Abstract
Exposure to pathogen-associated molecular patterns such as LPS can cause an immune refractory state in mammals known as endotoxin tolerance (ET), resulting in a decreased inflammatory response after pathogen contact. This ET concept was used to reduce the severity of an experimentally-induced clinical mastitis. Cows were pretreated with 1 µg LPS per udder quarter and challenged 72 h (group L72EC) or 240 h (group L240EC) later with 500 CFU Escherichia coli. Pretreated animals showed no leukopenia after challenge, no (L72EC), or only slightly (L240EC), elevated body temperature and significantly reduced systemic and local clinical scores compared with cows that were not pretreated. Whereas an increase of milk somatic cell count after the E. coli challenge was abrogated in L72EC animals, it was significantly delayed in the L240EC group. In both pretreated groups the bacterial load in milk was markedly reduced. Based on the expression of inflammation-related genes in lobulo-alveolar mammary tissue, the tolerizing effect of LPS pretreatment is based on the inhibited up-regulation of inflammatory (TNF-α, IL-6, CXCL8, CCL20) and anti-inflammatory genes (IL-10, IRAK-M). These findings indicate that the concept of ET may be usefully applied as mastitis prophylaxis facilitating a rapid response to microbial infection and avoiding dysregulated inflammation.
Introduction
Mastitis is one of the most significant diseases among dairy cattle worldwide. 1 Intramammary infections (IMI) with Escherichia coli frequently cause acute clinical mastitis and may result in severe disease with permanent tissue damage of the udder or even lethal outcome. Dysregulation of initial immune mechanisms is regarded as the main underlying reason for fatal cases. 2 Up to now existing concepts for prophylaxis and therapy of acute clinical mastitis have been unsatisfactory therefore novel approaches are required.
During intramammary E. coli infection, LPS is recognized in an orchestrated interaction of lipopolysaccharide binding protein (LBP), CD14, TLR4 and MD2, inducing the synthesis of pro-inflammatory genes via a tightly regulated signaling cascade. Dysregulation of these balanced molecular mechanisms may lead to hyperactivation of host cells by microbial products and septic shock. Repeated exposure to microbial pathogens or their constituents [pathogen-associated molecular patterns (PAMPs)] can cause a subsequent immune refractory state in mammals resulting in a decreased inflammatory response. 3 This phenomenon is known as ‘endotoxin tolerance’ (ET) and has been widely investigated in the human and murine system in connection with septic shock. 4 This refractory state of the host is not solely induced by LPS (endotoxin) but also by various other PAMPs.5,6 Although the molecular mechanisms are not fully understood, 4 PAMP-mediated tolerance is characterized by a transient inactivation of TLR-mediated induction of distinct inflammatory genes (e.g. TNF-α). In contrast, other TLR-driven genes encoding for antimicrobial effector molecules remain inducible in the state of ET. 7 Thus, PAMP-mediated tolerance is considered to be a potentially protective mechanism against septic shock. 3
In a bovine mastitis model, it was shown that experimental E. coli IMI, introduced twice with an interval of 14 d, was accompanied by a reduction of the acute phase response and severity of clinical signs during the second infection phase. 8 Induction of acute clinical mastitis by infusing 100 µg LPS into udder quarters prevented the establishment of E. coli IMI when the latter was applied 19 h after LPS infusion. 9 Recently, we reported that experimental E. coli mastitis in one udder quarter influenced the reactivity in a neighbouring udder quarter to a subsequent E. coli challenge. 10 Altogether these observations proved that cattle can be hyporesponsive when repeatedly exposed to PAMPs or Gram-negative infection. As there are no studies aiming at protection against IMI by targeting PAMP-mediated tolerance in cattle we decided to use the concept of ET to reduce the severity of an experimentally-induced clinical mastitis. The potential of a low dose intramammary LPS application to induce non- or hyporesponsiveness of the tissue to a subsequent controlled intramammary E. coli challenge was evaluated by recording clinical signs of mastitis. We tested how long the refractory state lasted in the udder. Protection against E. coli IMI and enhanced bacterial clearance by host mechanisms was evaluated by recovering E. coli from mammary secretions. Based on the expression of selected inflammation-related genes in lobulo-alveolar mammary tissue, we analyzed whether the tolerizing effect of LPS pretreatment was based on an inhibited up-regulation of inflammatory or an increased induction of anti-inflammatory and regulatory genes.
Materials and methods
Animals
This study included 26 healthy Holstein cows 3–6 months into their first lactation. The trials were conducted at the Clinic for Ruminants, LMU Munich (Oberschleißheim, Germany) with the approval of the ethics committee of the regional government of Upper Bavaria, Germany (No. 55.2-1-54-2531-108-05). Only animals without previous diagnosis of clinical or subclinical mastitis and a reported somatic cell count (SCC) <50,000/ml were included in the study. Before the trial, weekly cytobacteriologic testing of quarter milk samples ensured that milk contained <50,000 somatic cells/ml and was free of mastitis pathogens. Cows were milked twice daily at 12 h intervals and were fed a total mixed ration based on production level. To achieve comparable hormonal conditions, all animals were synchronized by insertion of CIDR® devices according to the manufacturer's specifications (Pfizer Animal Health, Berlin, Germany) so that at time point of tissue sample collection all animals were at the stage of estrus.
LPS and E. coli inoculum dose
Bovine mastitis isolate E. coli 1303 belongs to the major E. coli phylogenetic group A and has been used in previous studies.10,11 According to a multiplex PCR-based screening for virulence-associated genes of pathogenic E. coli, this strain does not represent an extra-intestinal or intestinal pathogenic E. coli isolate. Only the genes coding for type 1 fimbriae, F17 fimbriae, antigen 43, the ferric citrate siderophore system and the EAST1 toxin could be detected. Bacteria were kept cryo-conserved (Mikrobank-System Cryobank™, Mast Diagnostika, Reinfeld, Germany) for subsequent infections. Bacteria were plated on Columbia sheep blood agar (Oxoid, Wesel, Germany) and incubated (37°C) for 24 h. A few colonies were transferred to a tube of brain-heart infusion broth (Oxoid) and incubated for 6 h (37°C); then, a 100 µl sample was transferred to a tube containing 9.9 ml trypticase soy broth (Oxoid). Serial dilutions were made after 18 h to prepare the desired inoculum dose of 500 CFU/2 ml 0.9% sterile, pyrogen-free saline. The LPS of E. coli 1303 was prepared from the bacteria using the butanol extraction procedure and hydrophobic interaction chromatography, as described. 12 This was followed by a purification step with triethylamine and deoxycholate. 13 Finally the LPS was diluted in 0.9% sterile, pyrogen-free saline to a concentration of 0.2 µg/ml and kept stored in 5 ml aliquots at −80°C.
Milk samples, bacterial recovery and SCC
Throughout the trial, the milk secretion was macroscopically checked for alterations (clots, colour, consistency) every 12 h before regular milking. Simultaneously, quarter milk samples were aseptically collected in sterile vials for bacteriologic examination. Fifteen µl of quarter milk samples were plated on Columbia sheep blood agar, on Violet red bile agar and on Edwards agar (Oxoid). Plates were incubated for 48 h at 37°C and were examined daily for bacterial growth. The animals were milked with a quarter milker (WestfaliaSurge, Bönen, Germany) so that milk from individual udder quarters was collected in separate containers. This enabled determining the SCC with a Fossomatic 5000® (FOSS Electric, Hillerod, Denmark) in 20 ml of the main milk fraction of every individual quarter.
Clinical score for local and systemic signs
Systemic clinical score
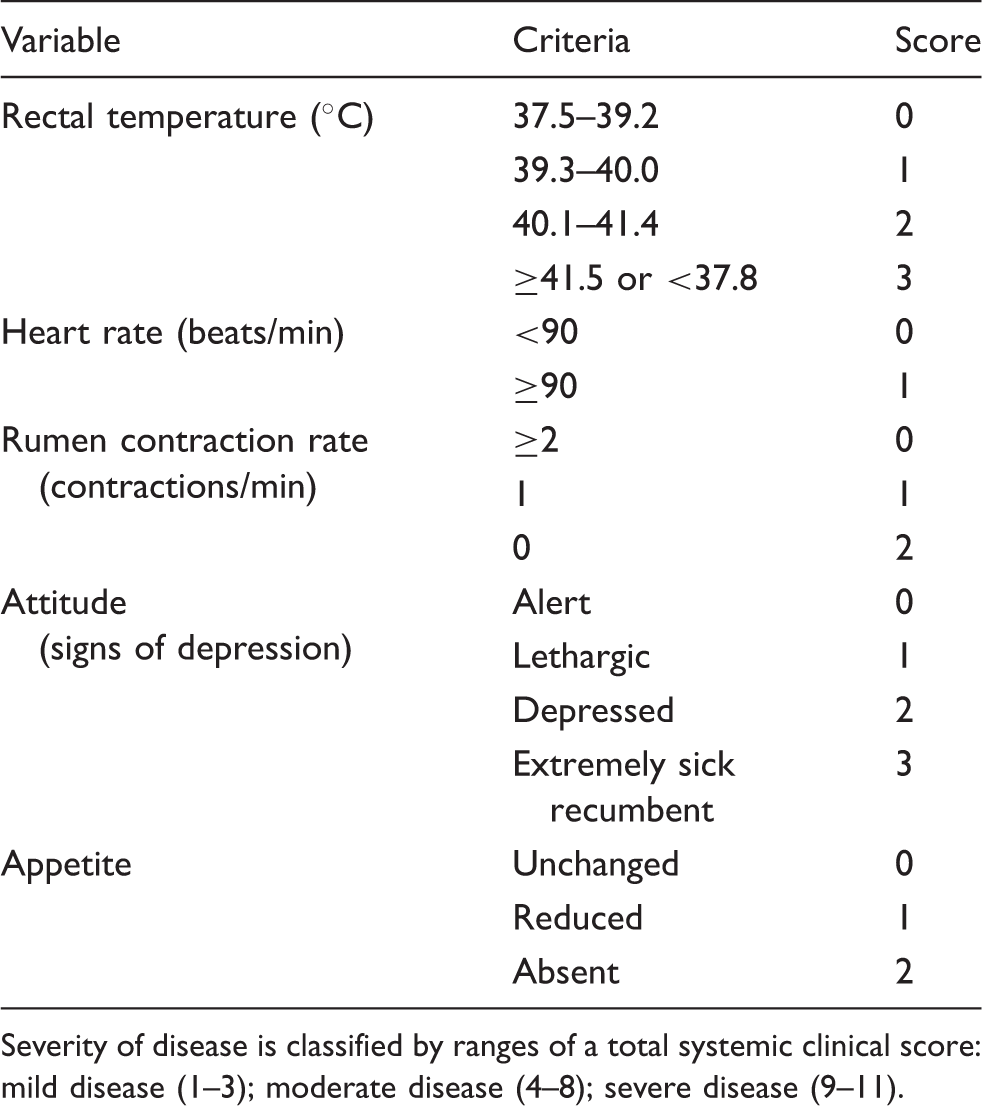
Severity of disease is classified by ranges of a total systemic clinical score: mild disease (1–3); moderate disease (4–8); severe disease (9–11).
Local clinical score
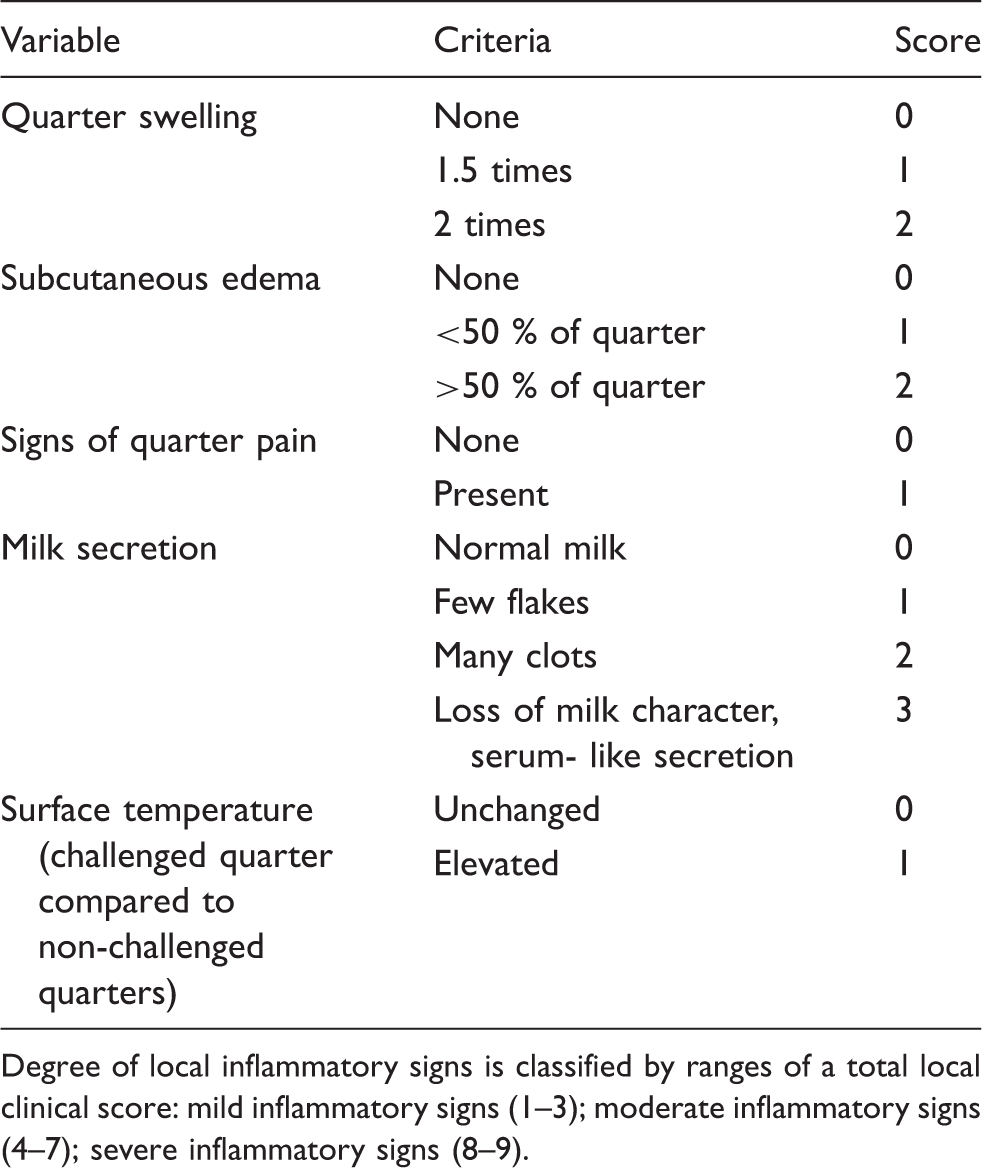
Degree of local inflammatory signs is classified by ranges of a total local clinical score: mild inflammatory signs (1–3); moderate inflammatory signs (4–7); severe inflammatory signs (8–9).
Intramammary LPS infusion, pathogen inoculation and sampling of blood and tissue
Before LPS infusion or E. coli challenge, all animals were entirely milked out after 20 IU Oxytocin (Veyx-Pharma, Schwarzenborn, Germany) were applied intravenously. Before intracisternal application of LPS or E. coli by inserting a teat cannula through the streak canal, the teats were cleaned and disinfected with 70% ethanol. E. coli challenge (500 CFU/2 ml sterile, pyrogen-free 0.9% saline) was carried out during estrus in one udder quarter per animal. The quarter with the lowest SCC was selected for challenge. The set-up of the animal groups is given in Figure 1. In total, 15 animals received intramammary LPS pretreatment in all quarters 72 h (group L72EC, n = 5) or 240 h (group L240EC, n = 10) before E. coli challenge. After completing the first five animals of the L240EC group, the group size was expanded to ten animals because of large interindividual differences in the clinical response to intramammary E. coli challenge. However, only samples from five animals of this group were analyzed by qRT-PCR for technical reasons. In the EC group (n = 5), animals received no LPS infusion before E. coli challenge in one udder quarter. An additional six animals received neither LPS infusion nor E. coli challenge and served as control animals in the qRT-PCR analysis to define baseline gene expression values.
Experimental set-up of intramammary LPS pretreatment and E. coli challenge. Animals received LPS pretreatment (circle, all udder quarters) 72 h (L72EC, n = 5) or 240 h (L240EC, n = 10 for clinical outcome, n = 5 for qRT-PCR analysis) prior to intramammary E. coli challenge (star, one udder quarter). Five animals received no pretreatment prior to E. coli challenge (EC, n = 5). Additionally, six untreated control animals (‘no treatment’) were sampled and only included in the qRT-PCR analysis. All samples were collected from lobulo-alveolar udder tissue from marked areas.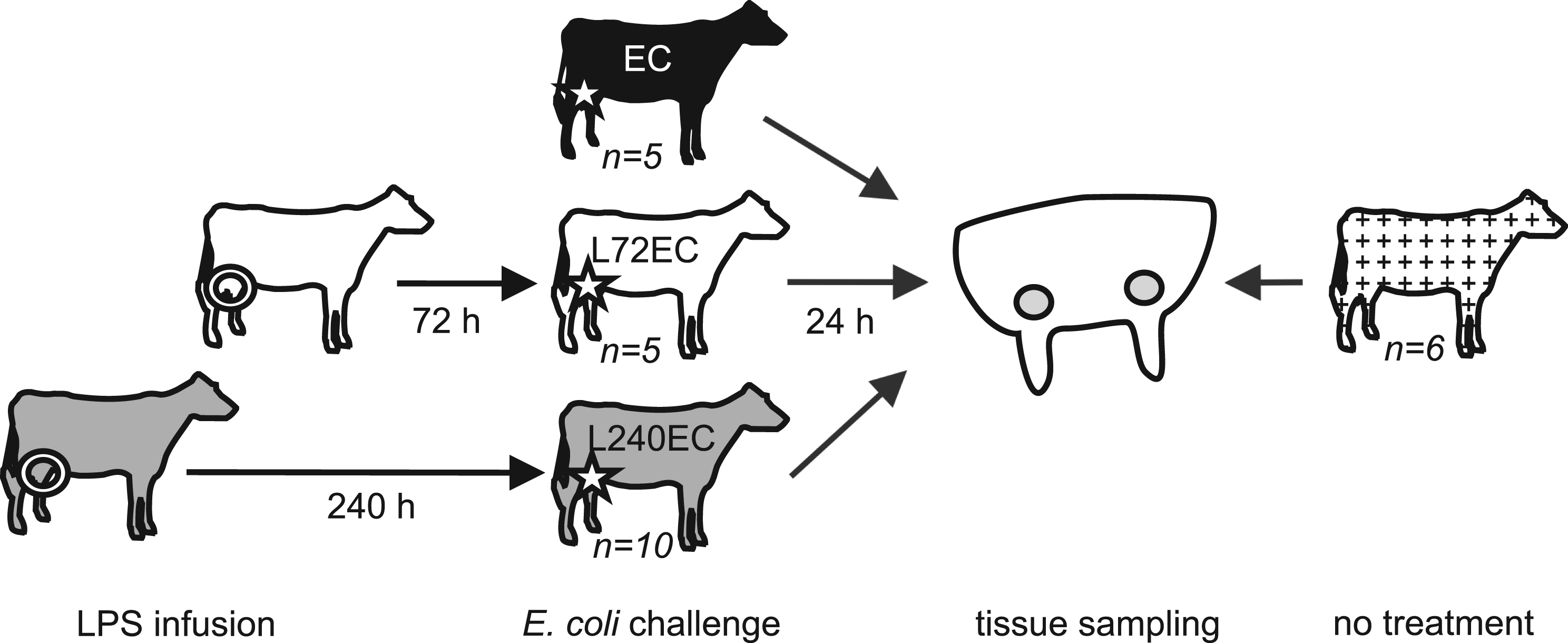
Blood samples (10 ml) were taken from the jugular vein aseptically using EDTA-Vacutainers (Becton Dickinson, Heidelberg, Germany) at 0, 6, 12 and 24 h after LPS infusion and E. coli challenge respectively. Leukocyte counts in blood samples were carried out with a Sysmex pocH100i (Sysmex, Norderstedt, Germany). At trial end (24 h after E. coli challenge), the cows were killed with a penetrating captive bolt gun followed by exsanguination. Tissue samples were aseptically collected from the slaughtered cows within 10 min after killing. A piece of tissue (5 × 5 × 5 cm) was removed from a deeper location of the udder quarter, 7 cm dorsal of the milk cistern, of which a smaller tissue piece (1 × 1 × 1 cm) was snap frozen in liquid nitrogen and served as original material for all further analyses.
RNA extraction and qRT-PCR
Oligonucleotide primers and fluorescence acquisition temperature for quantitative real-time PCR
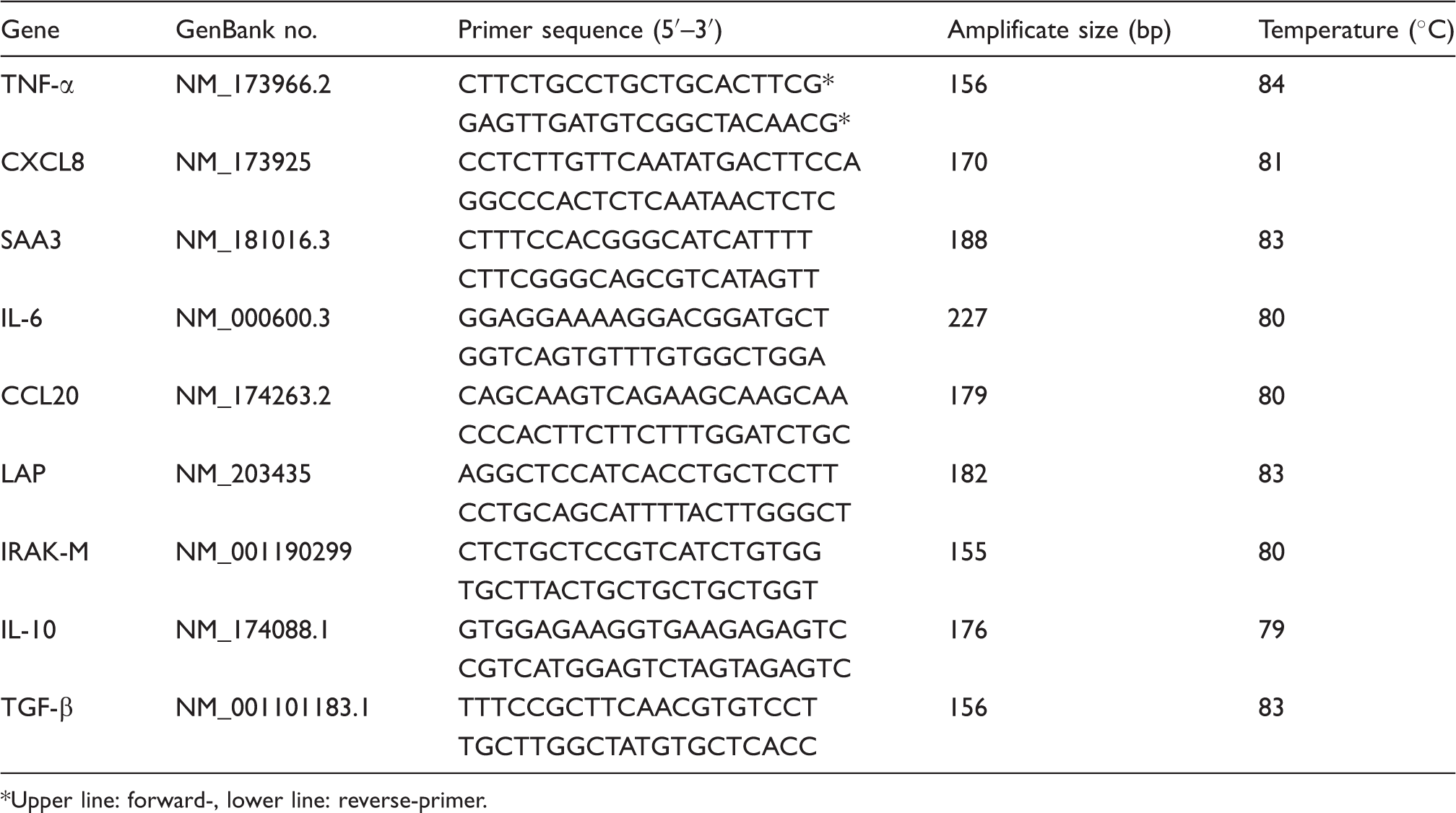
Upper line: forward-, lower line: reverse-primer.
Statistical analysis
Differences in rectal temperature, SCC and local clinical score between different time points owing to LPS infusion were compared using Mann-Whitney U-tests for pairwise comparisons. Differences between groups were compared by ANOVA with Tukey post hoc test for body temperature and blood leukocytes. Differences in SCC dynamics, systemic and local scores between groups were compared using mixed models (SAS, PROC MIXED), adjusting for repeated measures within animals and using Bonferroni for post hoc tests. Differences in bacterial recovery were tested by chi square test for significance. Gene expression was compared using Kruskal-Wallis-Test with post hoc Mann-Whitney U-test using Bonferroni correction for multiple testing (only four specific pair wise comparisons were investigated). Therefore P-values below 0.0125 were regarded as statistically significant.
Results
Clinical effects of intramammary LPS infusion
Rectal temperature, milk somatic cell count and local clinical score following intramammary LPS infusion
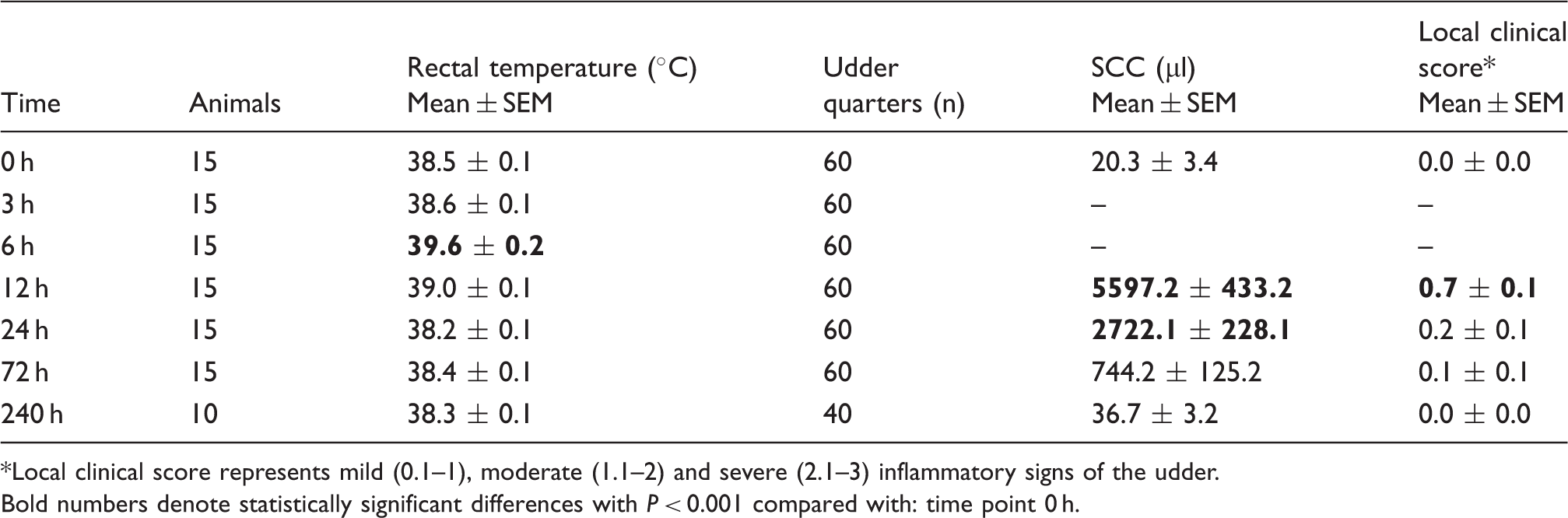
Local clinical score represents mild (0.1–1), moderate (1.1–2) and severe (2.1–3) inflammatory signs of the udder.
Bold numbers denote statistically significant differences with P < 0.001 compared with: time point 0 h.
LPS pretreatment reduces the severity of local and systemic clinical response after intramammary E. coli challenge
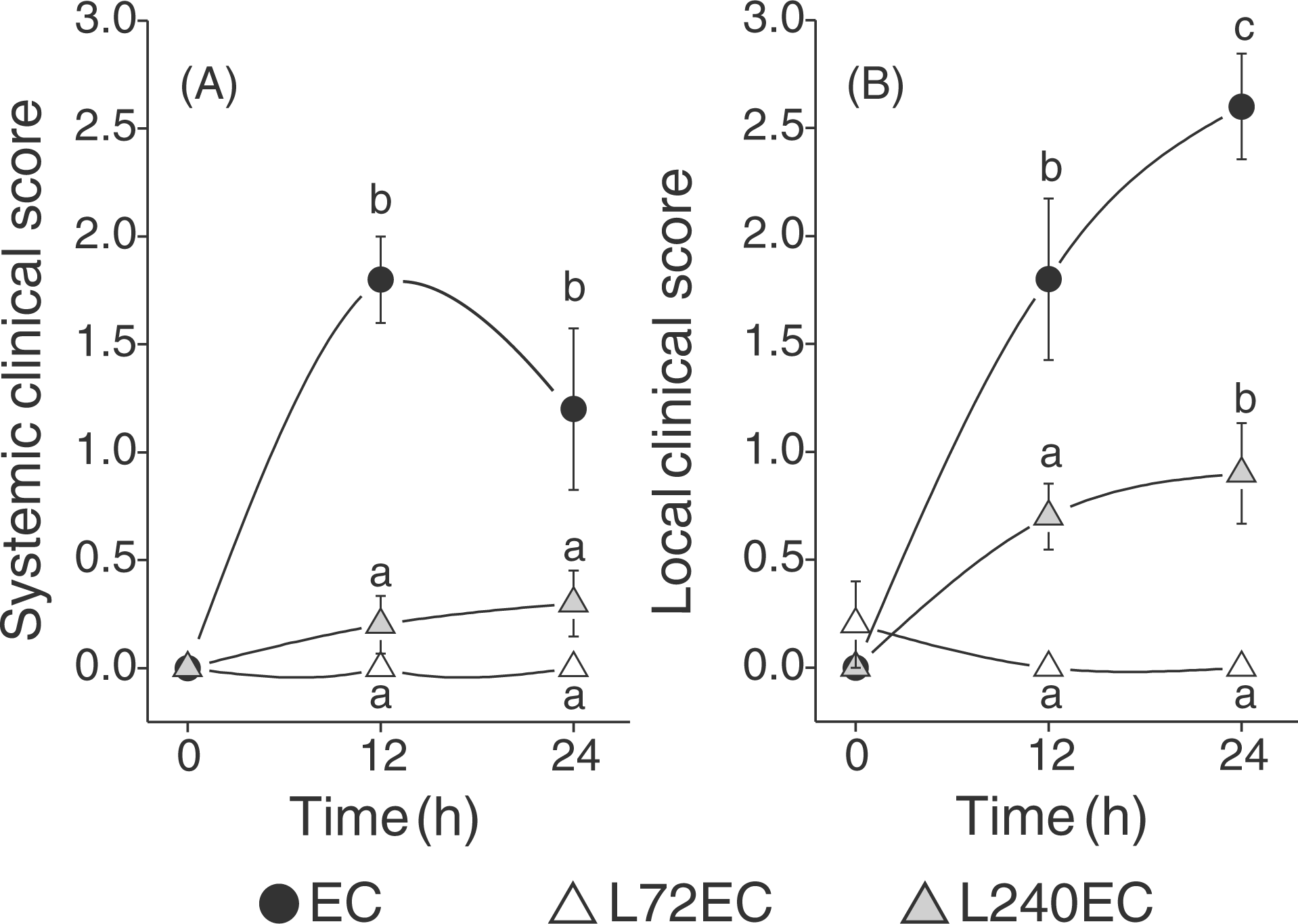
All animals without LPS pretreatment (EC) responded with acute clinical mastitis 12 h after intramammary E. coli challenge. Fever peaked with 41.1 ± 0.2°C after a period of 15–18 h following pathogen inoculation. This was accompanied by a significant leukopenia 12 h and 24 h after E. coli challenge (Figure 2). Animals that had received LPS pretreatment 72 h prior to E. coli challenge (L72EC) developed no fever and no alterations in circulating blood leukocyte numbers. In the group receiving LPS pretreatment 240 h prior to E. coli challenge, animals responded more heterogeneously: 5 out of 10 animals showed an increased body temperature (>39.2°C) but mean temperature did not exceed a maximum of 39.4 ± 0.3°C. There was a significant difference between pretreated groups compared with animals that had received no pretreatment for rectal body temperature and circulating blood leukocytes (Figure 2). Based on the classification of the systemic clinical score, all cows of the EC group showed a moderate clinical response towards intramammary E. coli challenge. Both pretreated groups showed only a mild systemic clinical score (Figure 3). Although a tendency could be seen that L240EC was more strongly affected, there was no significant difference to L72EC because of interindividual differences in L240EC. As early as 12 h after intramammary E. coli challenge, the three groups differed significantly in the characteristics of the local inflammation of the udder (Figure 3). These differences also lasted during the following 12 h. There were no signs of mastitis detectable in the L72EC group 12 and 24 h after intramammary E. coli challenge. In the L240EC group, local symptoms were mostly restricted to 5 out of 10 animals but still these 5 animals showed a significantly milder local clinical score than all animals of the EC group, but stronger symptoms than the L72EC group. Animals without LPS pretreatment invariantly showed udder swelling, subcutaneous edema and strongly altered milk secretion 24 h after intramammary E. coli challenge.
Body temperature (A), blood leukocyte counts (B) and SCC in milk (C) after intramammary E. coli challenge. Animals received intramammary LPS pretreatment in all quarters 72 h (L72EC, n = 5) or 240 h (L240EC, n = 10) before E. coli challenge respectively. One group received no pretreatment (EC, n = 5). In all animals, one quarter was challenged with 500 CFU E. coli at time point 0 h. Alterations in blood leukocytes are given as difference (Δ) to values at time point 0 h. SCC values are only given for E. coli challenged quarters. Values are means with SEM represented by vertical bars. ‘a’ and ‘b’ denote that differences are statistically significant between groups with P < 0.001 (body temperature), P < 0.01 (blood leukocyte counts, SCC for EC, L72EC) and P < 0.05 (SCC for L240EC). Systemic (A) and local (B) clinical score after intramammary E. coli challenge. Severity of systemic clinical signs is classified into mild (0.1–1), moderate (1.1–2) and severe (2.1–3) signs of disease. Local clinical score represents mild (0.1–1), moderate (1.1–2) and severe (2.1–3) inflammatory signs of the udder. Animals received intramammary LPS pretreatment in all quarters 72 h (L72EC, n = 5) or 240 h (L240EC, n = 10) before E. coli challenge in one quarter respectively. One group received no pretreatment (EC, n = 5). Data are mean values with SEM represented by vertical bars. ‘a’, ‘b’ and ‘c’ denote statistically significant differences for local and systemic score between the groups with P < 0.004 (A) or P < 0.04 (B) respectively.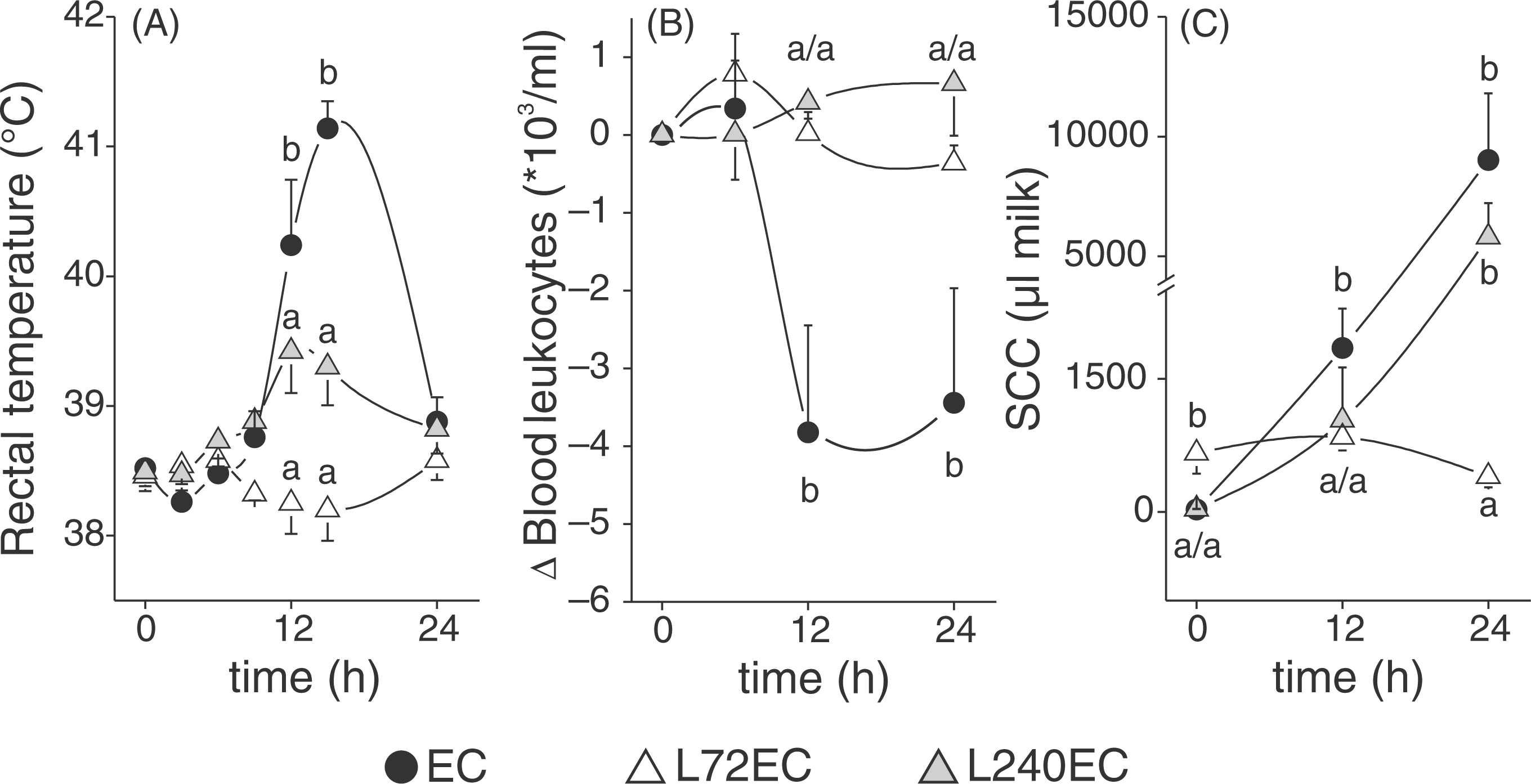
Somatic cell count in the milk of infected udder quarters increased more in the EC than in the L240EC group 12 h after E. coli challenge. L72EC showed no significant alteration of SCC in challenged udder quarters; however, SCC was still elevated (661,000 ± 229,000 cells/ml) at the time of E. coli challenge. Maximal values were detected 24 h after E. coli challenge in the EC and L240EC groups. Owing, to large variance these two groups did not differ significantly statistically, but they were both significantly different from the L72EC group (Figure 2).
Bacterial load is reduced after intramammary LPS pretreatment
Prior to E. coli challenge, the milk of all animals had repeatedly tested negative for major and minor mastitis pathogens. Milk samples were collected 12 h and 24 h following administration of 500 CFU of E. coli in one udder quarter of each animal. All animals that received no pretreatment tested positive for E. coli at every sample collection post-challenge, with consistently high numbers of bacteria (Figure 4). In the L240EC group, only 50% of inoculated quarters tested positive after 12 h with a decreasing tendency toward bacterial load and infected quarters 24 h post-challenge (Figure 4). In the L72EC group, E. coli was recovered only from the milk of one quarter 24 h after challenge. There was a persistently significant reduction of bacterial recovery in the L72EC group compared with the other groups, as well as reduced infected quarters in the L240EC group compared with EC 24 h after intramammary E. coli challenge.
LPS pretreatment reduces bacterial counts in milk after intramammary E. coli challenge. Percentage of E. coli positive udder quarters and ranges of CFU/ml 12 h and 24 h after intramammary E. coli challenge respectively. Animals received LPS pretreatment 72 h (L72EC) or 240 h (L240EC) prior to challenge or no pretreatment (EC). ‘a’, ‘b’ and ‘c’ denote statistically significant differences between the three groups with P < 0.05.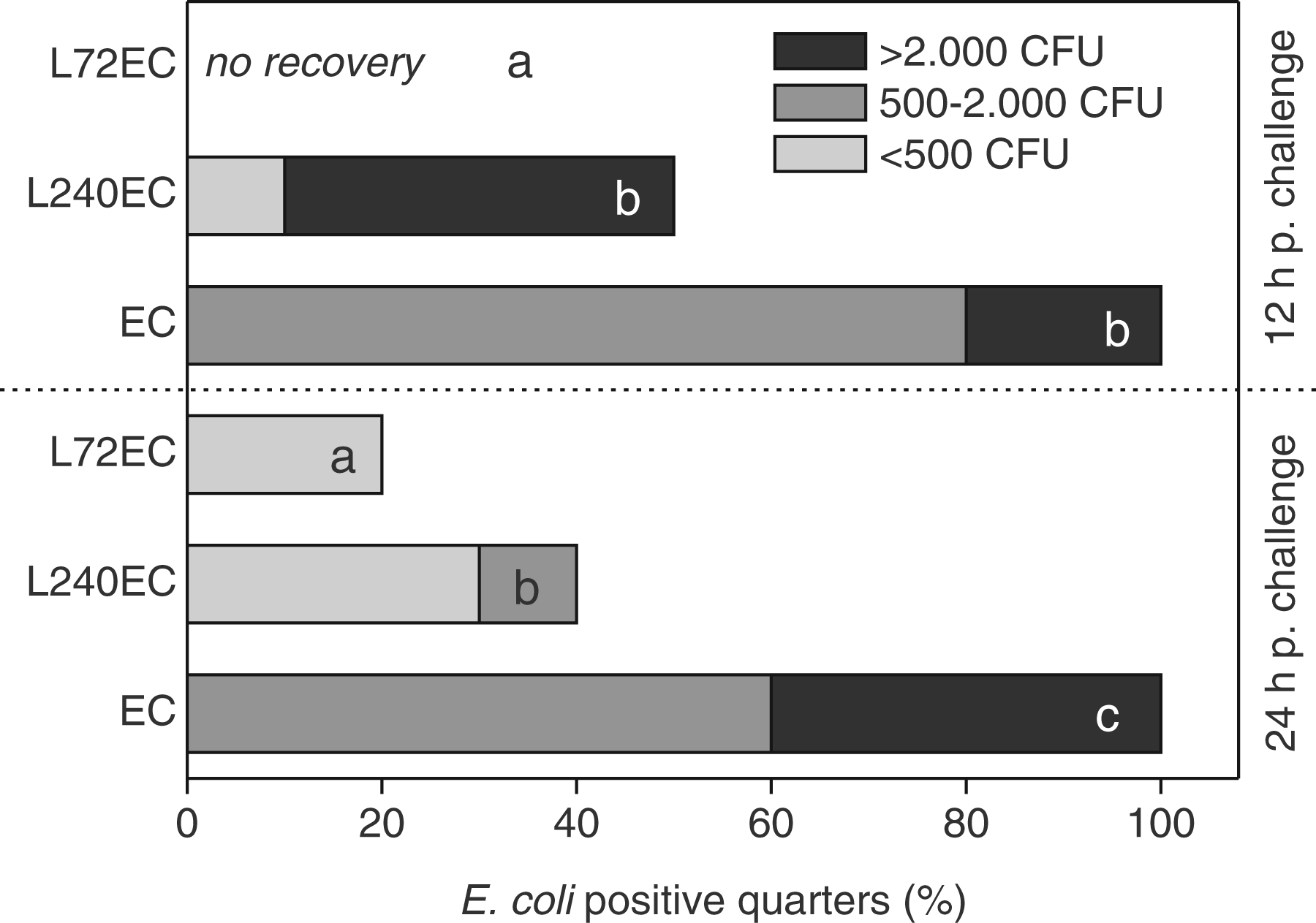
LPS pretreatment reduces the induction of inflammation related genes
The mRNA copy numbers, as measured in udder quarters 24 h after E. coli challenge, are given in Figures 5 and 6. Additionally, four udder quarters of six untreated animals were analyzed for determining baseline expression values for each gene. For IL-6, IL-10 and TGF-β, only one udder quarter of each of five animals was analyzed.
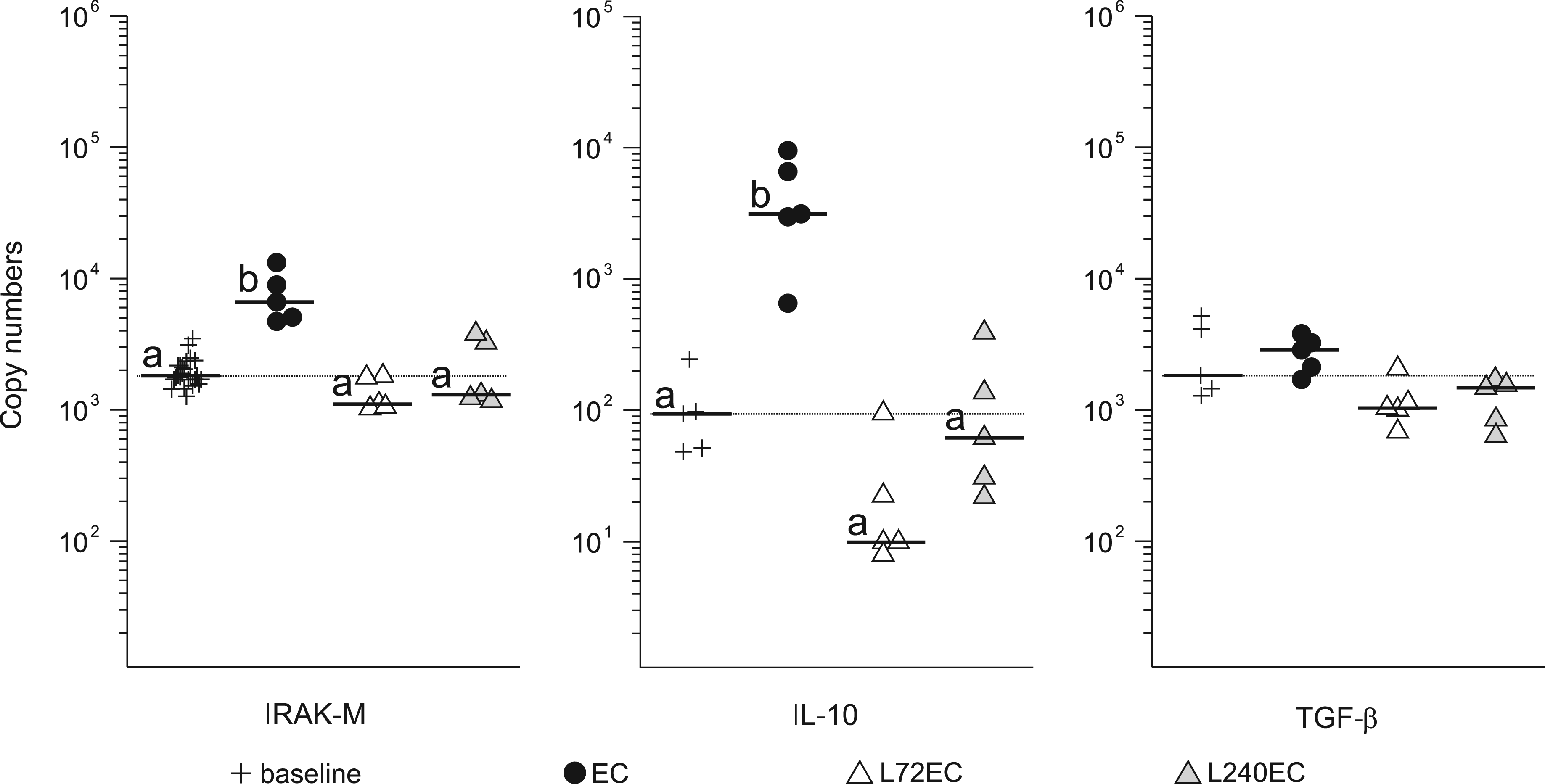
LPS pretreatment inhibits up-regulation of pro-inflammatory related genes in the udder after intramammary E. coli challenge. Absolute mRNA copy numbers obtained from qRT-PCR analysis are expressed as scatter plots of up-regulated genes in lobulo-alveolar mammary tissue 24 h after intramammary E. coli challenge. Animals received LPS pretreatment 72 h (L72EC, n = 5) or 240 h (L240EC, n = 5) prior to intramammary E. coli challenge or no pretreatment (EC, n = 5). Additionally, 24 udder quarters of 6 untreated control animals were used to define baseline levels. In the case of IL-6, only five udder quarters of five control animals were analyzed to define baseline levels. Each dot represents data derived from one sample; horizontal bar represents median values. Dotted line represents baseline expression values (median of group baseline). ‘a’ and ‘b’ denote statistically significant differences between groups with P < 0.0125. LPS pretreatment inhibits up-regulation of anti-inflammatory related genes in the udder after intramammary E. coli challenge. Absolute mRNA copy numbers obtained from qRT-PCR analysis are expressed as scatter plots of up-regulated genes in lobulo-alveolar mammary tissue 24 h after intramammary E. coli challenge. Animals received LPS pretreatment 72 h (L72EC, n = 5) or 240 h (L240EC, n = 5) prior to intramammary E. coli challenge or no pretreatment (EC, n = 5). Additionally, 24 udder quarters of 6 untreated control animals were used to define baseline levels. In the case of IL-10 and TGF-β, only five udder quarters of five control animals were analyzed to define baseline levels. Dotted line represents baseline expression values (median of group baseline). ‘a’ and ‘b’ denote statistically significant differences between groups with P < 0.0125.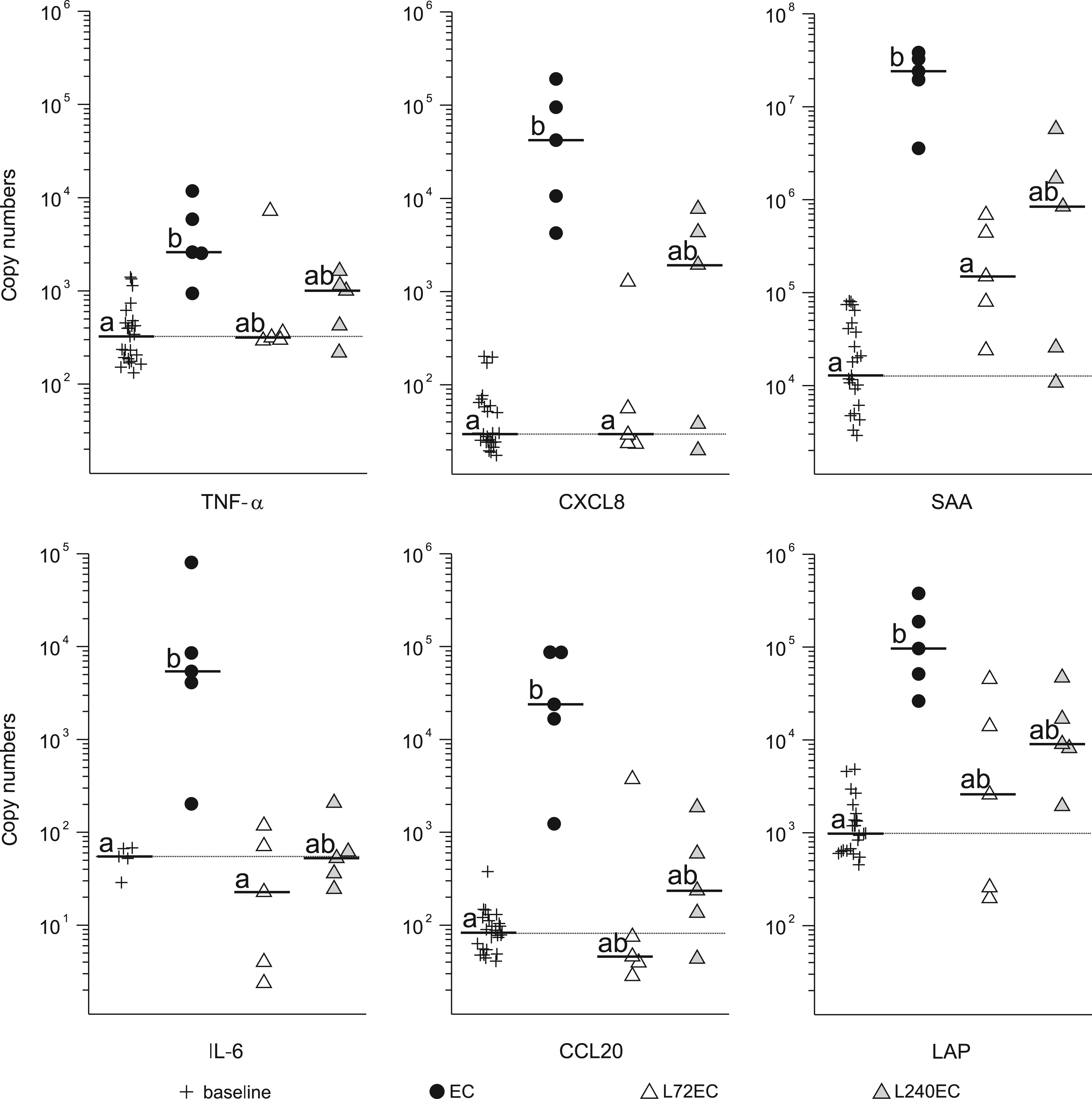
In animals that were not pretreated with LPS (EC), the mRNA abundance of TNF-α, CXCL8, serum amyloid A3 (SAA3), IL-6, CCL20 and the β-defensin lingual antimicrobial peptide (LAP) significantly increased over baseline values in mammary lobulo-alveolar tissue 24 h after E. coli challenge (Figure 5). In animals pretreated with LPS 72 h prior to E. coli challenge, the median mRNA copy numbers for all these genes did not differ significantly from baseline values. Compared with the EC group, expression levels of CXCL8, SAA and IL-6 were significantly different (Figure 5). In animals pretreated with LPS 240 h prior to E. coli challenge, the gene expression level ranged between those of groups EC and L72EC. All the genes were insignificant either to baseline values or to values of group EC, yet IL-6 differed near significance (P < 0.016) to group EC.
Among genes coding for anti-inflammatory and counter-regulatory products, IL-10, IRAK-M, but not TGF-β, were significantly up-regulated in the EC group. In animals pretreated with LPS (L72EC, L240EC), the E. coli challenge did not cause an altered expression compared with baseline values (Figure 6).
Discussion
Acute clinical mastitis in cows is a complex pathophysiologic reaction that has to be tightly regulated, keeping in mind both resolving the infection and limiting damage to the host. In cows developing acute clinical mastitis, the desired cascades begin with pathogen recognition, induction of an initial inflammatory reaction and subsequently anti-inflammatory regulatory pathways. To avoid over-exuberant inflammation the hypo-responsiveness towards repeated confrontation with microbial products seems to be an effective mechanism that markedly decreases the apparent immune response. 3 The aim of the current study was to evaluate this principle by reducing the severity of, or preventing an acute clinical, mastitis in cows. For the desensitization, we chose to infuse LPS into the udder as most of the studies about endotoxin tolerance in mice and humans were performed with this well-characterized PAMP. 4 Moreover LPS has been frequently applied intramammarily to study the pathogenesis of E. coli mastitis in cows.9,16 Our intention was that the immuno-modulatory pretreatment should not lead to signs of clinical mastitis. As toxicity of LPS can vary between different bacterial strains and LPS preparations, 17 the dosage for pretreatment was evaluated in pilot trials to guarantee that LPS from E. coli strain 1303 induced a mild host response (data not shown). When 1 µg LPS were infused in all four udder quarters, a slight, short-lived rise in body temperature and a moderate rise in SCC was observed (Table 4). In most published studies, 100 µg LPS were infused into udder quarters and induced an acute inflammatory systemic and local response in dairy cows.16,18 The clinical findings of our study showed that the LPS-induced response took place, but to a very limited extent. The general attitude of the animals, as well as milk yields and blood leukocyte counts stayed unaffected over the whole period of 240 h (data not shown). About 50% of infused udder quarters showed minimally-altered milk secretion (a few clots) in foremilk 12 h after LPS infusion, but were back to normal 72 h after LPS infusion. This moment was chosen for intramammary E. coli challenge in one group (L72EC). The SCC values at 72 h post-LPS pretreatment were significantly higher compared with baseline values (Table 4). To evaluate potential antimicrobial effects of an initially increased SCC on the applied E. coli inoculum dose in the L72EC group, the second group received an E. coli challenge 10 d after LPS pretreatment. At this time point, the SCC was back to baseline values (Table 4).
The inoculated strain, E. coli 1303, causes intramammary infection and acute clinical mastitis in cows.10,11 As it belongs to the phylogeny group A, this strain represents the majority of E. coli strains isolated from cases of bovine mastitis. 19 The five animals who received only an E. coli challenge (EC) showed almost a uniform acute inflammatory response, which is in line with previous observations. 10 This response included fever paired with blood leukopenia and a fast and high increase of milk somatic cells (Figure 2). The clinical symptoms indicating a systemic response peaked 12 h after challenge, while local symptoms were most apparent 24 h after challenge (Figure 3). The clinical scores were characteristic for cows responding moderately after intramammary E. coli challenge. 2
In contrast, animals that had received LPS pretreatment 72 h prior to E. coli challenge showed no systemic signs of clinical mastitis after inoculation of 500 CFU E. coli (Figure 3). This was also true for local signs except for one animal who showed two small flakes in milk and a second animal who showed an increased firmness of the udder parenchyma, both occurring 12 h after E. coli challenge. The absence of apparent inflammation was underpinned by lack of fever, leukopenia and SCC increase (Figure 2).
This clearly showed that the LPS pretreatment abrogated the clinical response towards an E. coli challenge. Whether this was caused by enhanced clearance of the inoculated bacteria by present milk cells or to a diminished inflammatory response of tissue cells is discussed below.
At the least, this effect was seen 72 h after LPS infusion. To test for how long the LPS effects lasted, group L240EC received an intramammary E. coli challenge 240 h after LPS pretreatment. In summary, this group displayed a higher degree of variability. Therefore we tested 10 animals for the clinical course of infection. The infection was able to establish itself, but the animals showed a clearly reduced disease severity in comparison to group EC. Other than a significantly reduced clinical score (Figure 3A), animals of L240EC showed no alterations in circulating blood leukocytes (Figure 2B), indicating that the systemic response was limited. Major differences from L72EC were shown when the SCC was compared. In contrast to the L72EC group, it had increased significantly in L240EC, but the SCC increase in the L240EC group was delayed in comparison with the EC group (Figure 2C).
It has been established that a delayed PMN influx into the udder can cause higher quantities of E. coli in milk and consequently increase the severity of disease. 20 However, although the SCC increase was delayed in L240EC, only in 50% of challenged udder quarters was E. coli recovered 12 h after challenge. Conversely, a fast SCC increase could not prevent E. coli multiplication in the EC group, where all udder quarters were positively tested for E. coli at every time point after challenge (Figure 4). This suggests that the number of immigrated milk phagocytes is not the sole reason for an inhibited growth of E. coli. This hypothesis is strengthened by the observation that E. coli bacteria could not be re-isolated from quarter samples of L72EC animals (except one quarter, Figure 4) although at 12 h after E. coli inoculation L72EC animals did not differ in SCC values from L240EC animals (Figure 2C).
As a result of the LPS pretreatment the SCC was still elevated (661,000 ± 229,000 cells per ml) in the milk of the L72EC animals (Figure 2C). This may have influenced bacterial growth by an increased initial presence of PMN. In other experimental mastitis models, however, cows with fairly high SCC values (up to 500,000/cells per ml) showed fast E. coli growth and developed clinical mastitis within 12 h. 21 In one L72EC animal E. coli was re-isolated 24 h after challenge, although it had been found negative 12 h before (Figure 4). This finding suggests that the initial inoculum dose had not been completely eliminated but that E. coli was able to multiply in this particular quarter.
As the number of somatic cells per ml of milk seemed not to be decisive for the observed LPS pretreatment effects on the clinical outcome of an E. coli challenge, it seems more likely that LPS pretreatment altered the response of the udder tissue towards E. coli.
To address this point we analyzed the expression of selected genes in tissue samples taken 24 h after E. coli challenge. In line with others’ findings, 7 animals pretreated with LPS and then challenged with E. coli displayed a significantly reduced expression of several genes related to the inflammatory process (Figures 5, 6). Interestingly, the reduced expression or the reduced up-regulation of genes not only affected the inflammatory cytokines TNF-α and IL-6 or the chemokine genes CXCL8 and CCL20, but also the anti-inflammatory gene, IL-10. Thus, the clinical tolerization to LPS not only inhibited the up-regulation of pro-, but also of anti-inflammatory genes. Interleukin-10 is only produced by specific cells, such as dendritic cells, monocytes and, notably, macrophages. The LPS pretreatment may have caused a distortion of the cell composition inside the udder possibly leading to a reduced number of IL-10 producing cells at the time of the E. coli challenge. This may be one reason for the observed absence of up-regulation of the IL-10 mRNA abundance in those udder samples. The type of broad-spectrum inhibitory effect became obvious when the expression of IRAK-M was analyzed, which has been described as a negative regulator of TLR signaling in murine macrophages. Also, IRAK-M knock-out mice proved to be less efficiently LPS tolerizable. 22 The LPS pretreatment of cows, however, did not result in enhanced expression of bovine IRAK-M (Figure 6), although it was induced in the EC group.
Altogether, the data obtained do not point to a selective inhibition of a set of genes, as was observed in studies with LPS-tolerized macrophages. 7 Whether there are still other genes or gene sets that are more highly expressed after LPS pretreatment and E. coli challenge was not further analyzed. Future transcriptome analyses studies could clarify the existence of inducible and non-inducible genes in LPS-tolerant cows and mammary cells.
After LPS pretreatment, we chose two different time points for subsequent E. coli challenge to see whether the protective effects in the udder are progressively reduced. The time-dependent decline of reduced protection was best seen in the local clinical score (Figure 3B) and in the bacterial load (Figure 4). Regarding these parameters, the L240EC group ranged between groups EC and L72EC. Systemically, the LPS pretreatment proved to be long lasting, with systemic clinical scores and the blood leukocyte counts being statistically identical between L72EC and L240EC animals. Surprisingly, this was not true for SCC values in milk (Figure 2C), where L240EC animals showed a delayed, but similar, increase after E. coli challenge compared with group EC, while this increase was totally absent in group L72EC. These differences between 72 h and 240 h post-LPS pretreatment may rely on the differential gene regulation. For instance, in udder tissue we observed a profoundly inhibited expression of IL-6 after E. coli challenge in group L72EC and a reduction in L240EC which was near significance (P < 0.016) after Bonferroni correction. This could explain why systemic effects were less apparent in these animals. In contrast, genes like CXCL8 coding for IL-8 which selectively attracts PMN appeared to be less inhibited in group L240EC (Figure 5).
In conflict with our findings, Rainard and Paape reported that repeated LPS stimulation sensitizes the bovine mammary gland, resulting in a boosted reaction to a following stimulus. 23 However, they infused LPS twice with an interval of 24 h. This interval may be too short to desensitize the udder tissue to a successive stimulus although Lohuis et al., who challenged cows with E. coli 19 h after a previous intramammary LPS infusion, found no inflammatory response. 9 These differences may be a result of the dose and type of the inoculated LPS. In summary, our findings are in line with those of Suojala et al. who showed that a consecutive E. coli mastitis with an interval of 2 wk was accompanied by a reduced acute phase response, although all animals developed acute clinical mastitis. 8
Intramammary LPS pretreatment of cows has been revealed to be effective in preventing and reducing the severity of clinical E. coli mastitis. Future studies should focus on minimizing the side effects of pretreatment on milk quality and extending the period of protection; thus, the concept of PAMP-mediated tolerance induction can be successfully applied to mastitis prevention in cattle.
Footnotes
Acknowledgement
This work was supported by Pfizer Animal Health and the German Research Foundation (DFG FOR 585).
Conflict of interest statement
The authors declare that there is no conflict of interest.
