Abstract
Alloiococcus otitidis is usually detected in children with otitis media (OM) by PCR as it is not often detected by routine culture. Our improved method for its isolation obtained A. otitidis from nearly 50% of 78 children with OM with effusion. The role of A. otitidis in pathogenesis of OM is unclear. This study tested two hypothesis: (1) that fresh isolates of A. otitidis would elicit pro-inflammatory cytokines from THP-1 monocytic cells equivalent to those induced by Streptococcus pneumoniae; (2) priming THP-1 cells with interferon-gamma (IFN-γ) a surrogate for virus infection, would enhance pro-inflammatory responses. Recent clinical isolates of A. otitidis, S. pneumoniae (ATCC 49619) and a blood culture isolate of S. pneumoniae (SP2) were used in the assays. Cytokines were quantified by BioRad bead assay and Luminex 200. IFN-γ priming enhanced cytokine responses. S. pneumoniae ATCC 49619 induced lower responses than SP2 for IL-1β, IL-6, TNF-α. A. otitidis LW 27 elicited higher IL-1β and TNF-α responses than either pneumococcal isolate. Small green colony types of A. otitidis induced higher responses than large white colony types for IL-8 and IL-1β. The hypothesis that A. otitidis elicits cytokines observed in middle ear effusions was supported; the need to use recent clinical isolates in studies of pathogenesis was highlighted.
Introduction
Inflammatory responses play a major role in pathogenesis of otitis media (OM) in response to infection by the major three otopathogens, Streptococcus pneumoniae, non-typeable Haemophilus influenzae and Moraxella catarrhalis. Cytokines have been identified in clinical material from patients with middle ear disease. 1 Our previous studies identified Alloiococcus otitidis as the predominant isolate from Indigenous Australian children (55%) and non-Indigenous children (38%) with symptomatic otitis media with effusion (OME). The common otopathogens were found in a very small number of children by either culture or molecular methods.2,3 Although A. otitidis has been identified by molecular methods in significant proportions of children with acute otitis media (AOM), 4 it is not often isolated from clinical material as the incubation period required for its detection is longer than those used in routine diagnostic laboratories.2,3 This has resulted in few studies on its role in pathogenesis of AOM or OME. A small number of studies have reported findings that support the hypothesis that A. otitidis is a pathogen. The organism activated lymphocytes and induced production of pro-inflammatory cytokines in similar amounts as the major middle ear pathogens.5–8 These studies indicated that A. otitidis contributed to the inflammatory reaction in the middle ear cavity; however, the use of type culture strains in these studies, not clinical isolates, has limited the strength of sound conclusions.
The investigation of cytokines directly in clinical specimens of middle ear effusions (MEE) collected from patients infected with A. otitidis was first reported by Harimaya et al. 9 These authors analysed the release of pro-inflammatory cytokines and chemokines in MEE of children with AOM from whom A. otitidis was detected by PCR. Results were compared with those for children with AOM due to S. pneumoniae. The control group consisted of patients with AOM from whose specimens no organism could be detected by culture or by PCR. Pro-inflammatory cytokines IL-8, IL-1β, IL-6 and TNF-α, and chemokines IFN-γ-inducible protein-10 and IFN-inducible T cell alpha chemoattractant were significantly increased in the A. otitidis group, as well as the S. pneumoniae group. The cytokine profiles for both organisms were very similar suggesting that A. otitidis has the potential to induce inflammatory reactions in the middle ear cavity similar to those elicited by S. pneumoniae.
In assessing the inflammatory responses to pathogens, there are various factors that can modify these responses and need to be controlled when investigating the potential virulence of an organism. Both genetic and environmental factors can contribute to variations in responses to microorganisms or their products. These include gender, 10 cigarette smoke, 11 concurrent symptomatic or asymptomatic virus infection,11–13 developmental stage, 14 hormone levels 15 and gene polymorphisms. 16 To minimise these effects, the THP-1 cell line was chosen as the model system to reduce the effects of sex and genetic background; to permit assessment of the effects of IFN-γ as a surrogate for virus infection; and to compare results obtained in earlier studies in which inflammatory responses were elicited by type culture strains of A. otitidis from THP-1 cells.5,6
THP-1 is a human monocytic cell line employed in previous studies on factors affecting inflammation and was chosen to examine differences in host responses to the collection of clinical isolates of A. otitidis obtained in previous studies. Both clinical and experimental reports indicated that the pro-inflammatory cytokines TNF-α, IL-1β, IL-6 and IL-8 were important mediators in OM; these were assessed for potential differences in their induction by the individual isolates of A. otitidis and in relation to the two colony types we described previously. 3
As virus infections are commonly associated with development of AOM 17 and can up-regulate cell surface antigens on the host cells that act as receptors for bacteria,18–20 this important risk factor was examined in the model system. IFN-γ is reported to differentiate and activate THP-1 cells. 21 Previous studies have reported IFN-γ production in response to viral stimulus in the range of 0.2–20 ng/ml for 5 × 105 PBMC/ml.21,22 The addition of IFN-γ to the experimental model within this concentration range was considered biologically relevant.
THP-1 cells do not normally express the receptor CD14, which, in conjunction with TLR4, is involved in initiation of responses to some antigens, such as endotoxin. THP-1 cells pre-treated with vitamin D3 (VitD3) expressed CD14 on the cell surface23,24 and significantly enhanced inflammatory responses to meningococcal endotoxin. 25 There was no information on what role, if any, CD14 played in induction of inflammatory responses to A. otitidis.
This study tested two hypothesis: (1) that recent clinical isolates of A. otitidis obtained from children with OME would elicit pro-inflammatory cytokines from a human monocytic cell line (THP-1) at levels equivalent to a classical Gram-positive otopathogen, S. pneumoniae; (2) that priming the THP-1 cells with IFN-γ as a surrogate for virus infection would significantly enhance cytokine responses in this model system. The objectives of this study were to determine:
which cytokines, in addition to IL-8 or IL-12 reported in previous studies, were elicited from THP-1 cells by A. otitidis; if the surrogate for virus infection (IFN-γ) enhanced inflammatory responses to the whole cells of A. otitidis; if the levels of cytokines elicited by A. otitidis were similar to those elicited by a known otopathogen S. pneumoniae; if there were differences in inflammatory responses induced by individual clinical isolates of A. otitidis obtained in our previous studies, particularly in relation to colony types.
Materials and methods
Materials and reagents
Cell culture media and reagents were sourced from Invitrogen (Melbourne, Australia). Chemicals were supplied by Sigma (Sydney, NSW, Australia) and were analytical grade unless otherwise noted. Solutions were made or diluted in MilliQ water (Millipore, Bedford, MA, USA) and autoclaved if heat stable. Reagents were thawed immediately before use in the stimulation assay and diluted as required in cell culture medium.
The THP-1 cell growth and maintenance medium (RPMI-10%) consisted of RPMI 1640 supplemented with foetal calf serum (FCS) (10% v/v), glutamine (100 µl 2.0 mM) and sodium pyruvate (100 µl 1.0 mM). Following preparation, the medium was filter sterilised (0.22 µl; Millipore) and stored at 4℃ for up to 14 d and pre-warmed to 37℃ before use. FCS was heat-inactivated at 56℃ for 30 min before use.
VitD3 was prepared in biological grade ethanol and stored at −20℃ in aliquots (10−4 M). To induce expression of CD14, THP-1 cells (2.5 × 105 cells/ml) were pre-treated with 10−7 M VitD3 for 72 h prior to use in stimulation assays. 25
CD14 expression of VitD3-differentiated THP-1 cells was confirmed by flow cytometry (Hunter Area Pathology Service, Newcastle, Australia).
IFN-γ was dissolved in PBS to obtain a concentration of 1.0 mg/ml, adjusted to pH 8.0 with sodium hydroxide according to manufacturer’s directions (Oxoid, Adelaide, SA, Australia). Subsequent dilutions were made in RPMI-10% and stored at −20℃.
THP-1 cell line
THP-1 cells were originally sourced from the American Typed Culture Collection (ATCC). They were stored in 1-ml aliquots (1 × 107 cell/ml) in 90% FCS, 10% DMSO (Ajax Finechem, Scoresby, VIC, Australia), 100 U/ml penicillin and 100 µg/ml streptomycin. Cells were frozen to −80℃ at a rate of −1℃ per min for a maximum of 4 h and then transferred to liquid nitrogen for long term storage.
Cell maintenance
THP-1 cells were washed by centrifugation at 250 g for 5 min then resuspended in pre-warmed RPMI-10%. Cells were maintained at log-phase growth (1 × 104–1 × 106 cells/ml) and incubated in CO2 (5%, v/v) at 37℃. Maintenance medium was renewed with pre-warmed RPMI-10% every 2–3 d. Cells were discarded after 18 wks of growth in log phase. For the stimulation assay, cells were washed, resuspended in fresh RPMI-10% and immediately transferred to cell culture plates before addition of stimulants.
Mycoplasma contamination detection
THP-1 cells were tested for mycoplasma contamination at intervals using the MycoSensor PCR Assay Kit (Stratagene, Sydney, NSW, Australia). Both cells and cell culture supernatant were tested according to the manufacturer’s instructions.
Cell-viability
THP-1 cells were diluted in 0.4% filtered trypan blue and viewed by light microscopy in a Neubauer counting chamber. A cell viability of > 85% was considered acceptable for the assays.
Formalin-killed bacterial cells
Clinical isolates of A. otitidis (n = 39), a reference strain of S. pneumoniae (ATCC 49619) and a strain of S. pneumoniae isolated from a blood culture (SP2) were grown on horse blood agar (HBA) (bioMérieux-Marcy l'Etoile, France). The colonies were transferred to separate tubes containing PBS with dilute formalin (0.5%, v/v) and incubated for 30 min at room temperature. An aliquot of the dense cell suspension was centrifuged at 3500 g (Heraeus-Megafuge 40, Scoresby, VIC, Australia) and the supernatants discarded. The bacteria were resuspended in sterile PBS and washed three times by re-suspension and centrifugation to remove traces of formalin. The cell count was adjusted to approximately 1 × 1010 following determination of the concentration in a counting chamber by light microscopy and confirmed by the McFarland opacity method. After the THP-1 cell count was established, appropriate dilutions of the killed bacterial cells were made aseptically in RPMI-10% to achieve biologically relevant bacteria to THP-1 cell ratios of 1 : 1 and 10 : 1. Time delay between stimulant preparation and subsequent addition to the THP-1 cells was minimal. A sample of each was cultured on HBA to detect evidence of contamination or survival of the organism.
Assays
The following assays were carried out to address the objectives.
The effect of bacterial dose and priming with VitD3 and IFN-γ
THP-1 cells incubated in RPMI-10%, with or without VitD3 for 72 h, were adjusted to 3 × 105 cells/ml in the growth medium. THP-1 cells (100 µl) were added to sterile 48- or 24-well tissue culture plates (Nunc, Scoresby, VIC, Australia). Stimulants were immediately added to the cell culture system. For negative controls, RPMI-10% was added in place of stimulants and wells treated the same as those to which stimulants were added. The plates were incubated at 37℃ in 5% (v/v) CO2 for 24 h.
At 12 h, cell culture samples (500 µl) were removed from each well of the tissue culture plate into separate microfuge tubes and centrifuged at 250 g for 5 min to obtain the supernatants. The samples were stored immediately at −20℃. For each experiment, assays were performed in triplicate; however, it was not possible assess all three samples owing to financial restrictions. Two samples for each experimental condition were tested for cytokine levels and the third sample reserved for repeat testing if there were significant differences between the initial two samples.
In the preliminary experiments to set up the model, the following stimulants were investigated: formalin-killed A. otitidis (a combined suspension of four clinical isolates) and S. pneumoniae ATCC 49619 at cell ratios of 1 : 1 and 10 : 1. These were used to stimulate THP-1 cells or THP-1 cells pre-incubated with VitD3. The same ratios were used to stimulate THP-1 cells to test the effect of priming with IFN-γ (10 ng/ml). Duplicate plates of cells, with or without VitD3 pre-treatment, were tested in parallel. Control wells contained RPMI-10%, and THP-1 cells.
Screening of individual clinical isolates for induction of inflammatory mediators
The experimental protocol developed was used to screen all 39 isolates of A. otitidis; formalin-killed organisms were tested at a ratio of 10 bacteria per THP-1 cell. These experiments used THP-1 cells pre-incubated for 72 h with VitD3 and primed for 24 h with IFN-γ (10 ng/ml) prior to addition of the bacteria. Samples were collected 12 h post-stimulation for assessment of cytokines. All experiments were carried out in triplicate, but owing to cost limitations, duplicate samples were tested for cytokines induced for each condition tested. Control wells containing THP-1 cells and RPMI-10% were tested in triplicate.
Analysis of cytokine response
Cell culture supernatants were measured for IL-1β, IL-6, IL-8 and TNF-α using a bead-based immunoassay kit (BioRad, Hercules, CA, USA). IL-10 was not assessed, as previous studies in our group had demonstrated that it was not induced from THP-1 cells under the conditions tested. The immunoassay was performed according to the manufacturer’s instructions. Supernatants were diluted 1 in 4 with RPMI-10% and tested in duplicate. A broad-range standard curve was generated using the standard provided with the kit. Bead median fluorescence intensity was detected with a Luminex 200 (Austin, TX, USA) using either low or high photomultiplier (PMT) setting. Cytokine concentrations (pg/ml) of unknowns were calculated from the standard curve using the Luminex 2.3 software (Luminex).
Cytokine variability
Previous experiments using this model showed no cytokine degradation between samples collected from the reaction wells and frozen within 30-min intervals, up to 90 min. All samples were frozen within 90 mins of collection. To reduce cytokine variability between triplicate samples, semi-automated pipetting was utilised to reduce variation.
Statistical analyses
The results for the cytokine assays were analysed by t-test on log10 of the data obtained in the experimental model.
Results
In preliminary studies with A. otitidis and S. pneumoniae ATCC 49619, there was no significant difference in inflammatory responses between THP-1 cells pre-incubated with or without VitD3; however, VitD3 pre-incubation was maintained for comparison with other ongoing studies.
Ratio of bacteria:THP-1 cell
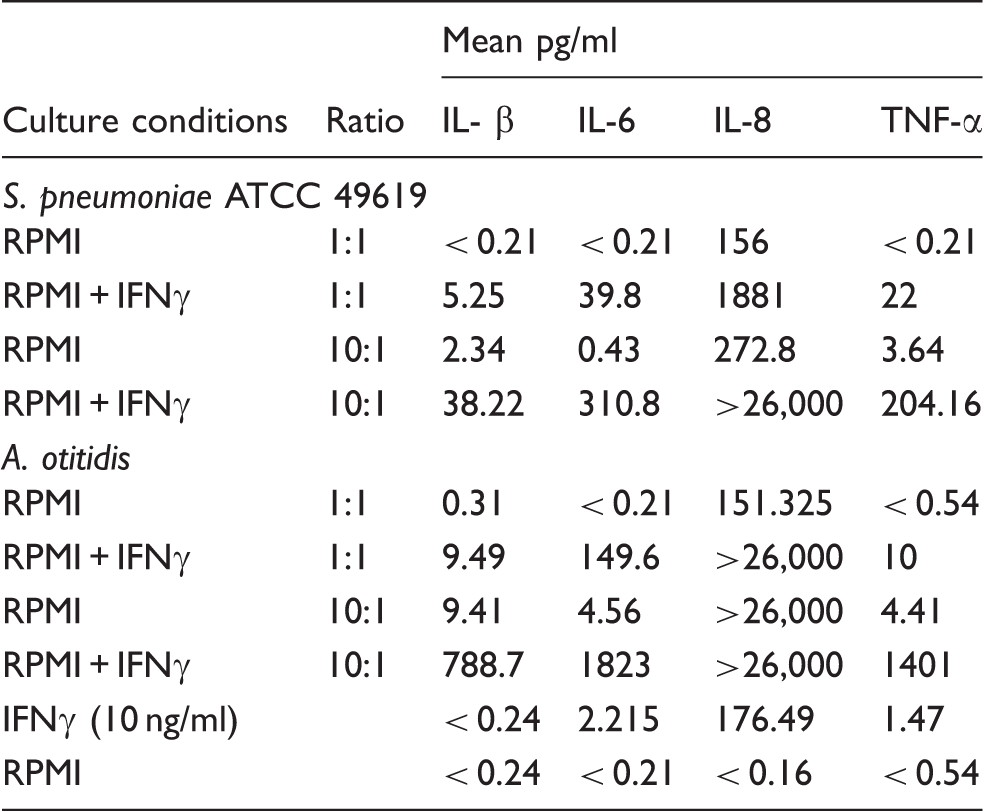
The effect of bacterial dose and priming with IFN-γ on cytokine responses elicited from THP-1 cells by S. pneumoniae (ATCC 49619) or the pool of four isolates of A. otitidis (mean of four experiments).
IFN-γ and inflammatory responses
Priming of THP-1 cells with IFN- γ alone increased some cytokines to levels detectable in the Luminex system: IL-6 from < 0.21 pg/ml to 2.2 pg/ml; IL-8 from < 0.16 pg/ml to 176 pg/ml; TNF-α from < 0.54 pg/ml to 1.47 pg/ml. Priming with IFN-γ significantly enhanced cytokine responses to both pneumococci and A. otitidis. The highest levels of cytokine production were observed with cells primed with IFN-γ and a bacteria:THP-1 cell ratio of 10:1 (Table 1). These were chosen as the conditions for further experiments.
Cytokines elicited by A. otitidis and S. pneumoniae
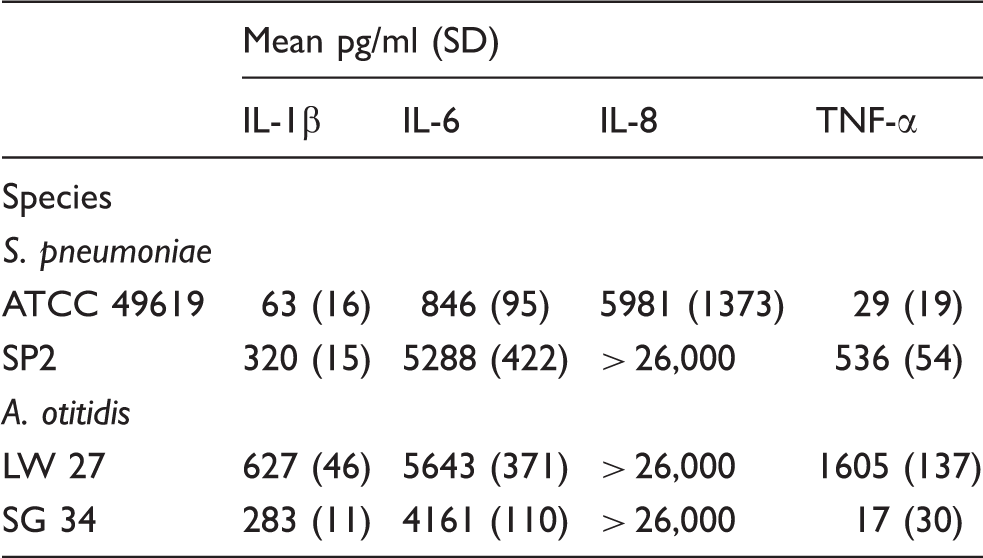
Mean (SD) of cytokine responses induced by S. pneumoniae or isolates of the two different colony types of A. otitidis, large white (LW) or small green (SG), from THP-1 cell primed with IFN-γ (mean of four experiments).
Responses induced by colony types of A. otitidis
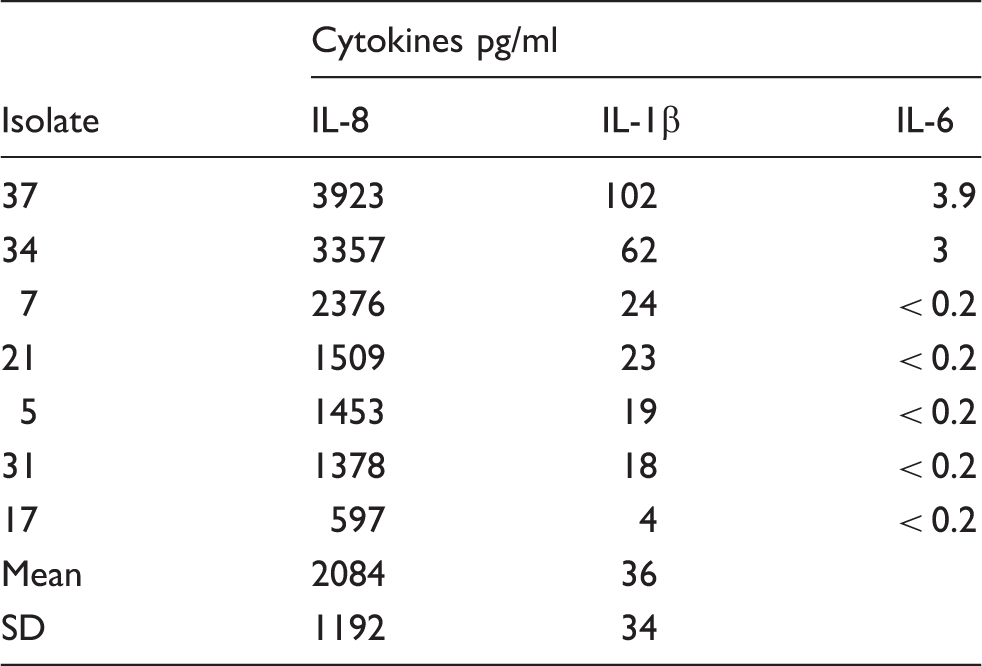
Cytokine responses elicited by isolates with the small green (SG) colony type (pg/ml).
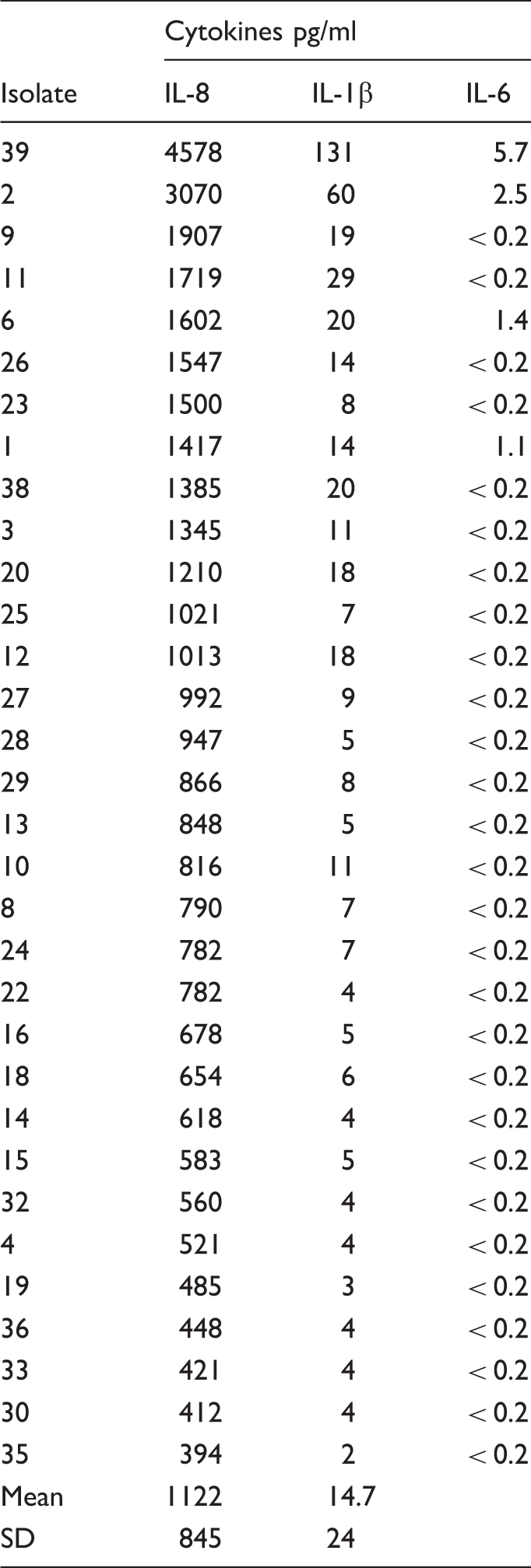
Cytokine responses elicited by isolates with the large white colony types (pg/ml).
Discussion
Advantages and disadvantages of the model system
To minimise the potentially confounding effects of genetic and environmental factors on cytokine responses to A. otitidis, the THP-1 cell line was chosen as the model system to provide a consistent background to compare cytokine responses to the individual A. otitidis isolates, differences in colony type and assessment of the effects of IFN-γ as a surrogate for virus infection.
One of the disadvantages and restrictions of this method was the high cost of the kits for detection of the cytokines; however, all those of interest could be tested in one small sample. The numbers of experiments performed were limited, but it was possible to obtain sufficient data for statistical analyses.
Cytokines stimulated by A. otitidis
While single cytokines had been assessed in previous studies of inflammatory responses elicited by A. otitidis (IL-8 or IL-12),5,6 this study assessed additional inflammatory markers identified in the MEEs of children in whose samples A. otitidis was identified by PCR. 9 All 39 isolates examined induced IL-1β, IL-8 and TNF-α. IL-6 was detected in preliminary experiments (Tables 1 and 2), but at the higher PMT level used for subsequent experiments to keep IL-8 within detection range, levels of IL-6 were below the lower limit of detection for most isolates. Pre-incubation with VitD3 did not affect either the type or quantity of cytokines tested, but this step was retained in our studies. In contrast to endotoxin, 25 expression of the CD14 antigen does not appear to affect responses of THP-1 cells to A. otitidis or S. pneumoniae.
Previous studies using a similar model indicated that cytokine levels obtained using live cells were higher than those in which inactivated bacteria were used as stimulants. 6 The comparison of responses to live and formalin-killed cells could not be tested. Because the studies were carried out using instruments in diagnostic laboratories, only killed bacterial cells could be used for testing whole organisms. In a subsequent study, cell-free filtrates were assessed to determine if extracellular materials contributed to the inflammatory responses. 26
Effect of priming with IFN-γ on inflammatory responses to whole cells of A. otitidis
For both pneumococcal and A. otitidis isolates, priming the THP-1 cells with IFN-γ significantly increased the cytokine responses. These observations are of interest in relation to susceptibility to and severity of OM. Upper respiratory viral infections often precede an episode of AOM. Previous studies found that virus- infected cells bound significantly higher numbers of the classical otopathogens than uninfected cells.18,19 In addition, IFN-γ enhanced the level of cytokine responses to both pneumococcal and A. otitidis isolates tested. While IFN-γ appears to play an important role in enhancement of cytokine responses, these findings indicate that the individual isolate tested is a significant factor in the responses elicited. Comparison with similar studies requires careful assessment of the species and strains used.
Comparison of cytokine responses elicited by A. otitidis and S. pneumoniae
In the preliminary studies (Table 1), levels of cytokines elicited by the pool of four isolates of A. otitidis were higher than the isolate of S. pneumoniae (ATCC 49619). While the recent clinical isolate of S. pneumoniae (SP2) elicited higher levels of the cytokines than the S. pneumoniae ATCC 49619 or SG 34 isolate, the LW 27 isolate of A. otitidis induced the highest levels of each of the cytokines (Table 2).
Inflammatory responses induced by individual clinical isolates of A. otitidis
Previous assessments of A. otitidis and inflammatory responses used only one isolate—a type culture strain—as the organism has been reported to be difficult to isolate.27,28 This is the first study to compare recent clinical isolates from different children for their abilities to induce cytokines.
Our observation that there are two different colony types (large white and small green) allowed us to test for differences associated with phenotypes of this species. The results for the last experiment were assessed in relation to colony morphology. In contrast to the results obtained for LW 27 and SG 34 in the preliminary experiments, as a group the SG colony types appeared to induce higher levels of IL-8 and IL-1β.
The results support the hypothesis that A. otitidis contributes to inflammation observed in OM and highlighted the need to use recent clinical isolates in studies of pathogenesis. Cytokine levels elicited by A. otitidis were equal to or greater than those elicited by pneumococci, an acknowledged Gram-positive otopathogen. These studies on the innate responses complement the observations of Harimaya et al. 29 in which IgG, secretory IgA, IgG2 and IgM specific for A. otitidis in MEE were detected by western blot analysis, indicating that A. otitidis is recognised by both innate and specific immune responses. The role of A. otitidis in the pathogenicity of both acute and chronic OM needs to be investigated and considered in treatment of these infections and development of preventive measures. While A. otitidis has been considered to be an outer ear commensal,30,31 it should be noted that a commensal in an area of the body that is not its natural habitat can behave as a pathogen. It is planned to use the methods developed for comparison of inflammatory responses elicited by isolates of A. otitidis from ear canal or nasopharynx with those obtained from MEEs.
Footnotes
Funding
The work was supported by grants from Hunter Children’s Research Foundation; Hunter Medical Research Institute; University of Newcastle; and Hunter Area Pathology Service Microbiology Trust Funds.
