Abstract
Periodontitis is a common infectious disease associated with increased risk for ischemic stroke though presently unclear mechanisms. In a case-control study, we investigated salivary levels of four periodontal pathogens, as well as systemic and local inflammatory markers. The population comprised 98 patients with acute ischemic stroke (mean ± SD, 68.2 ± 9.7 yrs; 45.9% women) and 100 healthy controls (69.1 ± 5.2 yrs; 47.0% women). Patients were more often edentulous and had fewer teeth than controls (13.8 ± 10.8 versus 16.6 ± 10.1). After adjusting for stroke risk factors and number of teeth, controls had higher saliva matrix metalloproteinase-8 (MMP-8), myeloperoxidase (MPO), IL-1β, Aggregatibacter actinomycetemcomitans, and serum LPS activity levels. Patients had higher serum MMP-8 and MPO, and they were more often qPCR-positive for A. actinomycetemcomitans (37.9% versus 19.0%) and for ≥3 periodontopathic species combined (50.0% versus 33.0%). We conclude that controls more often had evidence of current periodontal infection with higher periodontal pathogen amount, endotoxemia, local inflammation and tissue destruction. Stroke patients more often had evidence of end-stage periodontitis with edentulism and missing teeth. They were more often carriers of several periodontopathic pathogens in saliva, especially A. actinomycetemcomitans. Additionally, inflammatory burden may contribute to high systemic inflammation associated with elevated stroke susceptibility.
Introduction
Periodontitis is a common infectious disease in tooth-supporting tissues, which leads to chronic inflammation displayed locally 1 and systemically. 2 The symptoms of the disease include gingival bleeding, periodontal pocket formation, alveolar bone loss, and, finally—if left untreated—loss of teeth, which is considered as a sign of past periodontal disease. 3 Bacterial species associated with periodontitis typically include Gram-negative anaerobic bacteria, including Aggregatibacter actinomycetemcomitans, Porphyromas gingivalis, Prevotella intermedia and Tannerella forsythia. 4
Clinical periodontitis is associated with an increased risk for cardiovascular diseases through systemic inflammation as the most important etiopathogenetic link. 5 Potential biology underlying the association between these two diseases includes indirect and direct mechanisms. 6 In the indirect mechanism the mediators are inflammatory cytokines, which are produced by the gingival cells as host response to bacterial insult. In addition to the periodontal bacteria, 7 higher levels of host-originated enzymes, cytokines, proteases and Abs can be measured in saliva of periodontitis patients compared with periodontally healthy controls. 8 Matrix metalloproteinase (MMP)-8 or collagenase-2 is a catalytically complement endoprotease that can decisively process extracellular matrix components and non-matrix bioactive substrates causing tissue destruction and modulation of immuno-responses. MMP-8 in saliva and other oral fluids is regarded as a good biomarker of periodontitis, and in serum of systemic inflammation, 9 myeloperoxidase (MPO), by producing hypochloridic acid, can oxidatively activate latent MMP-8. 10 IL-1β can regulate the expression of MMP-8, MPO and tissue inhibitor of matrix metalloproteinase (TIMP)-1 by inflammatory and resident cells. 10
Direct mechanisms include bacteremia and infection of the vascular wall by periodontal pathogens. Among multiple bacterial species, DNA deriving from periodontal pathogens has been found in atherosclerotic plaques. 11 Through bleeding periodontal pockets, the dissemination of bacteria and their virulence factors is frequent when eating or brushing teeth. 12 Bacteremia, however, is difficult to measure, and surrogate markers, such as systemic Ab levels to periodontal pathogens and total endotoxin activity, are often used.
Association between clinical periodontitis and stroke has been found in both cross-sectional13,14 and prospective studies. 15 Several studies have reported an association between stroke and edentulism or missing teeth.16–18 However, the role of the periodontal microbiota, including etiologic bacterial species, in stroke is rarely investigated and still unclear. Even the results from serological studies are conflicting: two studies have reported a significant association with A. actinomycetemcomitans and P. gingivalis Ab levels,19,20 while one has shown an inverse association with high A. actinomycetemcomitans-leukotoxin-neutralizing Abs. 21 Increased subgingival P. gingivalis amounts have been reported in stroke patients. 22
We investigated if saliva periodontal pathogens and exposure to them are associated with the risk of acute ischemic stroke. In addition, the levels of local and systemic biomarkers of periodontitis representing endotoxemia, inflammation and tissue destruction were determined in this case-control study.
Materials and methods
Study patients
The present study population was a subsample of the ‘GenesiS’ study, an on-going case-control study on genetic, infectious and socioeconomic determinants of ischemic stroke. 23 Patients with a first-ever ischemic stroke between the age of 18 and 80 yrs living in the city of Ludwigshafen, an industrial city in the state of Rhineland-Palatinate (Germany), were consecutively asked for study participation. If they agreed, patients were examined and interviewed using a structured questionnaire. Additionally, blood samples were taken. Patients with clinical or laboratory signs of acute infection at time of stroke onset were excluded from the analysis.
Healthy control participants were selected from a 5% random sample of all inhabitants aged 18–80 yrs from the official population registry of the study area. They had to be free of previous stroke or myocardial infarction, which was ensured by self-report, review of any medical records (as provided by the patient) and results of the Ludwigshafen Stroke Study (LuSSt). ‘LuSSt’ is a prospective, on-going, population-based stroke register in the city of Ludwigshafen, which started on 1 January 2006. LuSSt includes all patients with incident stroke or transient ischemic attack. A detailed description of LuSSt has recently been published. 24 Possible control participants already registered in LuSSt were excluded. Cases and controls were matched for age (±2 yrs) and gender at a group level. Controls were examined parallel to patient recruitment if they gave informed consent to study participation. The participants were interviewed by trained nurses to collect information on smoking, number of remaining teeth and infection history, such as ‘Has a dentist diagnosed you with periodontitis?’. Between July 2010 and October 2011 patients (n = 100) and controls (n = 100) were additionally asked to provide paraffin-stimulated whole saliva samples. As recurrent stroke was retrospectively diagnosed in two patients, overall 98 patients and 100 controls were included in the present study. The study was approved by the ethics committee of the Landesärztekammer Rhineland-Palatinate.
Study definitions
Study definitions were similar to those being used in LuSSt.24,25 In brief, stroke was defined according to World Health Organization’s definition. 26 Stroke subtype classification was based on the results of brain imaging, discriminating between ischemic stroke, intracerebral hemorrhage or subarachnoid hemorrhage. Hypertension was diagnosed in case of antihypertensive medication on admission, a respective diagnosis by a physician before or if blood pressure was >140/90 mm Hg in two or more measurements >3 d after stroke. Diabetes mellitus was acknowledged in patients with a fasting blood Glc level >125 mg/dl, with anti-diabetics on admission or if a physician had previously made this diagnosis. Hypercholesterolemia was diagnosed in patients with fasting cholesterol levels >200 mg/dl or low-density lipoprotein-cholesterol >140 mg/dl or with cholesterol-lowering medication on admission. Atrial fibrillation (AF) was ascertained based on electrocardiography or Holter monitoring, or in case of a previous respective diagnosis. Current smoking was defined as current daily tobacco use (at least one cigarette, cigar or pipe). We defined coronary artery disease (CAD) as a history of myocardial infarction, coronary stenting or coronary artery bypass surgery, or a history of angina pectoris. In patients with a previous diagnosis of peripheral artery disease (PAD) or with a history of stenting or bypass surgery in the arteries of the lower limbs, or with a typical history of intermittent claudication of vascular origin PAD was diagnosed. 27 Body composition was analyzed by means of self-reported body mass index (BMI). 28
Serum and saliva determinations
Venous blood samples within 24 h of hospital admission and serum was separated by low-speed centrifugation. For saliva collection the participants chewed a piece of paraffin for 5 min and at least 2 ml of stimulated whole saliva was collected by expectoration during the same laboratory visit. Both serum and saliva samples were stored at −70℃ before analysis.
Serum IgA and IgG Ab levels were determined by multiserotype ELISA against the whole cell antigens of A. actinomycetemcomitans and P. gingivalis. 29 Two dilutions in duplicate were used for all samples and the results consisting of mean absorbances were calculated as continuous variables. The dilutions (v:v) for serum samples were 1:1500 and 1:3000 for A. actinomycetemcomitans-IgG, and 1:100 and 1:200 for A. actinomycetemcomitans-IgA and P. gingivalis-IgG and IgA. After the whole material was analyzed, the results were normalized according to the reference serum samples applied on each plate. The interassay coefficients of variation (CV%) were 5.4 and 5.8% for serum A. actinomycetemcomitans-IgA and IgG, 4.4, and 4.5% for serum P. gingivalis-IgA and IgG.
LPS activity was determined from the sera and the saliva supernatants by the Limulus amebocyte lysate assay coupled with a chromogenic substrate (HyCult Biotechnology, Uden, Netherlands). The CV% for the determinations was 6.7%. Most of the serum and saliva inflammation marker concentrations were determined by commercial ELISA kits according to the manufacturer’s instructions. The CV% for TIMP-1 (Amersham Biotrak, GE Healthcare, Buckinghamshire, UK), MPO (Immundiagnostik, Bensheim, Germany) and IL-1β (Amersham Biotrak) was 3.1, 7.7, and 7.9%, respectively. The serum MMP-8 concentrations were determined by a time-resolved immunofluorometric assay, as described previously. 30 The CV% was 7.7%. For the calculation of MMP-8/TIMP-1 ratios, the concentrations were converted to moles using molecular masses of 65 ku and 28 ku, as described previously.30,31
The saliva levels of four periodontal pathogens, A. actinomycetemcomitans, P. gingivalis, T. forsythia and P. intermedia, were determined by using single copy gene-targeted quantitative real-time PCR (qPCR), as described previously. 7 DNA was isolated from the pellets deriving from 500 µl of saliva using a ZR Fungal/Bacterial DNA KitTM (Zymo Research, Irvine, CA, USA) according to the manufacturer’s instructions. The amplifications were conducted in duplicate 25 -µl reactions using an Mx3005P Real-Time QPCR System (Stratagene, La Jolla, CA, USA), as described previously. 32 The results were analyzed using Mx3005P Real-Time qPCR System software and expressed as genomic equivalents (GE)/ml saliva.
As no clinical oral examination was available, saliva IL-1β and MMP-8 concentration, together with the qPCR results of P. gingivalis, were used to calculate the cumulative risk score (CRS) for having periodontitis. 1 IL-1β and MMP-8 results were divided into tertiles and P. gingivalis qPCR results into three groups: zero levels, below median levels and above median levels. The sub-scores were obtained by multiplication of the corresponding tertile/group values to result in 1–3, 4–9 and 10–27, which represented CRS with low (I), medium (II) and high (III) risk for having periodontitis, respectively.
Statistical analysis
In the analysis of baseline characteristics, a χ2-test was used to compare categorical data and the t-test was applied to analyze normally distributed continuous data. Continuous laboratory data were analyzed using a Wilcoxon signed-rank test. Laboratory data being significantly different between cases and controls in univariate analysis were additionally adjusted for diagnosis of hypertension, diabetes, CAD, PAD, AF, current smoking and number of teeth using multivariate logistic regression analysis. Not normally distributed parameters were log-transformed prior to multivariate analysis. Logistic regression analysis was used to calculate the odds ratio (OR) and 95% confidence interval (CI) for ischemic stroke. In multivariate logistic regression analysis, results were adjusted for aforementioned vascular risk factors and number of teeth. Spearman’s rho was calculated to analyse the correlation between continuous laboratory data and number of teeth. All tests were two-sided and the level of significance was set to 5%. Data were analyzed using SAS 9.1.3.
Results
Baseline characteristics.
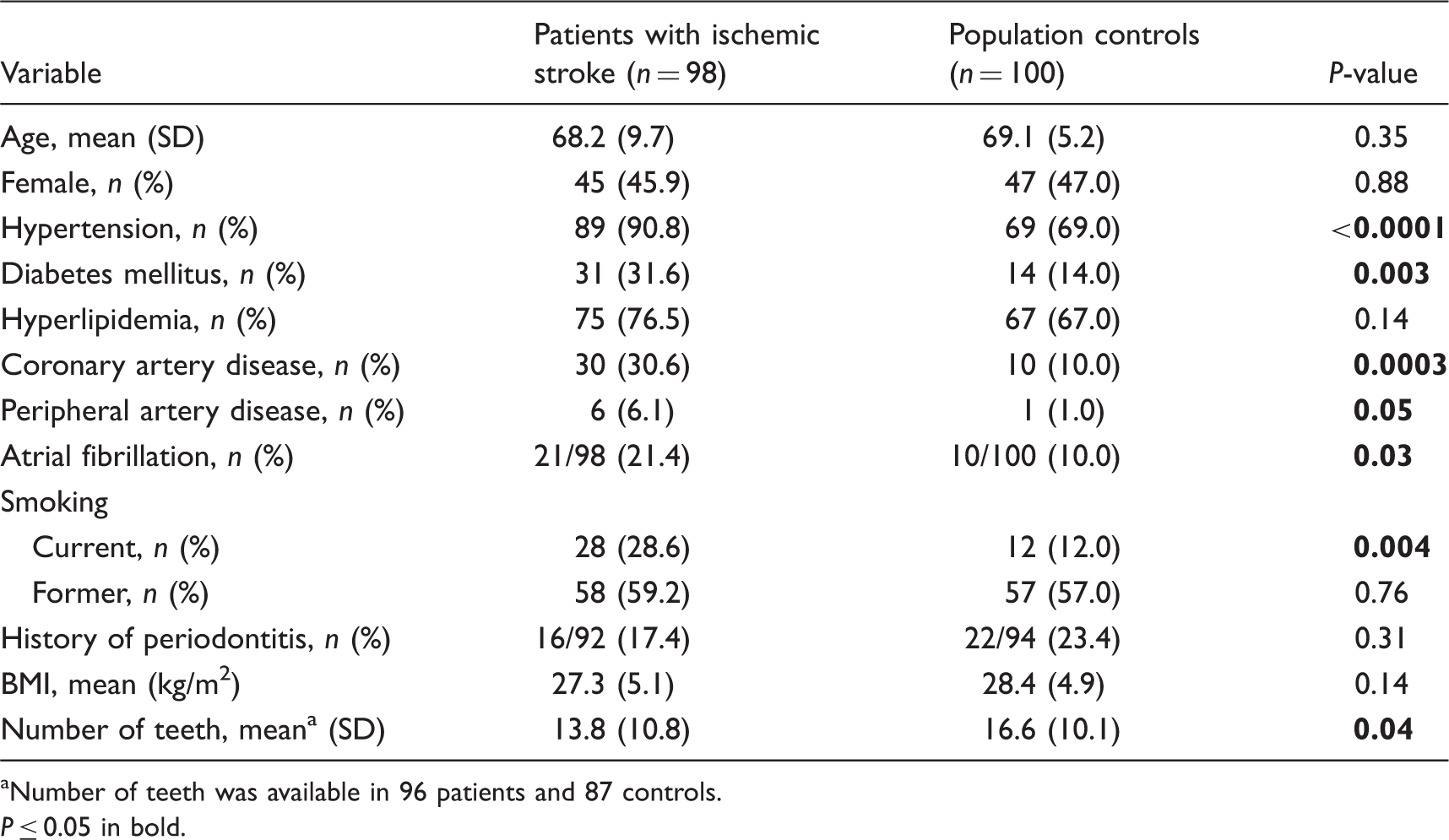
Number of teeth was available in 96 patients and 87 controls. P ≤ 0.05 in bold.
Seropositivity and Ab levels for periodontal pathogens.
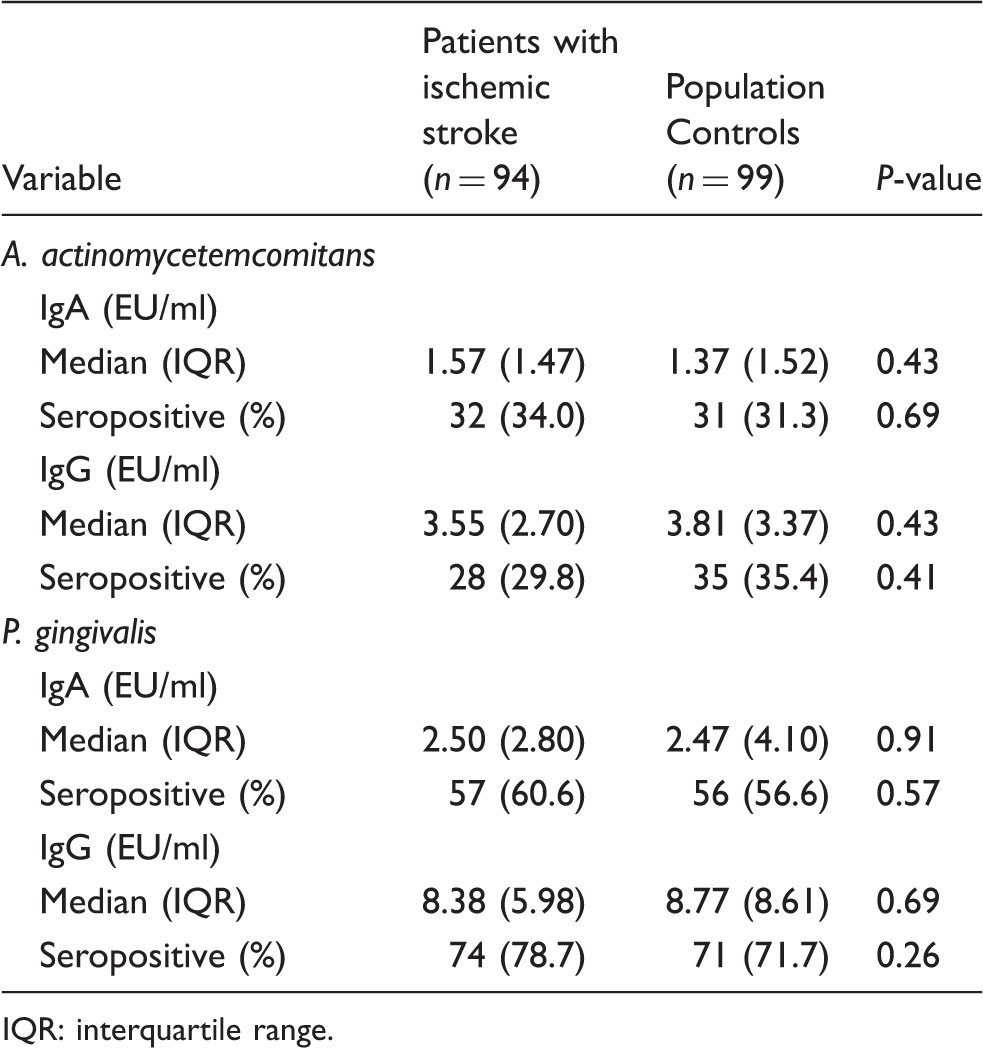
IQR: interquartile range.
Results of salivary qPCR analysis.
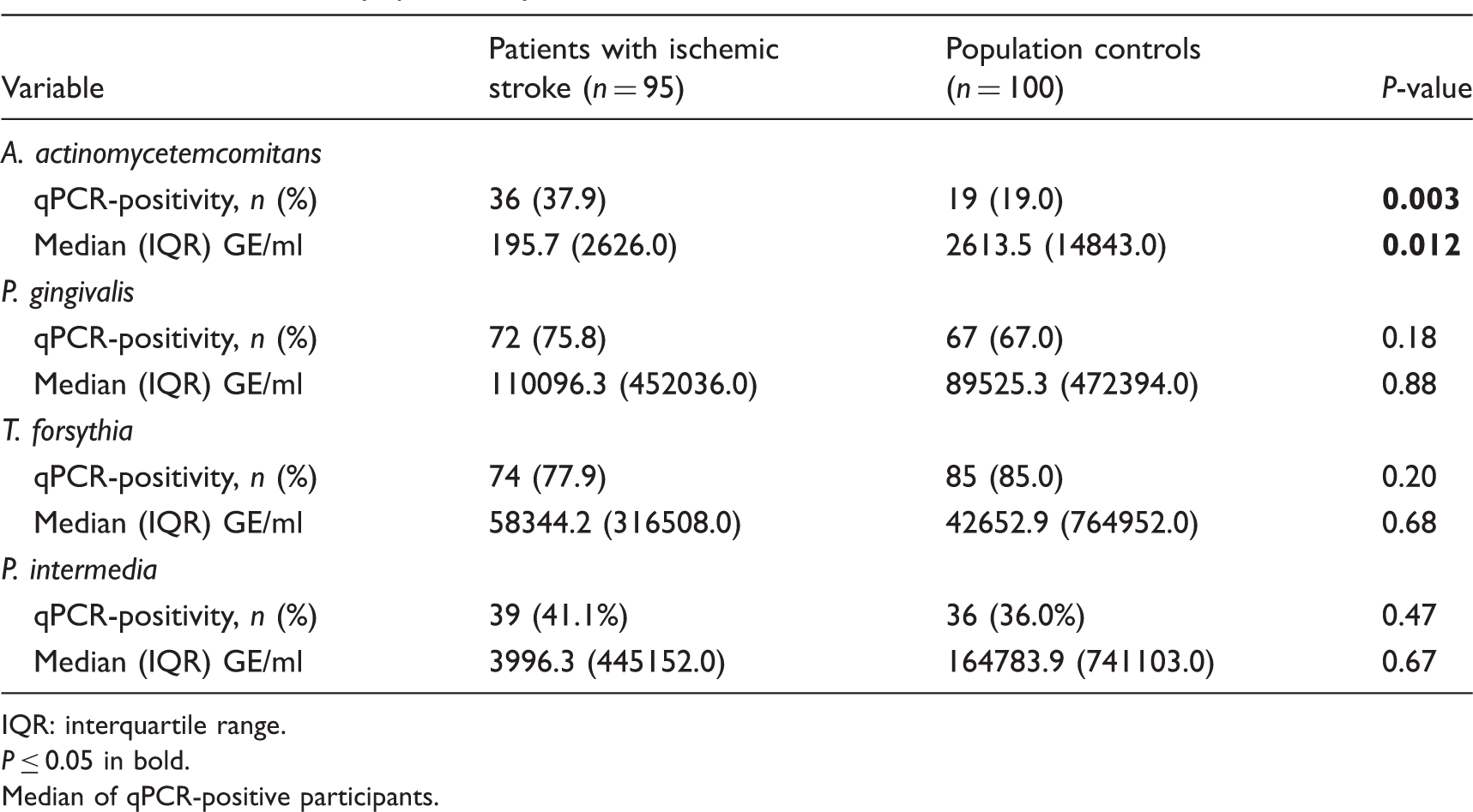
IQR: interquartile range.
P ≤ 0.05 in bold.
Median of qPCR-positive participants.
Markers of inflammation in patients and controls.
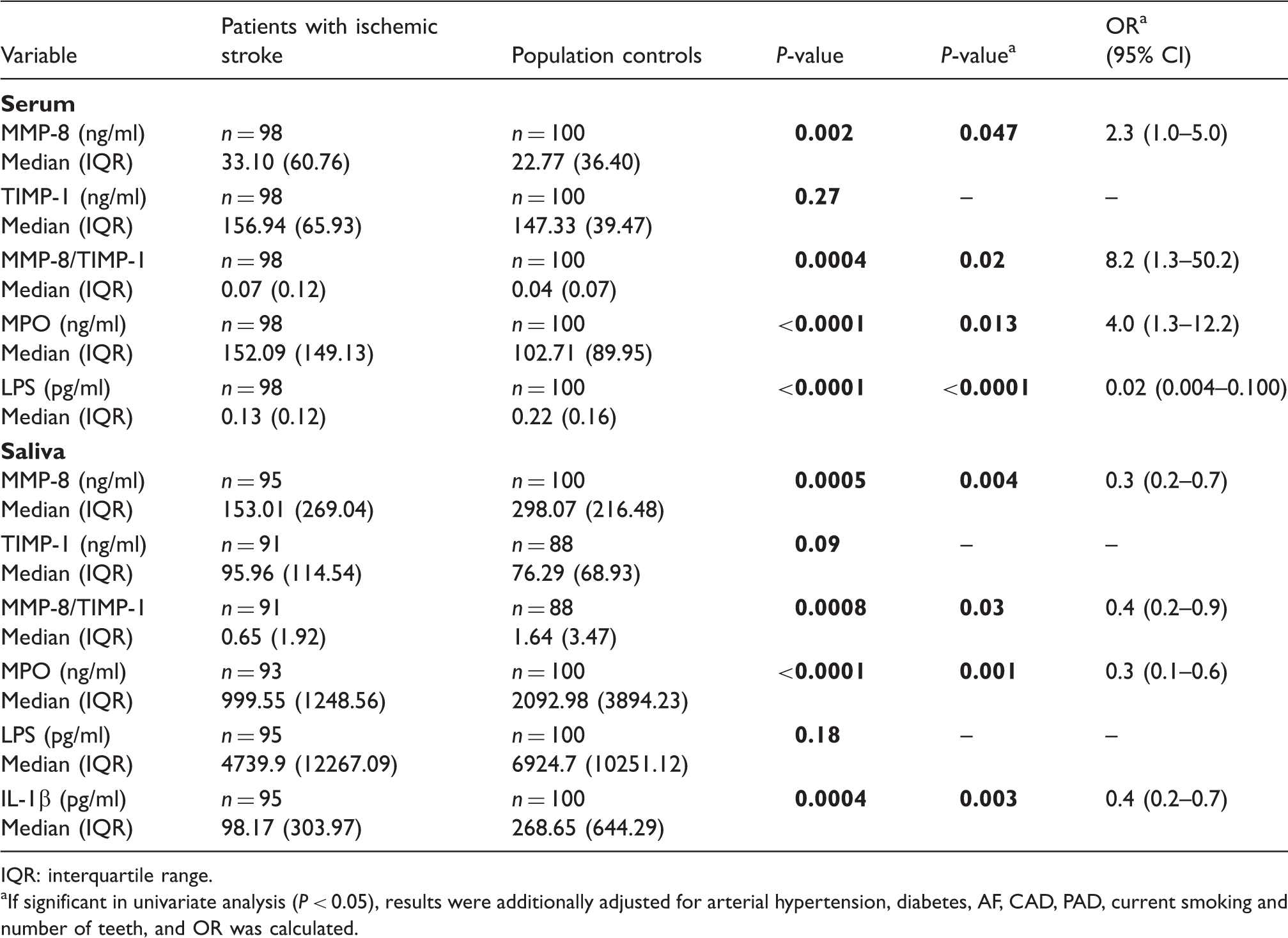
IQR: interquartile range.
If significant in univariate analysis (P < 0.05), results were additionally adjusted for arterial hypertension, diabetes, AF, CAD, PAD, current smoking and number of teeth, and OR was calculated.
CRS score for risk of having periodontitis in patients and controls.
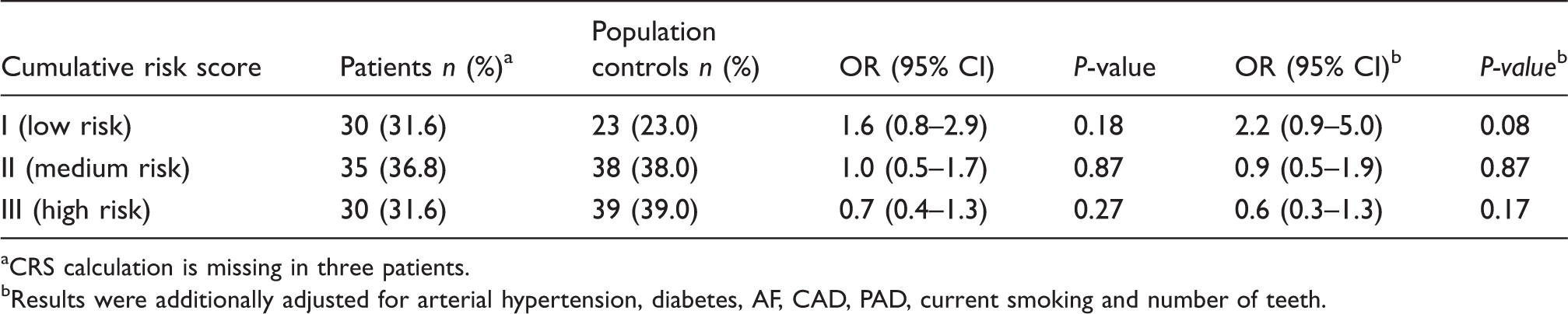
CRS calculation is missing in three patients. bResults were additionally adjusted for arterial hypertension, diabetes, AF, CAD, PAD, current smoking and number of teeth.
Discussion
The present case-control study investigates the association of periodontal pathogens, as well as systemic and local inflammatory markers in acute ischemic stroke. The study indicates that stroke patients more often have signs of past periodontal disease, namely edentulism and missing teeth, whereas control participants may more frequently suffer from current periodontal infection with endotoxemia and high biomarker levels of local inflammation, tissue destruction and periodontal pathogens. However, presence of A. actinomycetemcomitans alone, as well as multiple positivity for periodontal pathogens in saliva and high systemic inflammation, was associated with ischemic stroke. To the best of our knowledge, this is the first study on periodontitis in ischemic stroke patients analyzing periodontal pathogens by qPCR and Ab determinations, as well as markers of inflammation, both in serum and saliva. As study participants are well characterized, we were able to adjust for important confounders.
In the absence of clinical periodontal examination, we used saliva biomarkers to estimate the risk of having periodontitis. The selected periodontal pathogens were among the Gram-negative species that, according to the present consensus, are associated with periodontitis. 4 They all are members of the vast oral microbiota and usually present in higher amounts in periodontitis compared with periodontal health. 7
In the present study only the amount of A. actinomycetemcomitans among participants differed between patients and controls in the qPCR analysis; the median level was 13-fold higher in the controls. Also, as judged by other saliva biomarkers, IL-1β, MMP-8 and MPO, current periodontal infection was more evident in the controls. Notably, a positive correlation between saliva biomarkers and the number of teeth could be detected in our study. Thus, we adjusted for the number of teeth in multivariate analysis of biomarkers and CRS. Based on the CRS, there was a non-significant trend towards a higher risk of having periodontitis in controls. As periodontitis is characterized by dynamic states of microbial, inflammation and tissue degradation peaks, combining biomarkers of these events may reveal periodontitis more accurately than any of the biomarkers alone.1,33 The common pathogens associated with periodontitis, A. actinomycetemcomitans and P. gingivalis, can efficiently induce production of neutrophil-derived MPO and activation of MMP-8, thus inducing the bacteria–host destruction cascade. 34 High saliva MPO and MMP-8 concentrations observed in the controls suggest activation of the cascade and current periodontal tissue destruction.
Furthermore, the controls had higher systemic levels of LPS activity. The most probable sources of LPS in addition to Gram-negative bacterial infection are the gut and the oral cavity. It has been suggested that endotoxemia may derive from periodontitis and the access of bacteria or their parts to circulation through bleeding gums. 35 The median saliva LPS activities in the controls were also higher, although not significantly, suggesting that the endotoxemia was partly derived from the oral microbiota. However, the Limulus assay used for the determinations is not specific to any bacterial species, and strong conclusions on the origin of the serum LPS activity cannot be drawn. Nevertheless, endotoxin is a potent pro-inflammatory mediator in atherosclerosis. 36
The saliva carriage of A. actinomycetemcomitans associated with stroke
The bacterium was detected twice as often among patients than among control participants. A. actinomycetemcomitans displays a wide variety of virulence factors and heterogenic strains. 37 Periodontitis is usually considered to derive from an ecological imbalance in the commensal microbiota, but periodontitis patients with A. actinomycetemcomitans may harbor one especially aggressive clone. 38 It is possible that the patients of our study were infected early in life and represented the end stage of the disease; 41% of the patients had ≤10 teeth; the corresponding number for the controls was 26%. This hypothesis is supported by studies on serum anti-periodontal pathogen Abs in ischemic stroke. Similar to our results, a recent case-control study could not detect any differences between patients and controls in A. actinomycetemcomitans, P. gingivalis and P. intermedia Abs. 39 The mean age (SD) of patients was 71.3 (10.7) yrs, which was similar as in the present study, 68.2 (9.7) yrs, but notably higher compared with our earlier prospective study where IgA-seropositivity to A. actinomycetemcomitans was associated with increased stroke risk in patients aged 55.7 (5.7) yrs at baseline. 19 In addition to the association between stroke and A. actinomycetemcomitans positivity, carriage to multiple periodontal pathogens was associated with the risk of stroke. The idea of a ‘periodontal infectious burden’ being associated with cerebrovascular and cardiovascular diseases may arise and should be a subject of further studies.
Whereas saliva markers of inflammation were significantly higher in controls, higher serum concentrations of MMP-8 and MPO were detected in patients. Results were controlled for cerebrovascular risk factors, as they have been shown to significantly influence markers of inflammation. 40 Differences in inflammatory markers might derive from systemic inflammatory response to acute ischemic stroke, as shown previously. 41 Further studies might focus on MMP-8 and MPO in the course of ischemic stroke. In addition to post-stroke alteration, levels of inflammatory markers might also represent an elevated systemic inflammatory profile prior to stroke. 42 It is generally accepted that elevation of inflammatory markers (e.g. CRP, IL-6) is associated with increased stroke susceptibility. 42 Both, acute and chronic infections are among the conditions leading towards such increase in stroke risk, possibly via elevation of inflammatory markers.14,43,44 Although MMP-8 is mainly an indicator of current periodontal infection, its association with other infectious diseases and its role as a biomarker for cerebrovascular diseases should be of further interest.
The main limitation of our study is the absence of clinical oral examination, the gold standard of diagnosing periodontitis. Both information on ‘history of periodontitis’ and ‘number of remaining teeth’ was based on self-reporting in the interviews by trained research nurses. Recall bias may lead to under-reporting of the diagnosis of periodontitis. Number of teeth may also be challenging to self-report owing to possible radices, impacted teeth, implants, and fixed or removable prosthetics. It is known that the most common reason for tooth loss before middle-age is caries, whereas in elderly periodontal disease becomes the main cause for tooth extraction. 45 However, in the absence of dental charts, this information can not be obtained retrospectively. The number of teeth, however, is a major determinant for any biomarker of periodontitis and it should be taken into account. Furthermore, the study design was primarily explorative. As data on serum and salivary inflammatory markers and periodontal pathogens in stroke patients are rare, we did not perform sample size calculation.
Conclusion
The serum and saliva biomarkers indicate a higher prevalence of current periodontal infection among control participants, but end-stage periodontitis among stroke patients. The number of teeth is an important confounder, which needs to be controlled for in future studies. Elevated occurence of A. actinomycetemcomitans and proof of elevated periodontal pathogen burden among stroke patients suggests an effect of early infection (especially A. actinomycetemcomitans infection) on stroke risk. Higher serum inflammatory marker concentrations may derive from acute phase response following ischemic stroke, but they might also result from other acute or chronic infections leading to an increased risk of ischemic stroke.
Footnotes
Funding
Study was supported by a grant from the Deutsche Forschungsgemeinschaft (DFG; GR1102/6-1), the Paulo foundation (PJP), the Sigrid Juselius foundation (PJP), the University Central Hospital Research Foundation (TS; TT), and the Academy of Finland (PJP)
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
