Abstract
Periodontitis, a ubiquitous chronic inflammatory disease, is associated with reduced antioxidant defences and neutrophil hyperactivity in terms of reactive oxygen species (ROS) generation. Its phenotype is thus characterized by oxidative stress. We have determined the effect of antioxidant micronutrients ascorbate and α-tocopherol on neutrophil ROS generation. Peripheral neutrophils from periodontally-healthy individuals (n = 20) were challenged with phorbol myristate acetate, IgG-opsonised Staphylococcus aureus, Fusobacterium nucleatum or PBS in the presence and absence of micronutrients (50 µM). Total and extracellular ROS were measured by luminol and isoluminol chemiluminescence respectively. Total and extracellular unstimulated, baseline ROS generation was unaffected by α-tocopherol, but inhibited by ascorbate and a combination of both micronutrients. Fcγ-receptor (Fcγ-R)-stimulated total or extracellular ROS generation was not affected by the presence of individual micronutrients. However, the combination significantly reduced extracellular FcγR-stimulated ROS release. Neither micronutrient inhibited TLR-stimulated total ROS, but the combination caused inhibition. Ascorbate and the micronutrient combination, but not α-tocopherol, inhibited extracellular ROS release by TLR-stimulated cells. Such micronutrient effects in vivo could be beneficial in reducing collateral tissue damage in chronic inflammatory diseases, such as periodontitis, while retaining immune-mediated neutrophil function.
Introduction
Infectious inflammatory diseases are a major cause of morbidity and mortality, and share common mechanistic features of pathogenesis. The neutrophilic polymorphonuclear leukocytes (neutrophils) are vital players in both innate and acquired immune responses to invading microorganisms, but also contain a battery of potent cytotoxic species; inappropriate release of these can cause substantial collateral tissue damage.1,2 There is evidence in chronic inflammatory diseases, such as periodontitis, that neutrophils display baseline hyperactivity and hyper-reactivity to a second stimulus, 3 and such a neutrophil phenotype, when coupled with delayed tissue transit times, subverted efferocytosis or, indeed, tissue sequestration, 4 can lead to profound collateral tissue damage. As neutrophils are the predominant inflammatory immune cell within periodontal tissues, and specifically within periodontitis lesions, 5 periodontal inflammation is a model inflammatory–immune disease with which to explore such mechanisms, with a view to their therapeutic modulation.
Neutrophils recognize pathogenic organisms or their products via receptors that detect acquired and innate immune-complexed opsonins (e.g. Fcγ or complement C3b) and directly via pattern recognition receptors (e.g. TLRs) which bind to pathogen-associated molecular patterns. 6 Receptor engagement triggers a series of downstream events, including pathogen endocytosis into the phagolysosome, 7 activation of protein kinase-C (PKC) and MAPK cascades and the NADPH-oxidase enzyme complex, which generates the superoxide anion radical and other downstream reactive oxygen species (ROS). 8
Activated neutrophils release primary (azurophilic) granule constituent proteases, superoxide and myeloperoxidase into the phagolysosome, and also extracellularly following bacterial phagocytosis. 9 Generally, these products destroy infective bacteria and other phagocytosed material within the safe confines of the phagolysosome. Superoxide dismutates to hydrogen peroxide, which, in turn, forms a number of downstream reactive species, such as hypochlorous acid (via myeloperoxidase), the hydroxyl radical or peroxynitrite anion. 2 However, extracellular release of reactive enzymes and oxygen intermediates also leads to substantial collateral tissue damage, 10 which, in periodontitis, results in ground substance degradation, collagenolysis and pro-inflammatory cytokine production. 2
A variety of studies has shown higher amounts of neutrophil-derived products (elastase, lactoferrin, alkaline phosphatase) in active versus inactive periodontitis sites and in periodontitis patients relative to controls.11,12 Furthermore, significantly enhanced total ROS production, 13 superoxide formation 14 and degranulation of neutrophils, measured as elastase release, 15 have been demonstrated in peripheral blood neutrophils derived from patients with chronic generalized periodontitis following Fcγ-receptor (FcγR) stimulation. We have demonstrated recently an LPS-independent peripheral blood neutrophil hyper-reactivity (IgG-opsonised Staphylococcus aureus and Fusobacterium nucleatum) and a baseline unstimulated hyperactivity in periodontitis patients relative to healthy controls,3,13 which was not associated with elevated phox-gene expression. Moreover, whereas periodontal therapy reversed the Fcγ-R stimulated total ROS production in peripheral neutrophils from periodontitis patients, it had no effect upon unstimulated extracellular ROS generation.3,13 The neutrophil hyperactivity in periodontitis patients was characterized by a type-1 interferon gene expression signature and elevated plasma IFN-α concentrations, 16 and further work demonstrated a potential role for IL-8, granulocyte macrophage colony-stimulating factor (GM-CSF) and IFN-α in neutrophil hyperactivity, and for IFN-α also in neutrophil hyper-reactivity. 17
Consistent with the above, our previous studies have demonstrated that periodontitis is associated with lowered antioxidant defences18,19 and others have demonstrated increased levels of biomarkers of oxidative damage locally, within periodontal tissues and gingival crevicular fluid, and in the peripheral blood of periodontitis patients.20,21 Recently, peripheral blood leukocytes from periodontitis patients have also been shown to have reduced telomere lengths, with telomere length being significantly inversely correlated with age, oxidative stress indices and severity of chronic periodontitis. 22 Together these data suggest that boosting of local or systemic antioxidant systems may be beneficial in modulating neutrophil function and oxidative damage in periodontitis.
Ascorbate (vitamin C) and tocopherol (vitamin E) are major dietary low molecular mass antioxidant micronutrients reported to decrease oxidative damage to proteins, lipids and nucleotides in cell-free model systems. 23 The term micronutrient has replaced antioxidant in recent years in recognition that species traditionally classified as antioxidants, such as α-tocopherol, possess diverse antioxidant-independent molecular activities which may be health-beneficial. 24 By contrast, neutrophils pre-incubated with ascorbate show enhanced superoxide production, 25 as well as decreased IL-10 secretion, 26 indicating potential pro-inflammatory effects of ascorbate. By contrast, α-tocopherol, which alone appears to have no scavenging ability within cell free model systems, 27 can inhibit superoxide release through inhibition of PKC via the protein phosphatase 1 (PP1) or PP2A pathways, 28 but has no effect on IL-10 secretion. 26
Activation of discrete neutrophil receptors triggers ROS generation via different intracellular signaling pathways, and ascorbate and α-tocopherol may interfere with either ROS formation and/or scavenge ROS after their formation. Therefore, the aim of this study was to determine the effect of the antioxidant micronutrients ascorbate and α-tocopherol, both alone and in combination, on neutrophil ROS in the presence and absence of the Fcγ-receptor agonist, opsonized S. aureus and the TLR4 agonist F. nucleatum, both of potential importance in the pathogenesis of periodontitis.
Material and methods
Study populations
Periodontally healthy volunteers (n = 9–20, depending on stimulation, 8 males and 12 females; mean age = 33 ± 7 yrs, range = 21–45 yrs), defined by no visible signs of gingival inflammation, periodontal pocketing ≤4 mm and no interproximal attachment loss, were recruited from staff of the Dental Hospital. All volunteers were systemically healthy and exclusion criteria included a course of non-steroidal anti-inflammatory drugs or antimicrobial drugs within a three-month period prior to enrolment, pregnancy, and use of mouthwashes or vitamin supplements within the previous three-months. All were never smokers, had a negative history of current or previous recreational drug use, and had no special dietary requirements. Ethical approval for this work was granted by West Midlands Research Ethics Committee (LREC number 10/H1208/48). Informed consent to participate was obtained prior to participants completing a medical questionnaire.
Collection of blood and preparation of neutrophils
Venous blood was collected from volunteers from the ante-cubital fossa into Vacutainer™ lithium heparin (17 IU/ml) tubes and neutrophils isolated as described previously3,13 using a discontinuous Percoll gradient (δ = 1.079:1.098) followed by erythrocyte lysis (0.83% NH4Cl containing 1% KHCO3, 0.04% EDTA and 0.25% BSA). Isolated cells were re-suspended in PBS supplemented with glucose (1 mM) and cations (1 mM MgCl2, 1.5 mM CaCl2) at 1 × 106 cells/ml. Cell viability, typically >98%, was determined by dye exclusion (trypan blue).
Preparation of bacteria
Staphylococcus aureus (NCTC 6571) was prepared as described previously, 10 heat-treated (100°C for 10 min), opsonized 29 and stored as a 1.2 × 109 cell/ml suspension at −80°C. Fusobacterium nucleatum (ATCC 10953) was grown anaerobically at 37°C 30 and washed (three times) in H2O prior to storage at −80°C.
Enhanced chemiluminescence assay
All assays were performed as described previously.3,13,16 In brief, neutrophils (1 × 105) were placed in pre-blocked (PBS BSA 1%, overnight at 4°C) white microwells (Microlite2, Dynex, UK) with PBS supplemented with glucose and cations (35 µl) and luminol (3 mmol/l; 30 µl), for detection of total ROS production, or isoluminol (3 mmol/l; 60 µl) with 6 U HRP for the detection of extracellular ROS release. Neutrophils were incubated for 30 min to allow the cells to equilibrate (37°C, Berthold Technologies microplate-reader; LB96v: Bad Wilbad, Germany) in the microplate reader before addition of 10 µl ascorbic acid (1 mmol/l; Sigma, St Louis, MO, USA), α-tocopherol (1 mmol/l, Sigma) or a combination of the two (10 µl each). Cells were then stimulated immediately with either PMA (25 µl, 25 nM final conc.; Sigma), F. nucleatum (100 bacteria/neutrophil), opsonized S. aureus (300 bacteria/neutrophil) or PBS as control. Previous studies have demonstrated that un-opsonized S. aureus does not stimulate neutrophil ROS generation detectable by luminol or isoluminol chemiluminescence. 13 The final concentration of micronutrients in the test wells was 50 µmol/l, which was within (23–85 µmol/l ascorbate), or close to (12–42 µmol/l α-tocopherol), the reference ranges for these vitamins in plasma31–33 and within the concentration ranges used in published studies on their effects on neutrophil function. All analyses were performed in triplicate. Light emission in relative light units (RLU) was recorded 30 min pre-stimulation and 150 min post-stimulation, and the peak signal determined.
Statistics
Differences were determined by paired analysis using the Wilcoxon signed-rank test (two-tailed) using P ≤ 0.05 as the level of significance after Bonferroni correction 34 for multiple comparisons between control, PBS-treated cells and micronutrient-treated cells. All statistical analyses were conducted using Minitab statistical software.
Results
Summary of the effects of antioxidant micronutrients on neutrophil reactive oxygen species (ROS) responses to stimulation with PMA, IgG-opsonized S. aureus (FcγR agonist) and F. nucleatum (TLR agonist).
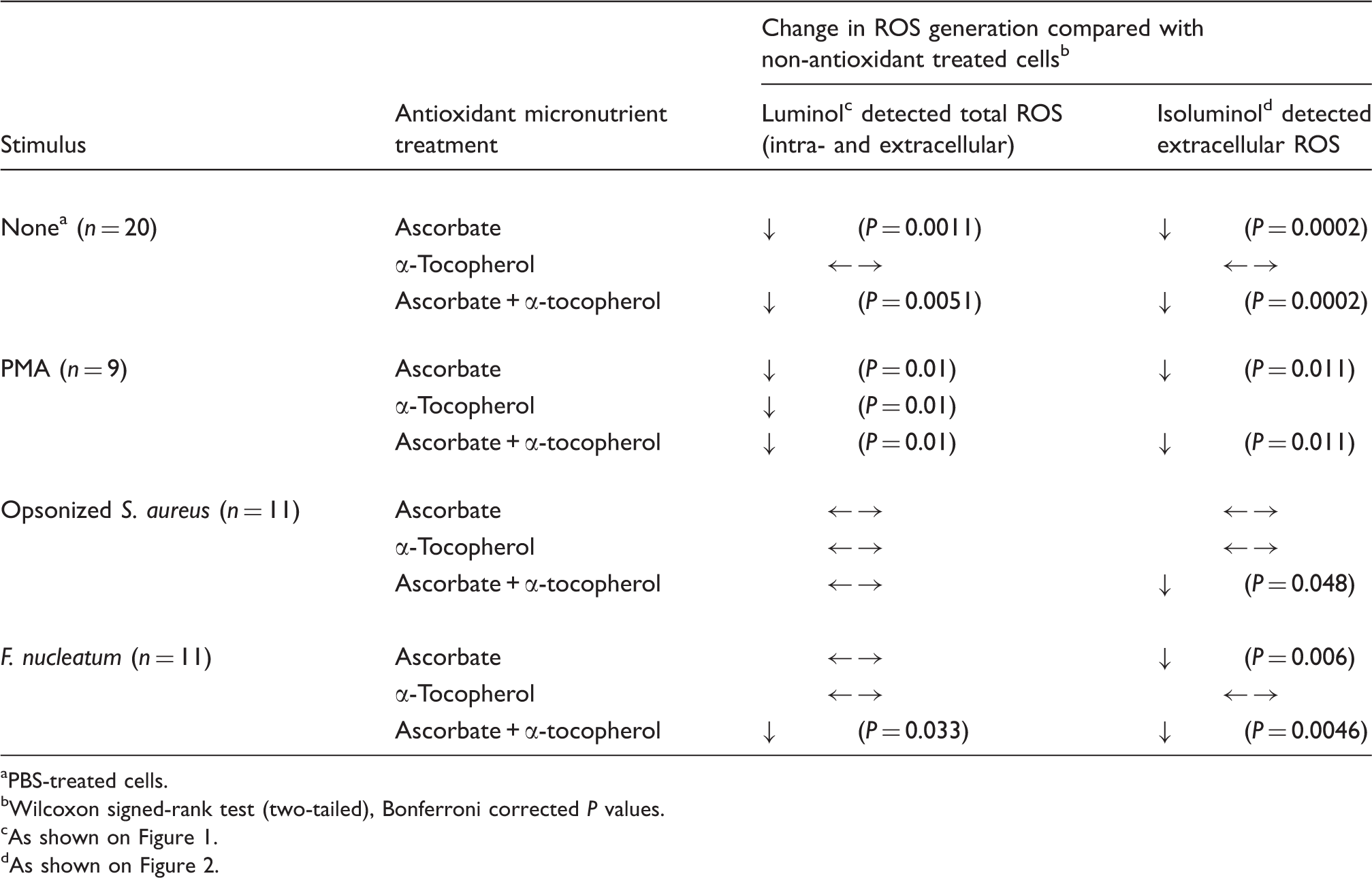
PBS-treated cells.
Wilcoxon signed-rank test (two-tailed), Bonferroni corrected P values.
As shown on Figure 1.
As shown on Figure 2.
Total ROS production detected by luminol chemiluminescence
Baseline, unstimulated ROS generation, as determined using vehicle-alone (PBS)-treated cells, was inhibited significantly by ascorbate (P = 0.0011) and the combination of ascorbate and α-tocopherol (P = 0.0051), but unaffected by α-tocopherol alone (Table 1, Figure 1A). By contrast, the antioxidant micronutrients, both individually and combined, inhibited Phorbol 12-myristate 13-acetate (PMA)-stimulated ROS generation (P = 0.01; Table 1, Figure 1B). Micronutrient effects on FcγR-mediated stimulation of ROS were, potentially, more complex. Ascorbate appeared to have a stimulatory effect, increasing mean (5.68 ± 0.69 × 104 RLU versus 4.81 ± 0.41 × 104 RLU) but not median ROS generation in response to opsonized S. aureus. This apparent increase was not statistically significant by paired analysis after Bonferroni correction (P = 0.053). α-Tocopherol alone and the combination of α-tocopherol and ascorbate had no detectable effect on FcγR-mediated stimulation of ROS. ROS generation stimulated by F. nucleatum was unaffected by the presence of α-tocopherol and non-significantly reduced by ascorbate (P = 0.063). By contrast, the combination of ascorbate and α-tocopherol significantly inhibited luminol-detected ROS generation in response to F. nucleatum (P = 0.033; Table 1, Figure 1D).
Luminol-detected total ROS production by neutrophils stimulated with (A) PBS (no stimulus; n = 20), (B) PMA (25 nM; n = 9), (C) opsonized S. aureus (n = 11) and (D) F. nucleatum (n = 11) in the presence of PBS (untreated control), ascorbate (50 µM), tocopherol (50 µM) and a mixture of ascorbate (Asc) and tocopherol (Toc) (50 µM each). Box and whisker plots show median, interquartile range, 5 and 95% confidence intervals; extreme values are represented as dots.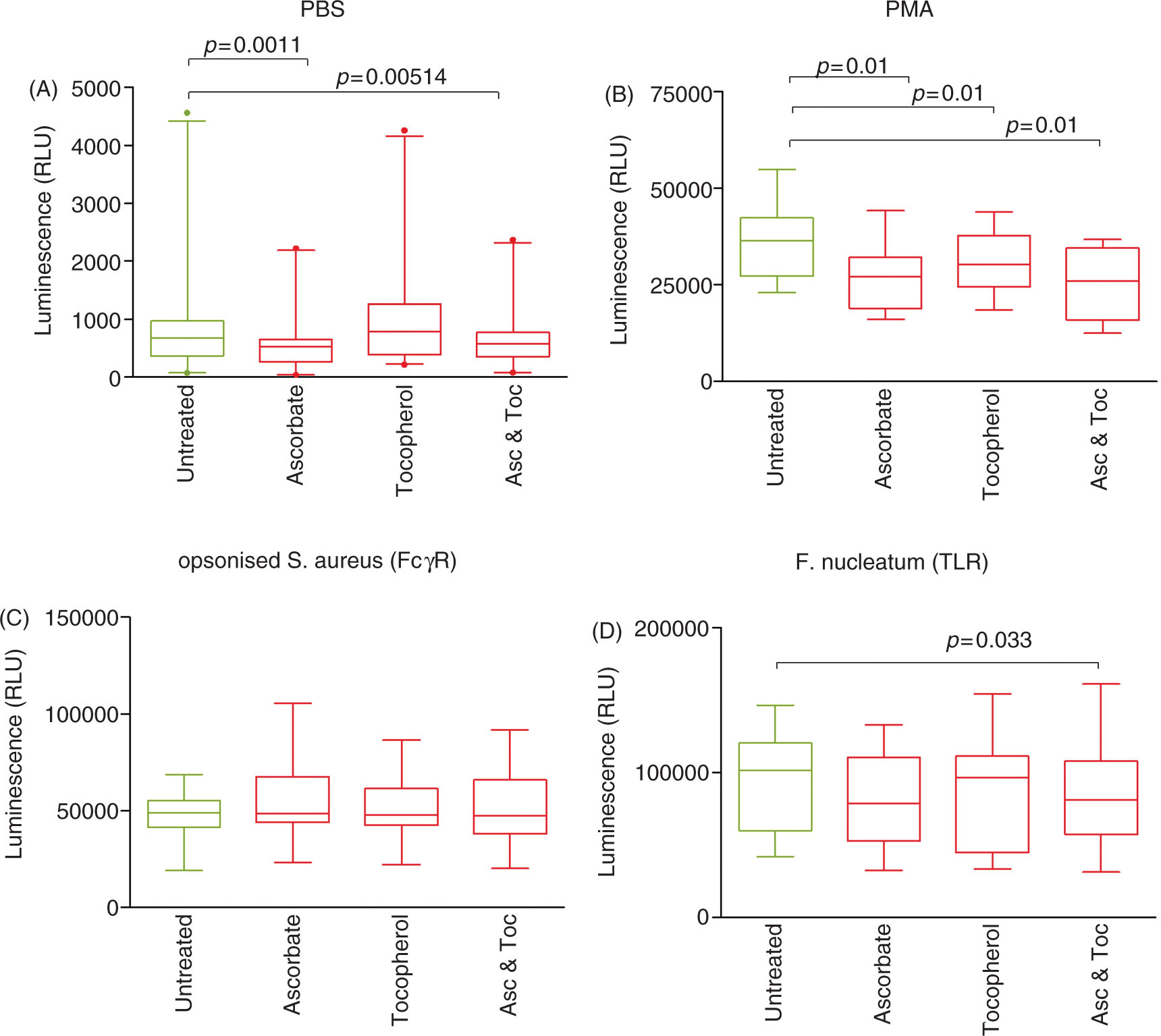
Extracellular ROS production detected by isoluminol chemiluminescence
The pattern of antioxidant micronutrient effects on extracellular ROS generation was similar to those detected by luminol chemiluminescence (Table 1, Figure 2). α-Tocopherol alone had no statistically significant effect on either unstimulated or stimulated extracellular ROS generation, although mean levels of ROS released from PMA-stimulated neutrophils were lower (4.91 × 104 versus 5.50 × 104 RLU; P = 0.053). Ascorbate alone and in combination with α-tocopherol inhibited unstimulated (P = 0.0002) extracellular ROS release. Similarly, F. nucleatum-stimulated ROS release was inhibited by ascorbate alone (P = 0.006) and in combination with alpha tocopherol (P = 0.0046). Although both mean and median levels of detectable extracellular ROS in response to opsonized S. aureus were lower in ascorbate-treated cells, this effect was not significant (P = 0.23; Table 1, Figure 2C). However, the combination of ascorbate and α-tocopherol, which did not affect luminol chemiluminescence, significantly inhibited isoluminol-detected ROS release after FcγR-stimulation (P = 0.048; Table 1, Figure 2C).
Isoluminol-detected extracellular ROS production by neutrophils stimulated with (A) PBS (no stimulus; n = 20), (B) PMA (25 nM; n = 9), (C) opsonized S. aureus (n = 11) and (D) F. nucleatum (n = 11) in the presence of PBS (untreated control), ascorbate (Asc; 50 µM), tocopherol (Toc; 50 µM), and a mixture of ascorbate and tocopherol (50 µM each). Box and whisker plots show median, interquartile range, 5 and 95% confidence intervals; extreme values are represented as dots.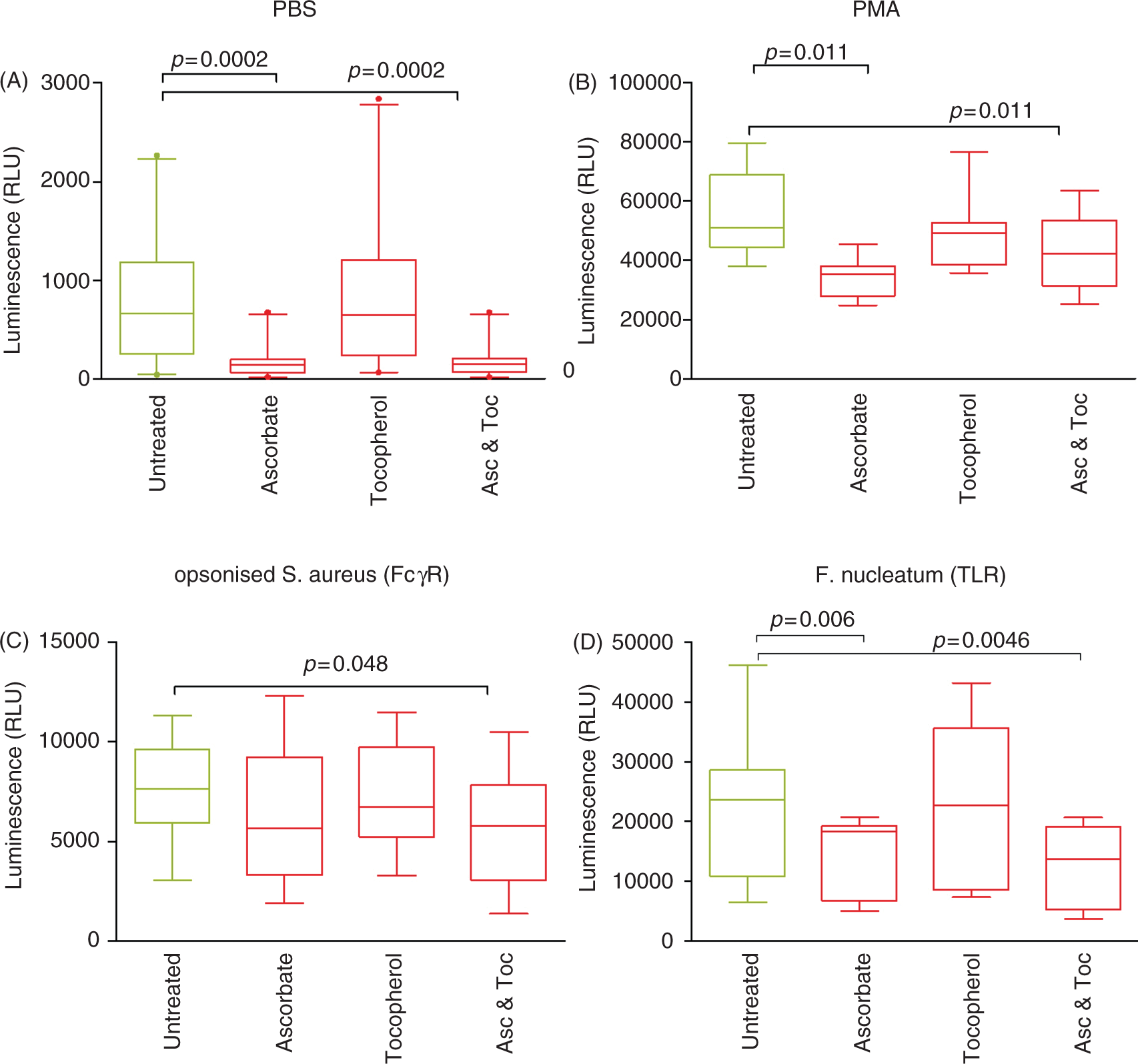
Discussion
Our data demonstrate that the antioxidant micronutrients ascorbate and α-tocopherol have the potential to lower both stimulated and baseline (PBS-stimulated) neutrophil ROS generation in our model system and thus have the potential to down-regulate cell function and collateral tissue damage induced by extracellular leakage of ROS. Importantly, the effects of these micronutrients on stimulated ROS generation were dependent upon the stimulus. Whereas ascorbate and α-tocopherol individually, and in combination, lowered levels of both total and extracellular ROS generated after PMA treatment, α-tocopherol alone did not alter detectable ROS generation in response to physiologically-relevant stimuli interacting via FcγR or TLRs. Significantly, the data suggest that a combination of these micronutrients is capable of reducing baseline and TLR-mediated total and extracellular ROS levels, while preserving intracellular ROS generation and reducing extracellular ROS in response to FcγR-stimulation. Ascorbate and α-tocopherol are known to recycle each other 35 and this may explain the increased antioxidant effect of the combination over ascorbate alone in this regard. In terms of periodontitis, this combined effect could be beneficial in reducing potentially damaging innate ROS generation within periodontal tissues that have been chronically stimulated by plaque components while retaining the ability to mount an unimpaired ROS response required for efficient intracellular killing/destruction of Ab-targeted bacteria, as well as reducing ROS release.
That ascorbate can exhibit pro-oxidant properties is well known 36 and more recent studies have demonstrated that it can generate hydrogen peroxide in extracellular fluid in vivo. 37 Furthermore, ascorbate has also been shown to have pro-oxidant effects on stimulated neutrophils.25,38 Thus, the presence of 0.1–1 mM ascorbate increased cumulative intracellular superoxide generation over 30 min, detected using dihydroethidium and flow cytometry, in neutrophils stimulated with arachidonic acid, fMLP, PMA or heat-killed, unopsonized Escherichia coli. These data contrast with our finding of significant antioxidant effects of ascorbate when neutrophils were stimulated with PMA and F. nucleatum. However, the published studies using dihydroethidium measured intracellular superoxide whereas we have used luminol-based detection systems. Luminol, which enters the cell, and isoluminol, which cannot pass through cell membranes, are oxidized exclusively by the superoxide-derived metabolites hydrogen peroxide and HOCl 39 formed by the activity of superoxide dismutase and neutrophil-specific myeloperoxidase.9,40 Thus, our data indicate that ascorbate has little effect on the generation of intracellular hydrogen peroxide and HOCl by neutrophils after stimulation with FcγR and TLR agonists, but significantly reduces their extracellular levels in response to TLR, but not FcγR, activation. OCl is a powerful, non-radical oxidant which plays an important role in intracellular bactericidal function 41 but, extracellularly, is highly reactive toward a range of biological substrates with the potential of causing significant pathology.9,40 We have also recently demonstrated that HOCl regulates the release of neutrophil extracellular traps (NETs), an antimicrobial strategy of emerging importance in innate immunity. 42
PMA acts directly upon the PKC signaling cascade as a diacyl glycerol analogue stimulating ROS production via mobilization of the p47 component of NADPH-oxidase, and our results with this stimulant are consistent with previously published data.27,43,44 Although PMA can provide information about the effects of a ROS scavenger upon ROS production it is not a physiologically relevant stimulus and therefore results obtained with its use do not always reflect what will occur in vivo. The biologically-relevant stimuli selected in our study (F. nucleatum and opsonized S. aureus) represent two arms of the immune response: F. nucleatum, which acts via TLR4 and 2, representing the non-specific (innate) immune response, and opsonized S. aureus, acting through FcγR, the specific immune response. Our data demonstrate that 50 µM α-tocopherol does not affect the intracellular or extracellular ROS responses of neutrophils to FcγR and TLR agonists. Similar findings of α-tocopherol-insensitive superoxide generation after fMLP and opsonized zymosan stimulation have been reported. 43 However, a more recent report suggests that α-tocopherol inhibits neutrophil superoxide responses to opsonized zymosan. 44
In this study we employed 50 uM ascorbate31,32,45 and α-tocopherol, 33 which were selected to represent physiologically-relevant concentrations. However, tocopherol can be increased 600-fold in lipid rich micro-environment. 46 Moreover, the reported micronutrient concentrations can be achieved easily through topical application, a route commonly utilized for adjunctive treatment of periodontal disease.
Neutrophils are important in the clearance of the initial/acute periodontal lesion; however, active resolution of neutrophilic inflammation is also vital in preventing collateral tissue damage, further activation of cytokine networks 47 and persistent chronic inflammation. Resolution of inflammation is a complex process involving a class switch in neutrophil phenotype, triggered by prostaglandin E2, and the subsequent generation of pro-resolving lipid mediators, such as Lipoxin A4.48,49 The latter antagonize neutrophil chemotaxis, stimulate monocytes towards a non-phlogistic inflammatory phenotype and activate neutrophil efferocytosis by macrophages, 50 thus actively resolving the inflammatory process and promoting healing. However, if the lesion is not resolved, either because of failed bacterial clearance or dysregulated inflammatory/resolution processes, the hyperactive or re-active state of circulating neutrophils3,13,16 may trigger exaggerated ROS and protease activity when bacteria or their virulence factors are encountered within the periodontal tissues. This may lead to collateral damage and periodontal tissue destruction. The lowering of innate ROS response pathways could be key to decreasing this collateral damage and subsequent release of damage-associated molecular patterns, 51 especially if specific opsonic neutrophil intracellular ROS activity was maintained. Our ex vivo data suggest the possibility that in vivo local delivery of ascorbate and α-tocopherol combined might fulfill such a modulatory role. Clinically, this may decrease periodontal tissue destruction while maintaining an innate immune response.
Conclusion
The combined effect of ascorbate and α-tocopherol inhibits innate (spontaneous and TLR-mediated) total and extracellular neutrophil ROS (hydrogen peroxide and HOCl) generation without affecting their intracellular generation in response to antibody labeled targets. Such a combination of antioxidant micronutrients may reduce collateral tissue damage caused by chronic non-specific, plaque-related neutrophil ROS release and oxidative stress. Further studies are required to unravel the molecular mechanisms involved.
Footnotes
Funding
Supported by Unilever Oral Care UK.
Conflicts of interest
This research has been financially supported by Unilever Oral Care UK.
A.E. Scott is employed by Unilever Oral Care UK.
