F4/80 staining. ROS production was expressed as the MFI of intracellularly-converted DHR123 to fluorescent rhodamine. Results are expressed as mean ± SEM (MFI) of 6–8 mice per group. This experiment was repeated twice. IS: LPS-immunosuppressed mice; IS-DHEA: DHEA-treated LPS-immunosuppressed mice; IS-MET: MET-treated LPS-immunosuppressed mice C: control mice. *P < 0.05 versus un-stimulated control; #P < 0.05 versus stimulated control.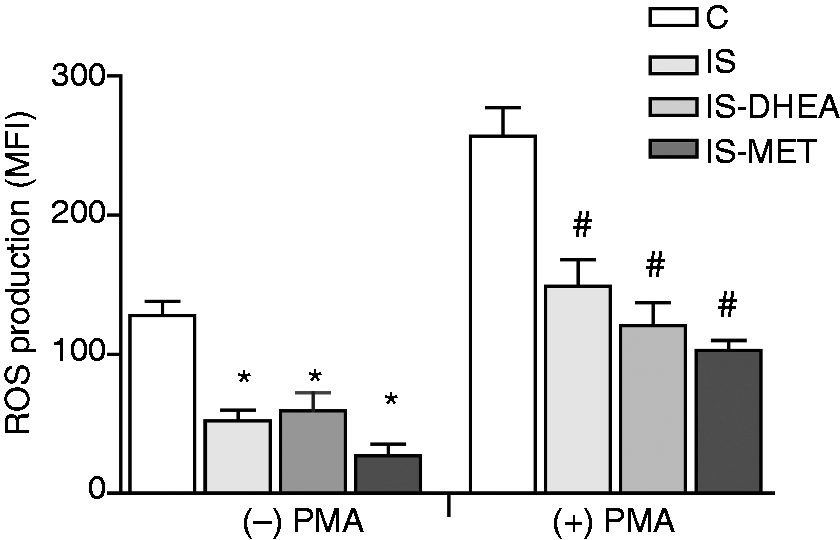
Discussion
The development of new therapies for sepsis has been particularly frustrating over the last 30 yrs, a period referred to as the ‘graveyard for pharmaceutical companies’. 16 This is evidenced by the results of more than 25 trials that have failed in ameliorating the disease. 34 This failure has been partly due to a lack of understanding of the pathogenic mechanisms driving sepsis. 8 In fact, most therapies for sepsis have focused on attenuating the initial inflammatory response, disregarding the secondary occurrence of an immunosuppression state developed during delayed phases,2,3 which is the cause of the inability to fight secondary infections in the post-septic period.8–10 In addition, it has been reported that even dormant viruses such as herpes simplex and cytomegalovirus can also be reactivated in these patients.22,35
Taking this into account, efforts to recover or preserve host immune functions has received much attention as the possible next major advance in the management of patients with sepsis.8–10 In agreement with this assumption, we have previously demonstrated that Mifepristone (RU486) is able to overcome the LPS-induced tolerance and immunosuppression at both the humoral and cellular levels,25,26 suggesting that glucocorticoids may be one of the main agents involved in immmunosuppression. Unfortunately, RU486 has other effects than inhibiting glucocorticoid activity, such as inhibiting progesterone or eventual undesirable effects. For this reason, in this report we intended to overcome LPS-induced immunosupression using two other agents involved in the glucocorticoid action: DHEA and MET.
DHEA is one of the most abundant adrenal sex steroid hormone precursors in human adults. Moreover, it exerts a variety of positive immunologic effects in a series of animal models, for example trauma, hemorrhage and sepsis.36,37 In these models, for instance, DHEA restores splenocytes proliferative capacity, increases the suppressed release of pro-inflammatory cytokines and reverts a DTH-depressed response.32,36,38–40
However, MET is an inhibitor of the 11β-hydroxylase enzyme (referred as to cytochrome P450 11B1), which is involved in the conversion of 11 deoxycortisol/deoxycorticosterone to cortisol/corticosterone in the human or rodent adrenal cortex, respectively. This inhibitor prevents the synthesis and subsequent secretion of glucocorticoid in plasma 41 and reduces the elevation of corticosterone induced by stress without affecting the basal level. 42 In addition, it also enhances the expression of pro-inflammatory cytokines mRNA still in the presence of plasma corticosterone. 43
The mechanisms by which DHEA mediates its beneficial effects on the immune system have not been clarified. However, it has been suggested that this drug acts directly on lymphocytes and monocytes via high-affinity DHEA-specific receptors in humans, as well as in mice.44–46
However, as DHEA is a precursor to androgens and estrogens, it was considered that the immune-enhancing effects observed following DHEA treatment could be mediated by the action of downstream steroids derivatives of DHEA.32,47,48 Moreover, there is evidence that DHEA and/or its metabolites may act as natural anti-glucocorticoids, counteracting the immunosuppressive actions of glucocorticoids.27,49 Blauer et al. 50 have demonstrated that DHEA antagonizes the suppressive effects of dexamethasone on lymphocyte proliferation. In addition, DHEA attenuates the cellular apoptosis rate, which could also be attributed to its anti-glucocorticoid activity observed in sepsis models. 33 In this study we described similar results between DHEA and MET. Taking into considerations previous findings with RU48625,26 and the results of the present work, we think that the immuno-enhancing effects of DHEA on restoring immune functions in our LPS immunosuppression model is related to its anti-glucocorticoid activities, which supports previous findings in the literature.
DHEA and MET were able to restore the capacity of Ab synthesis and the ability of Con A to induce T cell proliferation in immunosuppressed mice. It is important to note that although other authors have demonstrated that apoptosis of lymphoid cells would be one of the main mechanisms for immunosuppression, 51 our results demonstrate that this would not be the only mechanism because the partial recovery of immune competence by DHEA and MET—previously demonstrated with RU486 26 —shows that at least some of the cell(s) committed in the immune response were functionally affected, but not killed or eliminated by LPS treatment.
The improvement of cellular immune function by DHEA has been related to the reversal of sepsis-related DTH suppression. 36 In this case we demonstrated that not only DHEA treatment but also MET administration completely restored both DTH and CHS reaction in immunosuppressed mice. Although both responses are T-cell-dependent, each response is mediated by different effector cells. 52 Therefore, these treatments probably allowed the modulation of two different cellular compartments associated at the T cell immunity. Thus, our results suggest that glucocorticoids play an important role in the depression of the T-cell-dependent immune functions in an LPS-induced immunosuppression murine model.
Many authors have reported that DHEA exerts modulatory effects on mechanisms of innate immunity, such as pro-inflammatory cytokine modulation,32,39 on monocytes/macrophages or the increase of ROS production by neutrophils.53,54 Also, in accordance with these findings, DHEA reversed the suppressed release of pro-inflammatory cytokines by macrophages in trauma hemorrhage and experimental sepsis models.32,39 In this work we observed that DHEA treatment increased TNF-α in splenocytes and peritoneal macrophages from immunosuppressed mice. In contrast, the treatment with MET did not promote a significant increase of TNF-α levels in splenocytes or macrophages from immunosuppressed mice. This finding is in line with previous reports that demonstrated that in vivo treatment with MET did not modify the TNF-α levels induced by LPS. 55 However, other researchers have described an inhibition of LPS-induced TNF-α by MET. 56 With these last results in mind, some authors have suggested that an enzyme of the cytochrome P450 family might be involved in the signaling pathway leading to TNF-α production. This would be another effect of MET administration apart from the already described blockade of glucocorticoids (GC) synthesis.57,58 Furthermore, in the present study, we observed other alterations on innate effector mechanisms, such as an enhancement of the macrophage phagocytic capacity. Several reports have shown an exacerbated phagocytic capacity during endotoxin tolerance status using different cell models. However, there are some controversial aspects related to this augmented phagocytic capacity. In fact, while some authors consider that this may contribute to an accelerated clearance of microorganisms,59,60 others have demonstrated a poor antigenic presentation 61 and a reduced killing capacity by these macrophages. 62 In line with this, we observed a lower increase of ROS production in peritoneal macrophages from immunosuppressed mice. Other authors have consistently demonstrated an impaired respiratory burst in neutrophils both in endotoxin tolerance and in experimental sepsis models.11,63 Although in our experiments DHEA modulated the production of TNF-α in macrophages from immunosuppressed animals, no effects on phagocytic activity or ROS production were observed. Similarly, treatment with MET did not show effects on the innate immunity parameters that were evaluated. Future work is needed to make a final interpretation of the regulation of these effects.
Regarding cytometric and histological studies, we observed a diminished number of CD4 lymphocytes in spleen from immunosuppressed mice. These data are in agreement with other reports where an increment in the level of apoptosis has been demonstrated in this lymphocyte subset both in experimental sepsis model, as well as in septic patients.51,64 Furthermore, we also observed a reduced expression of MHC class II molecules on splenic macrophages, which has been associated with a diminished capacity for Ag presentation, consistent with a state of immunosuppression.11,61 Although no improvement was detected in the parameters evaluated by cytometry when the groups were treated with DHEA or MET, the histological studies revealed that both drugs promoted an architectural reorganization in spleen of immunosuppressed mice. Consequently, the administration of each drug partially restores the distinct components of the spleen (white and red pulp) to normal spleen histology, but this reorganization shows an irregular pattern.
Immunohistochemical studies revealed that both treatments promoted a partial improvement of the distribution and specific location of the different cellular compartments. Thus, these results suggest that the clear effect of DHEA and MET on functional variables such as humoral and cellular response could be due to an effect on the relocation of these populations in spleen architecture rather than to a quantitative recovery of cell populations. In addition, that proper distribution of effector cells seems to be critical for the recovery of the immune response.
In summary, the evidence observed in this study allows us to confirm the central effects of glucocorticoids in LPS-induced immunosuppression and indicates that the modulation of their action may be an important tool to, eventually, develop strategies to reinstall or reinforce an active immune response in late sepsis patients.
Whether our results can be extended to other states of immunosuppression exerted by the dual paradigm systemic inflammatory response syndrome/compensatory anti-inflammatory response syndrome to the fields of burn, shock and trauma is still unknown, but will deserve further study in the future.
Footnotes
Funding
This work was supported by grants from Agencia Nacional de Promoción Científica y Tecnológica (PICT-2005-38197), Consejo Nacional de Investigaciones Científicas y Técnicas (PIP-2010-2012GI; 11220090100606) and Fundación Alberto J. Roemmers.
Conflicts of interest
The authors do not have any potential conflicts of interest to declare.
Acknowledgements
We wish to thank Dr Julia Sabio y Garcia for critical reading and collaboration in redaction of the manuscript. We thank to Mrs Gabriela Camerano and Mr Héctor Costa for excellent technical assistance.
