Abstract
Leishmania parasites induce an immunomodulation by subverting the host immune response towards a CD4+ Th2 lymphocytic cell response that favors parasite persistence. Here, we report that after successful treatment of visceral leishmaniasis due to Leishmania infantum, an immune reconstitution syndrome revealing hip septic arthritis was associated with a switch from Th2 towards a Th1 cytokine profile, and a decrease in the level of immunomodulating factors, such as soluble HLA-G and indoleamine 2,3-dioxygenase (IDO) activity. We then measured IDO activity in a cohort of 39 patients and uninfected control subjects. Results showed significantly enhanced IDO activity in patients with visceral Leishmania infection, compared with uninfected control subjects (P < 0.001), but also compared with treated patients (P < 0.05). A decrease in IDO activity could constitute a relevant biomarker for the restoration of the immune response during visceral leishmaniasis.
Keywords
Introduction
Visceral leishmaniasis (VL) is a severe parasitic infection due to Leishmania infantum in the mediterranean basin, notably as an opportunistic infection during AIDS. 1 In immunocompetent hosts, Leishmania parasites induce an immunomodulation by subverting the host immune response towards a CD4+ Th2 lymphocytic cell response that favors parasite persistence. After successful treatment, CD8+ T cell expansion combined with a switch from a Th2 to a Th1 response are the hallmarks of the resolution of immunosuppression. 1 Only a few cases of ‘opportunistic infections’ during VL have been reported, i.e. tuberculosis and leprosy, which led to the debate of which infection underlined the other.2–4 More recently, the concept of immune reconstitution syndrome (IRS) and its immunological mechanism has emerged. IRS is well known during the course of AIDS disease and solid organ transplantation (SOT), and was reported to be associated with various bacterial and viral infections, and also fungal and parasitic infections.5,6 This syndrome has been described on the observation of unexplained severe concomitant infections, rising mainly in two situations: (i) after initiation of potent highly active antiretroviral therapy to treat HIV/AIDS and (ii) after reduction or withdrawal of immunosuppressive agents in SOT patients. In the literature, few cases of leishmaniasis that developed in the context of IRS were described, and all occurred in patients co-infected with HIV.5,7–9 Here, we report, for the first time, that VL can also be an underlying disease predisposing to the development of IRS. We investigated the immunologic environment through dosages of serum biomarkers, including cytokines, soluble histocompatibility-leukocyte-antigen-G (sHLA-G) molecule and indoleamine 2,3-dioxygenase (IDO) activity. IDO has been reported recently as an immunoregulatory enzyme increased in lymph node in cutaneous Leishmania major infection. 10 In addition, we determined IDO activity in blood samples of a retrospective cohort of VL and control patients in order to analyze its interest as a biomarker of immune suppression and reconstitution during VL.
Materials and methods
Patients
Case report of IRS after VL
A 75-year-old patient, living in France, presented with febrile hepatosplenomegaly and pancytopenia. The only noteworthy background consisted of right total hip arthroplasty for coxarthrosis 3 years ago. The patient also reported pain in the right hip appearing a few months prior to presentation, whilst holidaying in the South of France. VL is endemic in the south of France. A bone marrow aspiration was performed, which allowed the diagnosis of VL on a Giemsa-stained smear demonstrating intracellular amastigotes. The parasitic strain could be isolated by culture on Novy-McNeal-Nicolle medium and identified as L. infantum (Leishmania National Reference Center, University Montpellier 1). After treatment with liposomal amphotericin B at a dose regimen of 3 mg/kg at d 1, 2, 3, 4, 5 and 10, the patient presented with septic shock in relation to a systemic infection with a methicillin-sensitive Staphylococcus aureus, which was further proven to be related to a joint infection consecutive to the right total hip arthroplasty. Treatment included combined adapted anti-biotherapy and surgery. After 16 d of treatment with intravenous oxacillin and gentamycin, a negative culture of the abscess puncture was obtained. Treatment was switched to oral ofloxacin and rifampicin for 15 d and allowed a 1-stage revision of the acetabular cup without femoral stem changing. The total duration of anti-biotherapy was 3 months. A definitive cure of both VL and septic arthritis was confirmed after long-term follow-up of the patient, without relapse or sequelae. An immunological analysis was then performed in order to characterize this episode.
Cohort of VL patients and control participants
Forty-three blood samples from 39 participants were obtained retrospectively from Marseille and Rennes teaching hospitals (Table 2). Blood samples from VL patients were collected at the time of their primary diagnosis (n = 12 + IRS-VL patient) and during follow-up after treatment for four of them. Cases of HIV-VL co-infection were excluded from this study. The remaining 26 subjects were healthy blood donors. Local ethical committee approval was received for the study and informed consent of all participants was obtained. All samples were anonymized according to the French legislation for biomedical research and stored at −20℃ until use.
Cytokine assays
IL-12, IL-10, IL-8 and IL-6 blood levels were measured in order to characterize the inflammatory and cytokinic environment of the IRS-VL patients. These tests were performed with the patients’ sera at the acute phase of VL and 3 months after anti-parasitic treatment by flow immunocytometry using the human inflammatory cytokine CBA kit® (Beckman Coulter, Villepinte, France).
sHLA-G assay
The level of sHLA-G was measured with a specific ELISA method validated at the Wet Workshop for Quantification of sHLA-G (2004; Essen, Germany). Results were calculated from a standard curve prepared with serial dilutions of HLA-G5 purified from LCL-721–221-G5 cell-line culture supernatant and expressed in ng/ml as described previously. 10 This detection was performed with the IRS patient sera at the acute phase of VL and 3 months after anti-parasitic treatment.
IDO activity assessment
Systemic IDO activity was determined by measuring kynurenine and tryptophan levels (in µmol/l) by HPLC, using 3-nitro-
Statistical analysis
Data are expressed as mean ± SEM for each group of patients. Differences between groups were analyzed using non-parametric tests (Mann–Whitney test). Statistical analysis was performed using GraphPad Prism 5.02 software. Differences were considered significant when the P-value was < 0.05, and graduated as *(P < 0.05), **(P < 0.01) and ***(P < 0.001).
Results
Biomarkers of immune reconstitution after efficient treatment of the clinical case
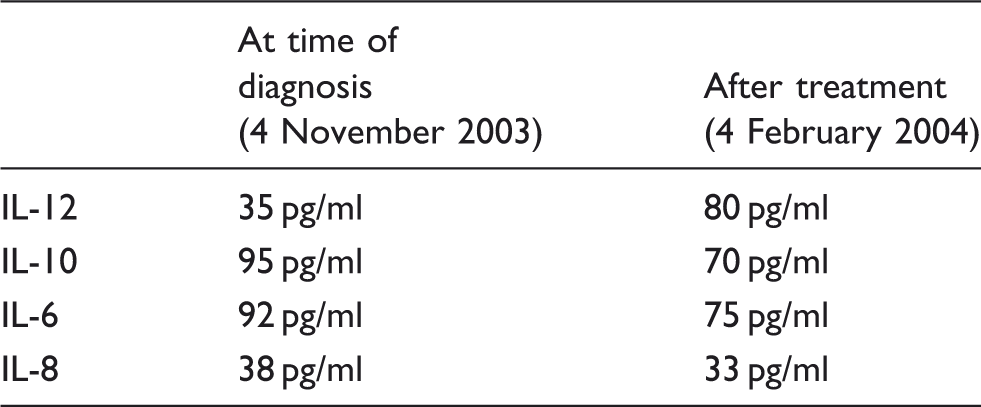
IL-12, IL-10, IL-8 and IL-6 levels were measured at the time of diagnosis and after treatment of the IRS-VL patient to characterize the cytokinic environment. During the acute phase of VL, the patient exhibited a high level of IL-10 (95 pg/ml) and IL-6 (92 pg/ml), and a low level of IL-12 (35 pg/ml; Table 1). After successful treatment with liposomal amphotericin B, IL-12 showed a twofold increase, whereas IL-10 and IL-6 decreased, suggesting a switch from a Th2 to a Th1 cytokinic environment. Unfortunately, IL-4 and IFN-γ could not be measured because of the limited volume of blood samples. Besides, IL-8 remained stable at a low level before and after treatment (38 and 33 pg/ml respectively). Regarding the level of sHLA-G, serum concentration level (85 and 89 ng/ml) during the initial phase was much higher than that usually observed during VL (a median sHLA-G concentration of 24 ng/ml was observed in a previous study) and similar to levels commonly observed during HIV infection.
10
We also observed that after efficient anti-parasitic treatment, sHLA-G levels progressively decreased in our patient to 20 ng/ml (plasma concentration in a control population
10
) 3 months after the end of therapy (Figure 1). Finally, systemic IDO activity was enhanced at the initial phase of diagnosis (0.331) and dramatically decreased after treatment (0.067).
sHLA-G levels determined by ELISA at different time points in the patient with IRS-VL. Decreasing titers were observed from onset of treatment (4 November 2003) to successful outcome (4 February 2004). Dotted line indicates baseline levels observed in healthy subjects. Cytokine blood levels in the patient with IRS-VL, determined by flow immunocytometry. Demographic and clinical characteristics of VL patients and healthy controls and results of plasmatic IDO activity (= ratio of kynurenine:tryptophan) (mean ± SEM). HBV : hepatitis B virus.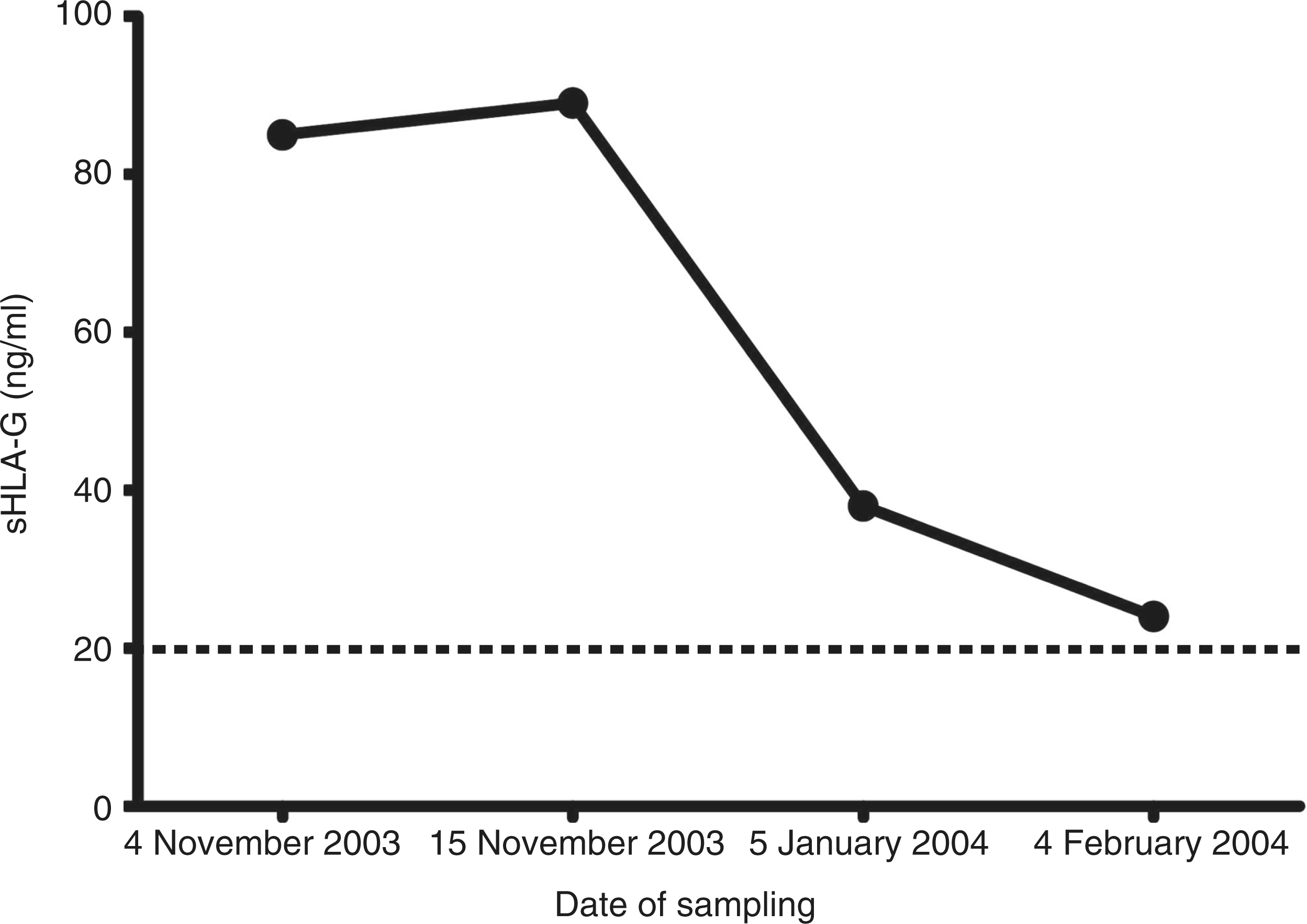

Enhanced IDO activity during VL
Systemic IDO activity was determined by measuring kynurenine and tryptophan in plasma samples of our patient and compared with samples obtained from 12 other VL patients at the time of diagnosis, 4 treated VL patients and 26 healthy participants. IDO activity was much higher in VL patients than in healthy participants (0.194 ± 0.03 vs 0.029 ± 0.001, P < 0.001). When a follow-up after treatment was performed, IDO activity had decreased significantly in treated patients (P < 0.05), but remained, however, at significantly higher levels than healthy controls (P < 0.01) (Figure 2).
IDO activity (= ratio of kynurenine:tryptophan) measured in the plasma of patients with visceral leishmaniasis before (VL; n = 13) and after treatment (treated VL; n = 4). Samples from healthy participants (n = 26) were used as controls (*P < 0.05; **P < 0.01; ***P < 0.001).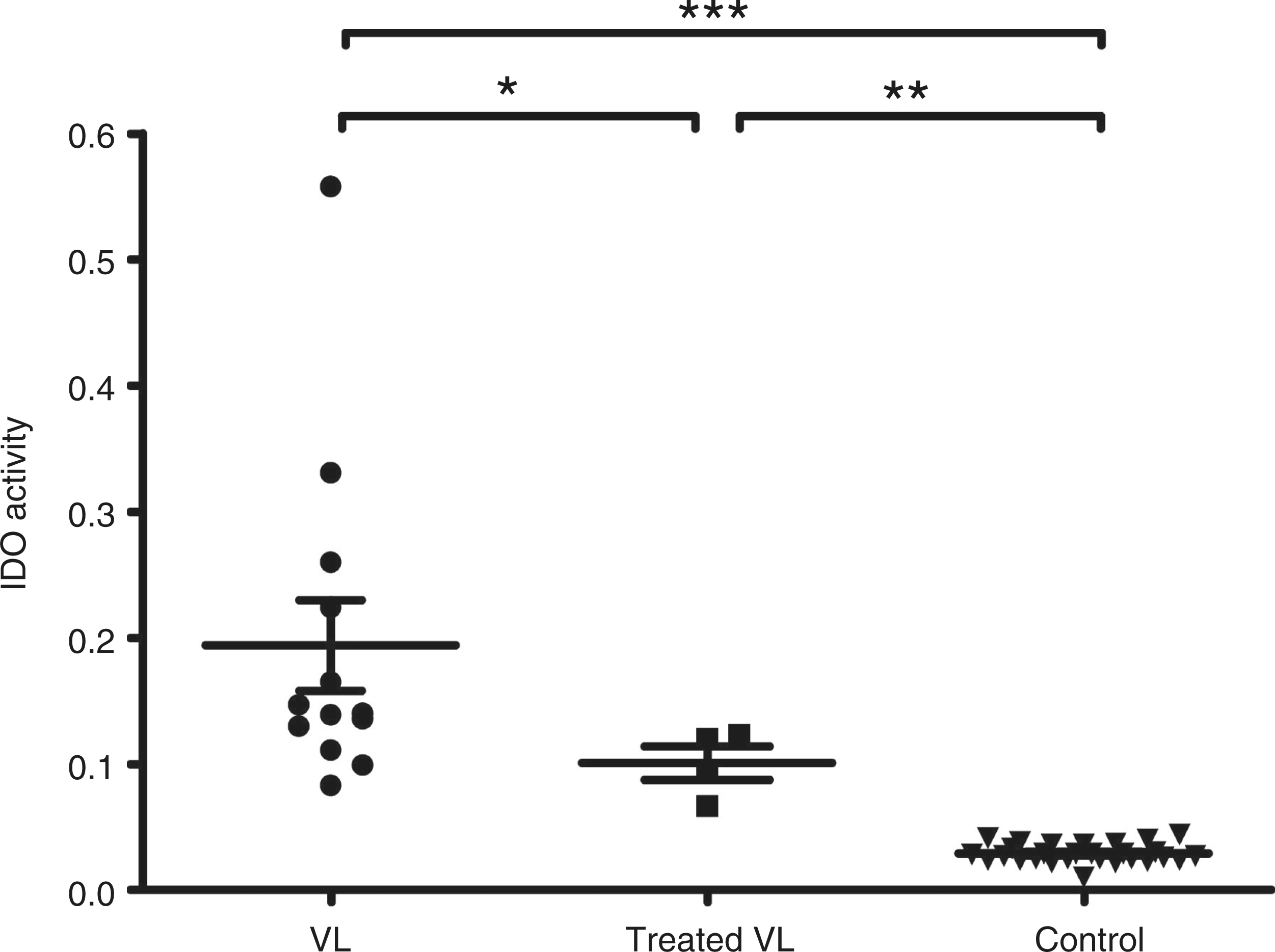
Discussion
We report here that the underlying background for IRS-related pyogenic infection is in our original clinical case the L. infantum infection itself, as investigations ruled out major causes of immunosuppression, i.e. hematological malignancy, solid cancer, HIV infection, other common causes of infection and diabetes. This observation can be explained by the immunosuppressive character of the Leishmania infection itself. Treatment with liposomal amphotericin B resulted in the regression of clinical signs and the return to normal of white and red blood cell and platelet counts, as reported previously. 12 The resulting increase in phagocytic cell function efficiency was associated with a shift from a Th2 response towards a Th1 cell response. This immune reconstitution enabled an inflammatory response with biological and clinical signs to develop, in response to a previously silent pyogenic infection. An original approach in this work was to measure the level of sHLA-G and systemic IDO activity. HLA-G are non-classical class I MHC molecules that have tolerogenic effects, such as inhibition of allogeneic T cell proliferation and natural killer cell-mediated cytolysis.13,14 Materno-fetal tolerance, hematological malignancies and also infection with HIV are characterized by high sHLA-G levels.13,14 We showed previously that patients display elevated levels of sHLA-G associated with a Th2 cytokine profile during VL. We therefore hypothesized that sHLA-G could contribute to the impairment of host resistance to Leishmania through suppression of NK cell-mediated cytolysis and reduction of IFN-γ production by T cells–two mechanisms that are essential in the control of Leishmania infection. 10 Here, we observed that sHLA-G may also represent an immune biomarker of successful treatment. Besides, it was shown that IDO can induce tolerance and favor chronic infections through suppression of T cell proliferation resulting from either anergy or T-cell death mediated by tryptophane depletion and/or toxic metabolites. 14 Two recent studies showed that during experimental L. major and human Leishmania guyanensis cutaneous infections, IDO is overexpressed locally and subsequently attenuates the T cell stimulatory functions of dendritic cells and suppresses local T cell response, particularly the regulatory T cell response.15,16 Here, we show, for the first time, that systemic IDO activity is detectable in the plasma of patients during visceral Leishmania infection and that the IDO pathway could also act as a molecular switch regulating host response. In a small number of patients, we also showed that after successful treatment, a decrease is observed, although it does not revert to the levels of healthy controls. This can be owing either to the average delay between diagnosis and time of sampling after treatment (a maximum of 3 months), but can also be a marker of asymptomatic carriage, even after clinical recovery. The limitation of our retrospective study is the small number of patients for which plasma was stored after treatment and thus available for dosage. However, these preliminary data encourage evaluation of the role of IDO in larger and prospective cohorts of patients, all the more so as, in a recent paper, Straten and Andersen proposed that IDO-specific T cells could, potentially, play a role in Leishmania vaccination in the near future. 17
Measurement of IDO activity and sHLA-G may thus constitute, together with Th1/Th2 cytokines levels, surrogate markers to indicate the resolution of VL, at least in immunocompetent patients. This point should be addressed in a prospective study aiming to characterize the kinetics of soluble biomarkers of the immune restoration after successful treatment compared to parasitological tools, such as direct examination, culture and anti-Leishmania Abs.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
