Abstract
We have previously reported that the co-culture of functional T-cell hybridoma B6HO3 with dying J774 macrophage cells infected with Listeria monocytogenes (LM) results in the production of IFN-γ by B6HO3 cells. Here, we explore the mechanism underlying this phenomenon. We found that IFN-γ production was dependent on IL-18, but that the dying LM-infected macrophages produced no more than 100 pg/ml of IL-18, much less than the amount of IL-18 required for stimulating B6HO3 cells to produce IFN-γ. Furthermore, IL-18 binding protein added to the co-culture was unable to easily gain access to IL-18 for neutralisation. B6HO3 cells formed cell conjugates with J774 macrophages, and IFN-γ-producing B6HO3 cells were spatially and temporally associated with LM-infected macrophage cell death that exhibited neither pyroptosis nor pyronecrosis. These results suggest that the IL-18 produced by dying LM-infected macrophages is released to the interface of the cell conjugates, thereby inducing B6HO3 cells to produce IFN-γ. Based on the present and also previous findings, we propose that IL-18 released from macrophages because of cell death caused by bacteria may be the primary cytokine that triggers the innate IFN-γ production that is required for activating the bactericidal functions of macrophages at early stages of bacterial infection.
Introduction
IL-18 (IL-1F4), one of the 11 members of the IL-1 family, which includes IL-1α, IL-1β, and IL-33,1–5 was first identified as an IFN-γ-inducing factor in the serum of mice with endotoxaemia. 5 However, because IL-18 by itself is a weak stimulator of IFN-γ production by NK and Th1 cells, and it markedly increases IFN-γ production in synergy with IL-12p70, it is generally thought that cooperation between IL-18 and IL-12p70, both of which are produced by macrophages and dendritic cells (DCs) through distinct signalling pathways, leads to the production of IFN-γ, which plays a crucial role in the defence against bacterial infection.1,4,6 The known functions of IL-18 are expanding and IL-18 is now recognised as a multifunctional cytokine. It is thought to be an important regulator of innate immunity and both Th1- and Th2-driven adaptive immune responses depending upon its cytokine milieu. 1 IL-18 is also implicated in the pathogenesis of a variety of autoimmune diseases. 3 Similar to IL-1β, IL-18 is synthesised as a 23-ku biologically inactive precursor and is cleaved into an 18-ku mature bioactive form mostly by inflammasome-activated caspase-1.4,7,8 However, in contrast to IL-1β, IL-18 is constitutively expressed in macrophages/monocytes,9–11 thus not requiring inflammatory stimulation for the production of its precursor. Both IL-18 and IL-1β lack signal peptides and are secreted through non-classical secretory pathways independently of the endoplasmic reticulum and Golgi apparatus.12–15 The biological activity of IL-18 is neutralised by a naturally occurring serum protein called IL-18 binding protein (IL-18BP), which possesses a high binding affinity for IL-18 and is thought of as a modulator of the Th1 cytokine response. 16
In a previous study, 17 we reported that when a functional T cell hybridoma, B6HO3, was co-cultured with dying J774 macrophage cells infected with Listeria monocytogenes (LM), the B6HO3 cells produced IFN-γ in an IL-12p70-independent and a cell–cell contact-dependent manner. Furthermore, a minor subset of αβ T cells and subsets of NK, NKT and γδ T cells, which are generically referred to as ‘innate lymphocytes’, 18 were found to produce IFN-γ in response to the cell death of J774 macrophage and bone marrow-derived macrophage (BMM) cells caused by bacteria such as Listeria, Shigella and Salmonella. 17 Moreover, when BMM cells and J774 macrophages were infected with Staphylococcus aureus (SA), an extracellular bacterium, BMM cells, but not J774 macrophage cells, underwent cell death, and only the dying SA-infected BMM cells induced B6HO3 cells and subsets of innate lymphocytes to produce IFN-γ. 19 On account of the close correlation between bacteria-infected macrophage cell death and IFN-γ production by innate lymphocytes upon their co-culture, we refer to this phenomenon as bacteria-infected macrophage cell death (BIMD)-mediated IFN-γ production. Furthermore, BIMD-mediated IFN-γ production, albeit at low levels, was observed with IL-12-deficient BMM cells infected with bacteria, indicating that both IL-12-dependent and IL-12-independent BIMD-mediated IFN-γ production pathways exist. 19 Furthermore, 20 h after LM infection in mice, a minor subset of αβ T cells and subsets of NK, NKT and γδ T cells in the spleen were found to produce IFN-γ with features similar to the IFN-γ producing cells in our in vitro co-culture system, thus suggesting that BIMD-mediated IFN-γ production may occur in vivo at an early stage of LM infection.20 In this study, we explore the mechanisms underlying BIMD-mediated IFN-γ production using the functional T-cell hybridoma B6HO3. We report that B6HO3 cells form cell conjugates with LM-infected J774 macrophages and that the small amount of IL-18 provided in dying LM-infected macrophages exerts its stimulatory effect at the interface of the cell conjugates, thus causing the IL-12-independent BIMD-mediated IFN-γ production in B6HO3 cells.
Materials and methods
Mice and cells
C57BL/6 mice were purchased from Oriental Yeast (Tokyo, Japan). BMM cells were grown from marrow cells according to the method described previously. 17 The functional T cell hybridoma B6HO3 has been described previously. 17 J774 macrophage cells were maintained in DMEM containing 10% FBS. Nylon wool non-adherent (NWNA) spleen cells were prepared as described previously. 19 Mice were used in accordance with the institutional guidance.
Bacteria
LM bacteria (EGD strain) were grown in tryptic soy broth overnight (16–20 h), washed twice with PBS and stored at −80℃ until use.
Abs and reagents
Anti-murine IL-12 Ab (goat polyclonal Ab), control goat Igs, recombinant murine IL-12p70 and IL-15 were purchased from PeproTech (Rocky, NJ, USA). Anti-IL-18 mAb (93-10C, rat IgG1), control rat isotype mAb (1H5, rat IgG1), and recombinant murine IL-18 were purchased from MBL (Nagoya, Japan). Recombinant IL-18BPd/Fc chimera protein (MM, 44.5 ku) was purchased from R&D Systems (Minneapolis, MN, USA). FITC-conjugated anti-mouse IFN-γ (XMG1.2) was purchased from eBioscience (San Diego, CA, USA). Anti-FcRII/III mAb (2.4G2) was purchased from BD Biosciences (San Jose, CA, USA). ATP was purchased from Wako (Osaka, Japan). LPS from Escherichia coli (O55:B5), ethidium monoazide bromide (EMA) and BSA were purchased from Sigma (St Louis, MO, USA). CA-074-Me (an inhibitor of cathepsin B) was purchased from Calbiochem (Darmstadt, Germany) and was dissolved in 200 mM DMSO. Ac-Tyr-Val-Ala-Asp-CHO (Ac-YVAD-CHO) (an inhibitor of caspase-1 and caspase-4) was purchased from Peptide Institute (Osaka, Japan) and was dissolved in 20 mg/ml H2O. Carboxyfluorescein diacetate succinimidyl ester (CFSE) was purchased from Life Technologies (Grand Island, NY, USA).
Cytokine assay
IL-18 and IFN-γ levels in culture supernatants were determined by ELISA using the ELISA kits from MBL and eBioscience respectively. The detection limits of the IL-18 ELISA kit and the IFN-γ ELISA kit were 25.6 and 15 pg/ml respectively. In some experiments, supernatants were concentrated by using Amicon Ultra 10K (Millipore, Billerica, MA, USA).
Macrophage cytotoxicity assay
Cytotoxicity was assessed by measuring the release of cytosolic lactate dehydrogenase (LDH) into supernatants according to the method described previously. 17 The relative LDH release was calculated as 100 × (experimental release – spontaneous release)/(total release – spontaneous release), where spontaneous release is the amount of LDH activity in the supernatant of uninfected J774 cells and total release is the activity in cell lysates.
In vitro stimulation of B6HO3 hybridoma cells and NWNA spleen cells with dying bacteria-infected macrophage cells
This method was described previously. 17 Briefly, J774 cells (1 × 105 cells per well) or BMM cells (8 × 104 cells per well) were cultured in a 96-well microculture plate overnight. After the microculture plate was washed with DMEM without antibiotics, LM bacteria were inoculated into each well of the microculture plate at a multiplicity of infection (MOI) of 50, and the plate was centrifuged at 800 × g for 5 min and then incubated at 37℃ for 1 h. B6HO3 cells (50 × 103 cells per well) or NWNA spleen cells (8 × 105 cells per well) were then added to the microculture plate, which was incubated at 37℃ for 20 h. During this incubation period 30–60% of LM-infected macrophages underwent cell death. 17 For the B6HO3 cells, culture supernatants were harvested and assessed for IFN-γ levels by ELISA. For NWNA spleen cells, intracellular IFN-γ staining was conducted and analysed on a FACScan (BD Biosciences, San Jose, CA, USA), according to the method described previously.17,19
In vitro stimulation of B6HO3 hybridoma cells with LPS-plus-ATP-stimulated macrophage cells
J774 cells (1 × 105 cells per well) were seeded into a 96-well microculture plate and cultured overnight in the presence or absence of 50 ng/ml LPS. The LPS-pretreated cells were pulsed for 20 min with 5 mM ATP, washed three times with medium, and then cultured for 20 h with or without B6HO3 cells (50 × 103 cells per well). Supernatants collected from cultures without B6HO3 cells were assessed for IL-18 levels and LDH release by ELISA. Supernatants collected from cultures with B6HO3 cells were assessed for IFN-γ levels by ELISA.
Cellular conjugate formation between J774 macrophage cells and B6HO3 cells
B6HO3 cells (1 × 106 cells/ml) were suspended in pre-warmed PBS containing 0.1% BSA and incubated at 37℃ for 10 min with 2.5 µM of CFSE, and then washed three times with DMEM containing 5% FBS. J774 cells (6 × 105 cells per well) were cultured overnight to adhere to the bottom of the wells of a 24-well microculture plate and then infected with LM (MOI: 50) in the same manner as described above. The CFSE-labelled B6HO3 cells (3 × 105 cells per well) were co-cultured with the J774 cells infected or uninfected with LM. After a 4-h incubation period the microculture plates were vigorously washed three times with PBS containing 10 mM EDTA to remove B6HO3 cells and then observed under a fluorescence microscope. For time-lapse microscopy, J774 cells were cultured overnight in a glass-bottomed dish (Iwaki, Tokyo, Japan) and then co-cultured for 5 h with CFSE-labelled B6HO3 cells. After non-adherent B6HO3 cells were washed away, the dish was placed in an inverted microscope with epifluorescence optics. An environmental chamber (Tokai Hit, Fujinomiya, Japan) was used to maintain the temperature at 37℃ and to provide an atmosphere of 5% CO2 in humid air. A fluorescence image was taken to identify hybridoma cells and then sequential bright field images were captured every 75 s. The images were assembled in a Quick Time movie format (six frames per second) with Quick Time Pro 7.6 (Apple, Cupertino, CA, USA).
Morphological observation of in vitro co-cultured cells
A culture glass was laid on the bottom of each well of a 24-well microculture plate and J774 cells suspended in culture medium without antibiotics were seeded into the wells (6 × 105 cells per well) and they were cultured at 37℃ overnight in a CO2 incubator. After each well was washed with DMEM without antibiotics, LM bacteria were inoculated into the wells at a MOI of 50 and the microculture plate was centrifuged at 800 g for 5 min and then incubated at 37℃ in a CO2 incubator. After a 1-h infection period, B6HO3 cells suspended in culture medium containing 100 µg/ml of gentamicin were added to the wells (3 × 105 cells per well). The microculture plate was incubated at 37℃ for 11 h in a CO2 incubator. GolgiStop (Monensin; BD Biosciences) was added for the last 6 h of culture. Culture glasses were then removed, rinsed in PBS and then 100 µl of PBS containing both the blocking Ab (anti-FcRII/III mAb) and 5 µg/ml of EMA were mounted on the culture glasses. The culture glasses were incubated at 4℃ for 20 min, washed carefully twice with PBS, and then placed at a distance of 10 cm from a fluorescent lamp for 10 min. The cells on the glasses were then permeabilised for 20 min by mounting 100 µl of permeabilisation solution (Cytofix/Cytoperm Plus kit; BD Biosciences) on the culture glasses. Then, FITC-conjugated anti-IFN-γ mAb was applied onto the culture glasses. After 30 min, the culture glasses were washed carefully twice with PBS and they were observed under a florescence microscopy.
Kinetics of LDH release from LM-infected J774 macrophage cells and IFN-γ production by B6HO3 cells co-cultured with dying LM-infected macrophages
J774 macrophages cells (1 × 105 cells per well) were cultured overnight in a 96-well microculture plate and infected with LM (MOI: 50) in the same way as described above. Then, they were co-cultured with B6HO3 cells (50 × 103 cells per well) for 20 h. Supernatants from LM-infected J774 culture and from the co-culture of LM-infected J774 cells with B6HO3 cells were collected every hour for the first 11 h. Supernatants were assessed for LDH and IFN-γ levels by ELISA. LDH release and IFN-γ production were the highest at 20 h. These maximum values were used as the control and represent 100% of LDH release and 100% of IFN-γ production. The percentage of LDH release and IFN-γ production compared with each control was calculated at each time point and plotted.
Caspase-1 and cathepsin B inhibition
J774 cells (1 × 105 cells per well) cultured overnight in a 96-well microculture plate were pretreated for 20 min with or without 20, 100 and 200 µM of the caspase-1 inhibitor ac-YVAD-CHO or for 60 min with 100 µM of CA-074-Me or vehicle (0.05% DMSO). Then, they were infected with LM (MOI: 50) in the presence of each inhibitor. After a 1-h infection period, LM-infected J774 cells were co-cultured with or without B6HO3 cells (50 × 103 cells per well) for 20 h in the presence of each inhibitor and the levels of IL-18, IFN-γ and LDH in culture supernatants were determined by ELISA.
Results
LM-infected dying J774 macrophage cells induce B6HO3 hybridoma cells to produce IFN-γ in an IL-18-dependent manner despite macrophages secreting an insufficient amount of IL-18 for inducing IFN-γ
To evaluate the role of IL-18 in BIMD-mediated IFN-γ production, B6HO3 hybridoma cells were co-cultured with dying LM-infected J774 macrophage cells in the presence of increasing amounts of neutralising mAb specific for IL-18 or an isotype control mAb (Figure 1). The effect of neutralising Ab specific for IL-12 on BIMD-mediated IFN-γ production was also examined. As shown in a previous study,
17
anti-IL-12 Ab did not inhibit IFN-γ production by B6HO3 cells (Figure 1A). In contrast, anti-IL-18 mAb completely inhibited IFN-γ production at a concentration of 10 µg/ml (Figure 1B), indicating that BIMD-mediated IFN-γ production by B6HO3 cells is dependent upon IL-18. The addition of control Abs to the co-culture system slightly reduced BIMD-mediated IFN-γ production by B6HO3 cells without dose dependency (Figure 1A, B). This reduction might be related to the possible occurrence of non-specific interaction between bacterial infected macrophages and control Abs in our in vitro co-culture system.
Effect of IL-18 neutralisation on BIMD-mediated IFN-γ production in B6HO3 cells, and the effect of IL-18 and IL-12p70 on IFN-γ production in B6HO3 cells. (A, B) B6HO3 cells were co-cultured for 20 h with dying LM-infected J774 cells in the presence of increasing amounts of anti-IL-12 Ab (♦) or control goat Igs (□) (A), or increasing amounts of anti-IL-18 mAb (♦) or control isotype mAb (□) (B). (C) B6HO3 cells were cultured for 20 h in medium containing 2.5 ng/ml IL-18 in the presence of increasing amounts of anti-IL-18 mAb (♦) or control isotype mAb (□). The levels of IFN-γ in culture supernatants were measured by ELISA and are shown as the percentage of each IFN-γ level compared to the IFN-γ level of the control culture without Ab. The control IFN-γ level was 3.2 ng/ml in A and B, and 13.2 ng/ml in C. (D–G) B6HO3 cells (D, E) and NWNA spleen cells (F, G) were cultured for 20 h with increasing amounts of IL-18 in either the absence (♦) or presence (○) of 10 pg/ml of IL-12p70 (D, F), or with increasing amounts of IL-12p70 (E, G). IFN-γ levels in culture supernatants were measured by ELISA (D, E), and the percentage of IFN-γ+ cells among NWNA spleen cells was evaluated by flow cytometry (F, G). Representative data from one of 2–3 independent experiments are shown.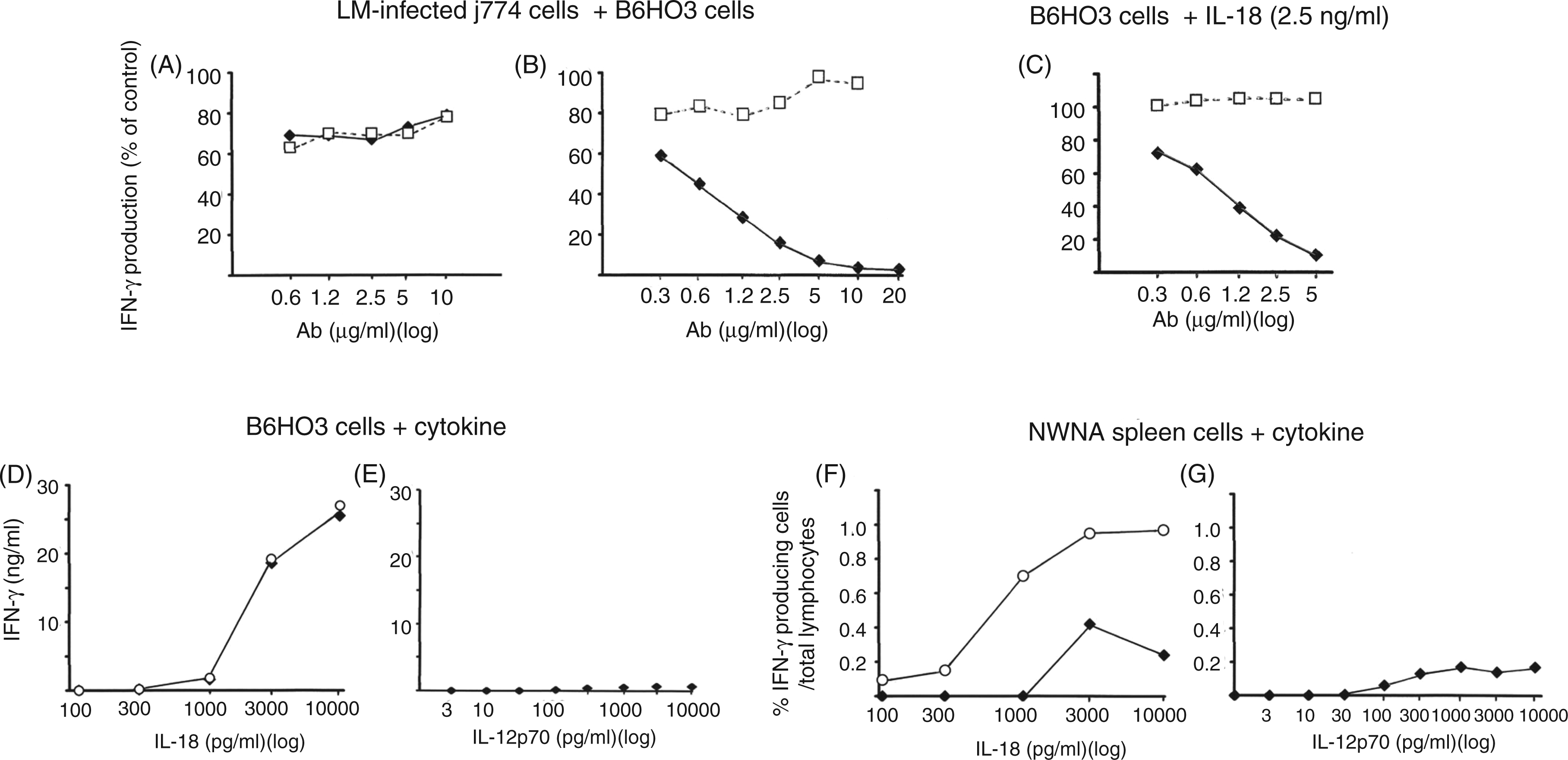
As B6HO3 cells produce IFN-γ when cultured in medium containing IL-18 (see below), the effect of anti-IL-18 mAb on IL-18-induced IFN-γ production in B6HO3 cells was examined by adding increasing amounts of anti-IL-18 Ab to the B6HO3 cells that were cultured in the presence of varying amounts of IL-18. An inhibition curve similar to that for BIMD-mediated IFN-γ production (Figure 1B) was obtained when the cells were cultured in the presence of IL-18 at a concentration as high as 2.5 ng/ml (Figure 1C).
The ability of IL-18, IL-15 and IL-12p70 to induce IFN-γ production by B6HO3 cells and NWNA spleen cells was further examined by culturing B6HO3 cells and NWNA spleen cells for 20 h in the presence of increasing concentrations of each cytokine (Figure 1D–G). B6HO3 cells responded to IL-18 at concentrations of more than 1 ng/ml with robust production of IFN-γ (Figure 1D). However, B6HO3 cells did not produce appreciable amounts of IFN-γ even in the presence of high concentrations of IL-12p70 (Figure 1E) nor did they respond to IL-15 with the production of IFN-γ (data not shown). A very small proportion of NWNA spleen cells produced IFN-γ in response to IL-18 (Figure 1F) and IL-12p70 (Figure 1G) at concentrations greater than 1 ng/ml and 100 pg/ml respectively. When a suboptimal concentration of IL-12p70 (10 pg/ml) was added to B6HO3 cell and NWNA spleen cell cultures together with increasing amounts of IL-18, a synergistic effect between IL-18 and IL-12p70 was observed with NWNA spleen cells (Figure 1F), but not with B6HO3 cells (Figure 1D). Overall, these results indicate that IL-18 by itself can stimulate B6HO3 cells to produce IFN-γ and that IL-18 was responsible for the IL-12p70-independent BIMD-mediated IFN-γ production in B6HO3 cells.
We next determined the amount of IL-18 produced by dying LM-infected macrophage cells after 20 h of culture. As shown in Figure 2, LM-infected J774 cells secreted approximately 60 pg/ml IL-18. As we had shown in our previous studies that dying LM-infected BMM cells also induce BIMD-mediated IFN-γ production,17,19 we also measured IL-18 levels secreted by LM-infected BMM cells (Figure 2). Similar to LM-infected J774 cells, LM-infected BMM cells secreted low amounts of IL-18. Compared with the results shown in Figure 1(D, F), it is evident that this low level of IL-18 is far below the threshold for inducing B6HO3 cells and NWNA spleen cells to produce IFN-γ. Thus, these results are compatible with our previous report that culture supernatants from dying bacteria-infected macrophages, when added to B6HO3 cell and NWNA spleen cell cultures, did not have the ability to induce IFN-γ production.
17
Compared with the results shown in Figure 1 (B, C), these results also indicate that an extremely large amount of anti-IL-18 mAb was necessary to neutralise the low amounts of IL-18 secreted by dying LM-infected macrophages in our in vitro co-culture system.
IL-18 production by bacterial-infected macrophages. J774 cells and BMM cells were infected with LM (MOI: 50) for 20 h, and IL-18 levels in culture supernatants were measured by ELISA. Data are shown as mean ± SD from three independent experiments.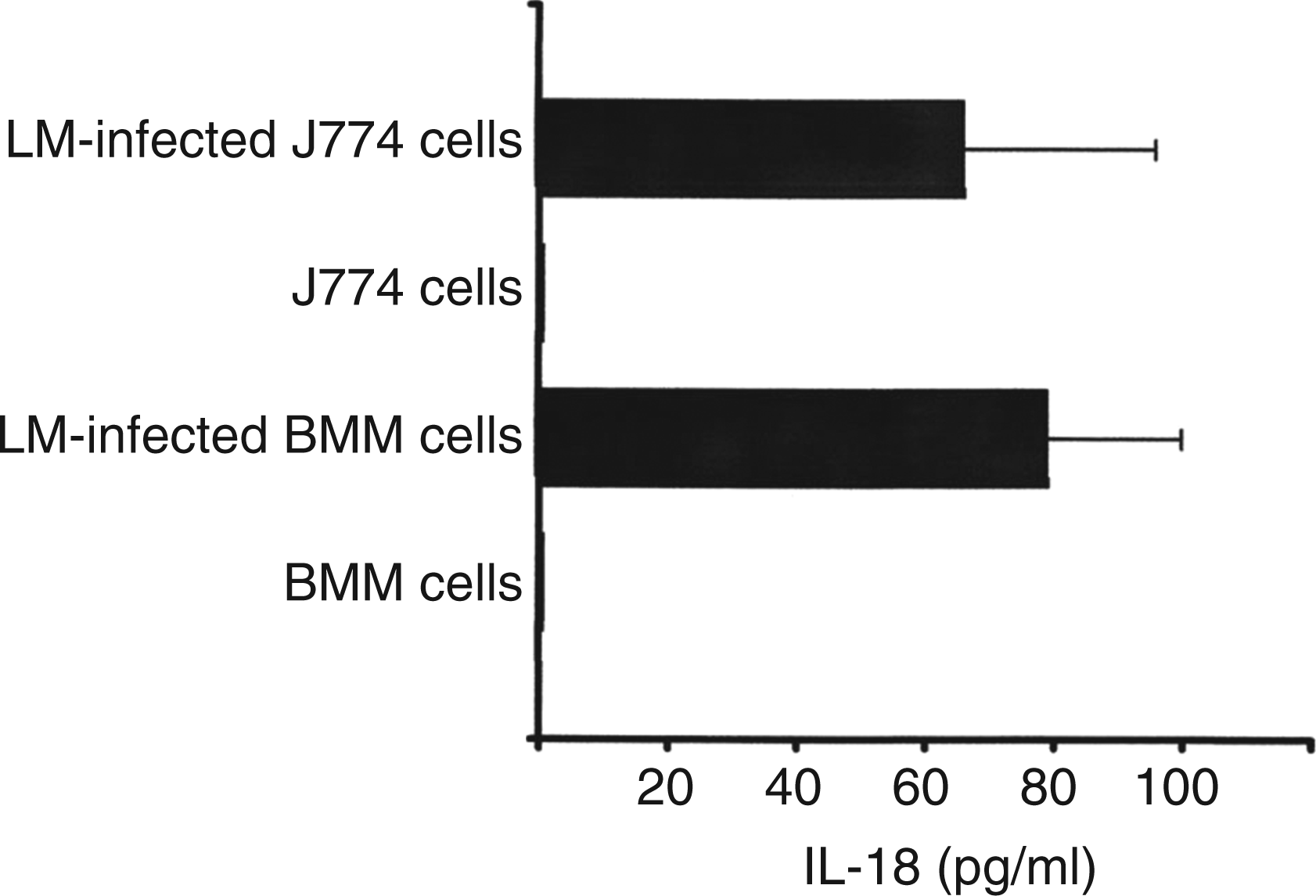
A large amount of IL-18 BP is required for interfering with BIMD-mediated IFN-γ production in B6HO3 cells
IL-18BP, which has a high IL-18-neutralising ability, is present in blood. To see the effect of IL-18BP on the BIMD-mediated IFN-γ production, we co-cultured dying LM-infected J774 macrophages cells with B6HO3 cells in the presence of increasing amounts of IL-18BPd/Fc (Figure 3A). As a control, the ability of IL-18BPd/Fc to inhibit IL-18-mediated IFN-γ production by B6HO3 cells was examined (Figure 3B). IL-18BPd/Fc inhibited BIMD-mediated IFN-γ production in B6HO3 cells with a 50% effective dose (ED50) of 1.5 µg/ml (95% CI 1.9–1.1) (∼34 nM) (Figure 3A), while the ED50 of IL-18BPd/Fc for the IL-18 neutralising effect on IL-18-induced IFN-γ production was as low as 30 ng/ml (95% CI 24–35) (∼0.7 nM) (Figure 3B). Together with the results shown in Figure 2, these results indicate that in our in vitro co-culture system an extremely large amount of IL-18BP was necessary for neutralising the small amounts of IL-18 secreted by dying LM-infected macrophage cells. This conclusion was similar to that of the experiment using anti-IL-18 mAb. Thus, these results imply that IL-18BP, as well as anti-IL-18 mAb, cannot easily gain access to the secreted IL-18 for neutralisation in our in vitro co-culture system. Furthermore, the comparison between the results shown in Figure 1B and in Figure 3A suggests that IL-18BP presented more difficulties for interfering with BIMD-mediated IFN-γ production on a molar basis and hence for neutralising the secreted IL-18 than anti-IL-18 mAb.
Effect of IL-18BP on BIMD-mediated and IL-18-mediated IFN-γ production in B6HO3 cells. (A) B6HO3 cells were co-cultured for 20 h with dying LM-infected J774 cells in the presence of increasing amounts of IL-18BPd/Fc chimera protein (0.003–10 ng/ml). (B) B6HO3 cells were cultured for 20 h with IL-18 at 2.5 ng/ml in the presence of increasing amounts of IL-18BPd/Fc chimera protein (0.003–4 ng/ml). IFN-γ levels in culture supernatants were measured by ELISA, and the percentage of the IFN-γ levels compared with that of the control culture was plotted. The mean IFN-γ levels in the control culture in (A) and (B) were 4.3 and 7.0 ng/ml respectively. Data are shown as mean ± SD from two independent experiments. ED50 of IL-18BPd/Fc for the IL-18 neutralising effect on BIMD-mediated IFN-γ production and IL-18-induced IFN-γ production is 1.5 µg/ml (95% CI 1.9–1.1) and 30 ng/ml (95% CI 24–35) respectively. ED50 was calculated by logit analysis.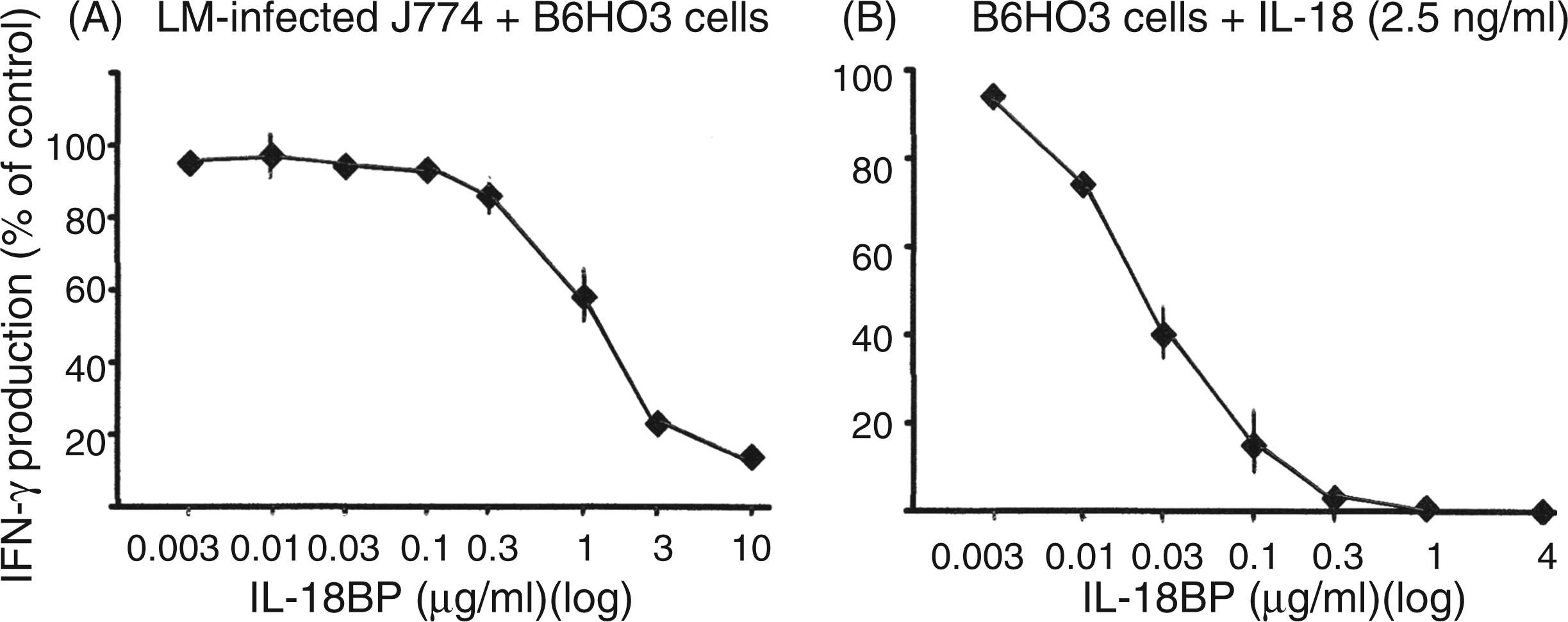
Cellular conjugate formation between B6HO3 cells and J774 macrophage cells, and positional and temporal relationships between IFN-γ-producing B6HO3 cells and dying LM-infected macrophage cells in the in vitro co-culture system
It is known that lymphocytes are capable of adhering to macrophages, even in the absence of antigenic stimulation, although the biological significance of this interaction remains unknown.21,22 Thus, we next examined the possibility that B6HO3 T-cell hybridoma cells and J774 macrophage cells might form cell conjugates. To this end, B6HO3 cells were stained with CFSE and were co-cultured with J774 cells or LM-infected J774 cells for 4 h in a 24-well culture plate. Then, the wells were washed vigorously with PBS containing 10 mM EDTA to remove B6HO3 cells and subsequently observed under a fluorescence microscope (Figure 4A, B). A number of CFSE-labelled B6HO3 cells (green) remained attached to LM-infected J774 cells (Figure 4B) and non-infected J774 cells (Figure 4A) in spite of the washing, indicating that B6HO3 cells adhered to J774 cells. Time-lapse imaging revealed that the attachment between non-infected J774 macrophage cells and B6HO3 cells lasted for several hours, during which time the attached B6HO3 cells were moving around on the macrophage cell surface (Supplementary Video S1). These results indicate that B6HO3 cells, indeed, form cell conjugates with J774 macrophage cells in the in vitro co-culture system.
Cellular conjugate formation between B6HO3 cells and LM-infected or non-infected J774 cells, and positional and temporal relationships between IFN-γ-producing B6HO3 cells and dead LM-infected J774 cells. (A, B) CFSE-labeled B6HO3 cells (green) were co-cultured with LM-infected (B) or non-infected (A) J774 cells for 4 h and washed away with PBS containing 10 mM EDTA, and then J774 cells were observed simultaneously by both fluorescent and visible light using a fluorescence microscope (100×). (C–E) LM-infected J774 cell culture (C), co-culture of LM-infected J774 cells with B6HO3 cells (D) and co-culture of non-infected J774 cells with B6HO3 cells (E) were conducted on culture glasses for 11 h, and they were stained with EMA (yellow) followed by intracellular IFN-γ staining (green), and then observed under a fluorescence microscope (100×). Representative data from one of two experiments are shown. (F) LDH release from LM-infected J774 cells and IFN-γ production by B6HO3 cells co-cultured with LM-infected J774 cells were measured over time in the supernatants and they reached their maximum values at 20 h. The percentages of LDH release and IFN-γ production at each time point compared to their respective maximum values are plotted. Representative data from one of two experiments are shown.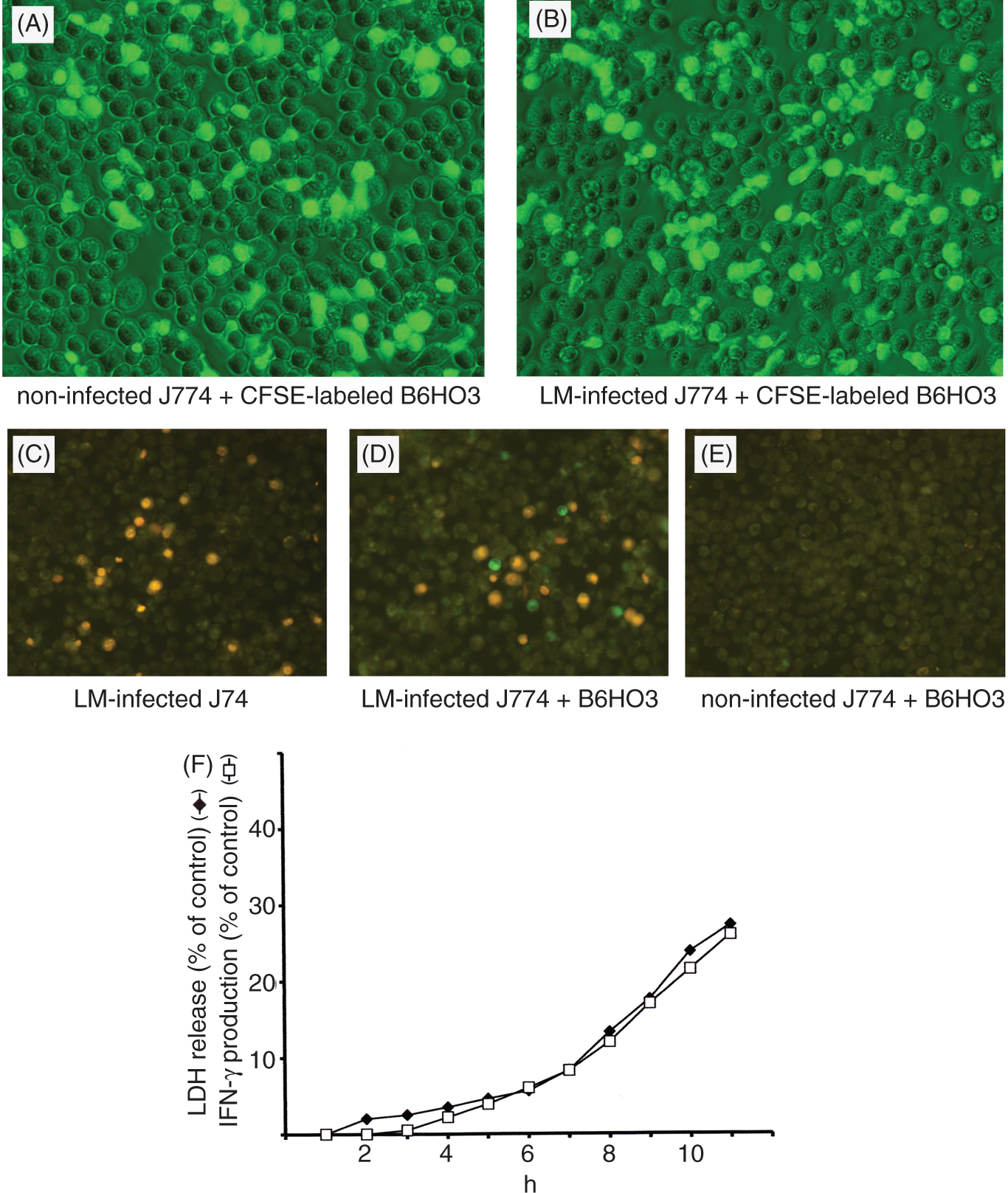
We further examined the positional relationship between IFN-γ-producing B6HO3 cells and dying LM-infected J774 cells in the in vitro co-culture system. For this, J774 macrophages were grown overnight on culture glasses, infected with LM and then co-cultured with B6HO3 cells. Eleven hours after co-culture, cells on culture glasses were stained with EMA followed by intracellular IFN-γ staining, and then observed under a fluorescence microscope (Figure 4C–E). EMA binds covalently to nucleic acids in cells that have compromised membranes, thus selectively staining dead cells. As shown in Figure 4D, most of the IFN-γ producing cells (green) were observed in close contact with dead EMA-stained J774 cells (yellow) (Figure 4D). No IFN-γ producing cells were seen in the co-culture of non-infected J774 cells with B6HO3 cells (Figure 4E) and the LM-infected J774 cell culture (Figure 4C). In parallel, the time course of LDH release from LM-infected macrophages and the time course of IFN-γ production by B6HO3 cells co-cultured with dying LM-infected macrophages were simultaneously examined. Figure 4F shows a strong correlation between the kinetics of LDH release and IFN-γ production, suggesting that there is a causal relationship between the cell death process in macrophages and IFN-γ production in B6HO3 cells. Overall, these results indicate that IFN-γ production in B6HO3 cells is intimately associated with cell conjugate formation between dying LM-infected macrophages and B6HO3 cells.
Both caspase-1-dependent and caspase-1-independent IL-18 maturation processes operate in dying LM-infected macrophages, and the mode of LM-infected macrophage cell death is neither pyroptosis nor pyronecrosis
We next evaluated the contribution of caspases-1 to IL-18 precursor cleavage by adding varying doses of the caspase-1 inhibitor ac-YVAD-CHO to LM-infected J774 cell cultures and to co-cultures of dying LM-infected J774 cells with B6HO3 cells (Figure 5). Furthermore, the effect of ac-YVAD-CHO on LDH release from LM-infected J774 cells was examined. ac-YVAD-CHO reduced IL-18 production in dying LM-infected J774 macrophage cells by a maximum of 70% (Figure 5A), indicating that IL-18 maturation occurs mainly through caspase-1-mediated cleavage. However, the result also indicates that caspase-1-independent IL-18 maturation pathways concomitantly operate, to a lesser extent, in the dying LM-infected macrophages. ac-YVAD-CHO suppressed BIMD-mediated IFN-γ production in B6HO3 to the same extent as its IL-18 maturation inhibition (Figure 5B). By contrast, ac-YVAD-CHO did not inhibit LDH release from LM-infected macrophage cells (Figure 5C), indicating that caspase-1 activation is not involved in LM-infected macrophage cell death. The same results were obtained when BMM cells were used instead of J774 macrophage cells (data not shown). We also examined the effect of the cathepsin B inhibitor CA-074-Me on the maturation of IL-18 and cell death in LM-infected macrophages (Figure 5D, E). Because CA-074-Me was toxic to B6HO3 cells, we were unable to examine its effect on BIMD-mediated IFN-γ production. CA-074-Me reduced IL-18 production in dying LM-infected J774 macrophage cells (Figure 5D) by approximately 44%, but it did not inhibit LM-infected macrophage cell death (Figure 5E). When the inhibitory effect of ac-YVAD-CHO on the maturation of IL-18 was examined in the presence of CA-074-Me, no additive inhibition of IL-18 maturation was observed (data not shown). Thus, cathepsin B seems to act upstream of caspase-1. Overall, the results indicate that multiple IL-18 maturation pathways operate in the dying LM-infected J774 cells and that the mode of macrophage cell death responsible for BIMD-mediated IFN-γ production is neither pyroptosis nor pyronecrosis.23,24
Effects of caspase-1 inhibitor ac-YVAD-CHO and cathepsin B inhibitor CA-074-Me on IL-18 production and LDH release from LM-infected J774 cells and on BIMD-mediated IFN-γ production by B6HO3 cells. (A–C) J774 cells were infected with LM in the presence or absence of various amounts of caspase-1 inhibitor ac-YVAD-CHO, and culture supernatants were assessed at 20 h post-infection for IL-18 production (A) and % LDH release (C) by ELISA. For IFN-γ production, J774 cells were infected with LM and co-cultured with B6HO3 cells for 20 h in the presence or absence of varying amounts of ac-YVAD-CHO (B). The IL-18 and IFN-γ levels in supernatants were measured by ELISA, and are shown as the percentages compared to the control level (black bar). The mean control levels of IL-18 and IFN-γ were 71 pg/ml and 4.1 ng/ml respectively. Data are shown as mean ± SD from two independent experiments. (D, E) J774 cells were infected with LM in the presence of CA-074-Me (100 µM) or vehicle (DMSO), and culture supernatants were assessed at 20 h post-infection for IL-18 production (D) and % LDH release (E) by ELISA. The percentage of the IL-18 level compared to the control (back bar) is shown (D). The mean control level of IL-18 was 55 pg/ml. Data are shown as mean ± SD from three independent experiments. *P < 0.01.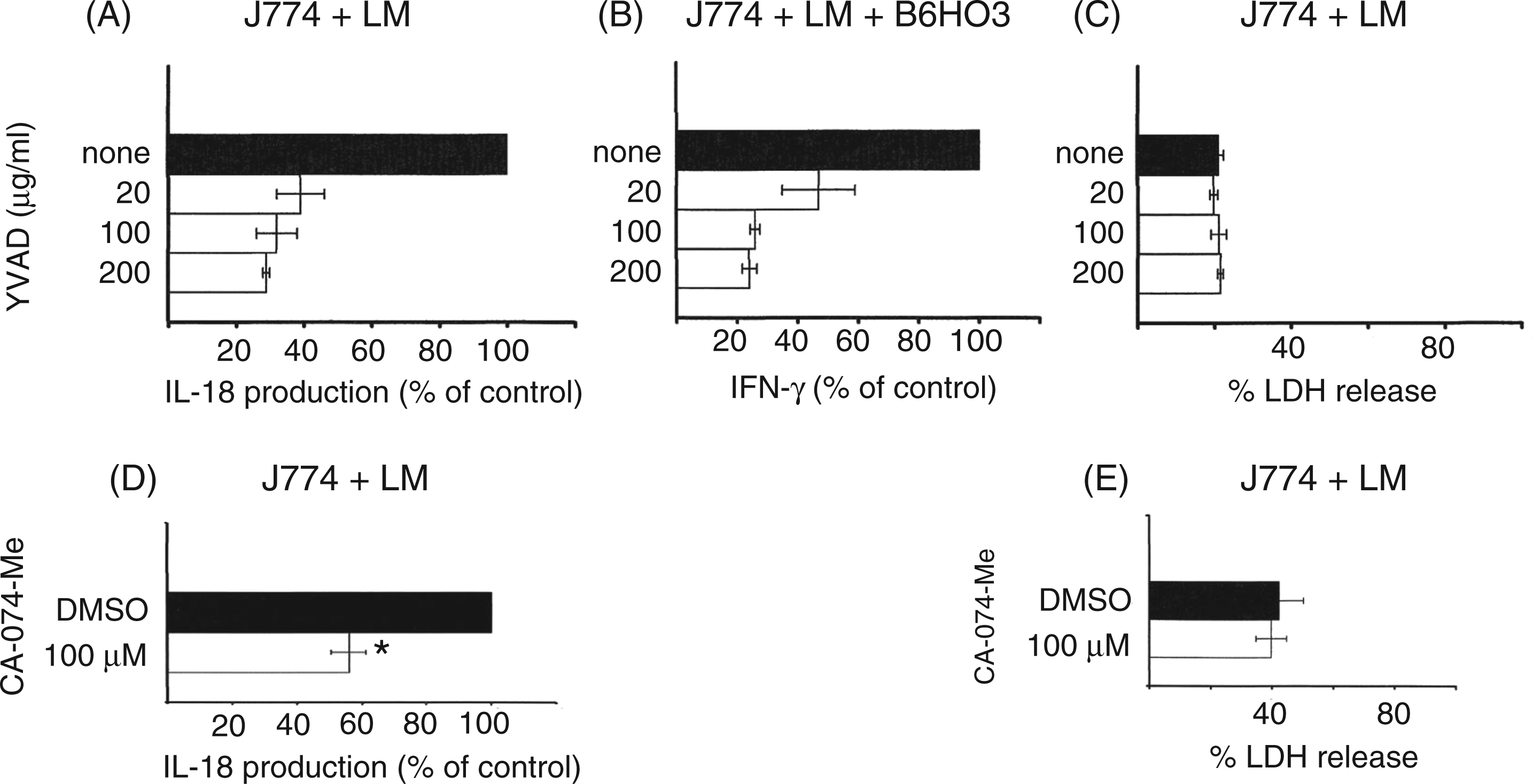
Co-culture of B6HO3 cells with LPS-plus-ATP-stimulated macrophage cells does not result in the production of IFN-γ by B6HO3 cells
It has been reported that LPS-pretreated macrophages, when pulsed with ATP, produce IL-18 through inflammasome-mediated caspase-1 activation and undergo cell death through a caspase-1-dependent or caspase-1-independent pathway.25–27 We next asked whether IL-18 produced by LPS-plus-ATP-stimulated macrophage cells can cause IFN-γ production in B6HO3 cells. B6HO3 cells were co-cultured for 20 h with either J774 or BMM cells that were treated overnight with LPS and then pulsed with ATP; the IFN-γ levels in the culture supernatants were examined (Figure 6). B6HO3 cells were also co-cultured with LM-infected J774 or BMM cells as a positive control. Simultaneously, IL-18 levels and LDH-release were measured in the supernatants from 20 h cultures of the LPS-plus-ATP-stimulated and LM-infected macrophages (Figure 6). In contrast to dying LM-infected macrophage cells, LPS-plus-ATP-stimulated macrophage cells did not induce B6HO3 cells to produce IFN-γ (Figure 6A), although LPS-plus-ATP-stimulated macrophages produced IL-18 and underwent cell death to the same degree as LM-infected macrophages (Figure 6B). The results indicate that the IL-18 produced by dying LPS-plus-ATP-stimulated macrophages cannot induce B6HO3 cells to produce IFN-γ.
IL-18 produced by LPS-plus-ATP-stimulated macrophages dose not induce B6HO3 cells to produce IFN-γ. (A) Co-culture of B6HO3 cells with LM-infected BMM or J774 cells and co-culture of B6HO3 cells with LPS-plus-ATP-stimulated BMM or J774 cells were conducted for 20 h, and IFN-γ levels in culture supernatants were measured by ELISA. (B) LM-infected BMM and J774 cells and LPS-plus-ATP-stimulated BMM and J774 cells were cultured for 20 h, and IL-18 production (upper) and % LDH release (lower) were evaluated in the supernatants by ELISA. Data are presented as mean ± SD from three independent experiments.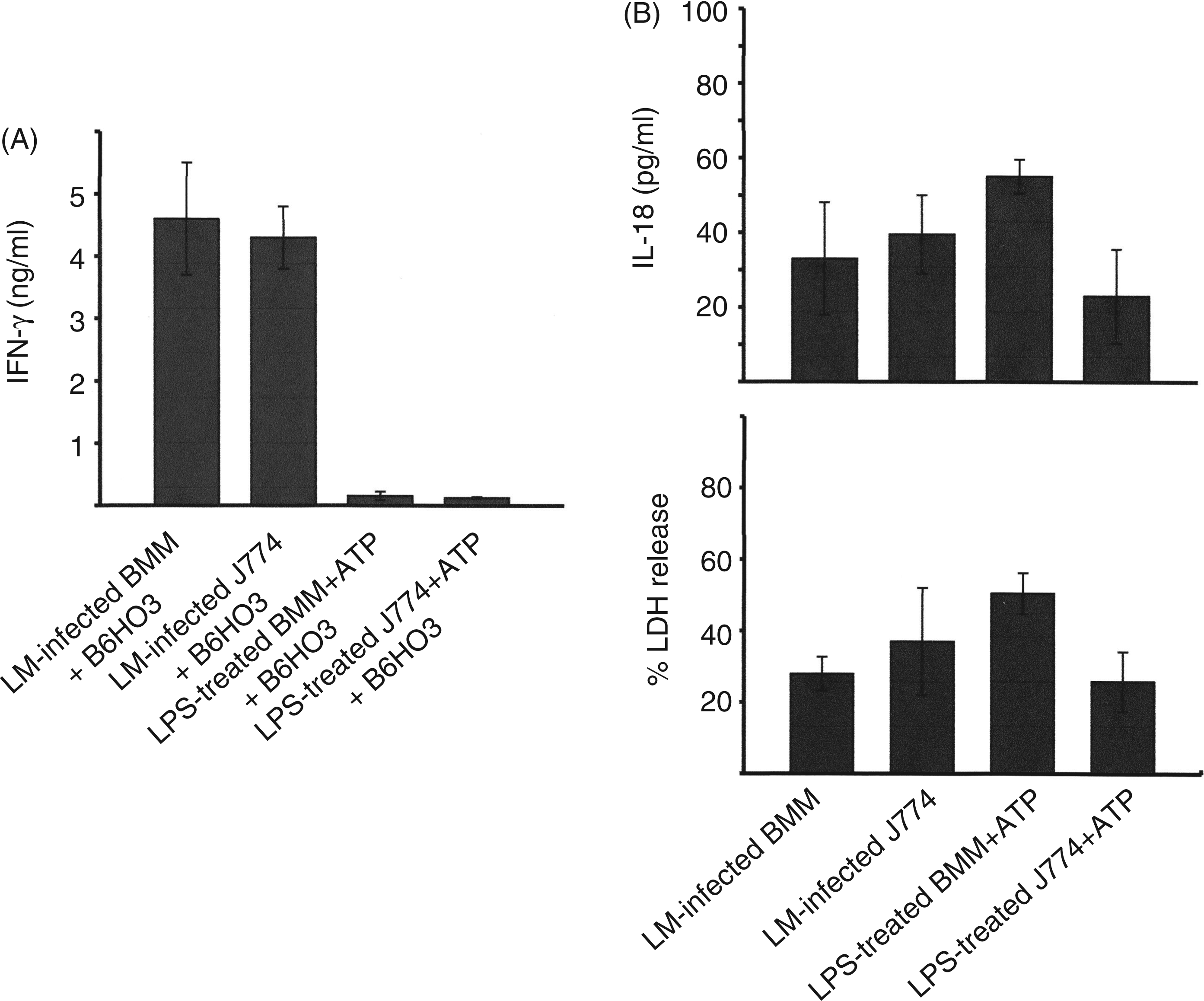
Discussion
In a previous study, 17 we showed that BIMD-mediated IFN-γ production by B6HO3 cells does not require de novo protein synthesis in LM-infected J774 cells. This observation suggests that some intracellular entity that exists intrinsically inside macrophages is responsible for inducing B6HO3 cells to produce IFN-γ. In the present study, we demonstrate that IL-18 is responsible for IL-12p70-independent BIMD-mediated IFN-γ production in B6HO3 cells. IL-18 mRNA and its precursor protein are reported to be constitutively expressed in macrophages/monocytes.9–11 Thus, IL-18 is consistent with the entity that we postulated in our previous study. However, bacteria-infected macrophages produce no more than 100 pg/ml of IL-18, the levels of which are far below the threshold (1 ng/ml) for inducing B6HO3 cells to produce IFN-γ. Cellular conjugate formation between LM-infected macrophages and B6HO3 cells and the close physical association of IFN-γ-producing B6HO3 cells with dying LM-infected macrophages may explain how low levels of IL-18 can induce B6HO3 cells to produce IFN-γ, i.e. IL-18 provided in dying LM-infected macrophages is thought to be exposed or released to the interface of the cell conjugates formed between dying LM-infected macrophages and B6HO3 cells, resulting in localised high concentrations of the cytokine that can transmit signals for producing IFN-γ. In addition, it is possible that some co-stimulatory molecule or some cytokine expressed in the bacteria-infected macrophages may be involved in potentiating IL-18-induced IFN-γ production at the interface of the cell conjugates. 28 Furthermore, cellular conjugate formation can also explain the observation that extremely high amounts of IL-18BP and anti-IL-18 mAb were necessary for neutralising small amounts of IL-18 in our in vitro co-culture system, i.e. it is conceivable that large molecules, such as IL-18BP and anti-IL-18 mAb, cannot easily permeate the interface of cell conjugates where IL-18 is released, thus requiring such high concentrations to neutralise IL-18. The reason why IL-18BP presents more difficulties for neutralising IL-18 at the interface of cell conjugates than anti-IL-18 mAb, despite the fact that IL-18BP has a high affinity for IL-18 and its molecular mass is lower than anti-IL-18 mAb, is not clear at present, however. It is possible that IL-18 has different binding sites for these two molecules, 29 and, thus, IL-18BP and anti-IL-18 mAb may exhibit different accessibilities to the IL-18 released in the distinctive milieu of the interface of cell conjugates.
The present results show that IL-18 is produced in dying LM-infected macrophages through both caspase-1-dependent and caspase-1-independent IL-18 maturation pathways. Caspase-1 activation is mainly mediated by inflammasome activation, which is triggered in response to diverse stimuli, including microbial pathogens, endogenous danger signals and exogenous crystalline particles.7,8,30 Endogenous danger signals include the release of the lysosomal protease cathepsin B into the cytosol because of lysosomal damage.7,30,31 The partial involvement of cathepsin B in the IL-18 maturation process, as shown in this study, suggests that inflammasome activation and hence, caspase-1 activation, can be caused not only by bacterial products but also by cathepsin B release into the cytosol because of phagolysosomal rupture. The important role of cathepsin B release in the activation of the NLRP3 inflammasome has also been reported by other investigators. 32 The mechanism of caspase-1-independent IL-18 maturation, however, is not clear at present. It is likely that phagolysosomal rupture may release some lysosomal proteases that have an ability to cleave pro-IL-18 into its active form, thereby leading to caspase-1-independent IL-18 maturation in the dying LM-infected macrophages. 8 Overall, among the multiple IL-18 maturation pathways observed in our dying LM-infected macrophages, the pathways activated by the release of lysosomal contents into the cytosol may be particularly important as a common denominator for the IL-18 activation mechanisms because a variety of different bacteria can induce BIMD-mediated IFN-γ production.17,19
IL-18 and IL-1β belong to the group of leaderless proteins that are released through non-classical secretory pathways, which are still not well characterised, and thus it is poorly understood how IL-18 is secreted. With respect to IL-1β secretion, extensive studies have been conducted though experiments using cells stimulated with LPS and ATP. 12 As the release of IL-1β is usually accompanied by significant cell death, cell lysis has been suggested as a mechanism of release. 33 However, it is known that IL-1β release can occur independently of cell lysis, prior to nonspecific release of cytosolic contents,12,15 and, thus, multiple different mechanisms of IL-1β release have been proposed.12–15 As with IL-1β release, the secretion of IL-18 is thought to be a complicated event that consists of multiple different mechanisms, including the release of the membrane-bound form of IL-18. 34 Our observation that IL-18 produced by LPS-plus-ATP-stimulated macrophages did not induce B6HO3 cells to produce IFN-γ suggests that the mechanism of IL-18 release in dying LM-infected macrophages differs from that in LPS-plus-ATP-stimulated macrophages. The secretion of IL-18 at the interface of the cell conjugates, which is responsible for BIMD-mediated IFN-γ production, may, therefore, be a unique mechanism, specifically restricted to bacteria-infected macrophages. Further investigation is required to clarify how IL-18 is secreted and how it exerts its IFN-γ-stimulatory effect at the interface of the cell conjugates in dying bacteria-infected macrophages.
Kinetic and morphological analyses confirmed that the IFN-γ production in B6HO3 cells was intimately associated with the cell death process in LM-infected macrophages in our in vitro co-culture system. It is well known that bacteria-infected macrophages undergo cell death in a variety of ways, such as apoptosis, oncosis, pyroptosis and pyronecrosis. 35 Caspase-1 activation is linked with a form of macrophage cell death called pyroptosis, which results in the release of bioactive IL-1β and IL-18, thereby inducing inflammation to elicit a protective host defence against infection. 23 Lysosomal membrane permeabilisation and the resulting leak of lysosomal contents into the cytosol are also linked with cell death. 36 Pyronecrosis is the cathepsin B-dependent cell death of macrophages and is characterised by the release of the pro-inflammatory mediator HMGB1. 24 Our results, however, demonstrated that although both caspase-1 activation and cathepsin B release were seen in dying LM-infected macrophages, the mode of the LM-infected macrophage cell death was neither pyroptosis nor pyronecrosis. Thus, the precise mode of cell death that leads to BIMD-mediated IFN-γ production is not clear at present. If the mode of bacteria-infected macrophage cell death is to be defined more appropriately by physiological and pathological consequences of cell death in addition to morphological and biochemical criteria, then our observations imply that there is a bacteria-infected macrophage cell death mode that is linked with the IFN-γ production in lymphocytes, thereby causing the activation of the bactericidal functions of macrophages rather than elicitation of inflammatory responses.
However, other investigators have reported that LM bacteria induce pyroptotic cell death in macrophages through multiple inflammasome-mediated caspase-1 activation pathways.37–40 The reason for this difference with our results is not clear at present. The same bacteria often induce distinct modes of cell death in macrophages depending on differences in infected macrophage origin and status, MOI and growth conditions of bacteria. Thus, there may be many reasons for the differences in results between our study and others. In particular, it should be underlined that the LM bacteria used in our experiments, probably owing to the differences in bacterial growth conditions, had the property of inducing cell death slowly, 17 while the LM used by others induced cell death within several hours of infection.37–40
IL-18BP, which possesses a high IL-18-neutralising ability, is present in blood 16 and it is thought to be a negative regulator of IL-18 function, thus restricting the influence of IL-18 in the tissue microenvironment. The circulating levels of IL-18BP in healthy individuals are reported to be in the range of 0.01–0.2 nM (0.5–7 ng/ml). 41 Our results show that the ED50 of IL-18BP for the IL-18 neutralising effect on BIMD-mediated IFN-γ production in B6HO3 cells was approximately 30 nM (∼1.5 µg/ml), far exceeding the circulating levels of IL-18BP. These results indicate that BIMD-mediated IFN-γ production cannot be negatively regulated in vivo by serum IL-18BP. In addition to IL-18BP, the blood possibly contains some other factors, such as soluble IL-18Rα and IL-18Rβ, that can interfere with the IFN-γ-producing activity of IL-18. 42 In fact, our preliminary results showed that serum had a strong IL-18-neutralising ability. BIMD-mediated IFN-γ production by B6HO3 cells was, however, resistant to the IL-18-neutralizing activity of serum, as well as to that of IL-18BP (data not shown). Thus, as has previously been reported, it is likely that BIMD-mediated IFN-γ production by subsets of innate lymphocytes can occur in the spleen early after LM infection.20
IL-18 was first identified as a cytokine with the ability to induce NK and Th1 cells to produce IFN-γ. 5 However, as IL-18 by itself is a weak stimulator of IFN-γ production by NK and Th1 cells in vitro, it is generally thought that IL-12p70, which is a heterodimeric cytokine consisting of IL-12p40 and IL-12p35, induces NK cells and memory-type CD8+ T cells to produce innate IFN-γ in conjunction with IL-18.1,2,43 Bacterial stimuli, however, are unable to induce macrophages and DCs to produce IL-12p70 unless these cells are primed by IFN-γ. 44 Thus, it is controversial whether IL-12p70 can be the initial signal for innate IFN-γ production and Th1 response to pathogens.44,45 In fact, there is evidence that a substantial amount of IFN-γ is produced in the absence of IL-12p70 at an early stage of LM infection in mice. 46 In addition, our previous studies have shown that BIMD-mediated IFN-γ production by innate lymphocytes occurs in the absence of IL-12p70 and that it may occur in vivo.19,20 In the present study, we conclude that the small amount of IL-18 produced through multiple IL-18 maturation pathways in dying LM-infected macrophages exerts its stimulatory effect at the interface of the cell conjugate formed between LM-infected J774 cells and B6HO3 cells, resulting in IL-12p70-independent BIMD-mediated IFN-γ production in B6HO3 cells. The IL-18 that is constitutively expressed in macrophages may, therefore, not be a mere co-stimulator for IFN-γ production, but it may rather be the primary cytokine that triggers the initial IFN-γ production in circumstances where resident tissue macrophages are killed by bacteria and IL-12p70 is not yet available at such an early stage of infection. Further evidence in support of this notion will be required for a better understanding of BIMD-mediated IFN-γ production, and further advanced study using the functional T-cell hybridoma B6HO3 will be instrumental in shedding light on the importance of the tissue microenvironment in which cytokines exert their physiologic functions in vivo.
Footnotes
Funding
This study was partly supported by a grant from Kitasato University School of Allied Health Sciences (Grant-in-Aid for Research Project, no. 2012-1011)
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
