Abstract
In our model system using the THP-1 monocytic cell line, whole heat-killed cells of Alloiococcus otitidis elicited several pro-inflammatory cytokines identified in ear effusions of children with otitis media (OM). Levels of these cytokines were equivalent to or greater than those elicited by a standard Gram-positive otopathogen, Streptococcus pneumoniae. The current study examined the hypothesis that extracellular material produced by A. otitidis might also contribute to the inflammatory responses in OM. Cell-free culture filtrates of recent A. otitidis isolates (n = 39) were tested for induction of pro-inflammatory cytokines from THP-1 cells primed with IFN-γ. The highest responses were from IL-8 followed by IL-1β, and the lowest from IL-6. Filtrates from nine isolates were treated with lysozyme or proteinase K to assess the nature of the extracellular stimulants. Peptidoglycan was not a major component eliciting the responses. There was no correlation between colony type or β-haemolysin production. Proteinase K treatment indicated extracellular proteins might induce the inflammatory responses, particularly the 70–75 ku band. Further studies on the role of the extracellular proteins of A. otitidis and cytokine responses in pathogenesis of ear infections are needed.
Introduction
The inflammatory responses play a significant role in the disease process and tissue damage associated with otitis media (OM). 1 A small number of early studies reported findings that support the hypothesis that Alloiococcus otitidis is a pathogen. The organism activated lymphocytes and induced production of pro-inflammatory cytokines in similar amounts as the major middle ear pathogens.2–5 While there was evidence that A. otitidis contributed to the inflammatory reaction in the middle ear cavity, the use of type culture strains in these studies, not clinical isolates, limited the strength of sound conclusions. The collection of fresh clinical isolates obtained in our epidemiological surveys have allowed us to initiate studies on how the slow-growing species A. otitidis contributes to the inflammatory responses involved in the pathogenesis of otitis media.6–8 In another article, we demonstrated that whole heat-killed cells of A. otitidis induced cytokine responses from THP-1 cells at levels equivalent to or greater than those elicited by Streptococcus pneumoniae, a recognised Gram-positive otopathogen. 8
There were two reports in which cell-free stimulants were tested in tissue culture assays by exposing THP-1 cells to diffusible material from live A. otitidis across a membrane. Only IL-8 and IL-12 were assessed in these studies;5,6 however, a wider range of cytokines associated with A. otitidis infections was detected directly in clinical material. 9
A soluble extract of A. otitidis [American Type Culture Collection (ATCC) 51267] elicited IL-12 from THP-1 cells. 2 Live organisms induced IL-12 from THP-1 cells; however, experiments in which it was tested in parallel with the classical otopathogens indicated there were marked differences in the stimulatory capacity. S. pneumoniae induced the lowest level and Haemophilus influenzae the highest. Induction of IL-12 in this system was reduced by treatment of the bacteria with glutaraldehyde or after heat treatment. The extract of A. otitidis was separated into four fractions by molecular mass. The 100-ku subfraction induced the highest levels of IL-12. 2 Using a two-chamber system and cell culture inserts that provided physical separation of bacterial cells from THP-1 cells, these investigators demonstrated that viable A. otitidis released substances that induced IL-12. Compared with levels of the cytokine induced by direct contact of the bacteria with the THP-1 cells, the IL-12 levels were lower. 2
IL-8 responses were investigated by similar methods in a parallel study by the same authors. 3 IL-8 was secreted from IFN-γ-primed THP-1 cells in the two-chamber system. This indicated that part of the IL-8 response might be induced by a diffusible substance released by the bacteria or soluble components of A. otitidis.
In the two-chamber system, IL-8 production from THP-1 cells in response to A. otitidis demonstrated a 33% reduction when only soluble material and not whole cells was used to stimulate cytokine responses. For the otopathogens tested in the system, similar reductions were noted: S. pneumoniae (23%), H. influenzae (65%) and Moraxella catarrhalis (63%). This suggested that part of the IL-8 and IL-12 responses might result from diffusible factors released by the bacteria, or soluble components of the organism itself; however, the nature of the stimulatory factor was not identified and these factors could differ for Gram-positive and Gram-negative organisms.
Compared with cytokine levels in the middle ear effusions (MEE) of patients in whose specimens no bacteria were detected by PCR, the levels of cytokines were significantly increased in samples from children infected with A. otitidis or S. pneumoniae. The following cytokines were identified in the patient samples: IL-8, IL-1β, IL-6, TNF-α, IFN-γ inducible protein-10 and monocyte chemoattractant protein-1. 9 This study indicated that a broader range of cytokines was elicited by A. otitidis than those previously assessed in vitro.
The objectives of this study were (1) to repeat previous observations on induction of inflammatory responses by cell-free filtrates of A. otitidis from 39 recent clinical isolates;6,7 (2) to determine if there were differences in responses elicited by cell-free filtrates of individual clinical isolates with reference to colony type or β-haemolysin production; (3) to determine the nature of components eliciting the inflammatory responses; (4) to compare the extracellular protein profiles of the clinical isolates; and (5) to determine if particular extracellular protein bands were associated with induction of inflammatory mediators.
Materials and methods
Preparation of extracellular filtrates of A. otitidis
A dense suspension of each of the 39 isolates was prepared in 10 ml of brain heart infusion (BHI) broth and incubated for 10 d at 37℃. The suspension was centrifuged at 3500 g for 10 min and the supernatant collected. This was filtered though a 0.22 -µm syringe filter (Millipore, Bedford, MA, USA) to remove free organisms and stored at −20℃. A control was prepared, which consisted of un-inoculated BHI treated in exactly the same way as the inoculated medium. The filtrates were warmed to 37℃ before addition to THP-1 cells.
Assessment of cytokine responses to filtrates
In preliminary experiments, the protocol described in the accompanying paper for induction of cytokines from THP-1 cells primed with IFN-γ was adapted to test their responses to cell-free filtrates of A. otitidis. 8 A pool of filtrates from eight isolates was tested in parallel with a heat inactivated (100℃ for 15 min) aliquot of the pool to determine if the stimulatory activity was heat stable. For the major experiment, the individual A. otitidis filtrates (50 µl) were added to THP-1 cells in individual wells; un-inoculated BHI (50 µl) was used as the control stimulant.
IL-8, IL-6, IL-1β and TNF-α were assayed by the Luminex system (Austin, TX, USA) as described previously. 8
Enzyme treatment of filtrates
To determine if the filtrate components inducing the inflammatory responses were protein or peptidoglycan fragments, aliquots of nine individual filtrates from isolates 30–38 were treated with lysozyme or Proteinase K. Lysozyme solution was prepared in RPMI (2.0 mg/ml) and stock Proteinase K (50 µg/ml) was diluted 1:150 in RPMI. Both were passed through a 0.22 -µm sterile syringe filter before use. Lysozyme was added to the filtrate in sterile glass tubes to the equivalent of 1.0 µl lysozyme in 1.0 ml of filtrate. The tubes were incubated at 37℃ for 30 min. Proteinase K was added to another series of aliquots in sterile tubes, 150 µl per 1.0 ml of filtrate. The tubes were incubated at 56℃ for 45 min, allowed to cool and placed on ice for 5 min.
Reagents for SDS-PAGE
Laemmli sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) buffer contained Tris base (30.3 g), glycine (144 g), SDS (10 g) and 1000 ml distilled water. Precision Plus Protein standards, Dual Color (BioRad) was used to visualise the proteins. The 2 × loading buffer contained bromphenol blue (0.0025 g), glycerol (2 ml), SDS (10%, 2.4 ml) and Tris base (0.606 g) in 5 ml (pH 6.8) made up to a final volume of 10 ml.
Concentration of filtrates and removal of lower molecular mass proteins
Initial gels using neat culture filtrates demonstrated a faint band around 70 ku; therefore, to examine the protein bands, the filtrates were concentrated 50 times using a gravity concentrator device (Prochem BJP-SP5, Brisbane, QLD, Australia) with a molecular mass cut-off of 7.5 ku. Each supernatant (5 ml) was processed in a separate concentrator.
Separation by electrophoresis and subsequent staining showed abundant low molecular mass proteins that obscured visualisation of larger proteins of interest. To improve resolution, a centrifugal filter (Millipore ultra-4; Millipore) with a nominal molecular mass cut off of 50 ku was used to remove the smaller proteins and further concentrate the larger proteins so that visualisation was possible.
Protein visualisation and interpretation
Proteins were separated by SDS-PAGE on Tris-HCl 4–20% 10-well mini-protein 3 apparatus Biorad (Hercules, CA, USA) at 200 V for 40 min. The proteins were stained using Coomassie brilliant blue NuSep (Sydney, NSW, Australia). The gels were de-stained in deionised water. The staining intensities can be used to estimate the molar ratios of the protein subunits of protein complexes. The gels were scanned on a flatbed scanner using Helena (Quick Scan 2004 v. 2). This gave a measure of relative protein concentration between the protein components.
Statistical methods
Cytokine responses were assessed on log10 transformed data by Student’s t-test; a value of P < 0.05 was set as significant.
Results
Cytokine production induced by cell-free filtrates of A. otitidis from THP-1 cells with or without pre-treatment with IFN- γ
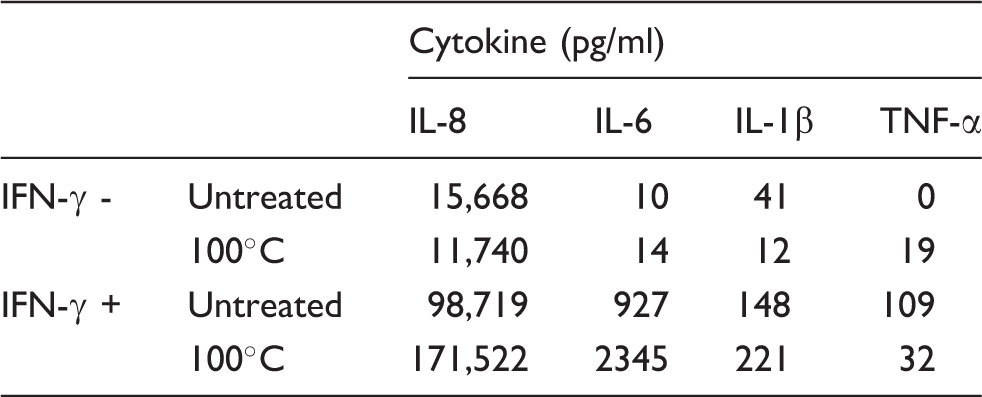
As with the whole cells, compared with THP-1 cells incubated in growth medium alone, priming with IFN- γ increased the levels of cytokines elicited by the untreated or heat-treated filtrates: IL-8 from six- to nine-fold; IL-6 from 9.2- to 167-fold; IL-β from 3.6- to 18-fold. TNF-α increased from undetectable levels to 109 pg/ml.
The effect of IFN- γ priming of THP-1 cells on cytokine production induced by pooled filtrates of A. otitidis with or without heat treatment (100℃ for 15 min). Average of two measurements.
Cytokines elicited from THP-1 cells by filtrates of the 39 A. otitidis isolates
Each of the 39 isolates elicited measurable levels of IL-8, IL-1β and IL-6 compared with the un-inoculated BHI control. The samples were assessed at a low photomultiplier tube (PMT) voltage to obtain IL-8 readings, which had been above the detection range when the samples were tested with settings using a high PMT voltage; this restricted the detection of IL-6 and IL-1β, which were present in lower quantities and detected more successfully using high PMT settings. The mean of IL-8 responses was 13,523 (SD = 5252; range = 4952–25,977). The mean of IL-1β responses was 1609 (SD = 646; range = 798–2589). The mean of IL-6 responses was 40 (SD = 14.5; range = 19–81).
Assessment of cytokine responses from filtrates obtained from isolates in relation to haemolysin production
The β-like haemolysin was a stable characteristic of the isolates that produced it. The α-haemolysis was variable, inconsistent and difficult to discern on some plates; therefore, non-haemolytic and α-haemolytic isolates were analysed as one group. For the 26 non-haemolytic or α-haemolytic isolates, the cytokine responses were as follows: IL-8, mean = 12,722, range = 4952–22,879 pg/ml; IL-1β, mean = 1522, range = 798–2255 pg/ml; IL-6, mean = 39, range = 19–81 pg/ml. For the 13 isolates producing β-haemolysis, the cytokine responses were as follows: IL-8, mean = 15,124, range = 10,214–15,977 pg/ml; IL-1β, mean = 1785, range = 919–2589 pg/ml; IL-6, mean = 42, range = 25–63 pg/ml. While the means for each of the cytokines were higher for the isolates producing β-haemolysin, the differences were not significant.
Assessment of cytokine responses from filtrates obtained from isolates in relation to colony type
For filtrates from isolates of the large white colony type (n = 32), the cytokine responses were as follows: IL-8, mean = 13,879, range = 5085–26,030 pg/ml; IL-1β, mean = 1635, range = 798–2589 pg/ml; IL-6, mean = 38.6, range = 19–81 pg/ml. For filtrates from the isolates of the small green colony type (n = 7), the cytokine responses were as follows: IL-8, mean = 11,810, range = 4952–25,977 pg/ml; IL-1β, mean = 1208, range = 225–2494 pg/ml; IL-6, mean = 45.7, range = 19–63 pg/ml. There were no significant differences between cytokine responses elicited by filtrates from the small green isolates compared with the large white isolates.
Cytokine responses elicited by cell-free filtrates treated with lysozyme or Proteinase K
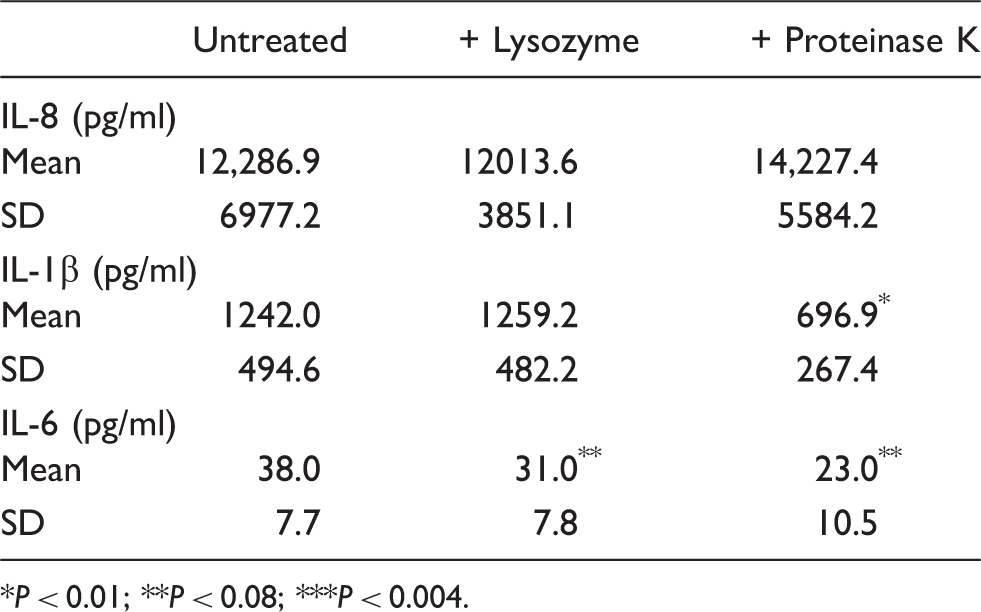
The effects of lysozyme and Proteinase K treatment of nine filtrates of A. otitidis on induction of cytokines from IFN-γ primed THP-1 (means of duplicate wells).
P < 0.01; **P < 0.08; ***P < 0.004.
SDS-PAGE of filtrates
In the initial experiments, faint bands were detected; however, the concentrated filtrates produced clearer bands after staining. In some gels, concentrators produced a large diffuse stained area on the gel in the range of 15–50 ku, but with little clarity. A clear band at about 70 ku was found in the majority of the filtrates. On subsequent gels, following attempted removal of the lower mass proteins, there was a distinct band of protein at 50 ku. It is postulated that this was due to polymerisation of low molecular mass components. Repeat runs following extra washing steps and pre-washing the concentrator device reduced the bands at 50 ku, but some low molecular mass proteins were still present.
On scanning the gels of 39 filtrates, 26 (70%) possessed a band equivalent to 75 ku and 7/39 (18%) showed a band corresponding to a range of 150–250 ku. Lower molecular mass bands around 15–50 ku, which were present in 35/39 of filtrates, might represent non-specific background substances derived from cell wall components; the BHI control lane did not show bands within this range.
Analysis of filtrates of A. otitidis by SDS-PAGE following treatment with lysozyme or Proteinase K
The results summarised in Table 2 indicated that proteins in the cell-free filtrates might be involved in inducing cytokine responses from THP-1 cells. The untreated cell-free filtrates for isolates 30–39 and their respective aliquots treated with either lysozyme or Proteinase K were combined, concentrated and the concentrated material run on PGE. There were no bands detected on the gels for lanes containing BHI or the Proteinase K-treated filtrate. Bands detected for the untreated filtrate and the filtrates treated with lysozyme showed no significant differences.
Analysis of protein bands in relation to β-like haemolysin and colony type
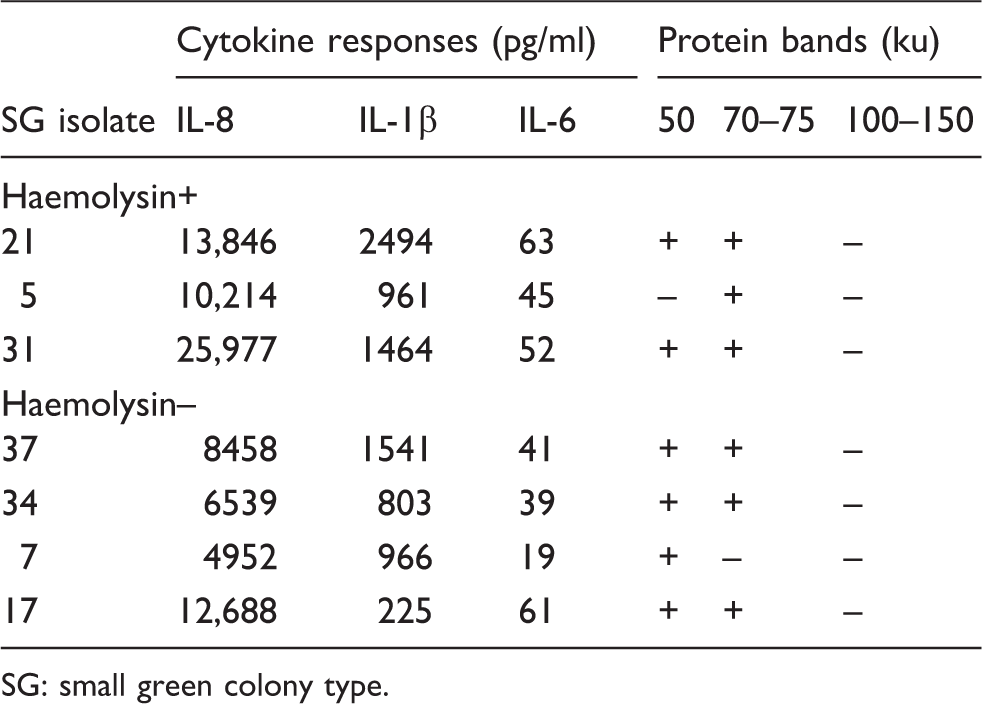
Cytokine responses induced by filtrates of seven isolates of A. otitidis with the small green colonies in relation to β-like haemolysin and cytokine production.
SG: small green colony type.
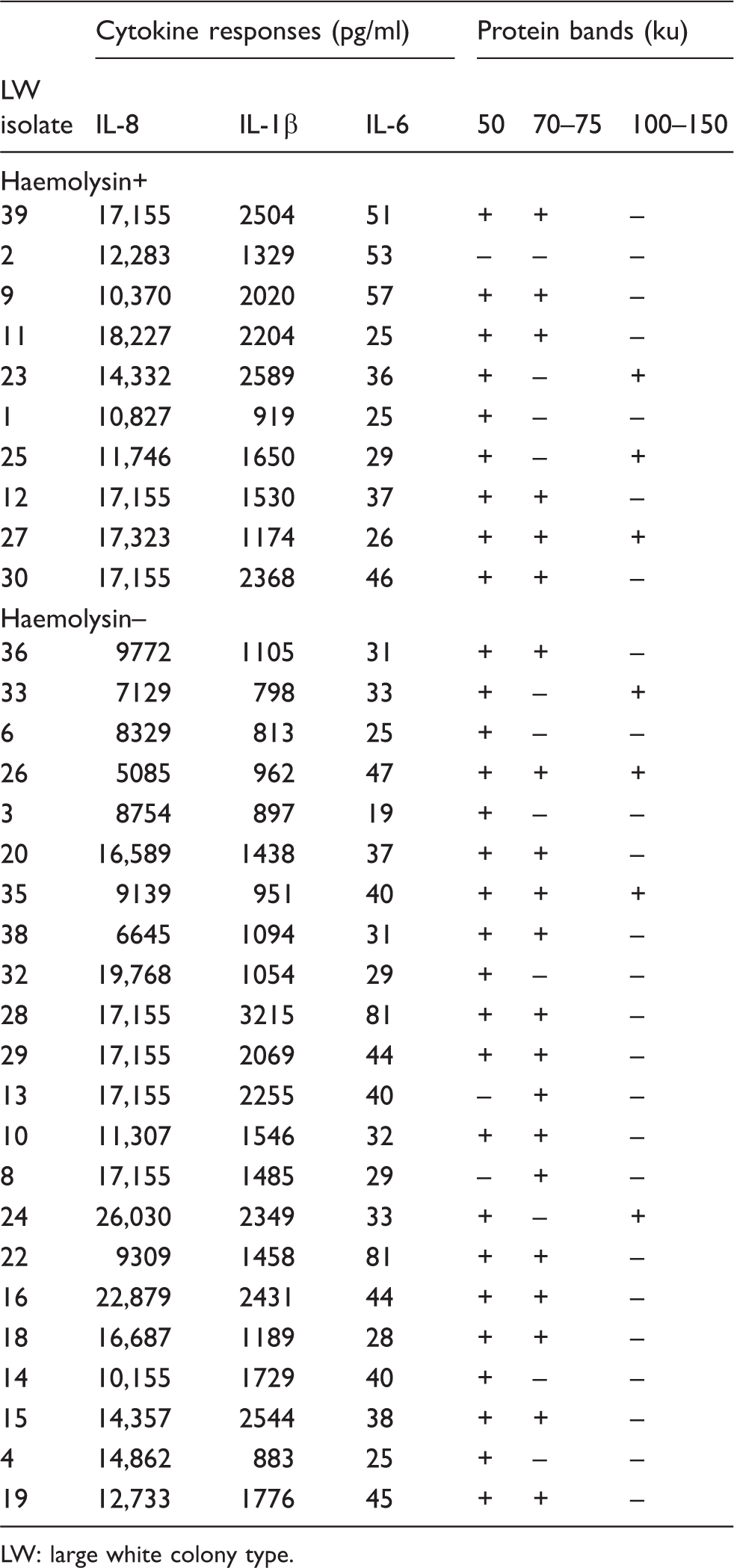
Cytokine responses induced by filtrates of 32 isolates of A. otitidis with large white colony types in relation to β-haemolysin and cytokine production.
LW: large white colony type.
There was no association with any of the major protein bands or the β-haemolysin.
Cytokine responses in relation to presence of extracellular proteins
The presence or absence of the major band at 70–75 ku was assessed in relation to cytokine levels elicited by filtrates containing this protein. There were 27 isolates with the 70–75 ku band and 12 without. The mean levels of IL-8 (14,008 pg/ml, SD = 5025) and IL-1β (1659 pg/ml, SD = 686) elicited by filtrates containing this band did not differ significantly from those elicited by filtrates without the band: IL-8 (12,431 pg/ml, SD = 5806), IL-1β (1331 pg/ml, SD = 618). For IL-6, the differences were significant. For filtrates containing the 70–75-ku band, the mean IL-6 response was 44 pg/ml (SD = 14.6); for the filtrates without the 70–75-ku band, the mean IL-6 response was 30.5 (SD = 9.5), P < 0.0002.
Discussion
A. otitidis has been identified in studies of children with both acute and chronic otitis media, mainly by PCR.10–12 Our previous studies indicated that extended incubation allowed sufficient time for A. otitidis to be detected.6,7 Because it has not been cultured by routine diagnostic methods, its relevance to these infections has been questioned. The current studies in our companion paper indicate that the species is capable of inducing pro-inflammatory responses equal to or greater than S. pneumoniae, 8 a classic Gram-positive otopathogen. The slow growth of the organism and presence of other species capable of producing β-lactamase is likely to reduce its susceptibility to penicillin, and a significant proportion of isolates was also resistant to macrolides used to treat otitis media.6,7 Failure to respond to treatment with these antibiotics and the absence of the usual otopathogens on culture should prompt investigation for A. otitidis, as nearly 80% of children in our studies referred for myringotomy had evidence of this species in middle ear exudates. This is the first study to attempt to identify components of recent clinical isolates of A. otitidis eliciting some of the pro-inflammatory responses detected in children with these infections.
Range of cytokines elicited by clinical isolates of A. otitidis
The first objective was to determine if cell-free filtrates stimulate inflammatory responses. In addition to IL-8 and IL-12 reported in previous studies, the results of this study found that IL-6, IL-1β and TNF-α were elicited from THP-1 cells. The costs of the detection kits did not allow for assessment of IL-12 or TNF-α in the repeat assays.
Induction of cytokines by individual filtrates of clinical isolates
Cytokine responses to cell-free filtrates of the 39 isolates showed some variation. Very high values of IL-8 were induced by most filtrates. This confirmed the results of combined filtrates tested in the preliminary experiments. Because many of the values obtained were equal to or above the upper limit of the standard curve, detailed comparison between the filtrates for IL-8 was limited. The results for IL-1β were consistently higher than levels elicited by BHI (range = 798–3215 pg/ml, mean = 1610 pg/ml. For IL-6, the responses were consistently lower than for IL-1B (range = 19–81 pg/ml, mean = 40 pg/ml). There were no significant differences in levels of cytokines elicited in relation to either colony type or production of β-like haemolysin.
Effects of enzyme treatment of the filtrates on induction of cytokines
Among the nine filtrates tested, treatment with either lysozyme or Proteinase K had no significant effect on induction of IL-8. For IL-1-β, lysozyme appeared to have little effect; however, Proteinase K decreased IL-1β responses in all but one of the filtrates tested, isolate 30 (P < 0.01). Although this result indicated breakdown of a protein component in the filtrate, there was no unique protein profile associated with the filtrate of isolate 30 compared with the other eight isolates tested.
Because of the low levels, results for IL-6 induced by the filtrates need to be interpreted cautiously; however, a trend similar to that for IL-1β was observed. Lysozyme had a marginal effect in reducing mean IL-6 responses compared with untreated filtrates (P = 0.065). Proteinase K treatment significantly reduced IL-6 production compared with the untreated filtrates (P = 0.004).
Extracellular proteins and biological activities
Results indicated that proteins in the cell-free filtrates might be involved in inducing cytokine responses from THP-1 cells. The untreated cell-free filtrates for isolates 30–39 and their respective aliquots treated with either lysozyme or Proteinase K were combined, concentrated and the concentrated material run on PGE.
Some technical problems were possibly responsible for the poor clarity of some gels. As the protein concentration increases in the gel, aggregates can form, which will be seen after staining. Frequent washes with PBS helped to minimise this. There might be some interference from preservatives used in the concentrator membrane following manufacture. The concentrators should be rinsed thoroughly prior to use.
The results of repeat gels were clearer after following these procedures; however, there was still evidence of small proteins around 25 ku.
The small green colony variants appeared to have a more uniform protein profile compared with the isolates producing the large white colonies. There was no association with any of the major protein bands and the β-haemolysin; however, the molecular masses of cytolysins of Gram-positive species are usually below the cut-off used to assess proteins in this study. 13
Comparison with results of previous studies
The numbers of previous reports on inflammatory responses or pathogenesis of A. otitdis have been limited and attract some criticisms. Most studies on detection of A. otitidis reported difficulty in its isolation; therefore, our recent clinical isolates were used to assess inflammatory responses. Previous investigators used A. otitidis isolates from type culture collections and often the source of the otopathogens tested was not given or referred to as clinical isolates, but no information regarding the site from which the species was isolated or the infection. Type culture strains have invariably undergone multiple subcultures and various storage conditions that might cause loss of typical characteristics of fresh isolates; comparison of the ATCC strain of S. pneumoniae and the recent isolate from a blood culture illustrated the importance of using recent clinical isolates. 8 Previous reports investigated induction of only one cytokine; therefore, except for IL-8, comparison with the current data is limited.
Human peripheral blood monocytic cells have been used in some studies of A. otitidis, but this model introduces potentially confounding genetic and environmental factors. These factors are not confounders for cell lines such as THP-1; 14 therefore, results obtained in the current study were compared with those reported previously for this cell line. Comparison of the current data with that from previous reports must be viewed with detailed knowledge of previous preparation methods and study conditions. In the current study, attempts were made to assess biologically relevant ratios of bacteria to THP-1 cells and concentrations of IFN-γ in the experiments. Previous authors used IFN-γ at concentrations (100 ng/ml) shown in previous studies to be outside ranges detected with virus-infected cells (1–10 ng/ml). Despite these differences, the patterns observed were similar to those reported previously, for example increased responses with cells primed with IFN-γ. The uniform background allowed comparisons of variations in responses elicited by the individual isolates and differences in phenotypes, which had not been done previously.
Previous studies used an ELISA method for cytokine measurement. Although this method has the advantage of lower costs, measurement of different cytokines has to be performed in separate conditions potentially increasing confounding factors. Authors who targeted only single cytokines avoided this problem. The Luminex system was not available in 2000 when the older studies were carried out. It has the advantage of simultaneous measurement of several cytokines from the same aliquot. The main disadvantages are high costs of the kits and the availability of a specialised instrument.
The cytokines tested in this study were examples of those identified in MEE of children with infections due to A. otitidis. Except for heat or chemical treatment of whole cells of A. otitidis, the only assessment of bacterial components involved in induction of inflammatory responses was lipoteichoic acid; this was obtained from Streptococcus pyogenes, not A. otitidis. 2
In conclusion, the presence of soluble materials in the cell free filtrates of A. otitidis elicited pro-inflammatory responses, as reported by earlier researchers. It was shown that peptidoglycan is not likely to be the major component eliciting the responses. There was no correlation between colony type or β-haemolysin production. Proteinase K treatment indicated extracellular proteins might induce inflammatory responses, particularly the 70–75-ku band. Further characterisation of the function and immunogenicity of this component is needed.
Footnotes
Funding
The work was supported by grants from Hunter Children’s Research Foundation; Hunter Medical Research Institute; University of Newcastle; and Hunter Area Pathology Service Microbiology Trust Funds.
