Abstract
Histologic chorioamnionitis, frequently associated with preterm births and adverse outcomes, results in prolonged exposure of preterm fetuses to infectious agents and pro-inflammatory mediators, such as LPS. Endotoxin tolerance-type effects were demonstrated in fetal sheep following repetitive systemic or intra-amniotic (i.a.) exposures to LPS, suggesting that i.a. LPS exposure would cause endotoxin tolerance to a postnatal systemic dose of LPS in preterm sheep. In this study, randomized pregnant ewes received either two i.a. injections of LPS or saline prior to preterm delivery. Following operative delivery, the lambs were treated with surfactant, ventilated, and randomized to receive either i.v. LPS or saline at 30 min of age. Physiologic variables and indicators of systemic and lung inflammation were measured. Intravenous LPS decreased blood neutrophils and platelets values following i.a. saline compared to that after i.a. LPS. Intra-amniotic LPS prevented blood pressure from decreasing following the i.v. LPS, but also caused an increased oxygen index. Intra-amniotic LPS did not cause endotoxin tolerance as assessed by cytokine expression in the liver, lung or plasma, but increased myeloperoxidase-positive cells in the lung. The different compartments of exposure to LPS (i.a. vs i.v.) are unique to the fetal to newborn transition. Intra-amniotic LPS incompletely tolerized fetal lambs to postnatal i.v. LPS.
Introduction
Histologic chorioamnionitis is associated with approximately 70% of preterm births <30 weeks gestation,
1
and chorioamnionitis contributes to adverse outcomes, including periventricular leukomalacia and bronchopulmonary dysplasia.2,3 To model repetitive systemic fetal exposures to inflammatory mediators, Duncan et al.
4
gave small daily i.v. doses of LPS to catheterized fetal sheep over 5 d and measured plasma IL-6 to assess the fetal inflammatory response. The LPS-exposed fetuses had large increases in IL-6 levels after the initial dose, attenuated IL-6 responses following a second dose and no IL-6 responses following subsequent doses, demonstrating endotoxin tolerance to i.v. LPS exposures.
5
Endotoxin tolerance is defined as a decreased inflammatory response to repeated doses of endotoxin following an initial response. Endotoxin tolerance generally is demonstrated
In contrast to systemic exposures, preterm fetuses with chronic and clinically silent chorioamnionitis will have prolonged epithelial exposures of chorioamnion, skin, gut, and lung to infectious agents and pro-inflammatory mediators, such as LPS.7–10 Fetal sheep respond to 4–100 mg intra-amniotic (i.a.) LPS with lung maturation,9,11 while doses <10 µg given by i.v. injection will kill the fetus, demonstrating clear differences in LPS responses depending on where the exposure occurs.
12
We previously showed that i.a. injections of LPS caused fetal lung inflammation because of direct contact of LPS with the fetal airspaces and a systemic fetal inflammatory response syndrome (FIRS).13–15 However, a second i.a. dose of LPS given 5 d after a first dose and 2 d before preterm delivery caused no increase in lung inflammation and a modest increase in selected indicators of FIRS. Intra-amniotic live
Materials and methods
In a pilot study with preterm lambs, we compared the effects of one i.a. LPS dose (10 mg) 5 d before delivery (
Animals and i.a. injections
All animals were studied in Western Australia with approval from the animal ethics/care and use committees of the Cincinnati Children’s Hospital Medical Center (Cincinnati, OH, USA) and the University of Western Australia. Time-mated ewes with singleton fetuses were randomly assigned to receive 10 mg injections of i.a.
The initial goal for ventilation was a tidal volume (VT) of 7 ml/kg with a peak inspiratory pressure limit of 40 cm H2O. The positive end-expiratory pressure and respiratory rate were set at 5 cm H2O and 50 breaths per min, respectively, and were not changed. The initial fraction of inspired oxygen (FiO2) was 0.40, using heated and humidified gas. Oxygen saturation, rectal temperature, blood pressure and heart rate were monitored continuously. Ventilator settings, vital signs and arterial blood gases were recorded at 15, 30, 60, 90 and 120 min of age. The VT was adjusted to achieve partial pressure of CO2 in arterial blood values between 50 and 60 mmHg. FiO2 was adjusted to an oxygen saturation target of 85–95%. Cord blood and blood collected from each lamb at 30 and 120 min were used for complete blood counts. Lambs were anesthetized with continuous infusions of propofol (0.1 mg/kg/min) and remifentanil (0.05 µg/kg/min) via the umbilical venous catheter.
At 30 min of life, lambs were also randomized to receive either 6 µg LPS or an equivalent 2 ml volume of saline by i.v. injection to yield four groups: i.a. Sal/i.v. Sal control; i.a. Sal/i.v. LPS; i.a. LPS/i.v. Sal; and i.a. LPS/i.v. LPS. The lambs were ventilated for 120 min and euthanized with pentobarbital. At the autopsy, tissue was snap-frozen for RNA analysis and fixed in 10% buffered formalin (pH 7.4) for histology. The upper lobe of the right lung was airway inflation fixed with 10% buffered formalin at 30 cm H2O pressure for 24 h.
Cytokine mRNA quantitation
Total RNA was isolated from the frozen lung and liver samples using a modified Chomczynski method. 21 The quantitation of mRNA was performed by RT-PCR after reverse transcription of the mRNA to yield single-stranded cDNA (Verso cDNA kit, Thermo Scientific, Waltham, MA, USA). cDNA was then used as a template with primers and TaqMan probes (Applied Biosystems, Carlsbad, CA, USA) specific to sheep sequences for IL-1β, IL-6, IL-8 and MCP-1. The values for each cytokine were normalized to internal 18 S rRNA values. Final expression data are represented as fold increase over the control value.
Protein analysis
Cytokine quantification was performed using custom sandwich ELISA as described previously.22,23 The following Ab sets were used: IL-1β [coating Ab—rabbit anti-ovine IL-1β and primary Ab guinea pig anti-ovine IL-1β (Seven Hills Bioreagents, Cincinnati, OH, USA)], IL-6 [coating Ab—mouse anti-ovine IL-6 (Chemicon # MAB1004) and primary Ab rabbit anti-ovine IL-6 (Chemicon #AB1839)], IL-8 [coating Ab—mouse anti-ovine IL-8 (Chemicon # MAB10445) and primary Ab rabbit anti-ovine IL-8 (Chemicon # AB1840)] and MCP-1 [coating Ab—rabbit anti-ovine MCP-1 and primary Ab guinea pig anti-ovine MCP-1 (Seven Hills Bioreagents)]. The detection Ab in all the assays was an appropriate species-specific HRP-conjugated Ab. The lowest detection limits and dynamic ranges were IL-1β and IL-6 (0.2–12.5 ng/ml), IL-8 (0.4–25 ng/ml) and MCP-1 (0.1–80 ng/ml). The correlation coefficients were 0.94–0.99 for all assays.
Immunohistochemistry
Immunostaining for myeloperoxidase (MPO) was performed after deparaffinization and rehydration of fixed lung tissue, antigen-retrieval using citric acid buffer, pH 6.0, with microwave boiling as reported. 24 Endogenous peroxidase activity was blocked with methyl alcohol/hydrogen peroxide. Non-specific interactions were inhibited with 4% goat serum during both primary and secondary Ab incubations. Sections were incubated with anti-MPO Ab (Cell marque, Rocklin, CA, USA) (1 : 500). Following overnight (15–16 h) incubation with the primary Ab at 4℃, sections were incubated with the appropriate secondary Ab for 30 min at 20–22℃ (room temperature, 1 : 200). Immunostaining was visualized using a Vectastain ABC peroxidase Elite kit to detect the antigen:Ab complexes (Vector Laboratories, Burlingame, CA, USA). Ag detection was enhanced with nickel–diaminobenzidine, followed by incubation with TRIS-cobalt to give a black precipitate. Nuclei were counterstained with Nuclear Fast Red for photo-microscopy. Blind scoring of inflammation in the lung was done by counting MPO-positive cells in 10 comparable non-overlapping high power fields for each animal (5–6 animals/group). The average cell count per animal was used to compute group averages and SE of the mean.
Data analysis
Results are reported as mean ± SEM. All comparisons were specified
Results
Blood pressure, oxygen requirement and lung function
Birth weight and cord pH were comparable among all lamb groups (Table 1). The fetal exposures to i.a. LPS increased the mean blood pressure following preterm delivery (i.a. Sal: 45.1 ± 2.6 vs IA LPS: 53.2 ± 1.9 mmHg, Intra-amniotic LPS prevented the i.v. LPS-induced blood pressure decreases, but increased the oxygen requirement. Physiologic variables: (A) systolic arterial pressure, (B) heart rate, (C) diastolic arterial pressure, (D) OI = (mean airway pressure*FiO2/pO2), (E) mean arterial pressure, and (F) pressure–volume curves. Grey hash line = i.a. Sal/i.v. Sal; black hash line = i.a. Sal/i.v. LPS; grey solid line = i.a. LPS/i.v. Sal; black solid line = i.a. LPS/i.v. LPS (* Description of lambs at birth and bronchoalveolar lavage at 2 h. BM: body weight; ND: not detected; BAL: bronchoalveloar lavage.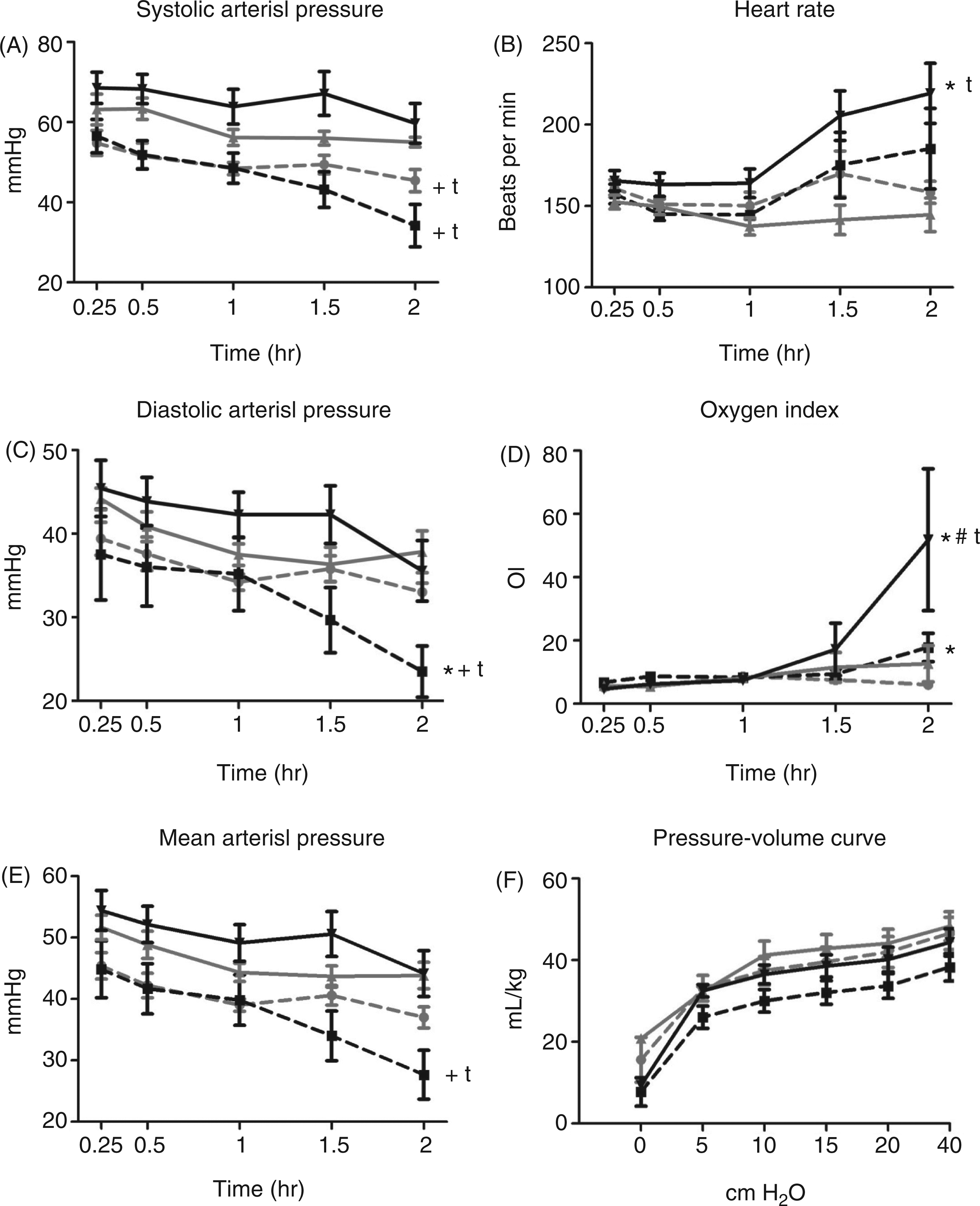

The differences in arterial pressures were accompanied by differences in heart rate responses. Intra-amniotic LPS/i.v. LPS animals had significantly higher heart rates and the i.a. Sal/i.v. LPS animals heart rates trended higher relative to the i.a. Sal/i.v. Sal and i.a. LPS/i.v. Sal groups (Figure 1B).
Oxygen index (OI) is a calculation that includes FiO2, mean airway pressure and partial pressure of arterial oxygen. The i.a. LPS/i.v. LPS lambs had a significantly increased mean OI at 2 h compared with the other groups of lambs that had stable oxygen requirements (Figure 1D). The OI for i.a. Sal/i.v. LPS lambs was higher than i.a. Sal/i.v. LPS lambs. The four groups of lambs had no differences in compliance as measured by the deflation limbs of pressure–volume curves (Figure 1F). There were no differences in body temperature, arterial blood pH or base deficit for the 2 h study period (data not shown).
Blood inflammatory changes
Intra-amniotic LPS significantly increased neutrophil counts in the cord blood compared to lambs receiving i.a. saline (Table 1). Intravenous LPS greatly decreased neutrophils at 2 h by about 10-fold, independent of the previous fetal exposures to saline or i.a. LPS, but owing to higher initial neutrophil count caused by i.a. LPS, neutrophil counts were at control levels at 2 h for the i.a. LPS/i.v. LPS lambs (Figure 2A).
Inflammatory changes in the blood. (A) Neutrophil and (B) platelet counts, and (C–F) plasma cytokine proteins measured by ELISA at 2 h of life. (*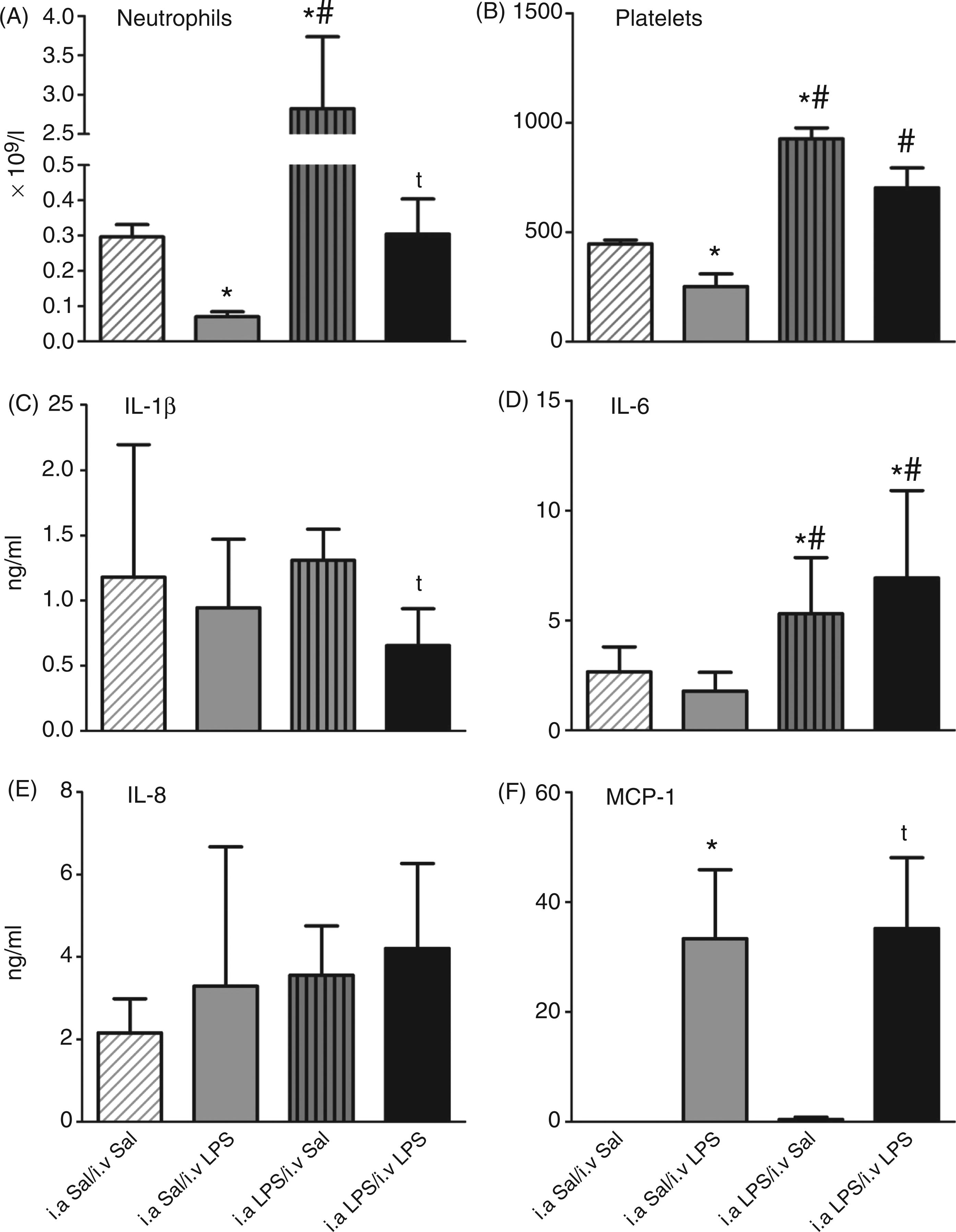
Similarly, cord blood platelet counts of lambs that received i.a. LPS were significantly elevated compared with lambs that received i.a. saline (Table 1). The i.v. LPS significantly decreased the platelets at 2 h, but fetuses that received i.a. LPS had significantly elevated platelet counts compared with i.a. Sal/i.v. LPS lambs (Figure 2B). Therefore, i.a. LPS protected the newborn from the decrease in platelets caused by i.v. LPS.
Monocyte chemotactic protein (MCP)-1 protein measured in the plasma by ELISA significantly increased following postnatal i.v. LPS, independently of the i.a. LPS fetal exposure (Figure 2F). In contrast, plasma IL-6 was increased by i.a. LPS (Figure 2D). IL-1β and IL-8 changed minimally with either i.a. or i.v. LPS (Figures 2C, E). There was no tolerance for plasma cytokine increases caused by LPS.
Tissue inflammatory markers
We also measured inflammatory cytokines in amniotic fluid, fetal lung fluid and bronchoalveolar fluid by ELISA, and cytokine mRNA in the lung and liver by real-time PCR. IL-1β, IL-6 and IL-8 proteins were significantly elevated in the amniotic fluid of animals that received i.a. LPS (Table 1). There was no difference in MCP-1 in amniotic fluid. Similarly, fetal lung fluid of animals exposed to i.a. LPS had significant increases of IL-1β, IL-8 and MCP-1. IL-6 was not measurable in fetal lung fluid. The i.a. LPS increased multiple pro-inflammatory mediators in the amniotic fluid and fetal lungs prior to the ventilation and i.v. LPS exposure.
IL-1β, IL-8 and MCP-1 proteins were elevated in bronchoalveolar lavage (BAL) after 2 h of ventilation from all animals compared with the i.a. Sal/i.v. Sal controls (Table 1). There were no differences among the three LPS-exposed experimental groups except that i.v. LPS increased MCP-1. Neither i.a. nor i.v. LPS changed bronchoalveolar IL-6. Cell counts on cytospins for BAL fluid showed increased neutrophils and monocytes in the i.a. LPS/i.v. Sal group.
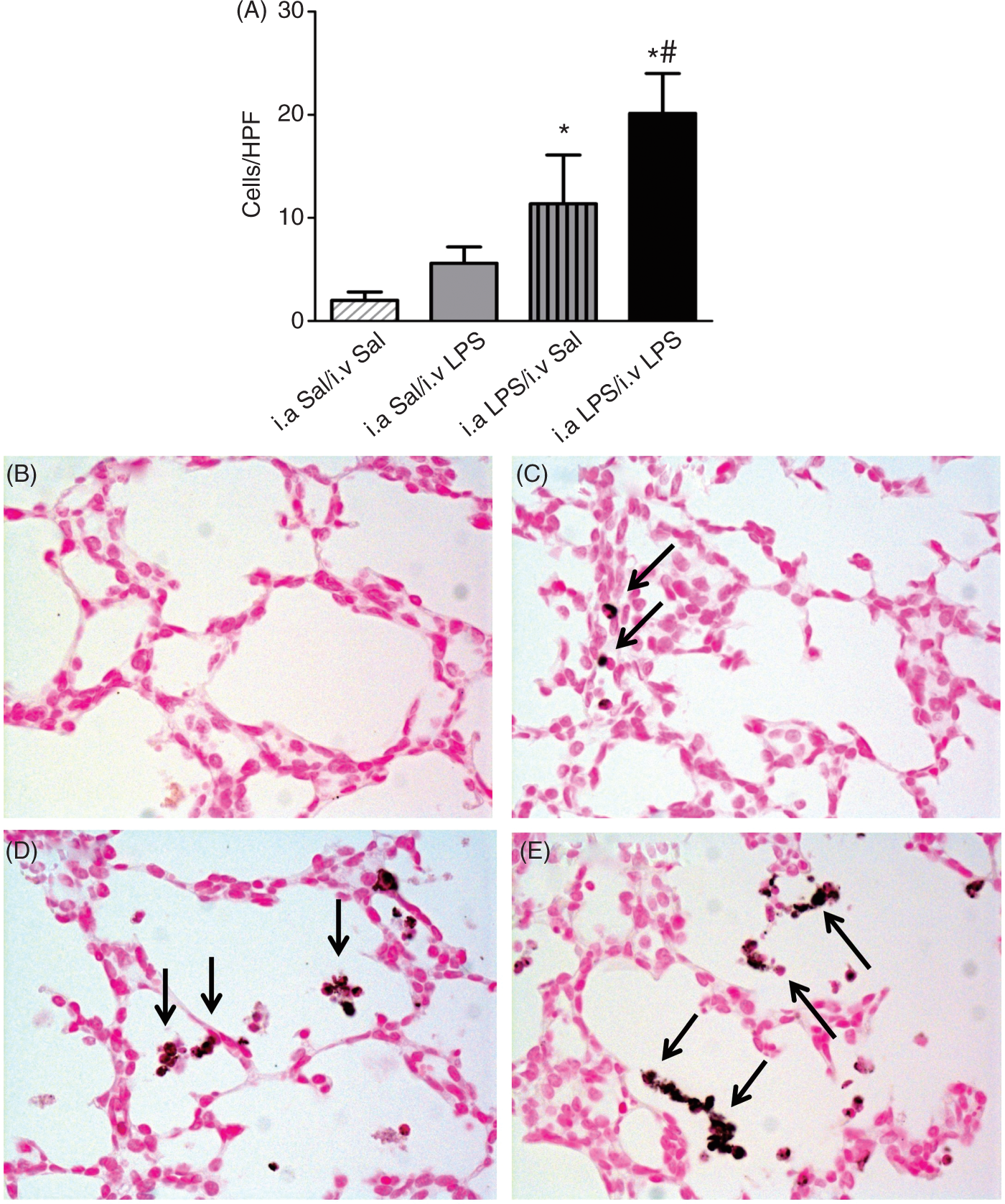
IL-1β, IL-6, IL-8, MCP-1 (Figures 3A, C, E, G) and TNF-α (data not shown) mRNA increased in the lung with a similar pattern of responses. All ventilated animals had significant cytokine increases than lung tissue from a comparison group of unventilated lambs. Both i.a. Sal/i.v. LPS and i.a. LPS/i.v. LPS had large and significant increases relative to i.a. Sal/i.v. Sal lambs and i.a. LPS/i.a. Sal lambs respectively. There were no differences in cytokine mRNA expression between i.a. Sal/i.v. LPS and i.a. LPS/i.v. LPS lambs, indicating no endotoxin tolerance effect on cytokine expression in the lungs. Ventilated Sal/Sal animals had few myeloperoxidase (MPO) positive cells in lung tissue (Figure 4A). The two groups that received i.a. LPS had significantly increased numbers of MPO-positive cells compared with i.a. Sal/i.v. Sal, and the i.a. LPS/i.v. LPS group had higher MPO-positive cells compared with i.a. Sal/i.v LPS. Thus, i.a. LPS primed the lung to increase MPO-positive cells in response to i.v. LPS.
Intravenous LPS exposure increased cytokine mRNA expression in the lung and liver. mRNAs were quantified for lung (A) and liver (B) IL-1β, (C, D) IL-6, (E, F) IL-8 and (G, H) MCP-1 by real-time PCR assays using sheep-specific primers and TaqMan probes. Each cytokine was normalized to 18 s ribosomal protein mRNA (internal control), and levels for each group were expressed as fold increase relative to controls (+ Intra-amniotic LPS exposure increased lung MPO-positive cells. (A) MPO-positive cells were quantified per microscopic high-power field (HPF). Representative 20 × photomicrographs are shown for MPO immunostaining in (B) i.a. Sal/i.v. Sal controls, (C) i.a. Sal/i.v. LPS, (D) i.a. LPS/i.v. Sal and (E) i.a. LPS/i.v. LPS. Arrows point to inflammatory cells in the lung in panels (C), (D) and (E) (*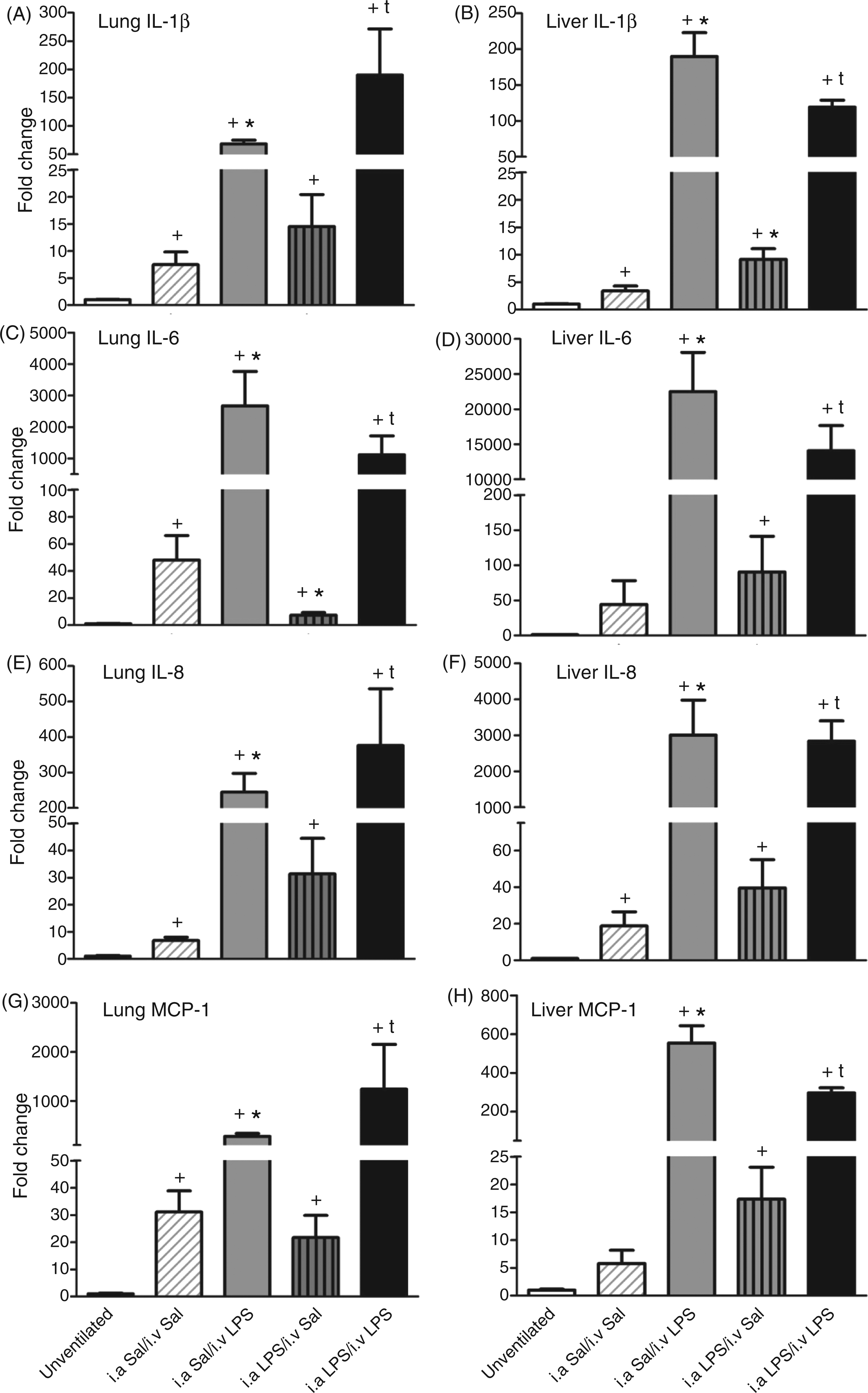
As a representative systemic organ, the liver IL-1β, IL-6, IL-8, MCP-1 (Figures 3B, D, F, H) and TNF-α (data not shown) mRNA also increased similarly to the lungs. All ventilated groups of lambs had higher liver cytokine mRNA than did tissue from unventilated lambs. The i.v. LPS exposure greatly increased mRNAs similarly and independently of the i.a. LPS exposure. There were no differences between i.a. Sal/i.v. LPS and i.a. LPS/i.v. LPS lambs. There was no endotoxin tolerance for liver inflammation.
Discussion
These experiments were designed to simulate the relatively common occurrence of postnatal Gram-negative sepsis following exposure to chorioamnionitis in a preterm infant on mechanical ventilation. 25 The interactions between three different inflammatory insults were tested: (a) antenatal exposure to LPS injected in the amniotic cavity; (b) postnatal exposure to LPS injected intravenously; and (c) postnatal exposure to mechanical ventilation. Of these, the exposure to mechanical ventilation needed to maintain postnatal life support in preterm lambs was common to the experimental groups. Our study should be interpreted in the context that the fetal responses to LPS exposure in the amniotic cavity are very different from responses to i.v. LPS injection. For example, i.a. LPS injections of doses ranging from 4 to 100 mg induce comparable lung inflammatory responses in the fetal sheep 14 and the fetuses do not have severe systemic responses. In contrast, 6 µg of LPS (a 10,000-fold lower dose), given by the i.v. route, caused a significant blood pressure decrease in this study, and similar repetitive doses caused endotoxin tolerance in fetal sheep. 4 The major findings from our study are that two exposures to i.a. LPS protected the fetus from decreased blood pressure and low neutrophil and platelet counts, but did not protect against pulmonary and systemic inflammation induced by postnatal i.v. LPS. Our study is unique in reporting the interactive adaptive effects between antenatal i.a. LPS and postnatal i.v. LPS in a preterm model.
Fetal inflammatory exposures can be conceptualized to occur in two major compartments: the amniotic and systemic. Chorioamnionitis induces inflammation predominantly in the amniotic compartment, which includes the amniotic fluid and the organs that come in physical contact with the amniotic fluid: the epithelia of the amniotic membranes, skin, airway and the gastrointestinal tract. Systemic inflammation includes inflammation in the blood and blood vessel endothelium, and systemic organs, such as liver, spleen and brain. The proximate cause of neonatal morbidities, such as white matter brain injury, is thought to be from systemic inflammation.
26
We previously reported that a prior exposure to LPS or live
Cardiovascular collapse is a significant component of septic shock. 31 Compared with adults, cardiac output in the neonates is more dependent on heart rate. 32 Our fetal lambs exposed to i.a. LPS had higher initial blood pressures than lambs exposed to i.a. saline, demonstrating effects of LPS on the fetal cardiovascular system. This is consistent with previous studies by Polglase et al. 33 and may partly explain the protection from the LPS induced decrease in blood pressure, but not completely. The blood pressure of the i.a. Sal/i.v. LPS lambs was significantly lower at 2 h relative to the i.a. Sal/i.v. Sal lambs. The blood pressure of the i.a. LPS/i.v. LPS lambs was not different than i.a. LPS/i.v. Sal lambs. If a higher initial blood pressure was the only determinant of the difference at 2 h, then we would expect a difference between i.a. LPS lambs after i.v. Sal or LPS. Although we did not measure cardiac output, the only group in which heart rate increased significantly after i.v. LPS was the i.a. LPS/i.v. LPS group. Interestingly, an increased heart rate after i.v. LPS is a characteristic response in the adult. 27 In the pilot single i.a. dose experiments, we did not observe cardiovascular protection. Several mechanisms have been suggested to explain tolerance against cardiovascular and endothelial dysfunction in adult animals. These include reduced production of pro-inflammatory cytokines 34 and NO. 35 However, in our studies the plasma cytokines were no different in the i.a. LPS/i.v. LPS cardiovascular tolerant group than the i.a. Sal/i.v. LPS group. We did not measure other mediators that could contribute to the blood pressure effects.
We previously reported that the preterm sheep fetus exposed to i.a. LPS (10 mg) induced tolerance to another 10-mg dose of i.a. LPS given 5 d later.
17
The tolerance was demonstrated by the decreased lung production of cytokines, activation of leukocytes, decreased liver acute phase response, and decreased responsiveness of blood and lung monocytes to an
This lung injury may be related to our finding of improved blood pressure in the i.a. LPS/i.v. LPS lambs. Polglase et al. 33 reported that lung arteriole muscularization following intra-amniotic inflammation may cause pulmonary hypertension. More lung injury following i.v. LPS demonstrated by MPO-positive cells in our animals potentially resulted in enough pulmonary hypertension to cause right-to-left shunting across a patent ductus arteriosus (PDA), leading to poor oxygenation, but with an improved systemic blood pressure. While respiratory distress syndrome could also lead to poor oxygenation, all of the lambs received surfactant to control for this variable.
Unlike the cytokine storm characteristic of adult systemic inflammatory response syndrome in sepsis and other conditions,
36
chorioamnionitis or i.a. LPS exposure induces a mild systemic inflammatory response.10,37 We also previously reported that i.a. LPS alone stimulated the bone marrow to increase circulating leukocytes and platelets.14,37 Consistent with these previous studies, we found increased neutrophils and platelets in lambs exposed to i.a. LPS (see Table 1). However, postnatal i.v. LPS induced a brisk reduction of circulating neutrophils (likely migration to tissues) and platelets, regardless of antenatal i.a. LPS exposure. Despite this reduction, neutrophils and platelets were at control levels in the i.a. LPS/i.v. LPS animals. Although mechanical ventilation alone induced a modest increase in IL-1β and IL-8 mRNAs in the liver, the major increases in IL-1β, IL-6, IL-8 and MCP-1 mRNAs in the liver were induced by postnatal i.v. LPS. Similarly, increases in plasma MCP-1 were detected after postnatal i.v. LPS. Intra-amniotic LPS did not blunt the cytokine increases either in the liver or in the plasma. The effects of chorioamnionitis on the fetal liver surprisingly showed persistence of inflammatory and metabolic changes for months in sheep.38–40 Our previous studies demonstrated that blood monocytes from preterm lambs exposed to two doses of i.a. LPS were refractory to LPS challenge
Suppression of immune function is a characteristic feature of endotoxin tolerance.5,40 While the decreased inflammation can be protective, decreased innate immune response can increase the risk for sepsis, as has been reported in the adult.41,42 Postnatal nosocomial sepsis is common among very low birth mass preterm infants. 43 While this sepsis has been attributed to the immaturity of the immune system of premature infants, recent evidence has demonstrated that prenatal inflammation affects the function of postnatal innate immunity.17,18 In an observational study, preterm infants with histologic chorioamnionitis had a lower monocyte MHC II expression and a higher risk of sepsis postnatally than infants not exposed to histologic chorioamnionitis. 44 However, early-onset sepsis did not increase the risk for late-onset sepsis in a large retrospective study of preterm infants in the USA. 45 Similarly, another retrospective study of 838 preterm infants in Western Australia reported that histologically diagnosed chorioamnionitis was protective for late onset sepsis. 46 The reasons for discrepant outcomes regarding susceptibility to postnatal sepsis after chorioamnionitis are unclear, but may be related to different patient populations, use of central catheters and other life support devices, and other practices that can influence the risk for sepsis.
A limitation of our study was the short observation period after birth. We might have found greater differences in physiologic or immunologic measurements after longer periods of time. Also, our ability to better characterize the complex effect of LPS in this model was limited by the absence of measurements of cardiovascular function, such as left ventricular output, the presence or absence of a PDA shunt, and measurements of pulmonary and systemic vascular resistance. The high oxygen requirement in the i.a. LPS/i.v. LPS group is probably explained by the increased lung inflammation with right-to-left shunting through the PDA, but that cannot be confirmed with the available data. Although there were large changes in cytokines in multiple organs, other mediators, such as prostanoids, reactive oxidants and components of the complement system associated with sepsis, were not measured, which further limits interpretations of the physiologic responses. Nevertheless, our study reports a novel finding that i.a. LPS protected against an i.v. LPS-induced rapid decrease in systemic arterial pressure, and fall in neutrophil and platelet counts.
Footnotes
Funding
This research was supported by grants HD 57869 and HD 72842 from the National Institutes of Health.
