Abstract
Indoleamine 2,3-dioxygenase (IDO) is a rate-limiting enzyme for the degradation of tryptophan (Trp) along the kynurenine (Kyn) pathway, and its increased activation is associated with immunologic disorders. Because the specific role of IDO activation is not yet completely clear, the aim of the present study was to establish a pig model of IDO activation for further research. The activation of IDO in pigs was induced experimentally by LPS stimulation
Introduction
Indoleamine 2,3-dioxygenase (IDO) is a rate-limiting enzyme for the degradation of tryptophan (Trp) along the kynurenine pathway to biologically active metabolites such as kynurenine (Kyn), kynurenic acid (Kyna) or quinolinic acid (Quin). This pathway of Trp metabolism is a link between the immune and the nervous system, and plays a crucial role in the regulation of the immune response. The IDO activation is one mechanism of innate immunity and has different physiological functions with a highly cell-type specific pattern of inducibility. IDO is expressed ubiquitously in a constitutive or inducible manner. 1 In many cell types it is induced by specific inflammatory stimuli, such as IFN-γ or LPS. 2 Moreover, LPS may also activate IDO by the production of TNF-α. 3 The depletion of Trp by IDO induction leads to suppression of intracellular pathogens and T-cell proliferation,4,5 and is associated with the development of depressive-like behavior in mice.6,7 An increased activity of IDO was also measured in a number of immunologic and neurologic disorders. For example, it was found that IDO is highly expressed in malignant human tumors leading to an effective immune escape by inhibiting T-cell responses, 8 and, in addition, an increased IDO activity in patients with sepsis is associated with a higher mortality risk. 9 An accumulation of Kyna or Quin was found in several neurologic disorders, for example Alzheimer’s disease, 10 schizophrenia 11 and Huntington disease, 12 suggesting neuroactive functions of these Trp metabolites. However, the immunosuppressive or neuroactive function of IDO activation in these disorders is not yet fully understood, and an animal model with high similarity of its Trp metabolism to that of humans would be necessary for further studies in this field. The pig is a species that closely resembles humans in anatomy, genetics and physiology, and is used increasingly as a model for human in biomedical research. Compared with rodents, pigs have obvious advantages with respect to the relevance for human pathophysiology.13,14 The size of pigs allows the use of instrumentation and monitoring devices similar to those used in humans, and the amount of blood allows for repeated blood sampling and comprehensive analyses. 15 Thus, relating to the current research in the therapeutic effect of IDO inhibition, experiments in pigs are much more likely to be predictive of therapeutic treatments in humans than experiments in rodents. Therefore, the aim of this study was to establish a pig model of IDO activation.
In the present study, we investigated IDO activation in pigs
Materials and methods
Animals
A total of 50 castrated male pigs from German Landrace, bred and raised in the experimental pig unit of the Leibniz Institute for Farm Animal Biology, were used in two experiments. All pigs received standard processing (oral iron supplementation and castration) within the first 3 d of life. At the beginning of the experiments, pigs were 7 wks old and weighed between 12 and 18 kg. Their health was checked continuously by visual inspection during the wk previous to the experiments. All the procedures involving animal handling and treatment were in strict accordance with the German animal protection law and approved by the relevant authorities (Landesamt für Landwirtschaft, Lebensmittelsicherheit und Fischerei, Mecklenburg-Vorpommern, Germany; LALLF M-V/TSD/7221.3-2.1-005/08, LALLF M-V/TSD/7221.3-2.2-014/09, LALLF M-V/TSD/7221.3-2.2-003/10).
Experimental design
Experiment 1: in vivo LPS administration
One wk before LPS administration, the animals were kept individually. To avoid stress, weighing of the pigs was carried out 3 d before starting the experiments. For the time of LPS and saline administration an approximated body mass (BM) was calculated. Each pen was equipped with a feeder and a nipple waterer. Pigs were fed
Experiment 2: ex vivo LPS stimulation in whole blood culture system
Seven-wk-old pigs were kept in group housing with free access to feed and water. Blood was taken as described above at 8:00 a.m. from 10 healthy untreated pigs. The cultivation of porcine blood in whole blood culture system was used for
Blood and tissue sampling
Blood samples were collected in ice-cooled tubes containing EDTA, subsequently centrifuged at 2000
TNF-α and IL-10 assays
Concentrations of TNF-α and IL-10 were analyzed in blood plasma and brain tissues using a commercially available pig ELISA kit (Invitrogen, Frederick, MD, USA). Analyses were performed according to the manufacturer’s instructions. The sensitivities of the TNF-α and IL-10 assays were 3 pg/ml. The intra- and inter-assay coefficients of variation (CV) of TNF-α were 6.2% and 8.2% respectively. The intra- and inter-assay CVs of IL-10 were 6.3% and 9.4% respectively. For cytokine analysis in hippocampus and PFC, samples were homogenized in ice-cold buffer containing a protease inhibitor cocktail and incubated at 4℃ for 60 min. Homogenate was centrifuged at 20,000
Quantification of plasma Trp and metabolites of the Kyn pathway
The method used in this study was described recently by Kiank
Western blot analysis
IDO protein expression was analyzed in plasma, supernatants of whole blood cultures and in several tissues. Tissues were homogenized with radioimmunoprecipitation assay buffer (50 mM Tris-HCl, 150 mM NaCl, 0.5% sodium deoxycholate, 10 mM EDTA, 1% SDS; pH 7.5) including a protease inhibitor cocktail (Sigma Aldrich). After ultracentrifugation at 40,000
Statistical analyses
Statistical analyses were performed using SAS software, version 9.2 (SAS, Cary, NC, USA). The data were evaluated by ANOVA using the mixed procedure. For the data from experiment 1, the model comprised the fixed effects treatment (LPS, saline), time (0, 1, 3, 6, 24 h), replication (2), their two-way interactions and the random effect sow. Repeated measures on the same animal were taken into account using the repeated statement in the MIXED procedure and a compound symmetry block diagonal structure of the residual covariance matrix. For the evaluation of the data from experiment 2, the differences between LPS-stimulated and unstimulated control samples (Δ) of TNF-α, Trp, Kyn, Kyna, Quin concentrations and Kyn/Trp were calculated. The model for the resulting difference variable ΔTNF-α, ΔTrp, ΔKyn, ΔKyna, ΔQuin and ΔKyn/Trp ratio comprised the fixed effect cultivation time (4, 6, 24 h) and the random sow effect. Least-square means (LS-means) and their standard errors (SE) were calculated and tested for each fixed effect in the models described earlier using the Tukey–Kramer procedure for all pair-wise multiple comparisons. Differences were considered significant if
Results
Experiment 1: IDO activation by in vivo LPS administration
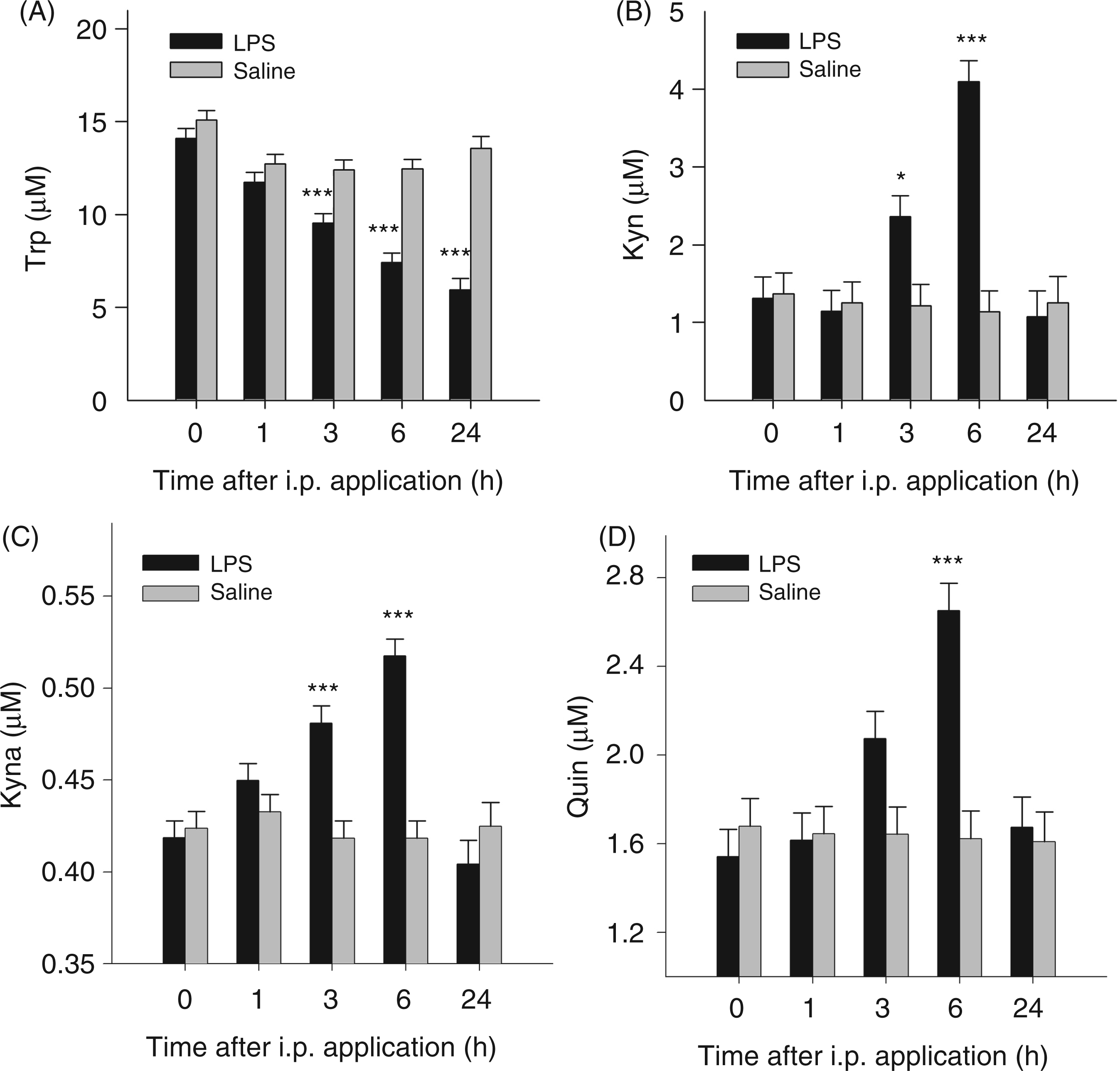
ANOVA revealed a significant effect of the interaction treatment × time on plasma concentrations of TNF-α, IL-10, Trp, Kyn, Kyn/Trp ratio, Kyna and Quin (for all parameters Plasma concentrations of TNF-α (A) and IL-10 (B) after i.p. LPS or saline administration. Cytokines were measured at different time points in blood plasma by ELISA. Results are presented as LS-means + SE. Significant differences between LPS- and saline-treated groups are shown for each time point. For time points 0, 1, 3 and 6 h, Tryptophan metabolites in blood plasma after i.p. LPS or saline administration. Trp (A), Kyn (B), Kyna (C) and Quin (D) were measured at different time points by tandem mass spectrometry. Results are presented as LS-means + SE and significant differences between LPS- and saline-treated groups are shown for each time point. For time points 0, 1, 3 and 6 h, Kyn/Trp ratio (A) in plasma after i.p. LPS or saline adminstration. Kyn/Trp ratio is presented as LS-means + SE and significant differences between LPS- and saline-treated groups are shown within each time point. For time points 0, 1, 3 and 6 h, 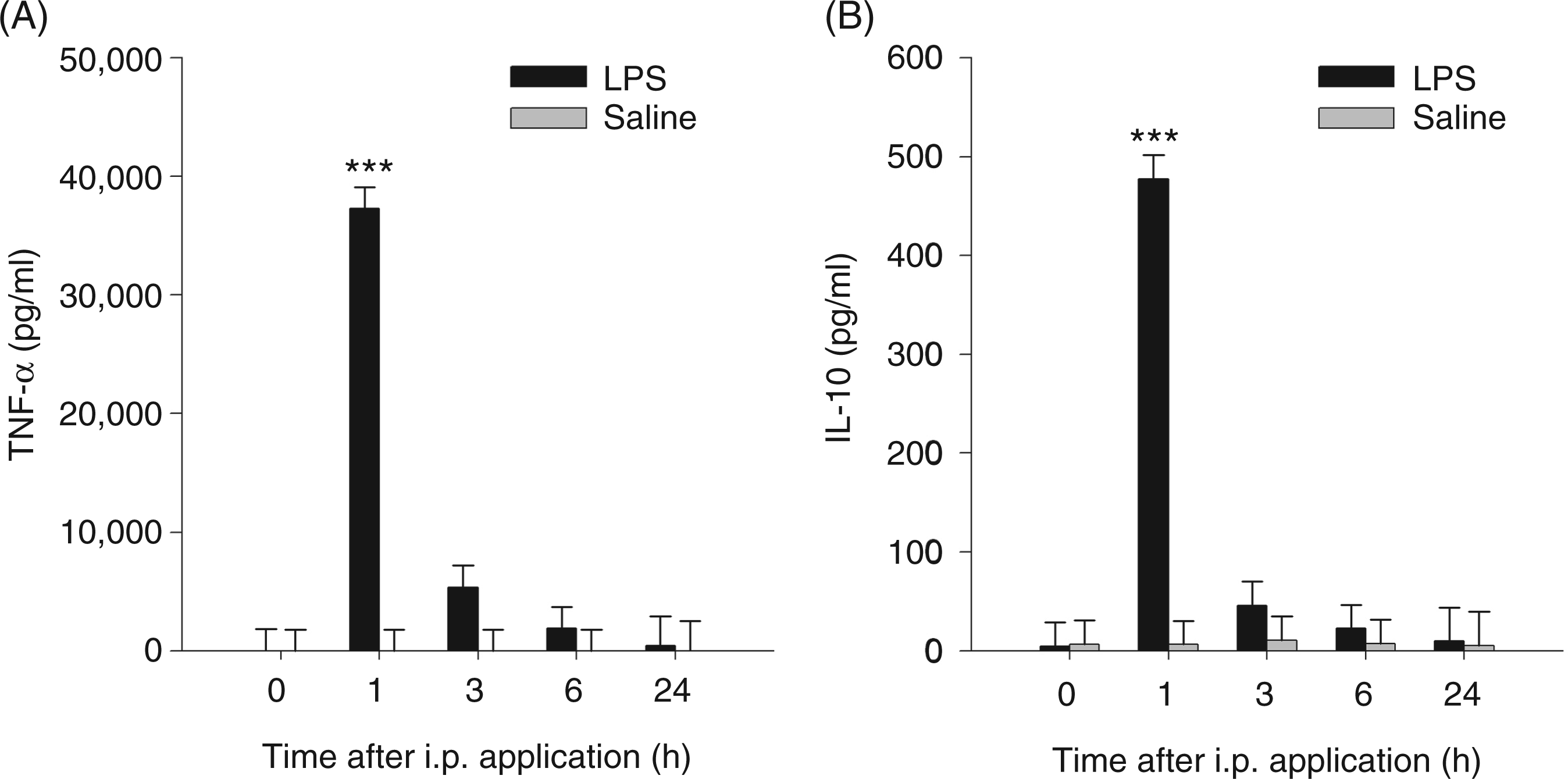
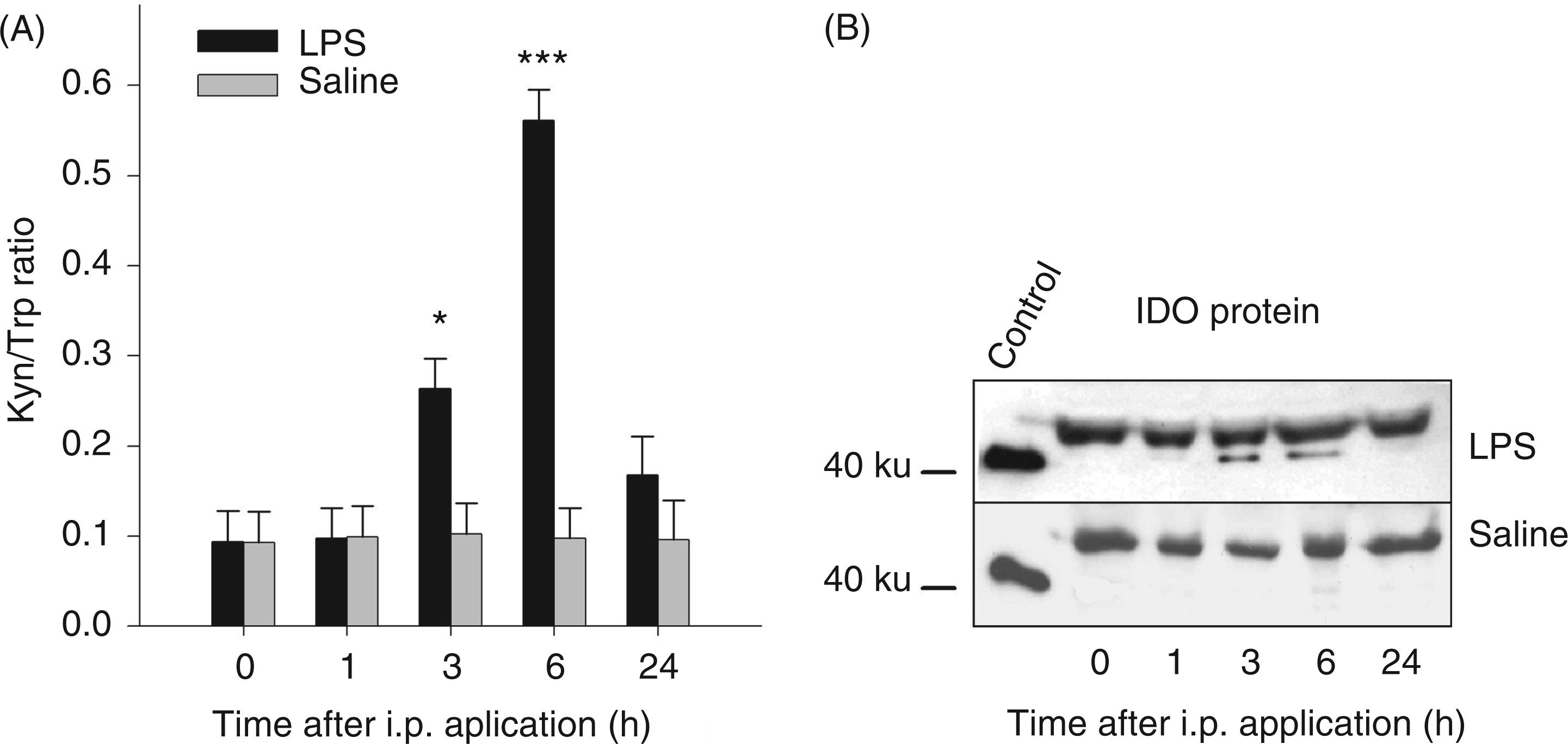
Western blot analyses showed that LPS affected IDO protein expression in blood plasma. One constitutive protein form with a molecular mass of approximately 50 ku was found in the plasma of LPS- and saline-treated groups at all times. A variant with ∼42 ku was induced 3 and 6 h after LPS administration (Figure 3B).
The IDO protein expression in peripheral and brain tissues was analyzed 6 h after LPS application. Western blot analyses of tissue supernatants showed LPS-induced IDO protein expression in the liver and lung, whereas IDO expression in muscle was not affected by the treatment (Figure 4A). In the liver, we found two forms of IDO protein. One variant with a molecular mass of ∼37 ku was detected in both LPS- and saline-treated groups, whereas a second form with a molecular mass of ∼34 ku was only detected in LPS-treated pigs. In the lung, LPS-treated pigs displayed one form of IDO protein with a molecular mass of approximately 34 ku, whereas no protein expression was found in saline-treated pigs (Figure 4A). Analysis of protein expression in different areas of the brain (amygdala, hippocampus, hypothalamus, pituitary and PFC) revealed no detectable IDO protein expression 6 h after LPS (Figure 4B). There were also no changes in concentrations of TNF-α and IL-10 in hippocampus and PFC 6 and 24 h after LPS injection (Table 1).
IDO protein expression in tissues after i.p. LPS or saline (sal) administration. Tissue samples were taken 6 h after treatment. IDO protein expression was detected by Western blot analysis in liver, lung and muscle (A), and in the brain areas amygdala (amy), hippocampus (hipp), hypothalamus (hyp), pituitary (pit) and PFC (B) 6 h after LPS. Protein (20 µg) was added to each lane; for loading control, β-actin was used. IDO (human): 293T lysate was used as a positive control. One representative experiment of three is shown. Concentrations of TNF-α and IL-10 in hippocampus and PFC after Data are expressed as LS-means ± SE. 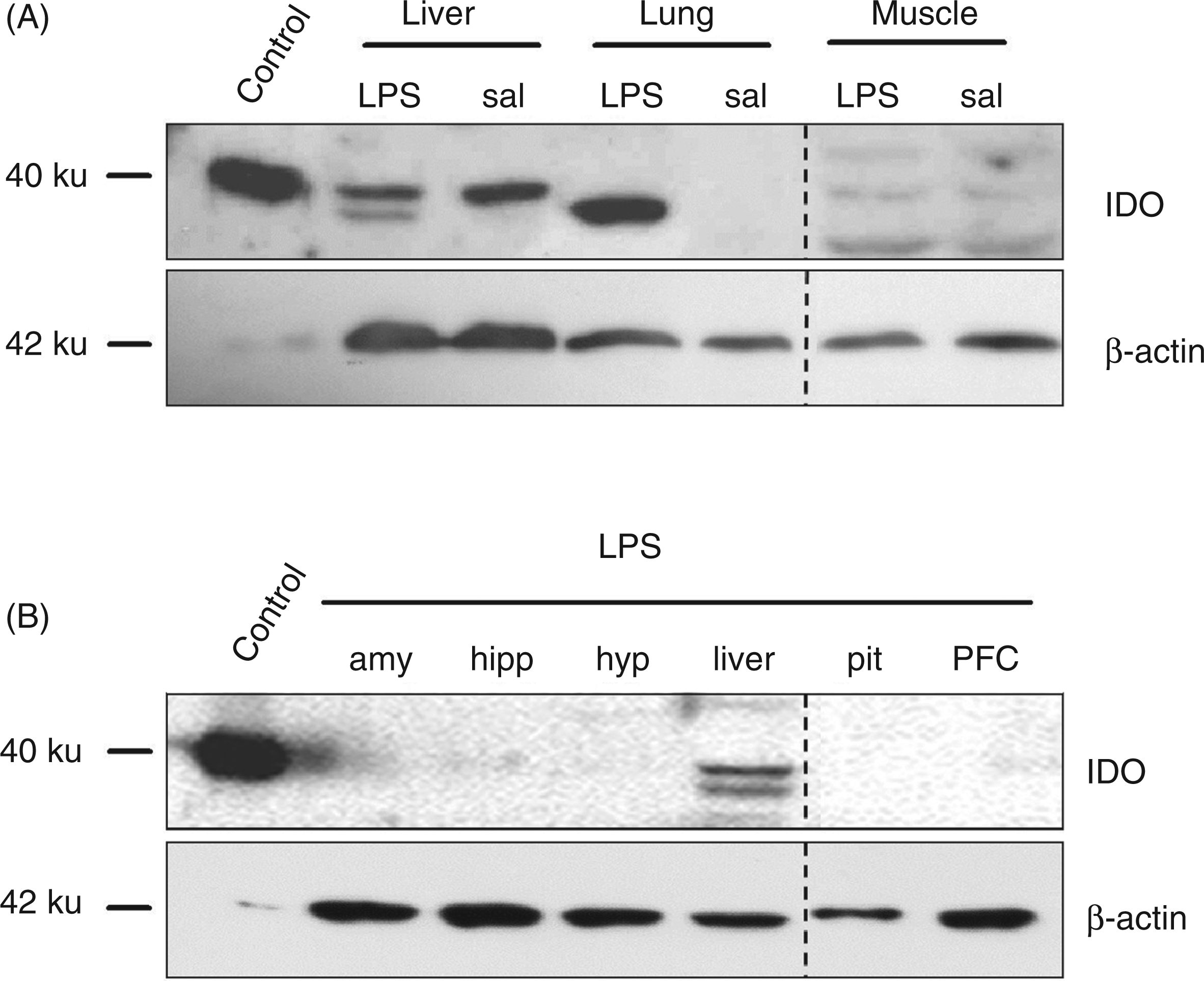
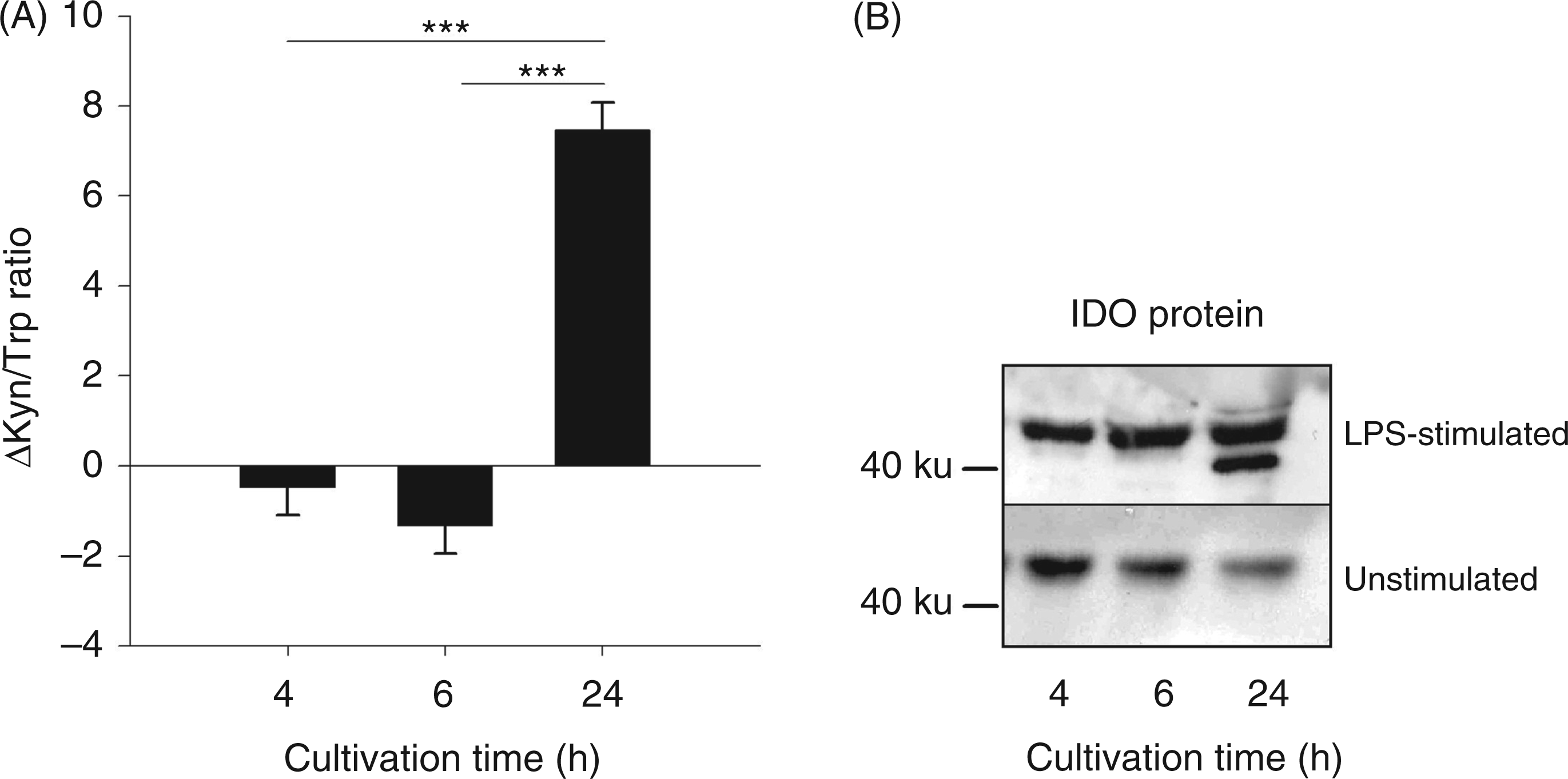
Experiment 2: IDO activation by ex vivo LPS stimulation
ANOVA revealed a significant effect of cultivation time on ΔTrp, ΔKyn, ΔKyn/Trp ratio and ΔQuin (for all parameters Concentrations of tryptophan metabolites in whole blood cultures. Blood was cultivated for 4, 6 or 24 h with or without LPS. The metabolites Trp (A), Kyn (B), Kyna (C) and Quin (D) were measured in supernatant by tandem mass spectrometry. Results are presented as LS-means ± SE of differences (Δ) between LPS-stimulated and unstimulated control blood samples: Kyn/Trp ratio (A) and IDO protein expression (B) in whole blood cultures. Kyn/Trp ratio is presented as LS-means ± SE of differences (Δ) between LPS-stimulated and unstimulated control blood samples. Proteins were detected by Western blot analysis. Protein (30 µg) was added to each lane. One representative experiment of three is shown. *** 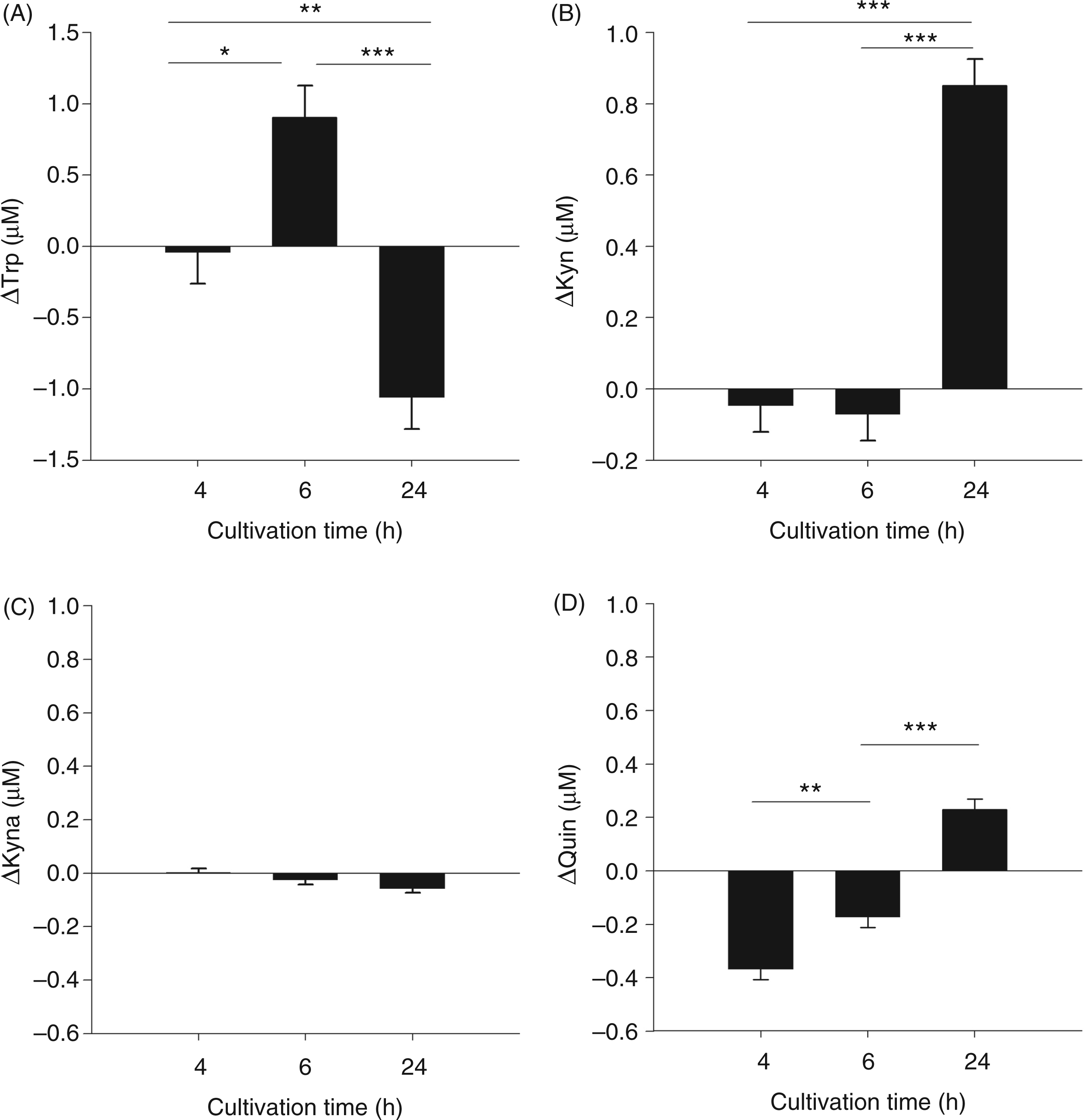
Discussion
The present study investigates the
The
Previous studies with pigs showed that the
It was shown that LPS can induce a release of cytokines in the brain.31–33 However, the results of the present study showed that the concentrations of TNF-α and IL-10 were unaffected by LPS treatment, at the time points measured, in the hippocampus and the PFC. It is possible that a single peripheral administration of LPS is not able to induce an increase of the cytokine concentrations in the brain. Alternatively, cytokine release in the brain follows a different temporary dynamics, which may not be covered by the time points chosen in the experiment.
Our data show that
In the present study, LPS application induced the accumulation of specific plasma Trp metabolites indicating the activation of the kynurenine pathway. The time courses of Kyna and Quin plasma concentrations were similar to that of Kyn after LPS administration. It was shown in gerbils that i.p. application of LPS together with galactosamine induced an increase of Quin in serum and tissues, and the magnitude of the Quin concentration also correlated with the increase of Kyn. 39 These findings indicate that Kyn is the rate-limiting substrate for the production of Kyna and Quin.
Western blot analyses showed that LPS induced IDO protein expression in blood and tissues. In plasma, the expression of the inducible protein form with a molecular mass of ∼42 ku correlated with the time of elevated plasma Kyn concentrations. In addition to the studies in blood, protein expression was measured in peripheral tissues and in the brain, detecting an LPS-induced IDO expression in the liver and in the lung, but not in the muscle and the analyzed brain areas. These results support previously-described findings of a tissue-specific IDO expression.26,40 Besides the inducible protein form, we detected a constitutively expressed IDO protein form in blood and liver. Western blot analyses in human eosinophils also detected two IDO forms with different molecular masses, one constitutive (∼45 ku) and one IFN-γ-inducible form (∼42 ku), 41 comparable with our results.
In addition to the
In summary, the present study characterizes
Footnotes
Funding
Acknowledgements
The authors are grateful to Dr Cornelia Müller for assisting in quantification of Trp and its metabolites. We thank the staff of the Research Unit Behavioural Physiology for technical assistance, particularly Birgit Sobczak and Regina Wal. Furthermore, we thank the staff of the experimental pig unit for their assistance with the handling of the animals.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
