Abstract
It was the aim of the present investigation to examine whether the stimulating effect of parathyroid hormone (PTH) on human periodontal ligament (hPDL) cell proliferation and differentiation would be enhanced by hPDL/T-cell interaction involving Wnt10b signaling as a mediating pathway. hPDL cells were cultured from healthy premolar tissues of three adolescent orthodontic patients and exposed to PTH(1-34) in monocultures or co-cultures with CD8+ T cells. At harvest, proliferation, alkaline phosphatase-specific activity (ALP), and osteocalcin production were determined by immunofluorescence cytochemistry, real-time PCR, biochemical assay, and ELISA. Wnt10b signaling was analyzed by the use of a specific WNT10b neutralizing antibody. PTH(1-34) stimulation of T cells significantly increased Wnt10b expression and production. Wnt10b exposure of hPDL cells enhanced proliferation and differentiation. PDL cells co-cultured with T cells showed a Wnt10b-dependent regulation of proliferation and differentiation parameters. The addition of a Wnt10b-neutralizing Ab to the co-culture medium resulted in a significant inhibition of the PTH(1-34) effect on proliferation, ALP-specific activity, and osteocalcin protein expression. Our findings provide novel insight into the mechanism of action of PTH on hPDL cells and establish the interplay of T cells and hPDL cells via the Wnt10b pathway as a modulating factor for the anabolic properties of the hormone in periodontal regeneration.
Introduction
Parathyroid hormone (PTH) represents a well-established anabolic option for the treatment of bony defects since it was shown to stimulate new bone formation and improve the microarchitecture of existing bone.1,2 From the rapidly developing field of osteoimmunology it is known that, particularly under inflammatory conditions, there is a tight link of regulatory processes between stromal and immune cells contributing to the transduction of the PTH effect.3–5 T cells were first implicated to be regulatorily involved in the anabolic response to PTH by Pettway et al., who demonstrated that intermittent PTH (iPTH) enhanced bone content in wild-type mice as opposed to the failure to induce vertebral bone growth in T-cell-deficient mice. 6 Further experiments using four strains of T-cell-deficient mice revealed that mice lacking T cells exhibited a blunted increase in bone formation and trabecular bone volume in response to iPTH that could be restored by adoptive transfer of T cells. 7 Regarding the mechanism by which T cells increase the anabolic response to PTH, Wnt10b secreted by CD8+ T cells upon PTH stimulation has been suggested to activate the Wnt signaling pathway to result in enhanced osteoblast differentiation and lifespan.3,4 By the number of existing types of immune cells it has been documented that Wnt10b signaling is most effective among the CD8+ T-cell population.3,4,7,8 This implication has been corroborated by the hampered effect of PTH on bone turnover in T-cell receptor-ß knockout mice reconstituted with T cells from Wnt10b knockout mice. 7 The analysis of the effects of PTH on the T-cell production of cytokines that regulate osteoblast function has revealed that iPTH does not change the expression of IL-1, −4, −6, −7, and TGF-ß, but slightly that of TNF-α. Furthermore, an age-dependent capacity of PTH to inhibit the production of Wnt inhibitors such as sclerostin was identified. 9 Thus, T-cell-mediated Wnt signaling in osteoblasts may represent a key mechanism by which PTH increases bone strength, providing further evidence for the crucial role of cross-talk pathways between T cells and mesenchymal cells in the mediation of anabolic effects of PTH.
In recent years, high-ranking publications provided accumulating evidence for the effectiveness of PTH in the craniofacial area and even pointed to an increased susceptibility of those target structures as compared to other regions of the body. 10 These findings raised the interest in the applicability of anabolic doses of PTH to support periodontal regeneration following trauma, inflammatory periodontal disease or orthodontic tooth movement. Animal experimental work indicated the capacity of PTH to limit the inflammatory infiltrate and also the extent of bone loss in ligature-induced periodontitis models.11,12 Shirota et al. demonstrated that reduced bone density and bone-to-implant contact in ovariectomized rats could be restored by iPTH administration. 13 In a recent clinical study, patients suffering from periodontitis benefitted from daily subcutaneous iPTH injections for 6 wk in addition to periodontal surgery as reflected by improved clinical outcomes, greater resolution of alveolar bone defects and accelerated osseous wound healing in the oral cavity. 14 Our group contributed to the recognition of periodontal ligament (PDL) cells as components of the tooth-supporting apparatus exhibiting osteoblastic traits and an osteoblast-like response to hormonal stimulation identifying them as a promising target in the attempt to support regenerative periodontal processes by PTH administration.15–24
Combining these lines of evidence, it was the aim of the present investigation to address the question of whether the stimulating effect of PTH is mediated through a cellular interaction of T cells and stromal cells in the area of the periodontal ligament. We hypothesized that CD8+ T cells would affect periodontal remodeling by modulating human PDL (hPDL) cell proliferation and differentiation. Furthermore, we speculated that this cellular interplay would involve T-cell-induced Wnt10b as a key mediator that could function as a potential target for the development of future treatment strategies.
Material and methods
All experimental protocols were reviewed and approved by the ethics committee of the University of Bonn (reference number 029/08).
PDL and T-cell culture
hPDL were cultured from the middle third of premolar roots to avoid contamination with gingival or apical tissue using the explant method. The three donors were aged between 12 and 14 yr and showed no clinical signs of periodontitis. The teeth had been extracted for orthodontic reasons and with informed written parental/guardian consent. Cells were cultured in DMEM containing 10% FBS and 0.5% antibiotics (diluted from a stock solution containing 5000 U/ml penicillin and 5000 U/ml streptomycin; Biochrom AG, Germany) at 37°C in an atmosphere of 100% humidity, 5% CO2, and 95% air. Fifth passage, non-pre-differentiated cells were seeded into 24-well plates (n = 6) at a density of 10,000 cells/well and cultured to subconfluence (∼70%). This seeding protocol was applied to immunocytochemistry, LRP5, alkaline phosphatase (ALP) and osteocalcin assays. Deviating seeding densities are detailed below. Prior to experimental use, hPDL cells were characterized by real-time PCR as described previously 17 and demonstrated to express several markers typical of cells of mesenchymal origin, including ALP, osteocalcin, TGF-ß, bone morphogenetic protein (BMP)-2 and BMP-4.
For T-cell experiments, cells from three different donors were extracted from buffy coats using a CD8+ extraction kit (Stemcell Technologies, Grenoble, France) resulting in ∼65% CD8+ T cells verified by flow cytometry (Beckman Coulter, Germany) (Figure 1). Cells were cultured in Roswell Park Memorial Institute (RPMI)1640 medium containing the same supplements as stated for hPDL cells above.
Basal and PTH(1-34)-stimulated Wnt10b expression in CD8+ T cells compared to hPDL cells. (a) Flow cytometry verification of CD8+ T-cell extraction from human buffy coats (red: CD8+ T cells ∼65%). (b) Basal Wnt10b mRNA expression by T cells was enhanced significantly by 10−9 M PTH(1-34) exposure for 48 h. (c) Wnt10b protein expression was enhanced following 10−9 M PTH(1-34) administration. In contrast, basal Wnt10b expression and production was not altered by PTH(1-34) in hPDL cell monocultures (b, c). Data were acquired from one of two separate experiments, both yielding comparable results. Each value is the mean ± SD for six independent cultures. *P < 0.05, experimental group vs. vehicle control.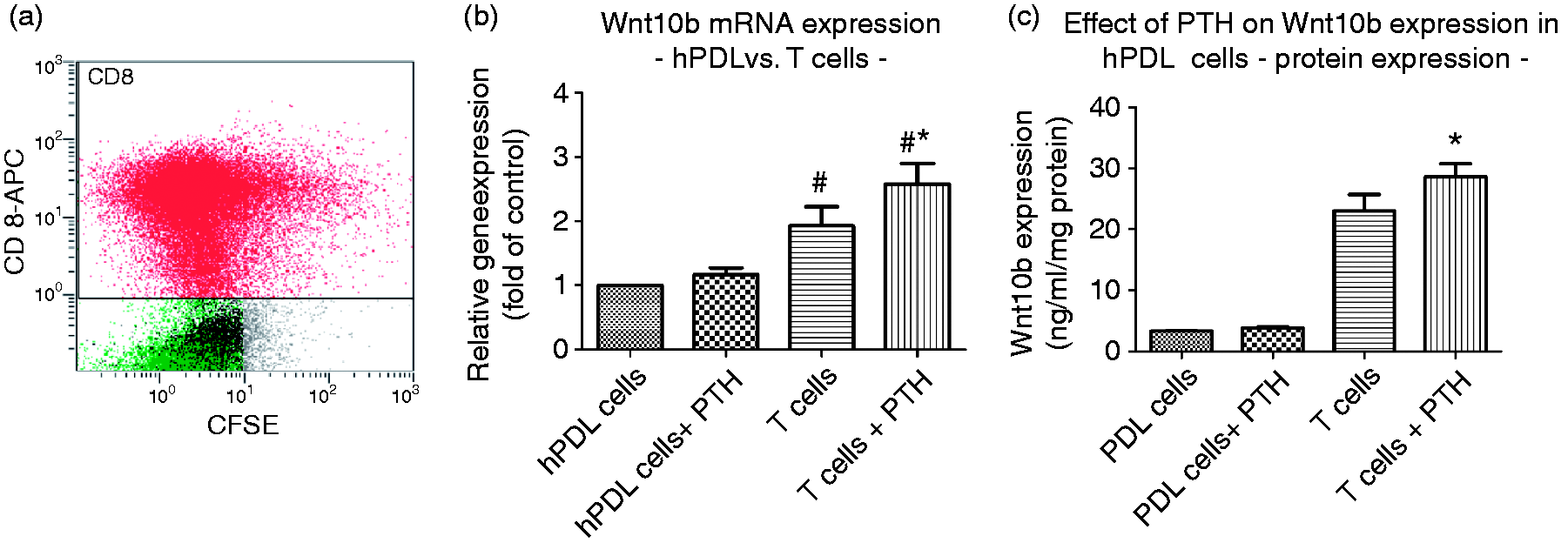
Effect of PTH(1-34) stimulation of T cells on Wnt10b expression
In a first step, T cells were stimulated with 10−9 M PTH(1-34) (Sigma Aldrich, Germany) for 48 h prior to determination of Wnt10b protein expression in the conditioned media by means of a commercially available ELISA kit specific for human Wnt10b (Hölzel Diagnostika, Germany) according to the manufacturer’s instructions. Changes in Wnt10b mRNA expression were analyzed by real-time PCR as described below.
Wnt10b effect on hPDL cell proliferation
Next, to examine the effect of Wnt10b on hPDL cell proliferation, the Wnt10b receptor LRP5 was demonstrated in hPDL cells by means of RT-PCR and immunfluorescence cytochemistry as detailed previously 15 and below, respectively. Thereafter, hPDL cells were stimulated with 50 ng/ml Wnt10b protein (GenWay-Biotech, USA) for 3 d. Vehicle-treated cultures (H2O) served as controls. A commercially available MTS assay (Promega, Germany) was carried out according to the manufacturer’s instructions to measure cell proliferation rate. In these experiments, the seeding density was 20,000 cells/well.
Influence of Wnt10b on osteoblastic differentiation of hPDL cells
Likewise, for proliferation, the impact of Wnt10b on hPDL cell differentiation was quantified at the transcriptional and protein level for ALP-specific activity as a marker for early and osteocalcin for late osteoblastic differentiation.
Real-time PCR
PTH(1-34)-induced changes in Wnt10b mRNA expression by T cells and Wnt10b-induced alterations in osteogenic marker gene expression in hPDL cells (seeding density for hPDL cells: 30,000 cells/well in six-well plates; T cells: 10000 cells/wells in 24-well plates) were analyzed by real-time PCR as described previously. 25 The primer sequences used were as follows: ALP sense 5′-AGA-GAA-AGC-GAT-GGT-GGA-TG-3′, antisense 5′-CGG-TGG-CAT-TAA-TAG-TGA-GAT-G-3′, 26 osteocalcin sense 5′-ATG-AGA-GCC-CTC-ACA-CTC-CTC-G-3′, antisense 5′-GTC-AGC-CAA-CTC-GTC-ACA-GTC-C-3′; LRP5 sense 5'-ACC-GGA-ACC-ACG-TCA-CAG-3', antisense 5'-GGG-TGG-ATA-GGG-GTC-TGA-GT-3', 27 Wnt10b sense 5'-GCA-CTG-TAT-TGC-TCC-TCC-CAC-TT-3'; antisense 5'-ATA-GGG-ACT-CCC-CAG-CCA-AA-3'. β-actin was used as an endogenous reference; sense: 5′-CAT-GGA-TGA-TGA-TAT-CGC-CGC-G-3′, antisense: 5′-ACA-TGA-TCT-GGG-TCA-TCT-TCT-CG-3′.
ALP protein expression
Following Wnt10b exposure for 48 h, hPDL cells were released from the culture surface by trypsinization at 37°C for 10 min. This reaction was terminated by the addition of DMEM containing 10% FBS. Thereafter, the cell suspension was centrifuged and the cell pellet resuspended in 0.01% TritonX-100. Changes in ALP-specific activity were measured in lysates of isolated cells as a function of release of paranitrophenol from paranitrophenylphosphate at pH 10.2 as described previously. 28 To ensure that changes in ALP activity did not simply result from changes in proliferation or apoptosis rather than from direct effects of Wnt10b, ALP data were calculated as a function of the protein content of the sample.
Osteocalcin protein expression
The level of osteocalcin in the conditioned media after 48 h of Wnt10b stimulation was assayed using a commercially available ELISA kit specific for human osteocalcin (IBL GmbH, Germany) according to the manufacturer’s instructions.
Immunofluorescence cytochemistry
To visualize the effect of Wnt10b on hPDL cell differentiation along the osteoblastic pathway, immunocytochemical staining was performed using either a 1:50 (40 µg/ml) working dilution of the primary Ab for ALP (rabbit anti-human; Quartett, Germany) or 1:25 (80 µg/ml) for osteocalcin (rabbit anti-human; Biozol, Germany) in combination with a secondary fluorescence-labeled (Cy-3) Ab (1:100 (1.5 µg/ml) working dilution; goat anti-rabbit; Invitrogen, Germany) and analyzed microscopically as reported previously. 29 Negative controls were run by (i) omitting the primary Ab, (ii) omitting the primary and secondary Ab and using TBS/BSA instead, and (iii) by substituting the primary Ab by an unspecific IgG. The same protocol was followed for detection of LRP5 in hPDL cells using a monoclonal rabbit anti-human Ab (Cell Signaling Technology Europe, The Netherlands) using a 1:100 (20 µg/ml) working dilution.
Co-culture experiments
To address the question of whether the interaction of hPDL and immune cells enhances the PDL cell response to PTH(1-34) via Wnt10b, experimental groups were stimulated with either 10−9 M PTH(1-34) alone or in combination with 500 ng/ml monoclonal Wnt10b Ab for 5 d (mouse anti-human, R&D Systems, Germany). Cell culture medium was changed on d 2. At harvest, supernatants were collected and stored at −80°C until further analysis. Changes in ALP and osteocalcin protein expression were quantified as described above.
To determine the effect on hPDL cell proliferation, cells were washed with PBS and images of green fluorescent hPDL cells were captured at a magnification of 40× using a fluorescent microscope (Zeiss, Germany). Cell number was counted using the freeware ImageJ (National Institutes of Health, Bethesda, MD, USA). Data from the co-culture experiments were compared to the findings in hPDL cell monocultures that were exposed to 10−9 M PTH(1-34) for 48 h prior to the analysis of proliferation, ALP and osteocalcin mRNA expression and protein production as described above.
Statistical analysis
Each data point represents the mean ± SD of six independent cultures per experimental group. Data were analyzed by analysis of variance (ANOVA) and statistical significance determined using Bonferroni’s modification of Student’s t-test for multiple comparisons or the Wilcoxon test. P-values < 0.05 were considered to be significant. The data are representative of two replicate experiments that both yielded similar results. Only one set of results from the two sets of experiments are presented.
Results
Effect of PTH (1-34) on CD8+ T cells
CD8+ T-cell isolation from human blood samples resulted in a ∼65% CD8+ T-cell suspension (Figure 1a). In a first step, the basal expression of Wnt10b in CD8+ T cells and its inducibility by PTH(1-34) were tested both at the transcriptional and protein level. At both levels, a basal Wnt10b expression in isolated human CD8+ T cells could be observed. Wnt10b mRNA expression was significantly increased (∼1.5-fold) upon 10−9 M PTH(1-34) stimulation for 6 h (Figure 1b). Similar to the transcriptional data, Wnt10b protein expression was significantly enhanced compared to untreated controls (Figure 1c).
Effect of PTH(1-34) on hPDL cell monocultures
In contrast to the findings in CD8+ T cells, the basal Wnt10b mRNA and protein expression was significantly lower in hPDL cells and not altered by 10−9 M PTH(1-34) (Figure 1b and c). However, proliferation, ALP-specific activity and osteocalcin production were all enhanced significantly following PTH(1-34) exposure of the monocultures (Figure 4).
Documentation of the presence of the Wnt10b receptor LRP5 in human PDL cells. Immunofluorescence staining using a Cy3-labelled secondary antibody confirmed the presence of the protein in hPDL cells (left panel); negative control was carried out by omitting the primary antibody and using TBS/BSA instead (right panel); magnification ×40.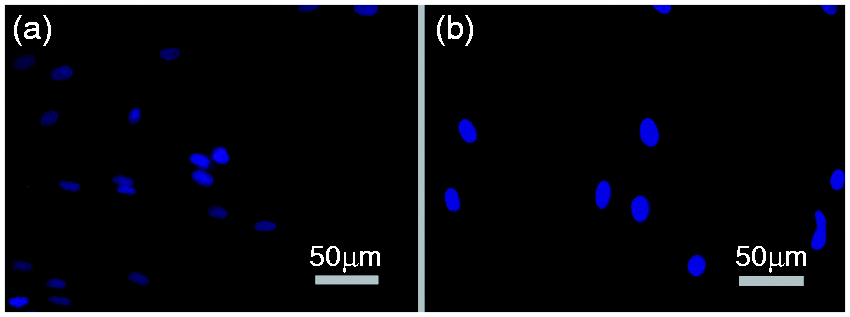
Verification of Wnt10b signaling receptor in hPDL cells
To provide evidence that hPDL cells carry the necessary receptor for Wnt10b signaling, the expression of LRP5 as an important receptor for Wnt10b was demonstrated by RT-PCR analysis (data not shown) and immunofluorescence cytochemistry (Figure 2).
Wnt10b stimulates the proliferation of hPDL cells. Human PDL cells were exposed to 50 ng/ml Wnt10b for 3 d prior to quantification of the proliferation rate by MTS assay. Data were acquired from one of two separate experiments, both yielding comparable results. Each value is the mean ± SD for 6 independent cultures. *P < 0.05, experimental group vs. vehicle control.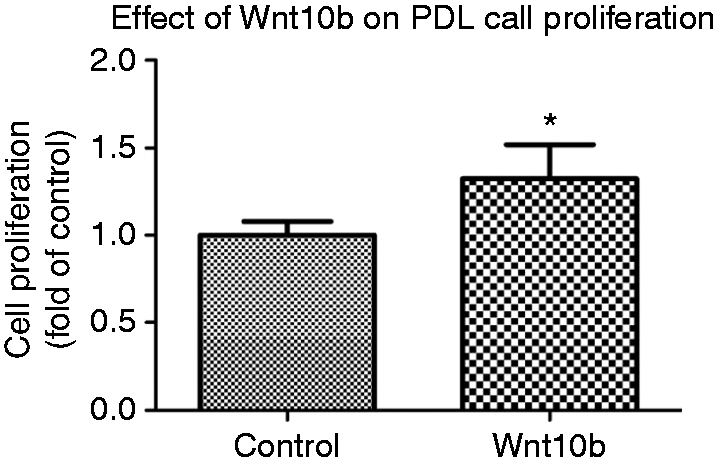
Effect of Wnt10b on hPDL cells
To prove that Wnt10b signaling is able to regulate important parameters associated with regeneration of the PDL, the isolated effect of Wnt10b on hPDL cells was analyzed in vitro. Following stimulation of hPDL cells with a concentration of 50 ng/ml Wnt10b protein, hPDL cell proliferation rate was enhanced by ∼30% compared to untreated controls (Figure 3). Similar to cell proliferation, early osteogenic differentiation parameters also were affected. Regarding ALP mRNA, a 2.5-fold increase could be observed (Figure 4a). A similar effect was demonstrated for ALP protein production. Compared to untreated controls, Wnt10b stimulation induced a ∼50% increase in ALP-specific activity (Figure 4b). Likewise, ALP, osteocalcin expression, as a marker of late osteoblastic differentiation, was significantly increased at the mRNA and protein level following Wnt10b stimulation (Figure 4c and d). Those quantitative data on mRNA and protein expression corroborated the visual impression obtained by immunofluorescence staining, where Wnt10b stimulation resulted in an enhanced ALP and osteocalcin immunoreactivity compared to control cultures (Figure 4c and f).
Effect of Wnt10b on alkaline phosphatase specific activity and osteocalcin expression in hPDL cells. The addition of 50 ng/ml Wnt10b to the culture medium for 3 d resulted in a significant increase in ALP mRNA (a) and protein expression (b) as determined by realtime PCR and biochemical assay. Immunocytochemical staining confirmed the quantitative data (c), magnification ×40. Similar results were obtained for osteocalcin expression (e) and production (e,f). Data were acquired from one of two separate experiments, both yielding comparable results. Each value is the mean ± SD for 6 independent cultures. *P < 0.05, experimental group vs. vehicle control. Role of Wnt10b for mediating the interaction of T and hPDL cells. Co-cultures of hPDL cells and CD8+ T cells were exposed to 10-9 M PTH(1-34) for 5 d in the presence or absence of 500 ng/ml Wnt10b neutralizing antibody. PTH exposure resulted in an increase of hPDL cell proliferation (a), ALP specific activity (b), and osteocalcin production (c). The addition of a neutralizing Wnt10b antibody inhibited the PTH effect or even leveled it out at control levels. Data were acquired from one of two separate experiments, both yielding comparable results. Each value is the mean ± SD for 6 independent cultures. *P < 0.05, experimental group vs. Wnt10b-Ab treated group; #P < 0.05, experimental group vs. untreated control.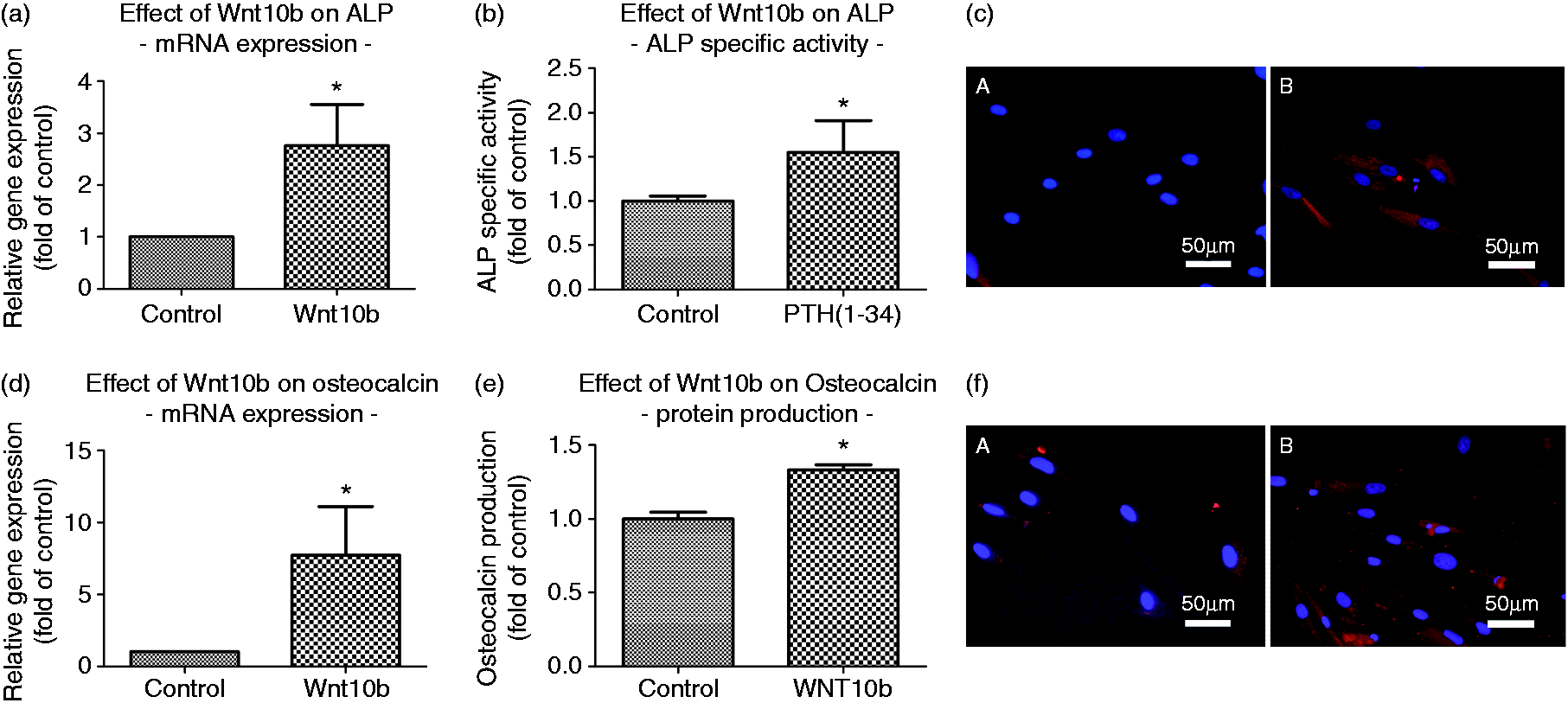
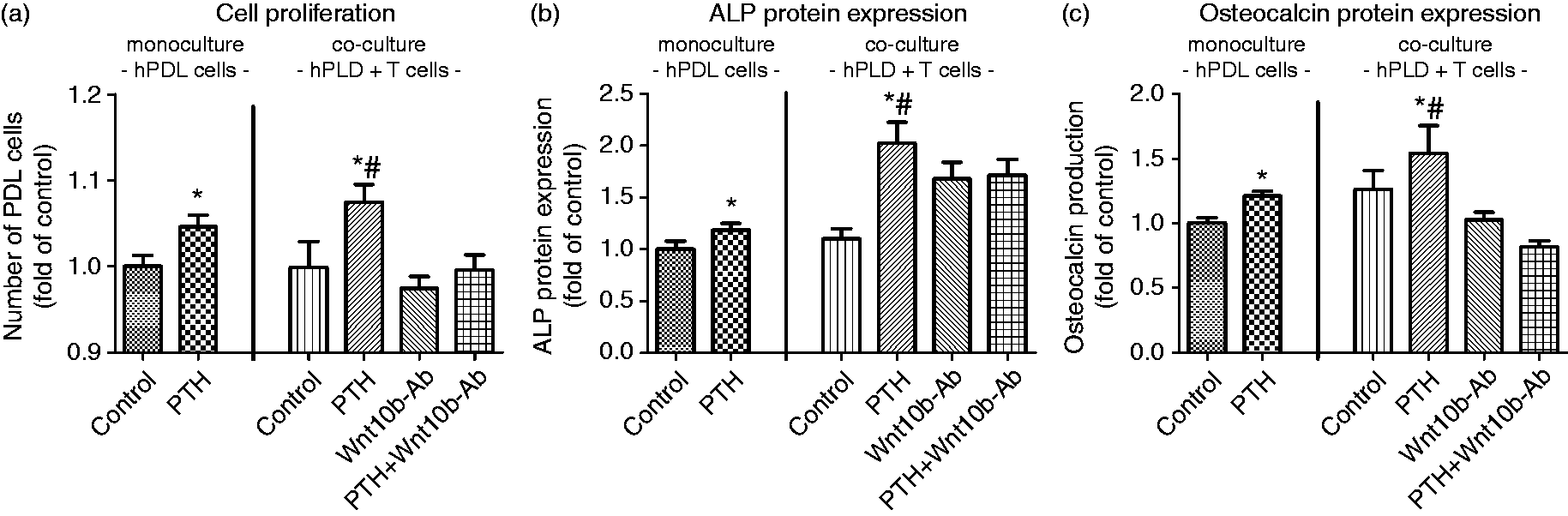
Wnt10b-dependent regulation of T-cell and hPDL-cell interaction
To test the physiological relevance of T-cell-released Wnt10b on hPDL-cell behavior and its enhancement following PTH stimulation, the observed findings of cell monocultures were transferred to a T-cell/hPDL-cell co-culture system.
Compared to the observations in hPDL-cell monocultures, the co-culture experiments revealed an increase in cell number and basal expression of both differentiation parameters. Likewise, an enhanced hPDL-cell sensitivity to PTH regarding the cellular response to T-cell-expressed Wnt10b and its effect on PDL-cell proliferation and differentiation was observed. Following PTH(1-34) stimulation, an increase in the number of cells as well as in ALP activity and osteocalcin protein expression was demonstrated.
To provide further evidence for Wnt10b regulation of the observed phenomena, a specific neutralizing Ab raised against Wnt10b was added to the co-culture medium. Following Wnt10b Ab challenge, the increase in proliferation, ALP-specific activity and osteocalcin expression seen upon 10−9 M PTH(1-34) stimulation was inhibited significantly or even leveled out at control levels (Figure 5a–c).
Discussion
The present study examined the interplay of hPDL cells and T cells regarding the anabolic response of hPDL cells to PTH and demonstrated an enhanced hPDL-cell sensitivity to hormonal stimulation in co-culture with respect to proliferation and differentiation as compared to PTH challenge of monocultures. Furthermore, Wnt10b was identified as a mechanistic link to mediate and amplify the PTH effect.
The PDL hosts a heterogeneous cell population ranging from cells with stem cell character to terminally differentiated epithelial cells, osteoblasts, cementoblasts and fibroblasts.30,31 The latter have been reported to share osteoblast-like traits and respond accordingly to hormonal stimulation.32,33 PDL cells were shown to express the PTH 1 receptor (1R) 29 and respond to anabolic doses of PTH with changes of many crucial cell functions including proliferation, differentiation, apoptosis, and the production of key molecules of bone remodeling.15–17 These findings are in line with the data obtained here in the monoculture experiments regarding proliferation and differentiation and strengthen the view of PDL cells as important regulators of periodontal remodeling. The detection of LRP5 in hPDL cells as one of the receptors for Wnt ligand binding indicates the cells’ ability to process Wnt signals and is supported by previous reports in PDL cells.34–36 Furthermore, it corroborates the osteoblast-like features of PDL cells and underlines the bidirectional nature of the interaction with immune cells under inflammatory conditions, such as periodontal disease, trauma or orthodontic tooth movement. Not only do PDL cells express pro-inflammatory cytokines that in turn mediate the attraction of macrophages to injured sites, 37 but apparently they respond to immune cell signaling via Wnt ligand activation. T-cell-driven Wnt10b production has been reported to be critical for the activation of Wnt signaling in osteoblasts and thus crucial for potentiating the bone anabolic activity of PTH. 4 In the absence of T cells, Terauchi and colleagues reported on a reduced capacity of PTH to induce stromal cell commitment to the osteoblastic lineage, osteoblast proliferation and differentiation, and mitigate osteoblast apoptosis. All of the anabolic actions of PTH were attributed to the ability of T cells to activate Wnt signaling in osteoblasts. 7 Therefore, we first checked the Wnt10b responsiveness in hPDL cells. Our results with increased proliferation, ALP-specific activity and osteocalcin production, at both the transcriptional and protein level, indicate that Wnt10b directs the differentiation of hPDL cells along the osteoblastic pathway. Therefore, it can be concluded that Wnt10b serves as a mechanistic link in T-cell/PDL cell interaction, too. The co-culture experiments we performed prove that this is indeed the case as evidenced by increased proliferation and differentiation of hPDL cells in the presence of T cells as compared to hPDL-cell monocultures under unstimulated conditions. The observation that an addition of Wnt10b Ab at least partially inhibited the stimulatory effect of T-cell/PDL-cell interaction further substantiates the above interpretation. Regarding the mediation of the PTH effect on hPDL cells, this apparently involves a direct and an indirect mechanism, with the former being transduced by the PTH receptor present on the PDL cell membrane, whereas the indirect effect is mediated via a PTH-induced enhanced production of Wnt10b by T cells, which then in turn affects hPDL cell physiology. Since the inhibition of Wnt ligand binding by addition of Wnt Ab to the culture medium did reduce, but, as noted for ALP-specific activity, not completely inhibit the PTH-induced effects, a Wnt ligand-independent activation of the Wnt receptors LRP5/638 and suppressed production of sclerostin, which acts as an inhibitor of the Wnt signaling pathway, also have to be considered.9,39,40 Another possibility is that, in addition to Wnt10b, PTH may induce the secretion of several other Wnt ligands.
Interestingly, regarding proliferation and early osteoblastic differentiation, co-culture with T cells seemed to sensitize hPDL cells for the subsequent PTH signaling, since the increase in cell number and ALP activity was more intense in hPDL cells harvested from co-cultures in comparison to monocultures. However, this synergistic effect was not visible for osteocalcin expression as a marker of late differentiation stages, indicating a differential response of hPDL cells that underlines their unique characteristics despite several features they share with osteoblasts.
In summary and in the attempt to put our findings into a clinical perspective, there is considerable agreement among all our data allowing for the conclusion that in periodontal remodeling there is a crosstalk between T cells and PDL cells involved in the mediation of the anabolic PTH effect. It results in an expansion of the pool of hPDL cells present in the local microenvironment that then further differentiate along the osteoblastic pathway and contribute to the regeneration of periodontal tissues that were damaged or lost in the course of inflammatory disease or dental measures such as orthodontic tooth movement. This information might be exploited in the future to refine and amend existing regenerative periodontal treatment strategies founded on the anabolic properties of PTH and the regulatory capacity of hPDL cells for the remodeling of the local microenvironment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the German Research Foundation (DFG) (grant number KFO 208, TP8, LO-1181/2-2) and the Medical Faculty of the University of Bonn.
