Abstract
The fatal H5N1 infection has a high mortality rate among infected patients. The pathogenesis of H5N1 viral infection is associated with the ability of the virus to induce apoptotic cell death. However, the molecular mechanism of apoptosis induced by H5N1 remains unclear. In the present study we demonstrate that H5N1 virus is able to up-regulate the expression of gene associated with retinoid and interferon induced mortality-19 (GRIM-19) in human monocyte-derived macrophages (hMDMs). GRIM-19 has been identified as a novel gene with apoptotic effects in virus-infected cells. The percentage of apoptotic cells is significantly decreased in H5N1-infected GRIM-19 depleted hMDMs, which is also associated with a decrease of BH3-interacting domain death agonist cleavage and apoptosis-inducing factor (AIF) release to the cytosol. These results suggested the involvement of GRIM-19 in apoptosis induced by H5N1 virus. Furthermore, neutralizing-IFN-β Ab is able to suppress GRIM-19 expression in H5N1-infected cells resulting in a decrease in apoptotic cell number, indicating that IFN-β secreted by H5N1-infected hMDMs regulates GRIM-19 expression leading to apoptosis. Altogether, the results presented here provide additional insight on the regulatory mechanism of H5N1 viral-induced apoptotic cell death in hMDMs.
Introduction
Since 2003, zoonotic transmission to humans of the highly pathogenic avian influenza H5N1 virus continues to pose a threat to human health with concerns regarding a possible human influenza pandemic. Rapid progression from severe pneumonia to acute respiratory distress syndrome is the primary cause of death in patients with H5N1 disease. The presence of lymphopenia, hypercytokinemia and hemophagocytosis is the prominent clinical manifestation of the disease.1–3 Apoptosis has been shown to be a cellular killing mechanism during H5N1 infection. This virus is able to activate apoptosis via both the extrinsic and mitochondrial intrinsic pathways through FAS-FASL/TRAIL and the release of mitochondrial apoptogenic proteins respectively.4–6 Triggering either the extrinsic or intrinsic pathway activates further downstream caspases—caspase-8, 10, 3 and 7—leading to apoptosis.
Additionally, cytokine dysregulation has also been implicated in the severity of H5N1 disease. Several lines of evidence have demonstrated that the level of several cytokines and chemokines, for example IP-10, IL-6, IL-8 and IFN-β, was markedly elevated in sera and lung of patients with H5N1 disease.7,8 Hyper-induced expression of cytokines and chemokines by H5N1 virus has been demonstrated in both in vitro9–11 and in vivo.12,13 Of interest, IFN-β, a type I IFN produced at a high level during infection, may serve as a first line of defense against viral infection by inducing expression of several antiviral proteins including PKR, MxA, ISG15 via the JAK/STAT pathway. 14 In addition to its antiviral function, the role of IFN-β in inducing apoptosis was also demonstrated. For example, apoptosis induced by encephalomyocarditis virus infection in mouse embryonic fibroblasts is suppressed in the presence of anti-IFNα/β Abs. 15 IFN-β can also up-regulate the expression of apoptotic genes involved in the cell death process, such as TRAIL16,17 and X-linked inhibitor of apoptosis (XIAP) associated factor-1 (XAF1). 18 These findings suggest that IFN-β induced by H5N1 virus infection may exert a role in modulating cell death.
Recently, gene associated with retinoid and interferon induced mortality-19 (GRIM-19) was identified as a novel protein that regulates cell death induced by IFN-β and its expression can be enhanced with a combination treatment containing retinoic acid. 19 GRIM-19 promotes apoptosis by inhibiting STAT-3 resulting in the down-regulation of anti-apoptotic proteins, such as BCL-2 and BCL-XL. 20 In addition, in the presence of IFN-β and retinoic acid, GRIM-19 has been shown to interact with HtrA2—a mammalian serine protease released from mitochondria—thereby amplifying cell death by decreasing the anti-apoptotic protein, XIAP. Furthermore, depletion of GRIM-19 reduces apoptosis in HIV-infected T-cells. 21 In this study, we demonstrated the essential role of GRIM-19, particularly induced by secreted IFN-β, in apoptosis induced by H5N1 virus in human monocyte-derived macrophages (hMDMs).
Materials and methods
Virus isolation
Virus used in this study was an A/open-billed stork/Nakhonsawan/BBD0104F/04 strain of H5N1 influenza virus which was propagated in Madin–Darby canine kidney cells, 22 and was the same strain demonstrated previously to induce apoptosis in hMDMs. 5 Virus stock was aliquoted and stored at –80 °C until use.
hMDM preparation
hMDMs were prepared from heparinized blood from healthy individuals by density gradient centrifugation using Ficoll-Paque (Lymphoprep™; Axis-shield PoCAS, Oslo, Norway) and purified by CD14+ MACS magnetic beads (MACS®; Miltenyi Biotech GmbH, Bergisch Gladbach, Germany) as described previously.
5
The cells were seeded onto tissue culture plates in Iscove's Modified Dulbecco’s medium (Gibco, Grand Island, NY, USA) supplemented with 10% heat-inactivated human AB serum (Sigma, St Louis, MO, USA) and 1%
siRNA transfection
Pre-designed siRNAs against GRIM-19 (sense: 5′-GGCCAUCAUGAAGGACTT-3′, antisense: 5′-GUCCUUCAUGAUGAUGGCTC-3′) were purchased from Ambion Inc. (Life Technologies, New York, NY, USA). hMDMs were transfected with 40 nmol siRNA-GRIM-19 using Lipofectamine2000 reagent, according to manufacturer’s recommendation (Invitrogen, New York, NY, USA). As controls, the hMDMs were transfected with the same amounts of negative siRNA control. After 24 h incubation, the expression of GRIM-19 mRNA and protein were determined by RT-PCR and immunoblotting respectively.
Infection of hMDMs
The human macrophages were infected at a multiplicity of infection (MOI) of 1, in the presence or absence of neutralizing IFN-β Ab 600 IU/ml or IgG isotype control (PBL InterferonSource, Piscataway, NJ, USA). At the times indicated, depending on the experimental design, cells were collected for apoptosis analysis by TUNEL staining, RT-PCR and immunoblotting.
TUNEL staining
Apoptotic cells were examined following fixation of the cells for at least 30 min with 4% paraformaldehyde in PBS. The cells were stained with TUNEL reaction mixture (Roche Diagnostics, Mannheim, Germany) for 60 min at 37°C in a humidified chamber, and then incubated with alkaline phosphatase-conjugated FITC-labeled Ab for 30 min at 37°C. The apoptotic cells were observed under a fluorescence microscope and the percentage of apoptotic cells were determined by counting at least 500 cells per experimental condition.
RT-PCR
Total RNA was extracted from infected cells according to the manufacturer’s instruction (illustra RNAspin Mini RNA isolation; GE Healthcare, Buckinghamshire, UK) and used for cDNA synthesis (AMV RT; Promega, Madison, WI, USA). Templates for RT-PCR were cDNAs synthesized from total RNA extracted from infected hMDMs. PCR was then performed using primer pairs specific for GRIM-19 (forward: 5′-ACTGCGAGTATGGCGGCGTC-3′; reverse: 5′-TGCAAGGTCCTCCGGTCGGT-3′), BCL-XL (forward: 5′- GAGGCAGGCGACGAGTT-3′, reverse: 5′-TTCCCATAGAGTTCCACAAAA-3′) and GAPDH (forward: 5′-ATGGGGAAGGTGAAGGTCG-3′; reverse: 5′- GGGGTCATTGATGGCAACA-3′).
Immunoblotting analysis
Preparation of cytosolic proteins
At the times indicated, the hMDMs infected with H5N1 were washed twice with PBS and treated with 5 mM EDTA at 37°C for 10 min. The cells were then centrifuged and cell pellets were permeabilized for 10 min in ice-cold buffer [20 mM HEPES pH 7.3, 110 mM potassium acetate, 5 mM sodium acetate, 2 mM magnesium acetate, 2 mM dithiothreitol, 0.1% Triton X-100 and complete protease inhibitor cocktail (Roche Diagnostics, Mannheim, Germany)]. The cell lysates were centrifuged at 14,000 g for 15 min at 4°C and the supernatants were solubilized with SDS-PAGE sample buffer (0.0625 M Tris-HCl, pH 6.8, 1% SDS, 10% glycerol, 5% 2-mercaptoethanol and bromophenol blue) before heating at 100°C for 10 min. The cytosolic fraction was determined for protein concentration by BCA assay (Pierce Biotechnology, Rockford, IL, USA) before being subjected to immunoblotting analysis.
Immunoblotting
Twenty micrograms of total protein from cytosolic fractions was electrophoresed on a 10–15% SDS-PAGE gel and then electro-transferred to a nitrocellulose membrane (Amersham Biosciences, Freiburg, Germany). The membrane was blocked with 5% milk for 1 h before incubating overnight with the appropriate specific monoclonal rabbit Abs to BID and AIF (Cell Signaling, Beverly, MA, USA), monoclonal mouse Abs to GRIM-19 (Santa Cruz Biotechnology, Dallas, TX, USA) or actin (Chemicon, Millipore Corp., Billerica, MA, USA). The blots were then allowed to react with horseradish peroxidase-conjugated goat anti-rabbit or goat anti-mouse immunoglobulin G (Pierce). Protein bands were detected by enhanced chemiluminescence, as recommended by the manufacturer (Roche).
Statistical analysis
The data were evaluated statistically by ANOVA. Significance was considered as a P-value < 0.05. All experiments were performed with at least three donors to account for any donor specific differences. Assays were performed at least three times and in triplicate.
Results
H5N1 virus induces GRIM-19 expression in hMDMs
To determine GRIM-19 expression in H5N1-infected cells, hMDMs were infected with H5N1 virus at MOI of 1 for 4, 8, 12 and 16 h before the expression of the GRIM-19 gene was analyzed by RT-PCR. The results showed that the virus was able to activate GRIM-19 expression within 4 h of infection in a time-dependent manner (Figure 1A). Consistent with its gene expression, the level of GRIM-19 protein detected by immunoblotting was observed at 8 h after infection and gradually increased to the end of the experiment (Figure 1B). These results suggest that H5N1 virus is able to induce GRIM-19 expression in hMDMs.
H5N1 activates GRIM-19 expression. hMDMs were infected with H5N1 virus at a MOI of 1, analysis of gene expression by RT-PCR (A) and protein by immunoblotting (B) for GRIM-19 expression in H5N1 virus-infected hMDMs at the times indicated. GAPDH and β-actin were used as an internal control for RT-PCR and immunoblotting respectively. The results shown are representative of three independent experiments.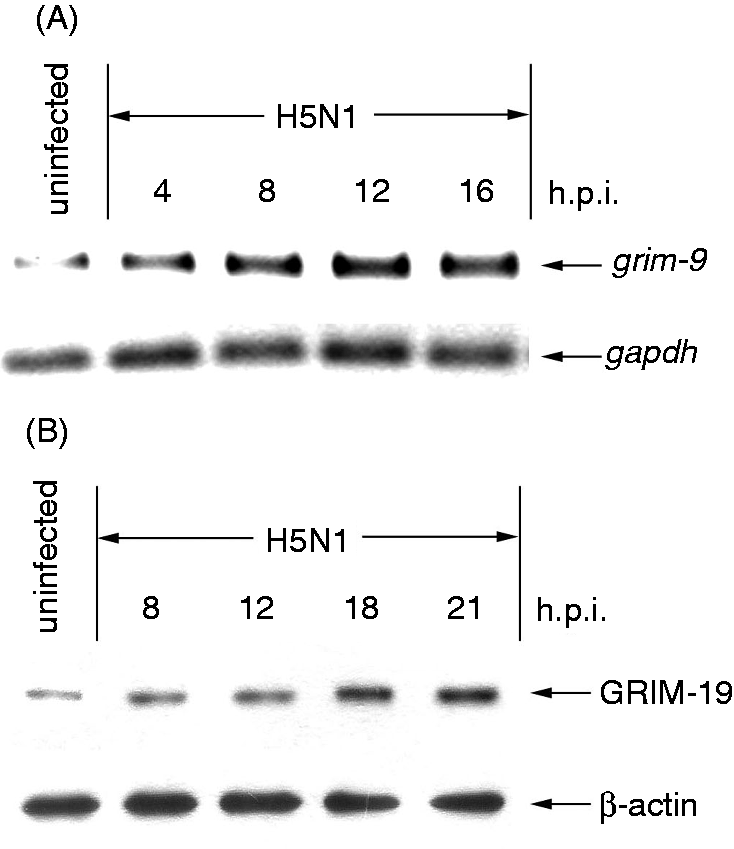
Apoptosis induced by H5N1 virus is suppressed in GRIM-19-depleted cells
To investigate the role of GRIM-19 in H5N1 virus-induced apoptosis, hMDMs were transfected with siRNAs against GRIM-19 or control siRNAs before the cells were infected with the virus. As shown in Figure 2A, the expression of GRIM-19 mRNA was markedly decreased in H5N1 virus-infected GRIM-19-depleted cells when compared with that of control cells. Similar to mRNA expression, up-regulation of GRIM-19 protein was also suppressed in the H5N1 virus-infected GRIM-19-depleted cells (Figure 2B). It should be mentioned that the percentage of H5N1 infection in transfected cells did not differ from that of the untransfected cells, as determined by immunofluoresence staining specific to influenza A nucleoprotein (>90% at 12 h after infection). To determine whether GRIM-19 is involved in apoptosis, GRIM-19-depleted cells were infected with H5N1 virus for 21 h before apoptosis was analyzed by TUNEL staining. As shown in Figure 2C, H5N1 virus could highly induce apoptosis up to 40% in untransfected or siRNA-control transfected cells. In contrast, when the expression of GRIM-19 was suppressed, the percentage of apoptotic cells was significantly reduced to 2% in H5N1 virus-infected cells. These results indicate that GRIM-19 plays a role in apoptotic induction during H5N1 infection.
Depleting GRIM-19 decreases apoptotic induction. siRNAs against GRIM-19 or negative control siRNA were transfected into hMDMs for 24 h before infection. The transfected cells were infected with H5N1 virus at a MOI of 1 before the expression of GRIM-19 gene and protein were determined by RT-PCR at 8 h.p.i. (A) and immunoblotting at 21 h.p.i. (B) respectively. GAPDH and β-actin were used as an internal control for RT-PCR and immnunoblotting respectively. (C) The number of apoptotic cells was determined by TUNEL staining after 21 h.p.i. Data were means and the SEM from independent experiments from three separate donors. *P < 0.05.
GRIM-19 depletion suppresses activation of apoptosis effector molecules
Our previous investigation demonstrated that H5N1 virus was able to induce cleavage of BID and the release of AIF from mitochondria to cytosol resulting in apoptosis in infected hMDMs.
5
In order to determine the effect of GRIM-19 on the apoptosis effector molecules, the GRIM-19-depleted hMDMs were infected with H5N1 virus at a MOI of 1 for 21 h, and the levels of BID degradation and AIF in cytosol were determined by immunoblotting. The data shown in Figure 3A demonstrate that the virus was able to trigger BID cleavage and AIF release to cytosol in siRNA control transfected cells. In contrast, the level of BID in H5N1-infected GRIM-19-depleted cells was maintained at a similar level as that observed in the uninfected control cells. In addition, only a trace of AIF could be detected in the cytosol of infected GRIM-19 depleted cells. These results indicated a role of GRIM-19 in apoptotic induction via BID cleavage and AIF release.
Depleting GRIM-19 affects apoptotic machinery effectors. (A) Detection of BID cleavage and AIF release in H5N1-infectred transfected hMDMs. hMDMs were transfected with siRNAs against GRIM-19 or control siRNAs before infection with H5N1 virus at a MOI of 1 for 21 h. Immunoblotting analysis of BID and AIF proteins in H5N1-infcted transfected cells was performed. Actin was used for the loading control. (B) Expression of BCL-XL mRNA in GRIM-19-depleted hMDMs after H5N1 infection. Human MDMs were transfected with siRNAs against GRIM-19 for 24 h prior to H5N1 infection. Cell lysates were harvested at 8 h.p.i. and the expression of GRIM-19 mRNA was determined by RT-PCR. The results shown are representative of three independent experiments.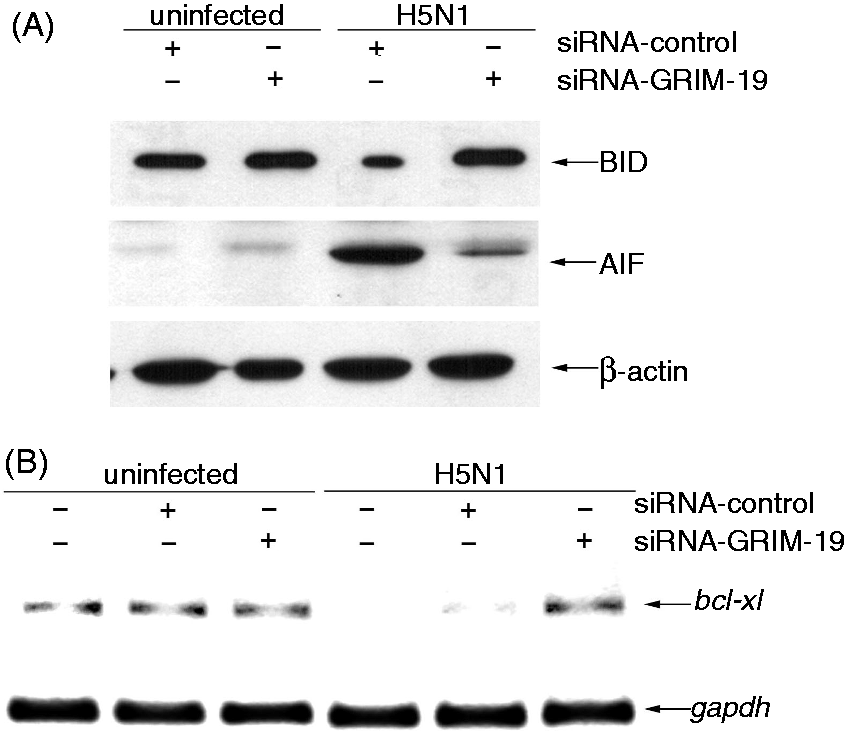
Because GRIM-19 expression has been shown to down-regulate the expression of anti-apoptotic protein BCL-XL during HIV infection, 21 the involvement of GRIM-19 in BCL-XL expression in H5N1 infection was also investigated. In this experiment, the GRIM-19-depleted hMDMs were infected with the virus for 8 h before the expression of BCL-XL was analyzed by RT-PCR. As shown in Figure 3B, the expression of BCL-XL was markedly down-regulated in both H5N1-infected untransfected hMDMs and H5N1-infected siRNA control transfected cells. Conversely, the expression of BCL-XL mRNA was detected in H5N1-infected GRIM-19-depleted cells. These results suggested the expression of GRIM-19 is associated with the down-regulation of BCL-XL in H5N1 virus-infected hMDMs.
IFN-β induced by H5N1 virus is involved in GRIM-19 expression
GRIM-19 was first identified as a novel cell death regulatory gene induced by IFN-β.
19
Current available evidence demonstrates that H5N1 virus is able to activate the expression of IFN-β in infected cells.10,23,24 Under conditions used in the present study, the secretion of IFN-β in the H5N1-infected hMDMs as detected by ELISA (R&D Systems, Minneapolis, MN, USA) increased to about 5000 pg/ml 24 h after infection, while in the uninfected cells the level of IFN-β secretion was less than 200 pg/ml (data not shown). To investigate whether IFN-β secreted from H5N1 virus infected cells can induce GRIM-19 expression, neutralizing IFN-β Ab (600 IU/ml) was added to the cells 1 h prior to virus infection. As shown in Figure 4A, in the presence of neutralizing IFN-β Ab, the level of GRIM-19 mRNA in H5N1 virus-infected cells was noticeably reduced throughout the time course when compared with that of the virus-infected cells in the absence of this Ab. We further investigated whether IFN-β plays a role in apoptosis induction. hMDMs were pretreated with neutralizing IFN-β Ab for 1 h prior to H5N1 infection and the number of apoptotic cells was subsequently analyzed by TUNEL staining at 21 h post-infection (h.p.i.). In the presence of neutralizing IFN-β Ab, the number of H5N1-induced apoptotic cells was significantly reduced to 27% compared with 40% in the untreated control cells (Figure 4B). Neutralizing IFN-β Ab itself had no effect on the viability of uninfected cells. It should be noted that in the presence of IgG isotype control, used as a negative isotype control in this study, the percentage of apoptotic cells were similar to that observed in the untreated cells. Altogether, these results suggest that IFN-β secreted from H5N1-infected hMDMs induces apoptosis via GRIM-19.
GRIM-19 expression is inhibited by neutralizing IFN-β Ab. (A) hMDMs were pretreated with neutralizing IFN-β Ab (600 IU/ml) 1 h prior to H5N1 infection at MOI of 1. The expression of genes was determined by RT-PCR at 4, 8, 12 and 16 h.p.i. GAPDH expression was used as an internal control. (B) The number of apoptotic cells was determined by TUNEL staining after 21 h.p.i. Data were means and SEM of independent experiments from three separate donors. *P < 0.05.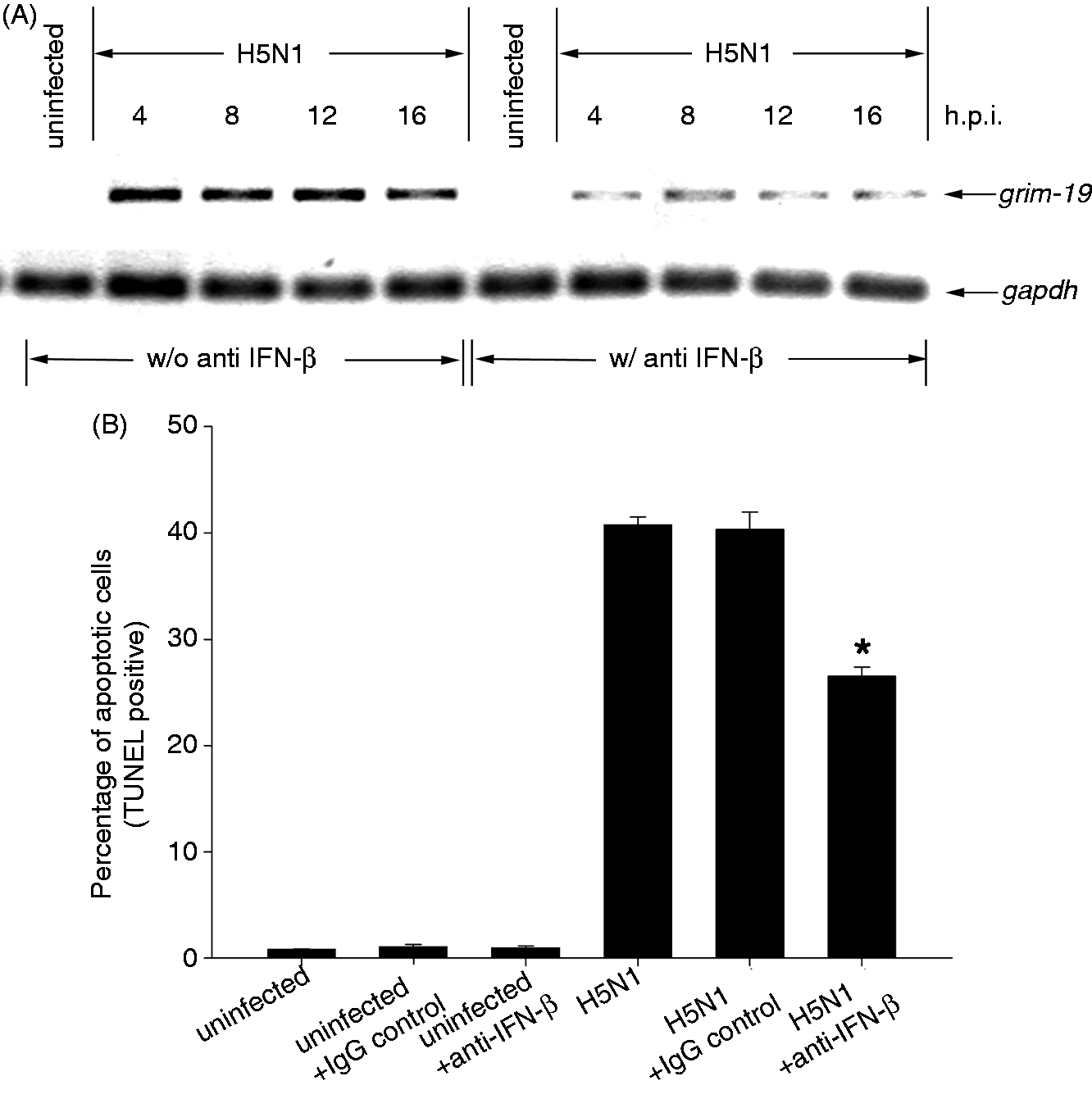
Discussion
Using a genetic technique, GRIM-19 has been identified as a novel apoptotic gene in the IFN-β/retinoic acid-induced apoptotic pathway. 19 In some viral infections, i.e. HIV, the up-regulation of GRIM-19 could be observed when T-cells were exposed to the virus, leading to cell death of HIV-infected cells. 21 In contrast, some viruses avoid apoptosis by producing viral protein that can interact and inhibit GRIM-19 activity. For example, viral interferon regulatory factor-1 protein produced by human herpes virus-8 interacts with GRIM-19 and blocks the ability of GRIM-19 to induce apoptosis. 25 These data indicate that GRIM-19 plays an important role in apoptosis induction during viral infections. In the present study, we demonstrated that H5N1 virus is able to induce GRIM-19 expression in hMDMs in a time-dependent manner (Figure 1). These results correlate with an increase in the percentage of apoptotic cell death over the course of infection. 5 However, the number of apoptotic cells induced by H5N1 was almost absent in GRIM-19-depleted cells (Figure 2C), thus suggesting that GRIM-19 plays an essential role in apoptosis induced in H5N1 virus-infected hMDMs.
Activation of apoptosis usually associates with several apoptotic effector molecules. Previously, we demonstrated that H5N1 was able to activate the mitochondrial apoptotic pathway resulting in cell death. 5 In this study, we extended our findings to demonstrate that activation of apoptosis effectors such as BID and the release of AIF from mitochondria were also diminished in infected GRIM-19-depleted cells (Figure 3A). Furthermore, the activation of caspase-10 and caspase-7 observed in H5N1-infected hMDMs 5 was also inhibited in the H5N1-infected GRIM-19 depleted cells (data not shown). These results suggested that GRIM-19 participates in the apoptosis of H5N1-infected hMDMs at a very upstream level. However, the exact mechanism of GRIM-19-induced apoptosis remains to be investigated.
Up-regulation of GRIM-19 has been demonstrated to be correlated with the down-regulation of BCL-XL, which is one mechanism that viruses use to induce host cell apoptosis. 21 For example, in HIV-infected patients, both BCL-2 and BCL-XL were found to be markedly decreased in HIV-specific CD8+ T cells when compared with those in the healthy donors, thus resulting in an increased sensitivity to FASL-induced apoptosis. 26 Severe acute respiratory syndrome coronavirus infection could induce apoptosis by encoding 7 a protein to interact with BCL-XL and interfered with BCL-XL function. 27 In the present study, we also demonstrated that H5N1 virus down-regulated the expression of BCL-XL in infected cells. Moreover, inhibition of BCL-XL down-regulation was observed in H5N1 virus-infected GRIM-19-depleted cells, suggesting that GRIM-19 plays a role in BCL-XL regulation (Figure 3B). Although, the regulation of BCL-XL during apoptosis has not been elucidated fully, it has been demonstrated that the expression of BCL-XL could be regulated by the STAT3 pathway. Silencing STAT3 in cancer cells results in down-regulation of BCL-XL leading to an increase in apoptosis.28,29 Furthermore, interference of STAT3 by GRIM-19 could down-regulate the expression of BCL-XL.20,30 Therefore, the down-regulation of BCL-XL in H5N1 virus-infected hMDMs might be due to the ability of virus to induce GRIM-19 expression to suppress STAT3 activity.
Dysregulation of cytokines induced by H5N1 virus is known to be also associated with the pathogenesis of the disease. Hyper-induction of cytokines, such as TNF-α, IP-10, IFN-α and IFN-β, could be detected in infected cells.7,8,10,11 As H5N1 virus was able to induce the expression of IFN-β, which is known to play a role as a first line defense against viral infection, cell destruction still occurred; therefore, IFN-β induced by H5N1 virus may play a role in apoptosis. In the present study, we demonstrated that the expression of GRIM-19 in H5N1 virus-infected cells was suppressed when the neutralizing IFN-β Ab was added (Figure 4A). These results indicated that the expression of GRIM-19 was associated with IFN-β secretion during H5N1 infection. Moreover, the number of apoptotic cells in H5N1 virus-infected hMDMs was decreased when neutralizing IFN-β Ab was added (Figure 4B), implying that IFN-β might exert another biological function in apoptosis. These results also correlated with the suppression of viral replication in the presence of neutralizing IFN-β Ab (data not shown). Consistent with our findings, inhibition of apoptosis induced by EMCV infection was observed in cells that were pretreated with anti-IFNα/β Abs. 15 EMCV-infected IFNAR-/- deficient cells were resistant to IFN-induced apoptosis indicating the involvement of type I IFNs in apoptosis induction. In addition, apoptosis of human cervical fibroblasts infected with either human influenza A or B viruses were inhibited with anti-IFN-β Ab. 31 These findings demonstrated that the induction of IFN-β during viral infections might contribute, at least in part, to virus-induced apoptosis. Taken together, our study indicates that GRIM-19 expression induced by H5N1 virus plays a role in apoptotic cell death. The data obtained from this study provide insights for the regulatory mechanism induced by H5N1 virus and an additional role of IFN-β leading to the pathogenesis of the disease. Further investigations on the relationship between apoptosis and type I IFNs should be considered.
Conclusions
This study elucidated a novel molecular mechanism of H5N1 virus-induced apoptotic cell death in hMDMs. GRIM-19 is involved in apoptosis induced by H5N1 virus and GRIM-19 might act at the upstream of the apoptosis pathway before caspase-10 activation.
Footnotes
Funding
This work was supported by National Institute of Health and National Institute of Allergy and Infectious Disease (YI-AI-5026-01).
