Abstract
Muramyl peptides (MPs) represent the building blocks of bacterial peptidoglycan, a critical component of bacterial cell walls. MPs are well characterized for their immunomodulatory properties, and numerous studies have delineated the role of MPs or synthetic MP analogs in host defense, adjuvanticity and inflammation. More recently, Nod1 and Nod2 have been identified as the host sensors for specific MPs, and, in particular, Nod2 was shown to detect muramyl dipeptide (MDP), a MP found in both Gram-positive and Gram-negative bacterial cell walls. Because mutations in Nod2 are associated with the etiology of Crohn’s disease, there is a need to identify synthetic MP analogs that could potentiate Nod2-dependent immunity. Here, we analyzed the Nod2-activating property of 36 MP analogs that had been tested previously for their adjuvanticity and anti-infectious activity. Using a luciferase-based screen, we demonstrate that addition of a methyl group to the second amino acid of MDP generates a MDP derivative with enhanced Nod2-activating capacity. We further validated these results in murine macrophages, human dendritic cells and in vivo. These results offer a basis for the rational development of synthetic MPs that could be used in the treatment of inflammatory disorders that have been associated with Nod2 dysfunction, such as Crohn’s disease.
Introduction
Peptidoglycan (PG) is a critical component of the cell wall of Gram-positive and Gram-negative bacteria. The PG heterogeneous polymer is composed of a sugar backbone comprised of alternating N-acetylglucosamine and N-acetylmuramic acid (MurNAc) residues. A peptide chain is attached to the MurNAc sugar, forming a muramyl peptide (MP), and these stem oligopeptides can be crosslinked to form the three-dimensional lattice structure of PG. The capacity of polymeric PG, as well as MP residues [and, in particular, muramyl dipeptide (MDP)], to induce both inflammatory and adaptive immune responses has been studied for decades. 1 Indeed, a wealth of scientific reports from the past four decades examine how chemical modifications of MDP would affect the ability of this compound to enhance host clearance of bacterial pathogens or function as an adjuvant for antigen-specific immune responses. 2
Following these studies on the immune properties of MPs, Nod1 and Nod2 were more recently identified as the critical cytosolic pattern recognition receptors (PRRs) that sense specific MPs in mammals, resulting in the induction of cellular defense pathways dependent on NF-κB, mitogen-associated protein kinase and autophagy.
3
Nod1 detects meso-diaminopimelic acid-containing MurNAc-tripeptide (Mur-TriDAP) found predominantly in Gram-negative bacteria,4,5 whereas Nod2 detects MDP found in both Gram-negative and Gram-positive bacteria.6,7 More detailed studies on the minimal structural requirements of the MP ligands needed for Nod1 or Nod2 activation revealed that the MurNAc sugar is not required for Nod1 activation as the
Recently, Nod2 was shown to bind directly to its MDP, 10 suggesting that this protein acts as a bona fide cytosolic receptor. It was also reported previously that an intact cellular endocytic pathway was critically required for the activation of both Nod1 and Nod2, indicating that cytosolic internalization of ligands was necessary for activation.11,12 The discovery of di- and tripeptide transporters located at the plasma membrane that are needed to internalize Mur-TriDAP and MDP further reinforces the idea that binding of MP ligands to Nod1 and Nod2 is essential for the induction of NF-κB signaling by these proteins. Specifically, it was shown that the oligopeptide transporter hPepT1 (also known as SLC15A1) acts as a specific transporter for MDP, 13 but not Nod1 ligands, 14 whereas the peptide transporter SLC15A4, which is expressed in early endosomes, is required for Nod1 stimulation by MurNAc-TriDAP and iE-DAP. 11 Finally, hPepT2 (also known as SLC15A2) was also found to be involved in MP internalization.15,16
Early studies have reported that the stimulatory capacity of MPs can be enhanced considerably by the addition of acyl chains of various lengths to the core of what are now known as the Nod1- and Nod2-activating ligands (i.e. iE-DAP or MDP respectively),17,18 and this property likely correlates with the enhanced capacity of acylated MPs to enter host cells by endocytosis. In addition, earlier studies (mainly from the 1970s and 1980s) provided detailed characterization of the structure/function relation for MPs and, in particular, MPs derived from MDP. However, since the identification of Nod2 as the cellular MDP sensor little effort was made to revisit these earlier studies and test the compounds from the synthetic MP libraries directly for their Nod2 stimulatory capacity. Here, we initially screened the Nod2-dependent NF-κB activation capacity of a library of 36 chemical derivatives of MDP and identified MurNAc-
Materials and methods
Reagents
List of tested muramyl dipeptide derivatives used in the current study. Structure information and working names are provided. Adjuvant refers to reported adjuvant capacity of the tested compounds, 31 whereas anti-infectious refers to previously reported ability to enhance host immunity against the pathogen Klebsiella pneumoniae. 32
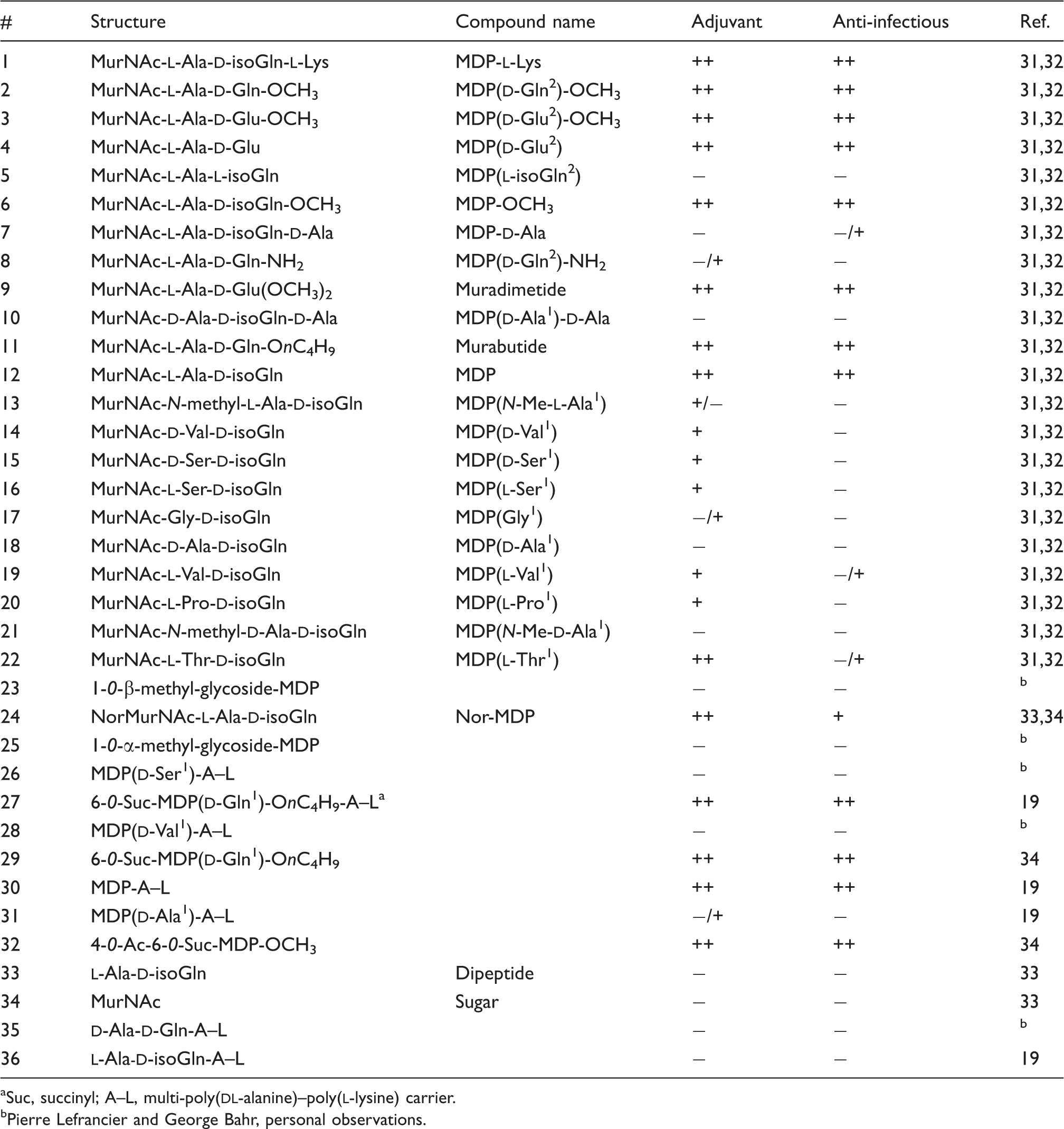
Suc, succinyl; A–L, multi-poly(
Pierre Lefrancier and George Bahr, personal observations.
Luciferase assays
NF-κB activation luciferase assays in cells over-expressing Nod2 were performed as described previously. 6 Briefly, HEK293T cells were transfected overnight with 30 ng of Nod2 and 75 ng of Igκ luciferase reporter plasmid. The cells were treated simultaneously with 1, 10, 100 or 1000 ng of the indicated muramyl peptide analog and the NF-κB-dependent luciferase activation was measured 24 h later on a luminometer.
Mice
C57BL/6 (Charles River) and Nod2−/− mice were bred and housed under specific pathogen-free conditions at the Center for Cellular and Biomolecular Research, University of Toronto. The University of Toronto Animal Ethics Review Committee approved all animal experiments. Sex-matched mice that were 6–10 wk old were used for experiments.
In vivo cytokine response
C57BL/6 (Charles River) and Nod2−/− mice were injected i.p. with the indicated dose of the MDP-derivatives, and serum was collected 2 h later. The concentration of the chemokine KC and the cytokine IL-6 in the serum was measured by ELISA (R&D Systems, Minneapolis, MN, USA) according to the manufacturer’s protocol.
OVA-specific Ab response
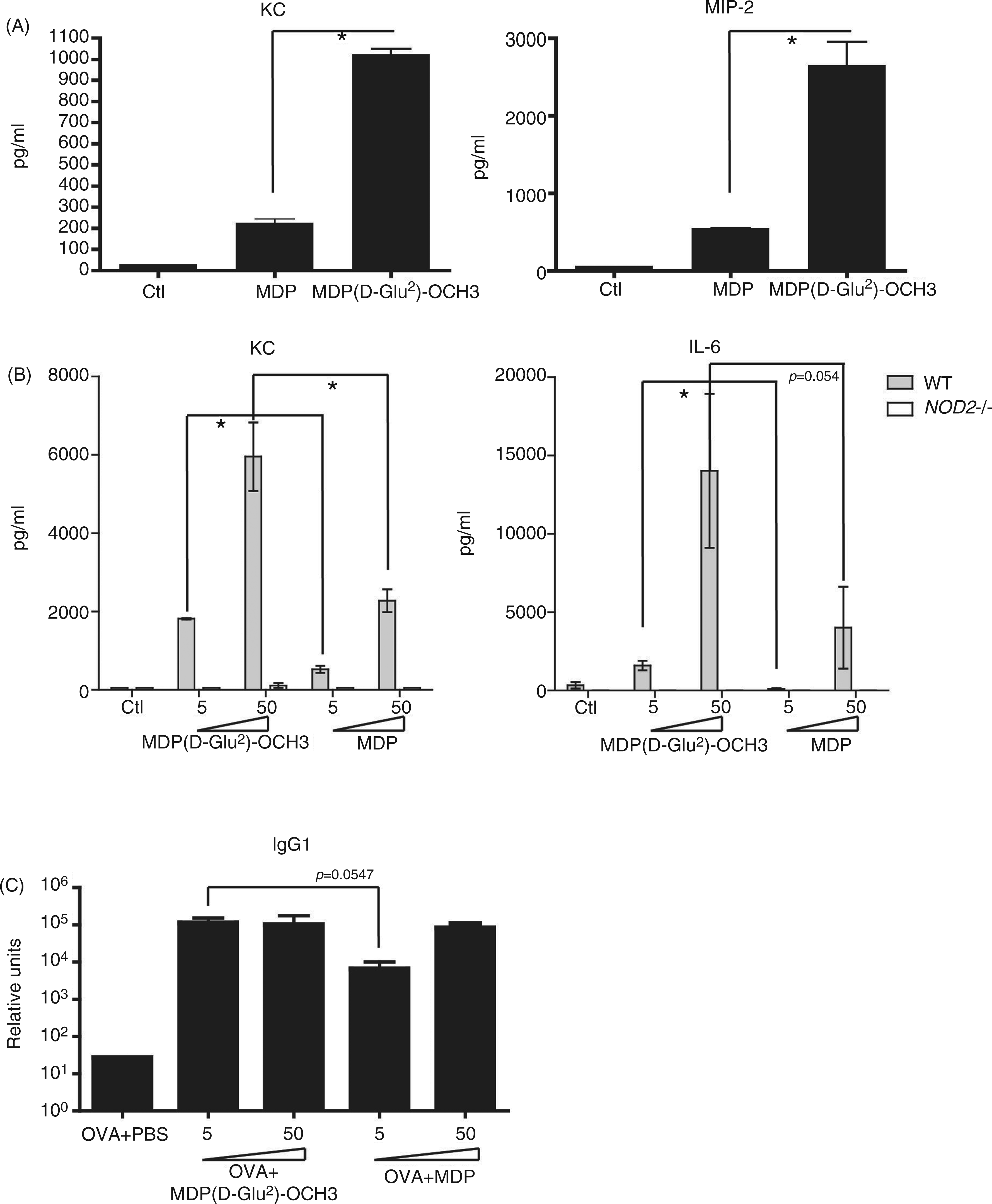
WT and Nod2−/− mice were immunized i.p. with a mixture of OVA (50 µg/mouse) and MDP, MDP(
Bone marrow-derived macrophage preparation
Bone marrow-derived macrophages (BMDM) were isolated from wild type (WT) and Nod2−/− mice, and cultured as described previously.
25
Briefly, total bone marrow cells were seeded at 5 × 106 cells in 10-cm dishes in 10 ml of DMEM [supplemented with 2 mM
Isolation of human dendritic cells
Monocytes were purified from human PBMCs of healthy donors using anti-CD14-conjugated magnetic microbeads (Miltenyi Biotec, Bergisch Gladbach, Germany). Immature dendritic cells (DC) were generated by culturing monocytes in RPMI 1640 Glutamax medium [50 µM 2-mercaptoethanol, 10 mM HEPES, 100 U/ml penicillin, 100 µg/ml streptomycin (Invitrogen), and 10% FCS (Biowest, Nuaille, France) with 100 ng/ml GM-CSF (Miltenyi Biotec) and 40 ng/ml IL-4 (Miltenyi Biotec)] for 6 d. Immature DCs obtained were 100% CD1a+/CD14−.
Statistical analyses
Student’s t-tests or Mann–Whitney tests were performed using Prism (GraphPad Software, La Jolla, CA, USA), and P-values < 0.05 using a two-tailed 95% confidence interval were considered significant.
Results
In order to characterize the Nod2-dependent responses to synthetic MDP analogs, we used a library of 36 MDP derivative compounds (which includes MDP, compound 12) which had been tested previously for their adjuvanticity and anti-infectious activity, as summarized in Table 1. Interestingly, many of the tested compounds that had a modification at the second amino acid of MDP, such as MurNAc-
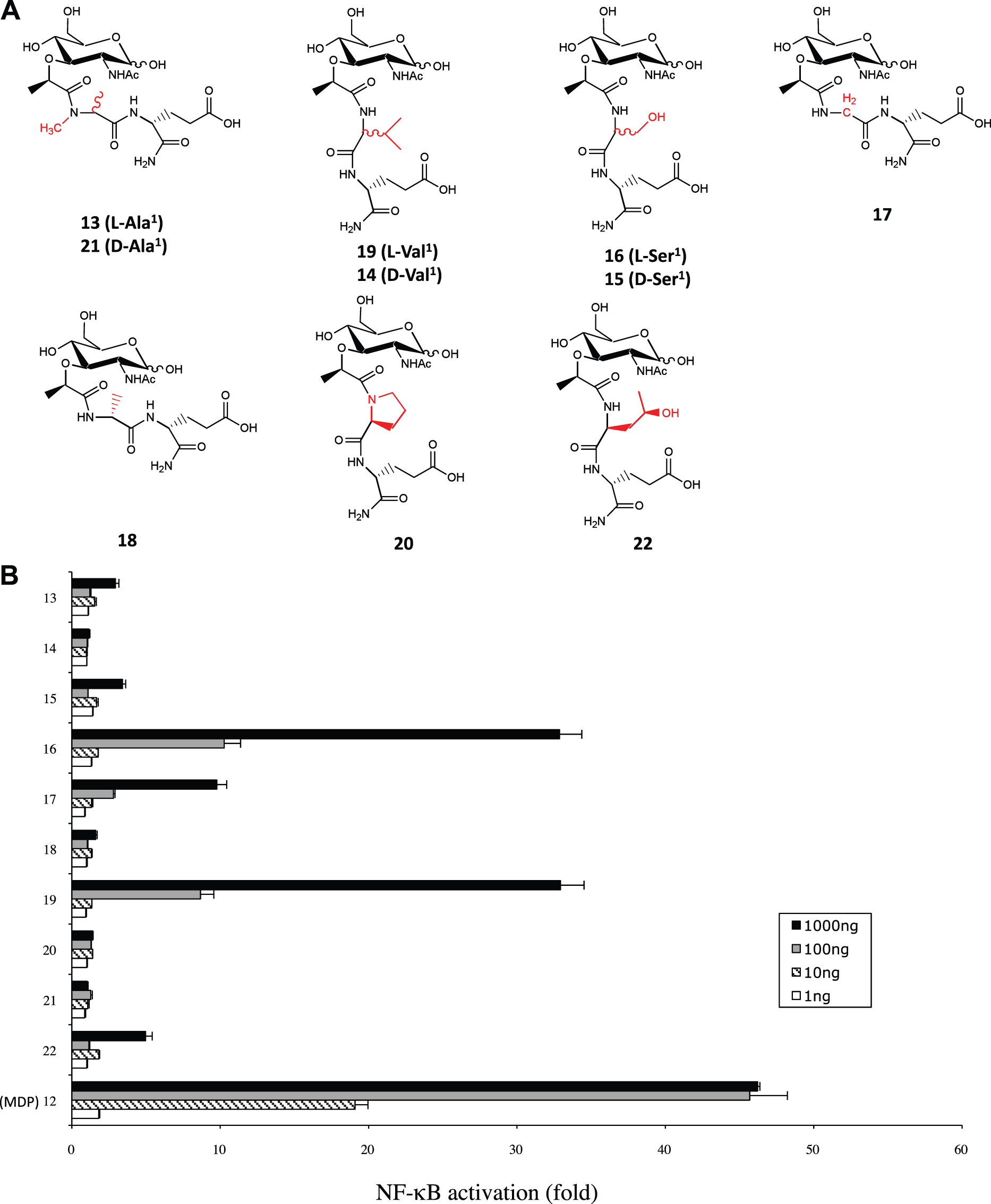
We now know that Nod2 is the PRR mediating host sensing of MDP; thus, we were interested in assessing the capacity of the 36 MDP-derivatives to activate Nod2 at the cellular level. To do this we used the now well-established Nod2-Igκ luciferase reporter assay in HEK293T cells, and these cells were stimulated with our MDP-derivative library. Overall, our screen revealed, as expected, an excellent correlation between Nod2-stimulating capacities of these MDP derivatives and their previously assigned anti-infectious and adjuvant properties (Figures 1 and 2; Supplementary Figures 1–3; Table 1). Interestingly, we determined that the same second amino acid-modified MDP-derivatives that were shown to be good antibacterial compounds and adjuvants (Table 1) were also strong inducers of Nod2-dependent NF-κB activation in vitro (Figure 1). Interestingly, MDP( NF-κB stimulatory capacity of MDP-derivative compounds modified at the second amino acid. (A Structures of compounds 1–11 (see Table 1 for working names), which represent MDP derivatives modified at the second amino acid. Modifications with respect to MDP structure (compound 12) are written in red. (B) HEK293T cells that were transfected overnight with Nod2 plasmid and Igκ luciferase reporter plasmid were stimulated with 1, 10, 100 or 1000 ng of compounds 1–12, with 12 being MDP and acting as a positive control. The bar graphs represent the fold NF-kB activation over unstimulated cells. Values depict the average of three replicates from two independent experiments. Error bars depict SEM. NF-κB stimulatory capacity of MDP-derivative compounds modified at the first amino acid. (A) Structures of compounds 13–22 (see Table 1 for working names), which represent MDP derivatives modified at the first amino acid. Modifications with respect to MDP structure are written in red. (B) HEK293T cells that were transfected overnight with Nod2 plasmid and Igκ luciferase reporter plasmid were stimulated with 1, 10, 100 or 1000 ng of compounds 12–22, with 12 being MDP and acting as a positive control. Values depict the average of three replicates from two independent experiments. The bar graphs represent the fold NF-kB activation over unstimulated cells. Error bars depict SEM.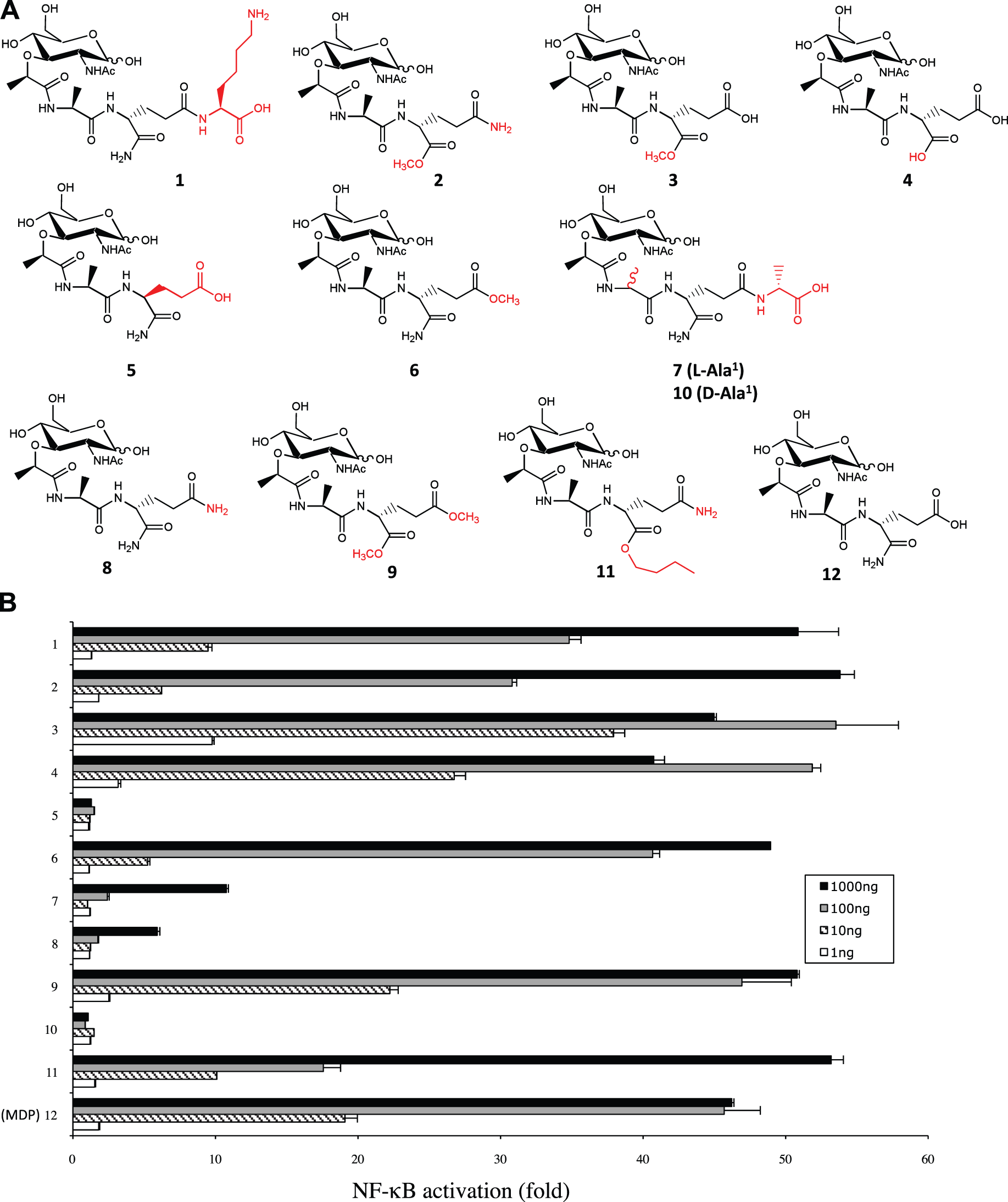
Given the previously reported capacity of MDP( In vitro and in vivo responses observed with MDP-derivative (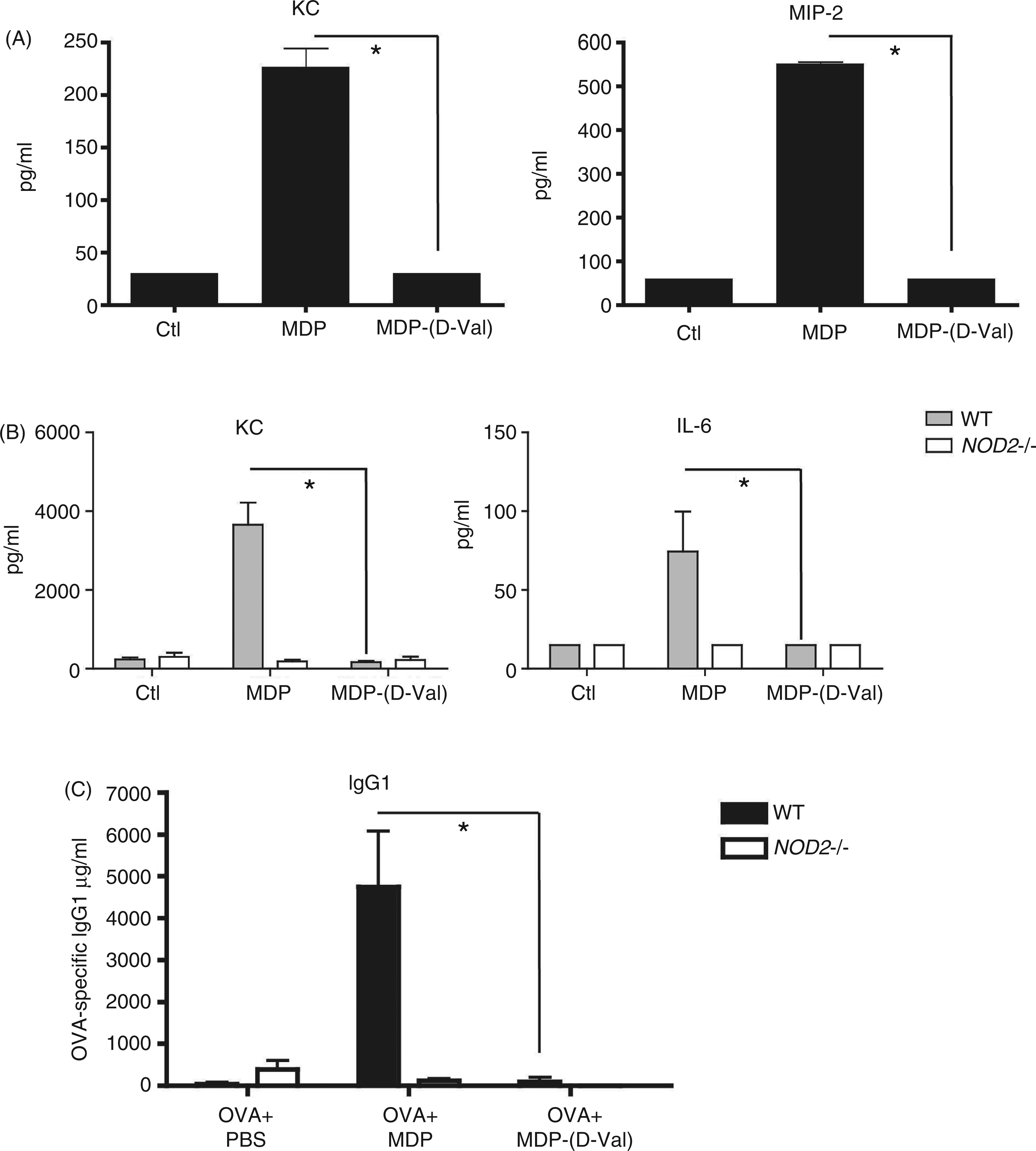
As we observed that MDP( MDP-derivative ( In vitro and in vivo responses observed with MDP-derivative (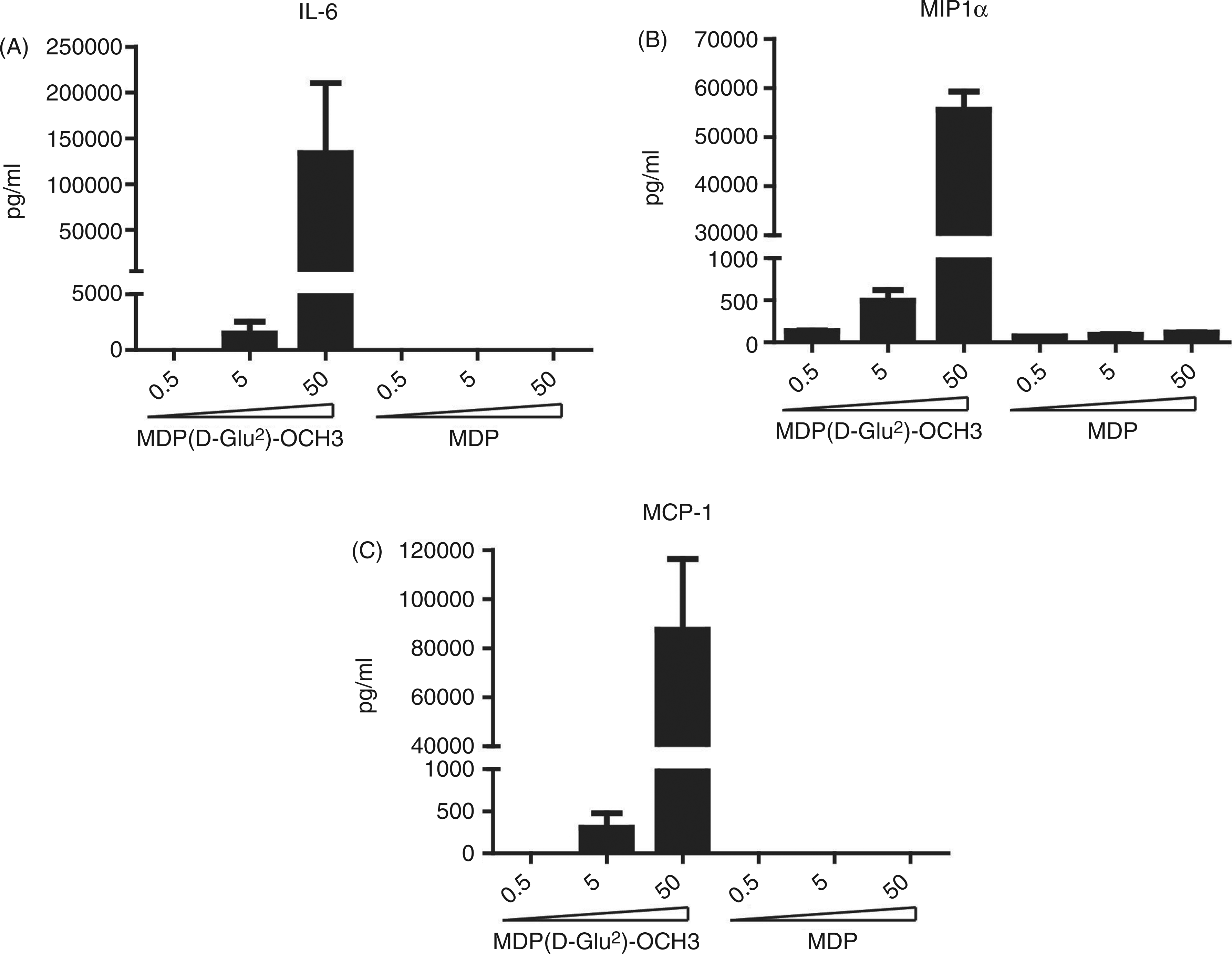
Discussion
In this study, we found that many MDP-derivative compounds modified at the first or second amino acid can stimulate Nod2 equally well as MDP, while one in particular, MDP(
We demonstrated that none of the MDP-derivatives that had the first amino acid of the
Previously, it was reported that the conversion of the N-acetyl group on the MurNAc sugar to a N-glycolyl group, which occurs naturally in mycobacterial species by the enzyme N-acetylmuramic acid hydroxylase, results in a MP with a greater Nod2-stimulating activity.
26
In the present study, we have identified what appears to be the first chemical modification to the core amino acids of MDP that results in an analog with increased stimulatory capacity on Nod2. With this proof-of-principle observation, we can now speculate that the relatively modest increase in stimulatory capacity identified here (approximately fivefold) provided by the replacement of the terminal amide of MDP by a methyl ester could be optimized rationally by targeting the α-carboxyl group of
Many of the mutations in Nod2 that have been associated with increased risk for developing Crohn’s disease (CD) result in Nod2 proteins that are either completely unresponsive to MDP, such as the 3020insC frameshift mutant,27–29 or have reduced signaling capacity, such as the newly identified S431L and N852S variants.
30
Therefore, it would be interesting to determine if administration of MDP analogs that exhibit increased Nod2 stimulatory activity, such as MDP(
Footnotes
Funding
S.E.G. and D.J.P. are funded by grants from the Canadian Institute of Health Research (CIHR) and the Crohn's and Colitis Foundation of Canada. S.J.R. is supported by Banting and Best Graduate Scholarship from CIHR. J.G.M. is supported by a fellowship from the French Foundation for Medical Research.
Acknowledgements
We would like to thank Armelle Bohineust for her help with the human DC experiments.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
