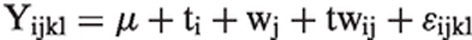Abstract
The study evaluated the effects of repeated oral exposure to LPS and lipoteichoic acid (LTA) on immune responses of dairy cows. Thirty pregnant Holstein cows were randomly assigned to two treatment groups. Cows received orally either 2 ml of 0.85% sterile saline solution (control group), or 2 ml of sterile saline solution containing three doses of LPS from Escherichia coli 0111 : B4 along with a flat dose of LTA from Bacillus subtilis. Blood and saliva samples were collected and analyzed for serum amyloid A (SAA); LPS-binding protein (LBP); anti-LPS plasma IgA, IgG and IgM; TNF-α; and IL-1. Results showed greater concentrations of IgA in the saliva of treated cows compared with the controls (P < 0.01). Treated cows had lower plasma concentrations of anti-LPS IgA, IgG and IgM Abs, and TNF-α than the controls (P < 0.05). There was a tendency for the concentrations of plasma LBP (P = 0.06) and haptoglobin (P = 0.10) to be lesser in the treatment group, although no differences were found in the concentration of plasma SAA and IL-1 (P > 0.10). Overall, the results of this study indicate that repeated oral administration with LPS and LTA stimulates innate and humoral immune responses in periparturient dairy cows.
Introduction
Dairy cows experience a state of immunosuppression during the periparturient period, which increases their susceptibility to various peripartal diseases. Furthermore, this attenuated immune function during the peripartum period increases the susceptibility to mastitis postpartum, which causes significant production loses to the dairy industry. 1 The reason(s) behind immunosupression in periparturient dairy cows is not well understood yet; however, several lines of evidence indicate that immune responsiveness decreases gradually in the prepartum period and reaches its lowest level immediately before parturition.2,3 There is a need to stimulate the immune competence of cows during the periparturient period because health status during this period is critical for the health and productivity of cows during the whole lactation.3,4
Early postpartum, cows encounter various immunogenic substances, such as LPS and lipoteichoic acid (LTA), which are important components of the Gram-negative and Gram-positive bacterial cell wall, respectively. 5 Additional exposure of mucosal layers to LPS and LTA occurs owing to accumulation of cell-free LPS or LTA in the rumen when cows are switched from a roughage-rich to a grain-rich diet6,7 at the onset of lactation. However, the virulence properties of those immunogenic compounds are highly influenced by the sudden switch in diet postpartum. For example, there is an abundance of Escherichia coli with a highly virulent conical-shape LPS moiety during high-grain feeding immediately after parturition, and it strongly binds to LPS-binding protein (LBP) to induce a high inflammatory response. 8 Furthermore, the E. coli infection paves the way for infection from other pathogens, and interacts with lactic acid-resistant bacteria, such as Bacillus subtilis, which produce LTA as a major immunostimulatory component. 9
Mucosal surfaces comprise the first port of entry for bacterial endotoxins and LTA. 10 Thus, developing a prophylactic treatment targeting the mucosal immune responses by co-stimulating with LPS and LTA might be of great interest. Mucosal immunity is primarily mediated by Abs of the IgA class, which is by far the most prominent isotype synthesized by the immune system. 11 The mucosal immune responses have been shown to strongly depend on the production of secretory IgA (sIgA) molecules.11,12 In fact, the interest in inducing mucosal immunity and, most importantly, in administering immunoAgs on the mucosal layers has increased recently. Furthermore, there is strong evidence that the mucosal sites of immunogen challenge influence the location of the IgA response. A recent report demonstrated that oral immunization induces protective mucosal immune responses, but suppresses systemic immunologic reactivity. 13 This kind of immunization stimulates secretory IgA responses at distant mucosal layers and develops sub-populations of regulatory T lymphocytes within the gut-associated lymphoid tissues, which inhibit the subsequent systemic responses to the same Ag. 14 Recently, we showed that oral treatment of cows with LPS was able to influence the pro-inflammatory responses and modulate production of anti-LPS IgM Abs in the plasma, as well as metabolic health status. 15 In addition, intra-mammary administration of LPS protected cows against experimental E. coli mastitis. 16
Despite tremendous progress in the study of the role of LPS on animal health only a few investigations have addressed the role of LTA on the etiopathogenesis of periparturient diseases of dairy cows. A recent study examined the effects of LTA from Staphylococcus aureus LTA, on initiation of clinical mastitis at the dose of 100 µg/quarter, and a subclinical inflammatory response at 10 µg/quarter. 17 Interestingly, another study showed that a challenge with E. coli-derived LPS and LTA from S. aureus induced a complex and robust immune response to pathogens. 18 Recent data from our work also showed enhanced IgA responses in the vaginal mucus of cows when they were orally challenged with LPS and LTA (unpublished data).
To the best of our knowledge, there are no reports dealing with cow’s responses to oral administration of LPS and LTA, as a prophylactic strategy against deleterious effects of these bacterial endotoxins. Therefore, we hypothesized that repeated oral exposure of the periparturient dairy cows to increasing doses of LPS and a flat dose of LTA before parturition might improve their innate and humoral immune responses against those toxic cell wall bacterial components and improve subsequent health status of dairy cows.
Materials and methods
Cows and experimental design
Thirty pregnant multiparous and primiparous Holstein dairy cows with body mass (BM) of 720 ± 30 and 600 ± 20 kg (mean ± SD), respectively, were blocked by parity, milk production, body condition score, disease susceptibility from previous year and the anticipated day of calving. Fifteen cows (10 multiparous and five primiparous) were randomly allocated to each group at 28 d before the expected day of parturition. Cows were orally administered either 2 ml of sterile saline solution (CTR) or 2 ml of sterile saline solution containing LPS (TRT) from E. coli strain 0111 : B4 at three increasing concentrations as follows: (i) 0.01 µg/kg BM once on d –28; (ii) 0.05 µg/kg BM twice on d–25 and –21; (3) 0.1 µg/kg BM twice on d –18 and –14 along with a flat dose of LTA from B. subtilis (i.e. 120 µg/animal) for 3 consecutive wks on the same days as LPS treatments. The initial crystalline E. coli LPS (from E. coli strain 0111 : B4) and B. subtilis LTA (both from Sigma-Aldrich Canada, Oakville, ON, Canada) containing 10 mg of purified LPS and LTA, were then dissolved in 10 ml of doubly-distilled water each, as suggested by the manufacturer, and stored in a refrigerator at 4℃. For administration to the animals, the daily dose was dissolved in 2 ml of saline and then introduced into the oral cavity of the cow using a disposable 5-ml syringe (Becton, Dickinson, Franklin Lakes, NJ, USA). Similarly, the same amount of carrier (i.e. 2 ml sterile saline; Sigma-Aldrich Canada) was orally administered to all cows in the control group. Doses of LPS used were based on previous research conducted with dairy cows by our team and on clinical and pathological responses to those doses, 15 whereas a dose study was conducted to determine the safe clinical dose of oral LTA to be used. 19 The lowest dose, 0.01 µg/kg BM, was chosen because previous experiments have shown minimal changes in the metabolism of dairy cows at this concentration, 15 whereas the highest dose was also selected owing to a maximum host response at this dose observed previously. 15 Furthermore, the induction of endotoxin tolerance is dose-dependent, and LPS is more effective in inducing endotoxin tolerance with increasing doses than LTA. The flat dose of LTA was selected based on a dose study conducted by us, indicating no effects to cow’s temperature, respiration rate, rumen contraction rate and feed intake. 19
The experiment lasted for 8 wks (i.e. 4 wks before and 4 wks after parturition) and cows were housed in tie stalls (122 × 200 cm) with free access to water throughout the experiment. Shortly before parturition, cows were transferred to the maternity pens (6.7 × 4.4 m) and returned to their stalls on the next day of parturition. Animals were fed once daily at 08:00 a.m. and milked twice at 05:00 a.m. and 15:30 p.m. in their stalls. All cows were fed the same close-up diet starting at 3 wks before the expected day of parturition. The close-up diet is usually offered to the dairy cows when they are close to parturition and contained approximately 20% concentrate on dry matter basis. After parturition, cows were gradually switched during the first 7 d to a fresh-lactation diet with higher proportion of grain (up to 50% on dry matter basis) to meet the energy demands for high milk production. All diets were formulated to meet or exceed the nutrient requirements of dry and early lactating cows, as per National Research Council guidelines. 20 Daily ration was offered as total mixed ration for ad libitum intake to allow approximately 10% feed refusals throughout the experiment. All experimental procedures were approved by the University of Alberta Animal Care and Use Committee for Livestock, and animals were cared for in accordance with the guidelines of the Canadian Council on Animal Care. 21 Veterinary supervision was provided to the animals throughout the experiment.
Sample collection
Blood samples were collected from the coccygeal vein on d –28, –25, –21, –14, –7, +7, +14, +21 and +28 around parturition for plasma haptoglobin, and once per week on d –28, –7, +7,and +28 around parturition for plasma serum amyloid A (SAA); LBP; anti-LPS plasma IgA, IgG, IgM; TNF-α; and IL-1. Blood samples of approximately 5–8 ml were collected in 10-ml glass tubes (BD Vacutainers; Becton Dickinson) with no additive. Blood samples were put immediately on ice, and centrifuged within 20 min (Rotanta 460 R; Hettich Zentrifugan, Tuttlingen, Germany) at 3000 g and 4℃ for 20 min. The plasma was separated and stored at –20℃ until analysis. No agitation or overreaction from cows was observed during the blood withdrawal. Feed intake was recorded daily during the entire experimental period. All disease and medication history was recorded for each cow throughout the entire experimental period.
Saliva samples were collected on d –28, –7, +7 and +28 around parturition using cotton swabs inserted between the cheek and the lower jaw, along the side of the mouth towards the back teeth until the swab was soaked. For collecting saliva, head movement of the animal was restrained using conventional restraining techniques (e.g. rope halter and held by a person). After collection, saliva samples were extracted from the cotton gauze using a 60-ml plastic syringe (Becton Dickinson) and then placed in a small sterile container, which was sealed securely and stored at –86℃ until analysis for total IgA. No preservatives or additional material were added to the saliva samples. Before assay, samples were centrifuged (Rotanta 460 R; Hettich Zentrifugan) at 1000 g and 4℃ for 20 min to remove any particulates.
Sample analyses
Concentrations of anti-LPS core IgA, IgG and IgM in the plasma were measured using a commercially-available ELISA kit EndoCab (HK504; Cell Sciences, Canton, MA, USA), using the methods described previously by Zebeli et al. 22 In brief, Abs directed against the core structure of endotoxin (EndoCab) are cross-reactive against most types of LPS, and are measured using a commercial sandwich EndoCab ELISA kit, which is a solid-phase ELISA with a working time of 2.5 h. The color developed was proportional to the amount of anti-endotoxin core Abs present in the sample. The absorbance was measured at 450 nm with a spectrophotometer (Spectramax 190; Molecular Devices, Sunnyvale, CA, USA). The minimum detection concentrations of IgG, IgM and IgA EndoCab Abs were 0.0125 GMU/ml, 0.055 MMU/ml, and 0.156 AMU/ml, respectively. The inter- and intra-assay coefficient of variations (CV) for the IgA, IgG and IgM anti-LPS Abs analysis were less than 10%.
Concentrations of haptoglobin in the plasma were measured with an ELISA kit provided by Tridelta Development (Greystones C., Wicklow, Ireland). According to the manufacturer, the minimum detection limit of the assay was 0.25 ng/ml, as defined by the linear range of the standard curves. All samples were tested in duplicate, and the OD was measured at 630 nm on a microplate spectrophotometer (Spectramax 190; Molecular Devices). The CV for the inter- and intra-assay analysis was less than 10% for all the samples tested.
Concentrations of LBP in the plasma were quantified with a commercially-available ELISA kit (Cell Sciences, Norwood, MA, USA). The Ab coated in the walls cross-reacted with bovine LBP. Plasma samples were initially diluted 1 : 1000, and samples with OD values lower than the range of the standard curve were tested with a lower dilution (1 : 500). The minimum detection limit of the assay was 5 ng/ml, as calculated from a standard curve of the known LBP values in human plasma. Samples were tested in duplicate, and the OD was measured at 450 nm on a microplate spectrophotometer (Spectramax 190; Molecular Devices). Inter- and intra-assay CV was less than 10% for this analysis.
Concentrations of SAA in plasma were determined by commercially-available ELISA kits (Tridelta Development) with monoclonal Abs specific for SAA coated on the walls of the microtiter strips originally described by McDonald et al. 23 Samples were initially diluted 1 : 500 and if some of the samples had OD values below the range of the standard curve they were reanalyzed in lower dilutions. The inter- and intra-assay CV for the SAA analysis was less than 10%. All samples were tested in duplicate and the OD values were read on a microplate spectrophotometer (Spectramax 190; Molecular Devices) at 450 nm. The minimum detection limit of the assay was 18.8 ng/ml.
Concentrations of TNF-α in the plasma were measured using commercially-available bovine ELISA kits (Bethyl Laboratories, Montgomery, TX, USA). Diluted samples and standards (100 μl) were incubated in the coated plate, followed by washing and incubation with 100 μl of detection Ab and HRP substrate for 1 h and 30 min, respectively. The incubation with each of these reagents was followed by washing four times. The detection Ab solution cross-reacts with the Abs attached to coated wells. The addition of 100 μl of 3,3′5,5′-tetramethylbenzidine solution allowed the enzymatic color reaction, and the color developed was proportional to the amount of anti-TNF-α Abs present in the sample. The absorbance was measured at 450 nm with a spectrophotometer (Spectramax 190; Molecular Devices). The minimum detection limit of TNF-α was 0.078 ng/ml. The inter- and intra-assay CV for the analysis of TNF-α was less than 10%.
Plasma IL-1 was determined by commercially-available bovine ELISA kits (Cusabio Biotech, Newark, NJ, USA). The assay is based on the competitive inhibition of an enzyme immunoassay technique. An Ab specific to IL-1 was pre-coated by the manufacturer onto microplate wells, and standards and samples were incubated with biotin-conjugated IL-1, which leads to a competitive inhibition reaction between IL-1 (standards or samples) and biotin-conjugated IL-1 with the pre-coated Ab specific for IL-1. Then, avidin conjugated to HRP was added to each microplate well and incubated after the substrate solution was added to the wells. The color developed was opposite to the amount of IL-1 in the sample. Further development of color was stopped by adding stop solution, and the intensity of the color was measured with a spectrophotometer (Spectramax 190; Molecular Devices) at 450 nm. The minimum detectable concentration of bovine IL-1 was at <125 pg/ml. The inter- and intra-assay CV for the IL-1 analysis was less than 10%.
Concentrations of total IgA in the saliva were measured using a commercially-available bovine ELISA kit (Uscn; Life Sciences, Houston, TX, USA). The procedure involved the basic principle of a sandwich enzyme immunoassay for the quantitative measurement of IgA in bovine saliva with a working time of 4 h. The microtiter plate provided with the kit was pre-coated with Abs specific to the IgA. The standards and samples were then added to appropriate microtiter plate wells with a biotin-conjugated Ab preparation specific for IgA, and then avidin conjugated to HRP was added. The color developed by the substrate was shown only in those wells which contained IgA, biotin-conjugated Ab and enzyme-conjugated avidin. The absorbance was measured at 450 nm with a spectrophotometer (Spectramax 190; Molecular Devices). The minimum detectable concentration of this assay was 0.78 ng/ml. The inter- and intra-assay CV for all the samples tested for total salivary IgA was less than 10%.
Statistical analyses
Data were analyzed using the MIXED procedure of SAS (v. 9.1.3; SAS Institute, Cary, NC, USA), as described by the following model
Results
Plasma and saliva anti-LPS immunoglobulins
Oral treatment of cows with LPS and LTA had an effect on the profile of total IgA in the saliva (Figure 1).Treated cows showed overall greater concentrations of salivary IgA than their control counterparts (P < 0.01). The factor time of sampling (P = 0.04), and treatment × wk interaction (P < 0.01) also affected salivary concentrations of IgA in this study. The control cows showed an unchanged and flat response of IgA in the saliva, whereas this variable was greater in the TRT cows starting from wk –1 onward (Figure 1).
Weekly variations of total IgA in saliva of multiparous and primiparous Holstein cows challenged with oral treatment of LPS–LTA (TRT; ▪) or saline (Control; ◊) (LSM ± SEM; n = 15; Trt = effect of treatment; Week = effect of sampling wk, Trt × Week = effect of treatment by sampling wk).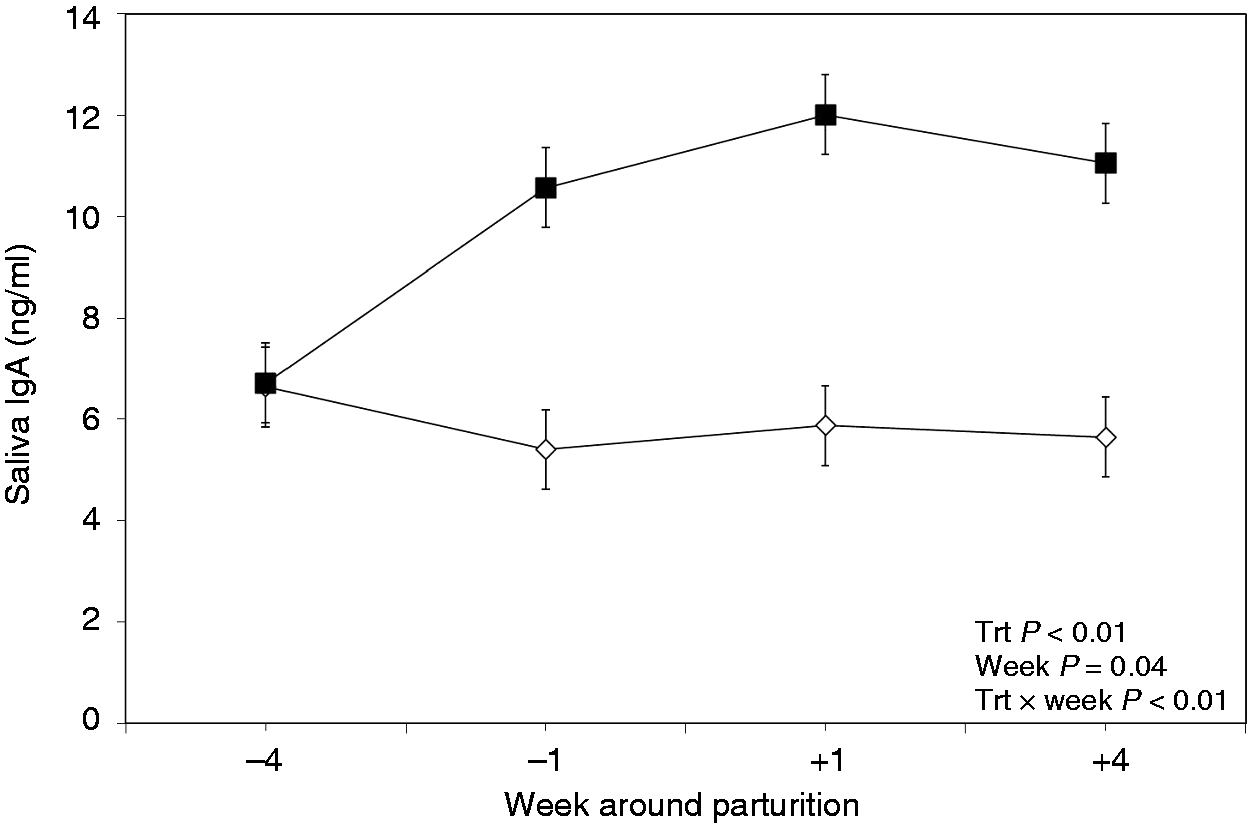
Results of the effects of oral LPS and LTA administration on plasma concentrations of anti-LPS IgA Abs are shown in Figure 2A. Results indicated differences between the groups, with the control group having greater concentrations of plasma anti-LPS IgA Abs (P < 0.01). However, the factor ‘measurement wk when blood samples were taken’ did not show an effect on plasma IgA (P = 0.22). Furthermore, no treatment × wk interaction was observed between the two treated groups regarding plasma anti-LPS IgA Abs (P = 0.74).
Weekly variations of anti-LPS IgA (A), IgG (B) and IgM (C) in the plasma of multiparous and primiparous Holstein cows challenged with oral treatment of LPS–LTA (TRT; ▪) or saline (Control; ◊) (LSM ± SEM; n = 15; Trt = effect of treatment; Week = effect of sampling wk, Trt × Week = effect of treatment by sampling wk). AMU: median units of IgA; GMU: median units of IgG; MMU: median units of IgM.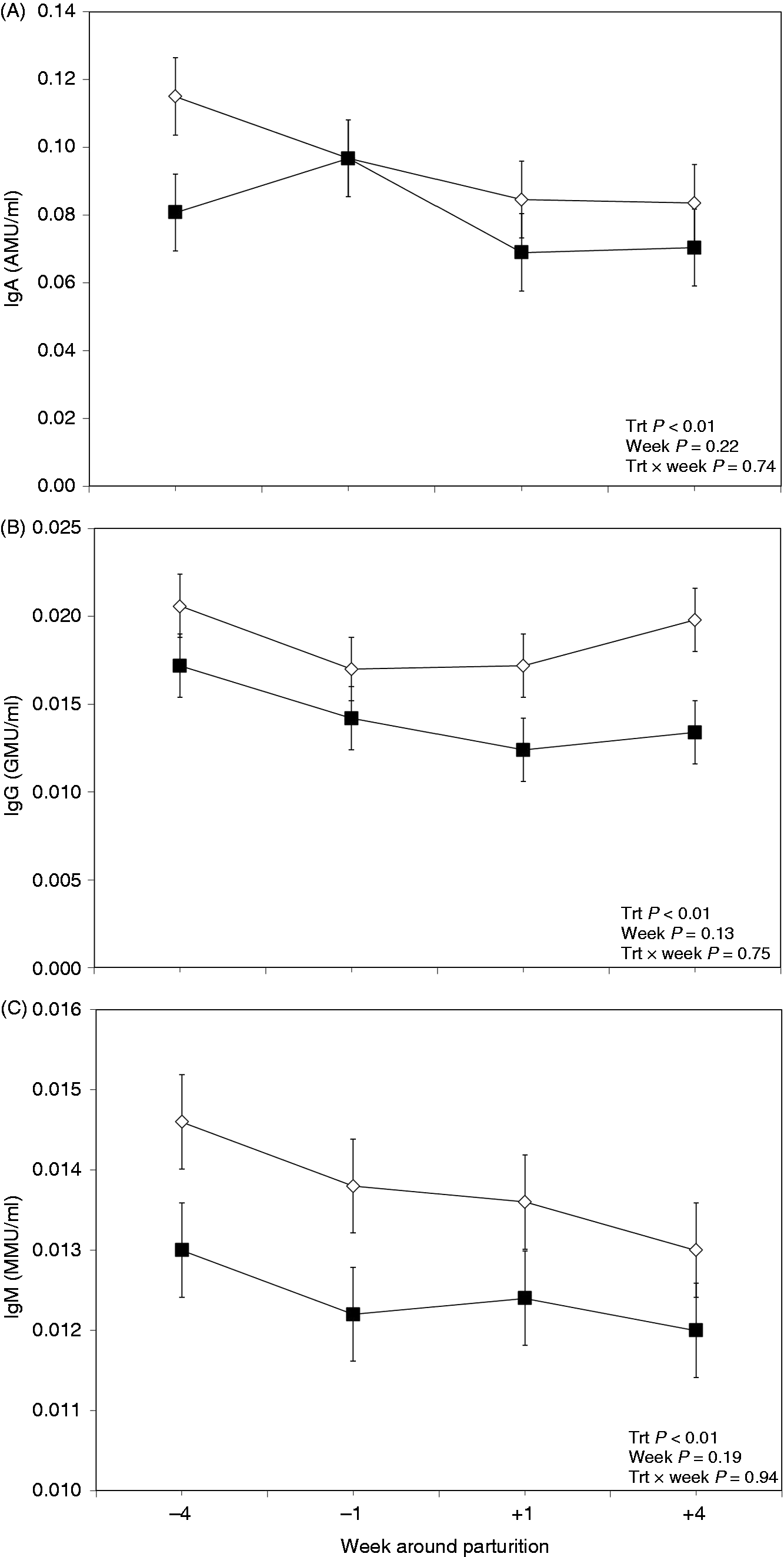
The group of cows treated orally with LPS and LTA had lower concentrations of plasma anti-LPS IgG Abs (P < 0.01; Figure 2B). However, no effect of wk (P = 0.13), or treatment × wk interaction was obtained regarding plasma anti-LPS IgG Abs (P = 0.75).
Concentrations of anti-LPS IgM Abs in the plasma were different between the treatment groups (P < 0.01; Figure 2C). The group of cows treated orally with saline had greater concentrations of plasma anti-LPS IgM Abs than the treated group. There was no effect of sampling wk (P = 0.19) or treatment × wk interaction with respect to plasma endotoxin IgM Abs (P = 0.94).
Plasma acute phase proteins
Treating dairy cows orally with LPS and LTA showed a tendency for lower concentrations of haptoglobin in the plasma (P = 0.10; Figure 3). Furthermore, no interaction between treatment and wk of sampling was evidenced for the concentrations of haptoglobin in the plasma (P = 0.27). Additionally, measurement wk did have an effect on plasma haptoglobin (P < 0.01).
Weekly variations of haptoglobin in plasma of multiparous and primiparous Holstein cows challenged with oral treatment of LPS–LTA (TRT; ▪) or saline (Control; ◊) (LSM ± SEM; n = 15; Trt = effect of treatment; Week = effect of sampling wk, Trt × Week = effect of treatment by sampling wk).
No differences between the two treatment groups were observed regarding concentration of SAA in the plasma (P = 0.17; Figure 4A), although sampling wk alone had an effect on plasma SAA (P < 0.01). However, data indicated no treatment × measurement wk interaction for plasma SAA (P = 0.60).
Weekly variations of SAA (A) and LBP (B) in plasma of multiparous and primiparous Holstein cows challenged with oral treatment of LPS–LTA (TRT; ▪) or saline (Control; ◊) (LSM ± SEM; n = 15; Trt = effect of treatment; Week = effect of sampling wk, Trt × Week = effect of treatment by sampling wk).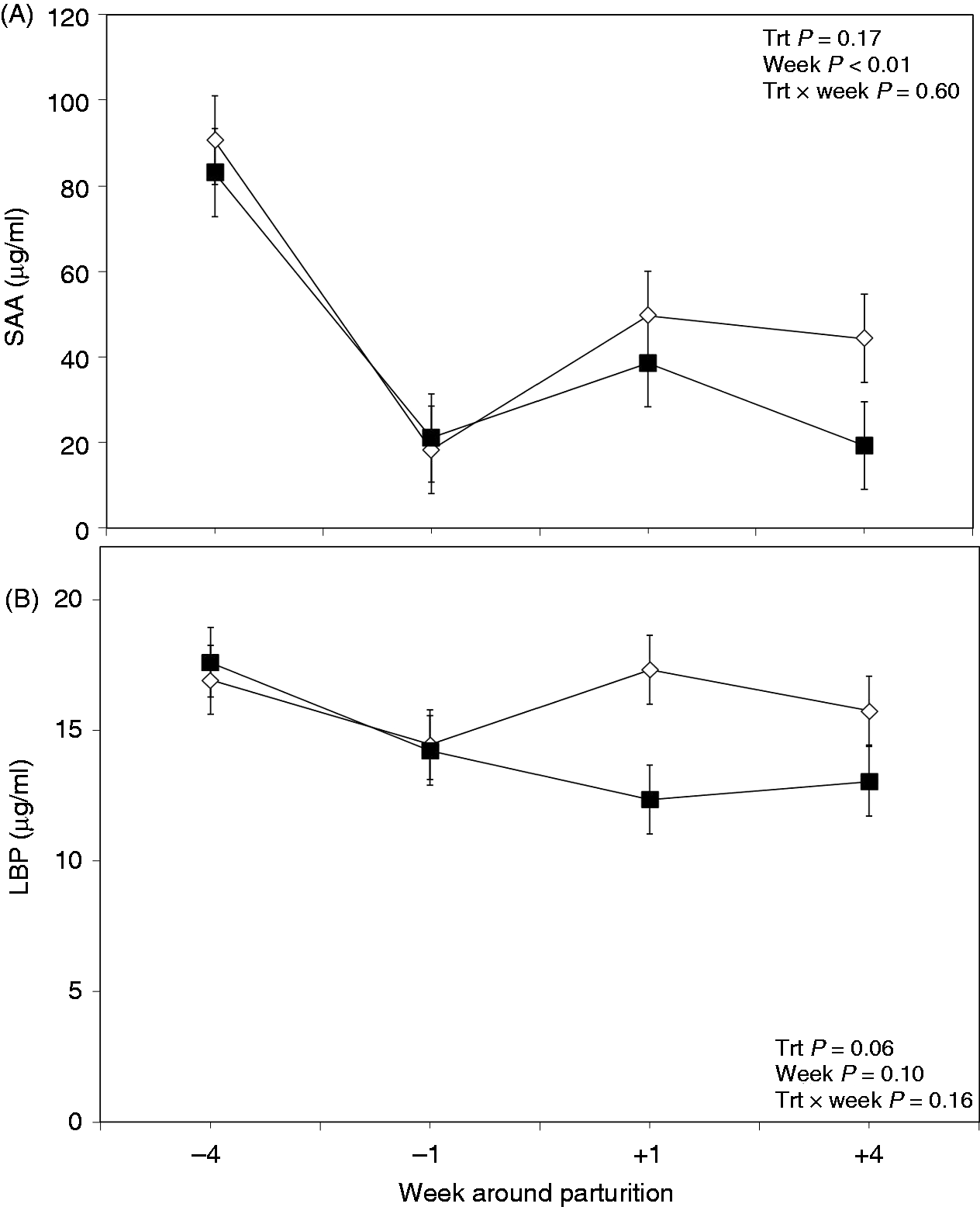
Data also demonstrated a tendency for the concentrations of LBP in the plasma to be lower in the treatment group (P = 0.06; Figure 4B). Moreover, results showed a tendency for the effect of wk of sampling on the concentrations of LBP (P = 0.10), although no treatment × wk interaction was obtained for the plasma LBP (P = 0.16).
Plasma cytokines
Results indicated differences for plasma TNF-α between the two treated groups, with the treatment group having lower concentrations before and after calving (P = 0.02; Figure 5A). However, no effect of wk of sampling was observed for plasma TNF-α (P = 0.87). Also, no treatment × measurement wk interaction was observed between the two treated groups regarding this variable (P = 0.89).
Weekly variations of TNF-α (A) and IL-1 (B) in plasma of multiparous and primiparous Holstein cows challenged with oral treatment of LPS–LTA (TRT; ▪) or saline (CTR; ◊) (LSM ± SEM; n = 15; Trt = effect of treatment; Week = effect of sampling wk, Trt × Week = effect of treatment by sampling wk).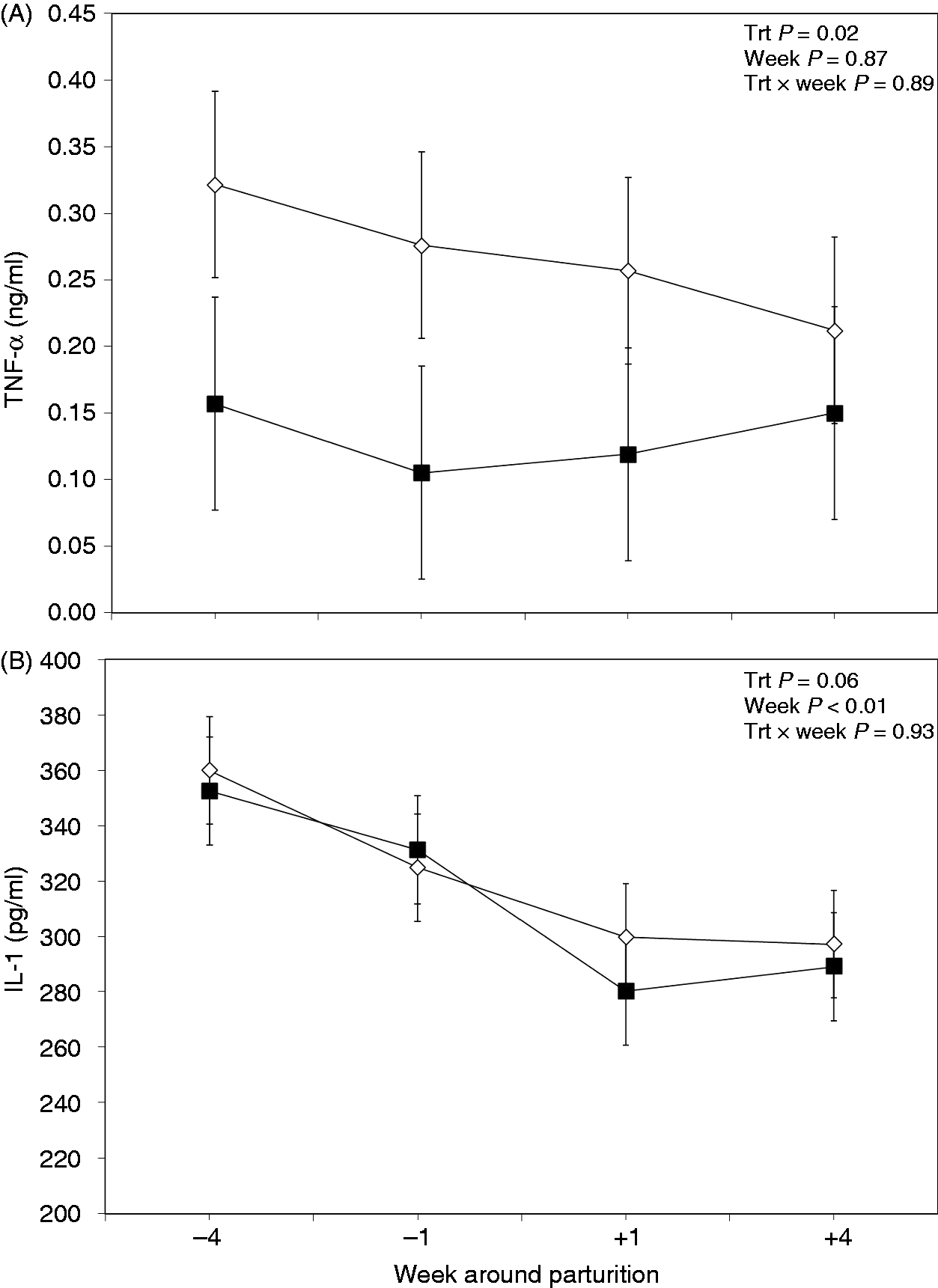
Concentration of IL-1 in the plasma did not differ between groups (P = 0.60; Figure 5B). However, the results showed an effect of time of sampling on plasma IL-1 (P < 0.01). Also, analysis of data indicated no treatment × sampling time interaction for plasma IL-1 (P = 0.93).
Discussion
The present study investigated whether repeated oral administration of LPS and LTA would affect the innate and humoral immune responses of periparturient dairy cows. Both LPS and LTA have been implicated in multiple metabolic and immune perturbations, as well as various metabolic and infectious diseases of dairy cows. 10 LPS and LTA are cell envelope components of Gram-negative and Gram-positive bacteria, respectively, with different mechanisms of action on host immune cells. LPS is known as a TLR4 ligand and LTA as a TLR2 ligand. Immune responses to those ligands are also different. 24 It is widely accepted that during the postpartum period dairy cows are suddenly exposed to increased concentrations of these immunogenic compounds—particularly LPS—and the underlying cause is the high-grain engorgement, which alters rumen microbial ecology. 25 For this reason administration of LPS and LTA was done in the oral mucosa of the dairy cows before this critical period in order to enhance the immune responsiveness of the cows by inducing Abs in the adjacent mucosal layers. Furthermore, repeated oral challenge with bacterial immunogens at different time points aimed at developing the state of mucosal immune tolerance and to lower responsiveness of the immune system when these cows are exposed to high concentrations of bacterial toxins postpartum. Indeed, data from this investigation supported our hypothesis and demonstrated modulation of several innate and humoral immune variables in dairy cows treated orally with both LPS and LTA. The mechanism(s) related to improved innate and humoral immune status of the treated cows deserves further investigation; however, the findings are discussed in more detail below.
The most important finding of this research was that total salivary IgA Abs were greater in the treated cows during the whole post-treatment measurement period. It is well known that oral immunization usually stimulates production of secretory IgA in the mucosal membranes, which are capable of inhibiting attachment of bacterial Ags to the luminal surface of the mucosal epithelia. 26 It is well known that IgA is the principal mucosal Ab class, and is synthesized by local plasma cells from the Peyer’s patches. 11 The results of the present study indicate development of better oral immunity in the LPS- and LTA-treated cows. A dichotomy exists between the systemic and mucosal immune responses, 27 and evidence is accumulating that mucosal immunity consists of stimulation of secretory IgA responses at different mucosal sites and inhibition of systemic responses, including plasma IgA production. 28 This might be the case in the present study, as circulatory IgA was lower in the treated cows. Thus, it might be speculated that following the oral administration of LPS and LTA, a protective immunity associated with the induction of mucosal Abs was elicited. However, it is obvious that treatment suppressed circulatory IgA Abs by effectively controlling the entrance of bacterial toxins at the mucosal sites without involvement of systemic primary humoral response. An earlier study demonstrated that repeated exposure to oral dosing with LPS dampens the Th1-type immune responses of the gut, and promotes oral tolerance in rats. 29 In addition, it was also observed that secretory IgA Ab responses were associated with systemic suppression of the humoral and T-cell responses to that specific Ag. 30
Another interesting finding of this study was that plasma anti-LPS IgG Abs were lesser in the LPS- and LTA-treated cows. These data are in agreement with previous findings from our team indicating that transition dairy cows treated repeatedly with increasing oral doses of LPS have lower anti-LPS IgG Abs. 15 It is speculated that the phenomena involved might be related to the prevention of translocation of those bacterial toxic compounds into the systemic circulation and the loss of systemic reactivity, which might involve not only the humoral responses but also T-cell-mediated reactions in the treated cows. It has been reported that regulatory T-cells, which suppress systemic T-cell responses, are involved in systemic tolerance and prevent the exhaustion of the immune system by the abundance of this Ab. 28 Furthermore, it is well accepted that plasma IgG is not as important in protection of the mucosal layers as is IgA, although some of its fractions may do so. 31
Data indicated lower concentrations of plasma anti-LPS IgM Abs, indicating the potential of this treatment to modulate primary humoral immune responses. Although the exact mechanism of action is not clear at present, it is speculated that the oral vaccination might down-regulate the pro-inflammatory immune reactivities and primary humoral immune responses by repeated exposure to two different bacterial stimulants. Again, the mechanisms behind this might be related to prevention of translocation of bacterial toxins into the host’s bloodstream. Interestingly, a recent study confirmed the existence of T-cell independent (TI) memory B cells in the context of vaccine administration of pure bacterial polysaccharides, which requires exogenous TLR. It was suggested that TI memory plasma cells may have an extended lifespan. 32 Results of the present study confirm the existence of memory B-cells after oral administration of bacterial polysaccharides. However, further research needs to be done to address the phenomenon of the longevity of memory against these bacterial toxins.
This study also showed lowered plasma TNF-α in the LPS- and LTA- treated cows. Results also showed that plasma IL-1 was numerically lesser in the treated cows; however, the difference did not reach significance. These data are indicative of a lower systemic inflammatory response in the treated cows, and are in agreement with results of the salivary IgA and plasma anti-LPS IgA, IgG and IgM Abs tests. It is known that translocation of the luminal bacterial toxic compounds into the systemic circulation stimulates the release of pro-inflammatory cytokines such as TNF-α, IL-1 and IL-6 by liver macrophages, 33 resulting in a state of inflammation in cattle. 6 Recent evidence suggests that mediators of inflammation are released from liver macrophages when activated by binding of endotoxin. 34 This results in enhanced secretion of a number of acute phase proteins (APP) like LBP, SAA and C-reactive protein, whose functions are to neutralize the effects of circulating LPS. 6
Our data support the hypothesis that repeated oral application of LPS and LTA played an important role in attenuation of the acute phase response (APR) as indicated by the lower concentrations of LBP and haptoglobin in the plasma of the treated cows. 35 It is well established that plasma APP are part of a general non-specific immune response, and translocation of endotoxin into the systemic circulation stimulates the release of those proteins from liver hepatocytes under the influence of pro-inflammatory cytokines like IL-1, IL-6 and TNF-α.6,33
The main role of LBP in the plasma is to bind circulating endotoxin and facilitate its clearance either by the macrophage or lipoprotein pathways. 3 Interestingly, the lower concentrations of haptoglobin in the treated cows in the present study is indicative of reduced translocation of bacterial toxins into the bloodstream immediately before and after parturition, showing the ability of this new approach in maintaining the mucosal barrier functions against these bacterial toxins.
Haptoglobin has the ability to selectively antagonize LPS effects by suppressing monocyte production of TNF-α, IL-10 and IL-12, and increased concentrations during an APR may generate a feedback that attenuates the release of cytokines, protecting against endotoxin harmful effects. 36 Repeated oral treatment with LPS and LTA showed no effect on the overall concentration of SAA in the plasma, although it should be pointed out that there were lower concentrations of SAA during the weeks following parturition. The greater concentrations of SAA in the control group are indicative of presence of an inflammatory state in those cows potentially related to increased circulating endotoxin. The main function of SAA is to bind and neutralize LPS, and transport it to the liver hepatocytes for excretion through bile.3,37
Conclusions
In conclusion, repeated oral administration with LPS from E. coli 0111 : B4 and LTA from B. subtilis modulated innate and humoral immune responses in transition dairy cows. Treatment enhanced the total concentration of salivary IgA Abs and lessened the concentrations of several plasma markers of APR, including LBP and haptoglobin in peripartarturient dairy cows. Moreover, cows treated orally with LPS and LTA had lesser concentrations of plasma anti-LPS IgA, IgG and IgM Abs, and the pro-inflammatory cytokine TNF-α. Altogether, data suggested that oral application of LPS and LTA before parturition might play a role as a booster of mucosal immunity against two main bacterial endotoxins in periparturient dairy cows.
Footnotes
Funding
Financial support for this research was provided by Alberta Milk (Edmonton, AB, Canada), Alberta Livestock Industry Development Fund (Edmonton, AB, Canada), and Natural Sciences and Engineering Research Council of Canada (Ottawa, ON, Canada), which is gratefully acknowledged.
Acknowledgements
We are grateful to the technical staff at the Dairy Research and Technology Centre, University of Alberta, for their help and care of the cows.
Conflict of interest
The authors declare no conflict of interest.