Abstract
The mammalian ribonucleaseA family comprises a large group of structurally similar proteins which are secreted by a range of tissues and immune cells. Their physiological role is unclear. It has been suggested that some of these RNases contribute to host defence, notably eosinophil-derived neurotoxin, eosinophil cationic protein, eosinophil-associated RNases, RNase4, angiogenin (RNase5), RNase7, RNase8 and bovine seminal RNase. This review summarises data supporting the involvement of these proteins in host defence, focusing on their antimicrobial, cytotoxic and immunomodulatory activities. The extent to which the data support possible mechanisms of action for these proteins is discussed. This compilation of findings and current hypotheses on the physiological role of these RNases will provide a stimulus for further research and development of ideas on the contribution of the RNases to host defence.
Introduction
The mammalian ribonuclease family consists of a large number of proteins found in a wide range of mammals. Its most well-characterised member is RNaseA, found in the bovine pancreas. Members of this family share a similar primary sequence, and a common disulfide-bonded tertiary conformation has been observed in all structurally-characterised examples. 1 For some time, the RNase family in humans was considered to comprise eight RNases: pancreatic RNase (also known as RNase1), eosinophil-derived neurotoxin (EDN/RNase2), eosinophil cationic protein (ECP/RNase3), RNase4, angiogenin (RNase5), RNase6 (k6), skin-derived RNase (RNase7) and RNase8.1,2 Analysis of the human genome sequence has revealed the existence of five additional RNases, which are named RNases 9–13. 3 All the mature proteins of this family are 14–16 kDa in size and contain 6–8 cysteine residues that are an important part of their tertiary structure. Some, but not all, contain a motif (CKXXNTF) that is associated with catalytic activity. 1 The structure and enzymatic activity of some of the family members have been characterised. RNaseA has been extensively studied, and its structure and enzymatic activity are very well characterised. 4 However, the biological functions of most of the members of this family are still poorly understood. Members of the family are found in a range of sources that could suggest a role in host defence, for example mucosal secretions such as milk,5,6 seminal fluid,7,8 saliva9,10 and tears. 11 In addition, members of this family are also expressed in several types of immune cells, including eosinophils,12,13 neutrophils, 14 monocytes, 15 and macrophages. 16 Major organs, such as brain, 17 liver 18 and kidney,19,20 also contain RNases, and many of these have a host defence function. A range of host defence-related activities have been reported for members of RNase family, including cytotoxic,21,22 helminthotoxic,23–25 antibacterial,26–29 antiviral30–33 and anti-Candida activity. 5
Analysis of RNase sequences between species has revealed that the mammalian RNase family is one of the most rapidly evolving families in mammals. 3 Interestingly, gene duplication appears to have occurred for several other immune-related gene families, for example the major histocompatibility complex, immunoglobulins and T cell receptors,34–37 suggesting a common evolutionary pressure on the host defence system.
Members of the RNase family proposed to have a host defence function.
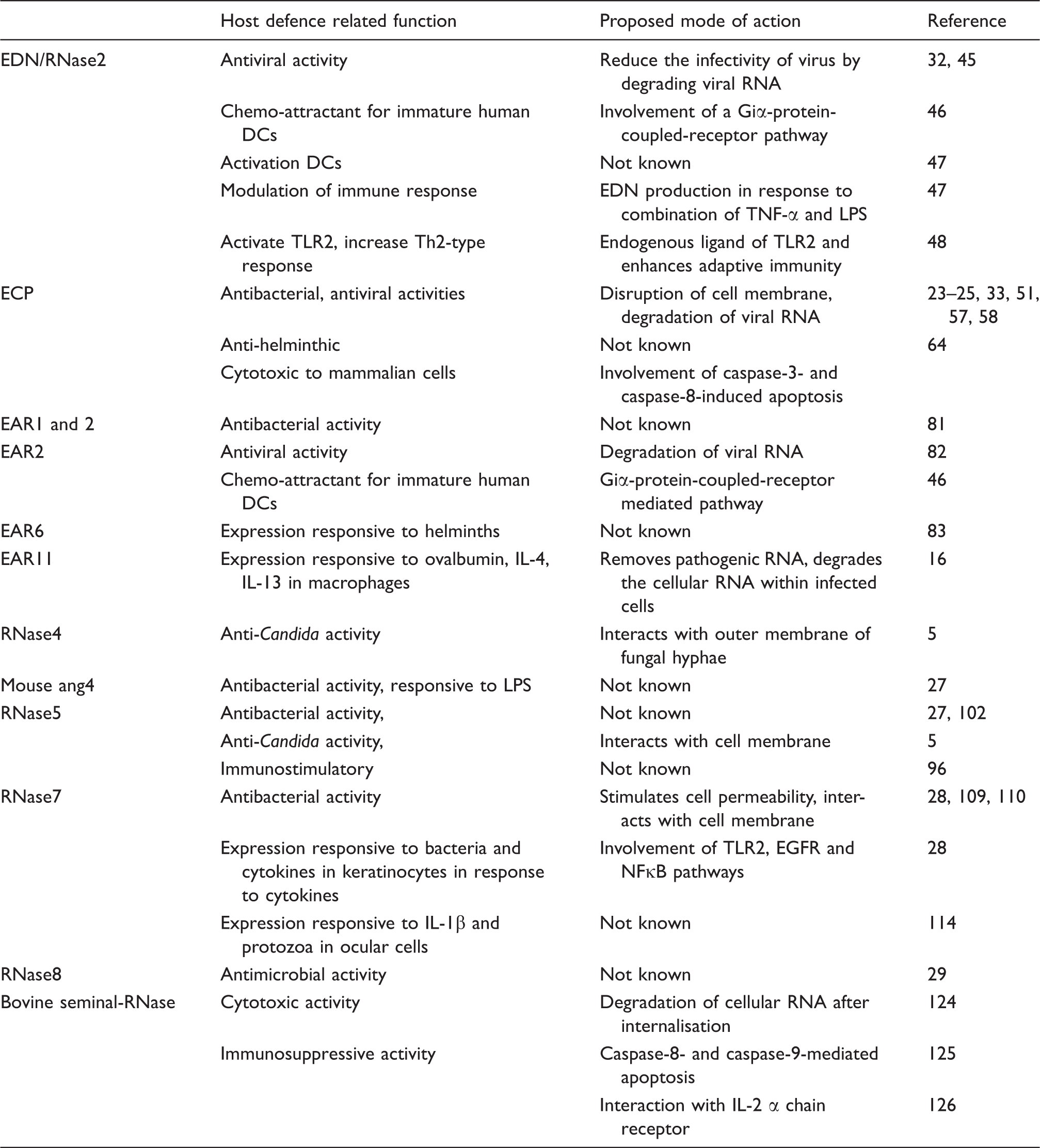
EDN/RNase2
Eosinophils are specialised cells of the circulating innate immune system. They migrate into various epithelial tissues, including the gastrointestinal, genitourinary and respiratory tracts in response to chemokine signals produced during some types of infection. Upon activation, eosinophils release a number of effector proteins which function to neutralise the pathogen. One of the four major secretory proteins of human eosinophil secondary granules is EDN (also known as RNase2). Its neurotoxic activity was reported as long ago as 1933, when it was found that interacerebral injection of a human lymph node suspension can cause neurotoxic syndrome in rabbits. 39 This neurotoxic effect was confirmed to be owing to a protein present in the infiltrating eosinophils and named eosinophil-derived neurotoxin. 14,40 EDN was isolated and shown to possess ribonuclease activity.41,42 However, it is not clear whether the ribonuclease activity of EDN is required for its neurotoxic activity or other biological functions. The physiological role of this neurotoxic activity remains unclear.
A number of host defence-associated activities have been proposed for EDN.43,44 These include reduction of the infectivity of respiratory syncytial virus (RSV) and HIV in cell culture.32,45 Other commonly observed activities of innate immune effector proteins, such as antimicrobial or anti-helminthic activity, have not yet been demonstrated for this protein. Interestingly, ablation of the catalytic activity of recombinant EDN through site directed mutagenesis suggests that its antiviral activity is dependent on its ribonuclease activity. 45 It has also been suggested that EDN can act as a chemoattractant. Yang and colleagues demonstrated that recombinant EDN stimulates migration of human immature dendritic cells (iDCs). 46 Pre-treatment of these cells with pertussis toxin blocked this activity, suggesting the involvement of a guanine nucleotide binding receptor pathway. 46 In addition to the chemoattractant properties of EDN, these authors also showed that both native, as well as the recombinant form of EDN can induce the maturation and the activation of cultured human DCs, as evidenced by the production of pro-inflammatory cytokines and chemokines and also up-regulated expression of the surface markers CD83, CD86 and HLA-DR. 47 These authors also demonstrated that human monocyte-derived macrophages challenged with a combination of LPS and TNF-α produced EDN in time-dependent manner. 47 Although the mechanisms by which EDN exerts its effects have not been identified it is conceivable that EDN activity may involve activation of multiple pathways. It has been shown that EDN is an endogenous ligand for the pathogen recognition receptor, TLR2, and that it can enhance an antigen-specific Th2 polarised response in vivo in mice, as evidenced by the production of IgG1 and other cytokines. 48 These authors have, therefore, suggested that EDN acts as an alarmin based on its ability to facilitate antigen recognition by the immune system.
The limited investigations to date mean that few specific molecular pathways can be proposed to explain the observed activities of EDN. It is conceivable that its neurotoxic activity could be manifested through activation of TLR2 as TLR2 is expressed in brain cells; however, this has yet to be confirmed. Activation of TLR2 by EDN may trigger an inflammatory response that results in these cells being eliminated by killer cells as part of the innate immune response. Alternatively, EDN may mediate activation of cell death pathways so as to reduce the infectivity of a virus or other infectious agent. Yet another possibility is that EDN may be internalised into infected cells, where it could degrade the viral RNA, so reducing the infectivity of viruses. Furthermore, it is also possible that EDN may be produced at the site of infection by eosinophils and monocytes/macrophages and help in modulating the immune response. These intriguing possibilities need further experimental verification.
ECP/RNase3
ECP, which is also known as RNase3, is another member of the RNaseA family that is found in the secondary granules of eosinophils. 49 The sequence of ECP is most similar to EDN and it appears that in humans the two genes have arisen through an evolutionarily recent gene duplication. 50 ECP possesses antibacterial, 51 anti-helminthic, antiviral and cytotoxic activities at micromolar concentrations in in vitro assays, leading to the suggestion that it contributes to host defence. 52
ECP has been shown to have cytotoxic activity against various mammalian cell lines, including those derived from blood, epidermis and mammary,21,53 as well as being toxic to parasites, and inhibiting viral replication. 52 Lehrer et al. demonstrated that ECP can kill both Escherichia coli and Staphylococcus aureus, but this was dependent on the temperature, pH and the ionic strength of the medium, as well as the growth phase of the bacteria. 26 Interestingly, recombinant protein containing ECP signal peptide and fused with RNaseA (rECPsp-RNaseA) has been reported to have cytotoxic activity against E. coli and Pichia pastoris, but not against mammalian cells. 54 It was proposed that this signal peptide may play a role in tissue remodelling. 55 Some reports have suggested that the cytotoxic property of ECP is independent of its ribonuclease activity, which is 10-fold lower than that of EDN.42,56 These somewhat contradictory reports mean that the exact nature of the cytotoxic activity, including its molecular basis, remains unclear.
The antimicrobial activity of ECP is somewhat better understood. Young and colleagues demonstrated that ECP can form transmembrane pores in an artificial lipid bilayer and suggested that this is a crucial step in its bactericidal activity. 57 Carreras and colleagues have shown that aromatic (tryptophan) and basic (arginine) amino acids present on the surface of ECP play a crucial role in the disruption of the outer and inner membrane of E. coli. 51 The role of individual tryptophan residues in the membrane destabilisation activity of ECP was explored through a mutagenesis approach. This suggested that the tryptophan at position 35 inserts into the lipid membrane, which destabilises and triggers the aggregation of the lipid layer, leading to the disruption of the membrane. 58 Thus, it appears that the surface exposed aromatic and basic amino acids of ECP facilitate membrane disruption through a carpet-like mechanism, as reported for other antimicrobial peptides.59–61 It is likely that this is a key step for both the bactericidal as well as the cytotoxic activities of ECP. While this provides one possible molecular level explanation of how ECP activity may be manifested at the molecular level, it should be noted that other activities besides membrane destabilisation could also contribute to the antimicrobial and cytotoxic activity of ECP. Recently, it has also been shown that ECP can bind to LPS and peptidoglycan with high affinity, and that this may also be important for its bactericidal activity. 62 This suggests that besides membrane disrupting activity, ECP may also act as an opsonin and could facilitate stimulation of inflammatory pathways. This possibility has not been actively investigated to date.
Like EDN, ECP has also been reported to have antiviral activity. 33 Domachowske and colleagues demonstrated that recombinant human ECP reduces the infectivity of RSV group B (RSV-B) and that this antiviral activity is dependent on its ribonuclease activity. 33 This finding suggests that, like EDN, ECP may also contribute to host defence against viruses.
Eosinophils have been reported to be cytotoxic against helminthic parasites in both in vitro and in vivo assays.24,63 It has been suggested that eosinophil-mediated killing of parasites is dependent on eosinophil granule proteins such as ECP. 49 ECP has been demonstrated to be highly toxic against the helminth parasite Schistosoma mansoni and on molar basis ECP is the most potent anti-helminthic protein compared with other eosinophil granule proteins. 23 ECP has also been reported to have cytotoxic activtiy against other disease-causing helminths, including Brugia pahangi and Trichinella spiralis.24,25 However, the underlying mechanism of ECP-mediated parasite killing is not very well understood.
ECP has been reported to induce apoptosis in airway epithelial cells. 64 However, the exact molecular mechanism for this is still unclear. Treatment of bronchial epithelial cells with ECP has been shown to induce the production of TNF-α and trigger apoptosis via a caspase-8 dependent pathway 64 through an unspecified mechanism. It has been shown that ECP binds specifically to a class of cell surface receptors—the heparan sulphate proteoglycans—and thereby becomes internalised through macropinocytosis.65,66 This has been suggested as a potential mechanism for how ECP gains entry into a cell. One possibility that has been suggested is that cell death is then initiated inside the cell by activation of a caspase cascade. 64 Alternatively, it has been suggested that ECP causes cell death by forming aggregates on the cell surface and by initiating the caspase-3 pathway without internalisation. 53 These studies suggest that ECP plays an important role in inducing apoptosis in cells and that apoptosis could be the major mechanism for ECP mediated cytotoxicity. However, further work is required to confirm this and shed light on its mechanism.
ECP has been associated with a range of inflammatory diseases (reviewed by Venge 67 ). Deposits of ECP have been found in affected tissues in inflammatory disorders such as bronchial asthma and Crohn’s disease, and the level correlates with the progression of tissue damage.68,69 The increased level of ECP in body fluids during inflammation is currently used as a clinical marker for the diagnosis of chronic inflammatory diseases.70,71 The physiological significance of this has not yet been elucidated. It is possible that ECP modulates inflammatory signalling or, alternatively, that elevated levels are the consequence of a cellular response elicited as part of the chronic inflammation that occurs in these diseases.
Inflammatory diseases such as asthma are often accompanied by elevated numbers of eosinophils in the affected tissues. 72 Apoptosis of epithelial cells occurs in these diseases as part of the process of lung remodelling and restoration of function. 73 It is possible that ECP plays a role in this. Eosinophils migrate towards infected epithelial cells and have been proposed to release effector proteins and other molecules from their granules.74–76 This could induce apoptosis of infected or damaged cells and also contribute to neutralising any pathogen that is present. The question of whether ECP plays a role in facilitating an immune response at the onset of the infection or plays an effecter role by killing the pathogen and the removal of damaged cells has not been resolved. Similarly, it is not clear whether the cytotoxic activity of ECP observed in cultured cells is physiologically relevant in an intact organism.
Eosinophil-associated ribonucleases
The eosinophil associated ribonucleases (EARs) are a subgroup of the RNaseA family that are found in the granules of eosinophils in rodents. They have close sequence similarity to EDN and ECP, and have been previously proposed to play a role in host defence. 77 At least 13 EARs that are expressed as full length proteins have been identified in mouse and 8 have been identified in rats.13,77–80 However, aside from their presence in eosinophils and their pattern of expression, their physiological roles are not very well understood. 77 At least some of the EARs appear to have antimicrobial activity. Rat EAR-1 and EAR-2 have been shown to inhibit the growth of E. coli and S. aureus at concentrations as low as 0.3 µM. 81 Antiviral activity has also been reported for the EARs. Moreau and colleagues demonstrated that recombinant EAR-2 reduces the infectivity of pneumonia virus of mice (PVM) in cultured respiratory epithelial cells. 82 However, EAR mRNA and protein has been reported to be reduced in murine lung tissue following challenge with PVM, 82 which is not the expected pattern of regulation for an innate immune effector protein. These authors hypothesised that the EAR down-regulation could be a strategy used by the virus to evade the host defence system. A molecular level explanation of how EARs may act is, to date, lacking. One possibility that could be experimentally verified is that the EARs act as opsonins by interacting with viral particles, as well as cellular receptors, thus triggering phagocytosis of the viral particle by phagocytes.
Other activities of the EARs have been reported. Mouse EAR-2 has been shown to have chemotactic activity on human and mouse dendritic cells in vitro, as well as in vivo. 46 Expression of EAR-6 increases in mouse spleen and liver tissue following challenge with S. mansoni, and this has been suggested to contribute to defence against helminthic parasites. 83 However, association with host defence based solely on observed changes in abundance during infection needs to be treated with caution, as this could be a peripheral aspect of a multifaceted response to infection. It has been demonstrated that EAR-11 transcripts appear in mouse lung tissue only after ovalbumin challenge and that this induction is dependent on CD4+ T cells. 16 These authors showed that broncho-alveolar macrophages constitutively express EAR1/2 and are the main source of EAR-11 transcripts in lung tissue of ova-challenged mice. Challenging the mice with Th2 cytokines (IL-4 or IL-13) resulted in higher expression of EAR-11 in the lungs of the challenged mice, supporting the involvement of EAR11 in a type 2 response. This study also demonstrated that challenging mice with ovalbumin or IL-13 results in an increase of RNase activity in the lung. It is possible that the EARs contribute significantly to this increase in ribonuclease activity and that RNase activity may be important for host defence during lung infections.
The physiological role of the EARs is largely unresolved. One possibility is the EARs may help in maintaining the homeostasis of lung by removing pathogenic RNA. Alternatively, during inflammation in the lung, specific EARs are expressed which help in regaining the homeostasis of the lung by degrading the cellular RNA within infected cells, so initiating cell death. Overall, the precise mechanism of action of any of the above described activities is still largely unknown.
Angiogenin/RNase5 and RNase4
Angiogenin, which is also known as RNase5, was first isolated from a human tumour cell line based on its ability to promote blood vessel growth. 84 RNase4, which was identified much more recently than RNase5, is very similar in sequence to RNase5. As with EDN and ECP, there appears to have been a series of gene duplications in different lineages during mammalian evolution, giving rise to multiple RNase5-like genes in the mouse (RNase5/ang1, ang2, ang3, ang4, ang6), as well as a distinct set of RNase5-like genes in cattle (RNase5/ang, ang2, ang3). 3 The structure of RNase5 is distinct from that of other RNases by having only three instead of four paired cysteine residues. Both RNase4 and RNase5 have ribonuclease activity, but the activity of RNase5 is significantly less potent than that of RNase4. Another distinctive feature of RNase5 is that it cleaves substrate into oligonucleotides rather than single nucleotides, which is the case for RNase4 and RNaseA. 85
RNase5 has been associated with a wide range of physiological processes, including tumorigenesis, reproduction, regeneration of damaged tissue, inflammatory bowel disease (IBD) and host defence.86–89 However, the underlying mechanisms for how RNase5 might influence these processes are unclear. RNase5 is constitutively expressed in a range of tissues and cell types. 90 Its pattern of expression is very different from other angiogenic factors, for example vascular endothelial growth factor (VEGF), and a molecular-level explanation for this activity of RNase5 is lacking. The physiological role of RNase5 is likely much broader than angiogenesis alone.
Several studies have suggested that RNase5 plays a role in host defence. 5 RNase5 levels increase in serum as a result of inflammation and mouse ang4 has been shown to be secreted from Paneth cells in response to bacterial LPS.27,89,91 Human RNase5/angiogenin and mouse ang1 and ang4 have been reported to have antimicrobial properties at nanomolar concentrations. 27 The biological significance of this anti-bacterial activity of angiogenin has been questioned. 92 Recently, it has been shown that RNase5 purified from cows’ milk has growth suppression activity against a dimorphic fungal pathogen, Candida albicans, and is particularly potent against the hyphal form. 5
RNase5 has been reported to function as an immunomodulator by interacting with a range of immune cells.93–95 It has been shown that RNase5 can inhibit the degranulation of polymorphonuclear cells (PMNL) at nanomolar concentrations. 93 It is possible that this activity may serve to inhibit uncontrolled degranulation of PMNL and contribute to the host’s overall return to a normal homeostatic condition. Shcheglovitova and colleagues demonstrated that RNase5 stimulates the levels of pro-inflammatory cytokines in human blood leukocytes when these cells were stimulated with phytohemagglutinin. 96 Human mast cells express RNase5 and store it in their granules. 95 These cells secrete RNase5 in response to a range of pathogen-associated substances, including E. coli LPS, peptidoglycan (PGN) and flagellin. 95 While the physiological role of the RNase5/angiogenin proteins is far from clear, together these findings suggest some possibilities that could form a focus for further work. For example, it is conceivable that RNase5 could be secreted by immune cells in response to certain pathogens and help to modulate immune responses. The presence of RNase5 in milk and its expression in intestinal tissue could conceivably help in establishing an interaction between gut microbes and the host, thereby contributing to optimal gut function.
RNase4 is very similar in sequence to RNase5. 5 In cattle, both RNase4 and RNase5 share the same promoter but are, nevertheless, independently expressed, with distinct patterns of expression between tissues (Wheeler, Maqbool and Gupta, J Mol Evol, in press). Compared to RNase5, RNase4 has not been well characterised to date, although there is some data associating RNase4 with host defence. The major sites of expression in cattle appear to be the liver, intestine, testes and mammary gland—all of which have a significant host defence function. RNase4 has been shown to suppress the growth of C. albicans, although it is not as potent as RNase5. 5 Overall, the physiological functions of RNase4 are largely unknown and good evidence for a role in host defence is lacking at this time.
RNase4 and RNase5 appear to be able to bind to nucleic acids (S.K. Gupta, unpublished data). Therefore, it is conceivable that these RNases may act as immunomodulatory proteins by interacting with pathogen-derived nucleic acid and facilitate their presentation to innate immune receptors within the cell. Several such nucleic acid response receptors have been described, including TLR3, TLR7, TLR9, RLRs and other DNA receptors.97–99 It has been suggested short length dsRNA is more stimulatory to the receptors compared with full length dsRNA. 100 Therefore, it is possible that the characteristic RNase activity of RNase5, which results in the formation of short oligos, is important for the recognition of exogenous nucleic acids by one or more of these receptors.
RNase7
RNase7 is another member of the RNaseA family that has been associated with host defence. 101 This RNase was first identified and purified from human skin, 28 which is a significant site for host defence. It is one of the major proteins secreted by keratinocytes and is the most abundant RNase of human skin.28,102 It has been proposed that, along with other components secreted by human skin, RNase7 may contribute to the host defence of cutaneous epithelia.101,103 However, its physiological role is likely to extend beyond skin as RNase7 has also been found to be expressed in a range of other host defence associated epithelial tissues, such as the urinary tract and the respiratory tract, as well as the kidney.20,104
RNase7 has been shown to possess broad spectrum microbicidal activity against a range of microbes. 101 Both native and recombinant RNase7 have bactericidal activity at micromolar concentrations against several Gram-negative and Gram-positive bacteria, including E. coli, S. aureus, Pseudomonas aeruginosa and Enterococcus faecium, as well as some fungal species.28,105 Of particular interest, native RNase7 was found to be lethal against a vancomycin-resistant isolate of E. faecium at concentrations as low as 30 nM, 28 suggesting it could have potential as an alternative to traditional antibiotics. The recombinant form of RNase7 also possesses similar bactericidal activity against E. coli and S. aureus. 106 A recent study demonstrated that RNase7 levels are increased in skin within 2 h of S. aureus challenge and this controls the growth of S. aureus on the skin surface. 107 Together, these findings suggest that RNase7 may contribute to the host defence of skin by controlling the colonisation of pathogens as a first line of defence against infection.
The mechanism for the bactericidal activity of RNase7 is not fully understood. It has been shown that members of RNaseA family with bactericidal activity contain a larger proportion of positively-charged residues on the surface of the protein. 108 RNase7 possesses four clustered lysine residues which have been shown to be critical for its bactericidal activity. 108 Torrent and colleagues demonstrated that human RNase7 interacts with synthetic lipid vesicles through electrostatic attraction and stimulates rapid leakage of their contents.109,110 This suggests RNase7 may act to disrupt bacterial membranes in a manner similar to other well-characterised antimicrobial proteins and peptides such as the bactericidal/permeability-increasing protein. 111 A recent study demonstrated that RNase7 can bind to LPS, as well as an outer membrane protein of P. aeruginosa, OprI. 112 These authors postulated that RNase7 dissociates an LPS-OprI complex, possibly by eliminating N-terminal fatty acids from OprI and that this detaches OprI from the membrane, which causes membrane permeability. It was suggested that RNase7 then forms a complex with the soluble OprI and is internalised into the cell where it may trigger cell death. The veracity of this membrane destabilisation mechanism has not yet been experimentally verified.
Several studies have shown that RNase7 expression is responsive to external stimuli relevant to host defence. It has been shown that the treatment of primary keratinocytes with TNF-α, IL-1β, IFN-γ or heat-killed bacteria results in increased expression of RNase7 in these cells. 28 In another study it has been shown that Staphylococcus epidermidis induces the expression of RNase7 in kerakinocytes in a TLR2-, EGFR- and NFκB-dependent manner. These authors suggested that RNase7 contributes to the innate immune defence of the skin. 113 It has also been shown that RNase7 expression increases in response to IL-1β in the cells of the ocular surface and that this is mediated through the MAPK pathway. 114 Another study demonstrated that the abundance of RNase7 mRNA increased in corneal epthelial cells in response to a protozoan challenge, 115 further suggesting a role for RNase7 in host defence in the eye. RNase7 is also expressed in human hair follicle epithelial cells when they are challenged with lipoteichoic acid (LTA), protein A or LPS, as visualised by immunohistochemistry, 116 suggesting that in some cells TLR-mediated pathways could be involved in the induction of expression. Taken together, these findings suggest that RNase7 is expressed in response to a range of stimuli in tissues that have a host defence function. However, the precise physiological role that this RNase plays and the particular signalling mechanisms involved still remains unclear.
RNase8
RNase8 was first identified as a gene that is expressed prominently in the placenta. 117 So far, no specific function has been identified for RNase8 and it remains little studied. However, some data point to a role in host defence. It has been shown that RNase8 exhibits microbicidal activity against a range of pathogenic bacteria, including multi-drug resistant pathogens at micro- to nanomolar concentrations. 29 No specific mechanism of action has been proposed for this activity. RNase8 has a high degree of amino acid similarity with RNase7 suggesting that both these proteins may share the same mechanism of bacterial killing. It has been long known that some pathogens can pass through the placenta to the fetus. 118 One possible physiological role for RNase8 is that it contributes to placental host defence by killing any pathogens from the maternal circulation and so preserves a sterile environment for the fetus.
Bovine seminal RNase
Bovine seminal RNase (BS-RNase) is most similar in sequence to bovine pancreatic and bovine brain RNase, all of which are most similar to RNase1 in other species. BS-RNase has a distinct pattern of expression among tissues, being found only in seminal fluid. 8 It is the only RNase reported to exist as a homodimer in its natural form.8,119 Despite its presence at a site where host defence is important, the knowledge of its involvement in host defence is very limited.
BS-RNase has been shown to be selectively cytotoxic to a range of cancer cells.22,120,121 Lee and colleagues demonstrated that the substitution of three residues (G38K/K39G/G88R) increases cytotoxic activity of BS-RNase in K-562 cells. 22 This activity was even higher with the monomeric variant of BS-RNase, presumably because the monomer has reduced affinity to ribonuclease inhibitor (RI) present in the cells. These authors suggested that the activity is at least partly owing to cationic residues in BS-RNase, which increases its affinity towards anionic mammalian cell membranes. In support of this, substitution of glycine 38 for a lysine residue was shown to increase cytotoxic activity against transformed fibroblasts. 122 The G38K substitution results in the protein binding more strongly to cell membranes than the wild type owing to stronger electrostatic interaction. In turn, this may lead to an increased rate of internalisation by endocytosis. 123 It has been suggested that the cytotoxic activity of BS-RNase is the result of triggering apoptosis after internalisation of the protein into cells and subsequent degradation of cellular RNA.124,125 However, the exact mechanism underlying the cytotoxic activity of BS-RNase is still unclear.
BS-RNase has also been shown to have immunosuppressive activity. It has been reported to suppress the activation of proliferating lymphocytes.126,127 Furthermore, treatment of activated T-lymphocytes with BS-RNase resulted in reduced expression of IL-2 α chain receptor, suggesting that BS-RNase manifests its immunosuppressive effect by interfering with IL-2 receptor expression on T lymphocytes. 126 Another study demonstrated that the presence of BS-RNase caused phytohemaglutinin-stimulated lymphocytes to undergo apoptosis, as indicated by observations typical of apoptosis, including DNA fragmentation, cell shrinking, chromatin migration and disorganised mitochondria. 128 BS-RNase has been shown to be selectively cytotoxic to thyroid carcinoma cells and to induce apoptosis in this cell line in a caspase-8 and caspase-9 dependent manner. 125
The extent of the cytotoxic activity of BS-RNase appears to vary according to the cell type.125,129 The cytotoxic activity of onconase (a RNase2-like protein isolated from amphibians) has been shown to be dependent on the route of internalisation into the cell. 130 It seems that the route of internalisation of RNases may be responsible for their differential cytotoxic activity in different cell types.125,130 These possibilities provide a focus for further research.
Conclusions
Significant progress has recently been made in understanding the physiological role of the mammalian RNase family of proteins. In particular, this has led to the hypothesis that some members of the family contribute to host defence. Although these suggestions are based on limited data and our mechanistic understanding of these activities is not strong, there are three lines of evidence which support this hypothesis. Firstly, their expression pattern in various tissues and in response to immune stimulation is consistent with a role in host defence. Secondly, in vitro assays indicate some family members have antimicrobial, antiviral and cytotoxic activities. Thirdly, they have immunomodulatory activity. Together, these data suggest a family of proteins that are expressed in the appropriate locations, with the relevant activities, to both combat infection and modulate the inflammatory response. The evidence obtained to date is consistent with any of three broad mechanisms: (i) the RNases could act as innate immune effector proteins through their antimicrobial activities; (ii) they could degrade extracellular RNA so as to prevent it from activating innate immune pathways; and (iii) they could act as opsonins by binding to extracellular nucleic acids and facilitate interactions with innate immune receptors such as TLR3, TLR7, TLR9 or other cytosolic nucleic acid sensing receptors in a manner analogous to the role of LPS binding protein in facilitating interaction of LPS with TLR4. These possibilities are summarised in Figure 1. To validate these concepts, further experimental evidence is required. Controlled studies comparing activities between the different RNase family members would allow functional differences between the proteins to be examined in a consistent manner. Consideration should also be given to assessing function under experimental conditions which mimic the physiological conditions prevailing at its site of secretion in vivo, for example the presence of serum. Furthermore, many of the observed activities have been reported only once, so confirmatory studies would be of value. Clarification of these questions will advance our understanding of the roles of the RNases in host defence and thereby contribute to an improved understanding of the biology of innate immunity. Furthermore, new insights could be gained into how the RNases could be used therapeutically to improve health. In addition, as some of the RNases are present in secretions from which they can be readily extracted, such as milk, the activities of some of the RNases may also prove useful as ingredients in functional foods.
Schematic overview of the proposed mechanisms of action for the RNases in host defence. The numbered processes are as follows: (1) constitutive production and secretion of RNases; (2) induced production and secretion of RNases in response to microbial challenge; (3) direct antimicrobial activity and leakage of cellular contents; (4) RNase activity to clear microbial RNA; (5) DNA-binding activity of RNases facilitate recognition of microbial DNA to innate immune receptors; (6) characteristic RNase activity of RNase5 produces short fragments of viral RNA which are then recognised by innate immune receptors; (7) EDN or ECP secreted by eosinophils degrades viral RNA; (8) receptor-mediated uptake of EDN or ECP induces apoptosis; (9) EDN or ECP degrade microbial RNA released from infected and dying cells; (10) RNases produced by the liver are secreted into blood and lyse circulating microbes.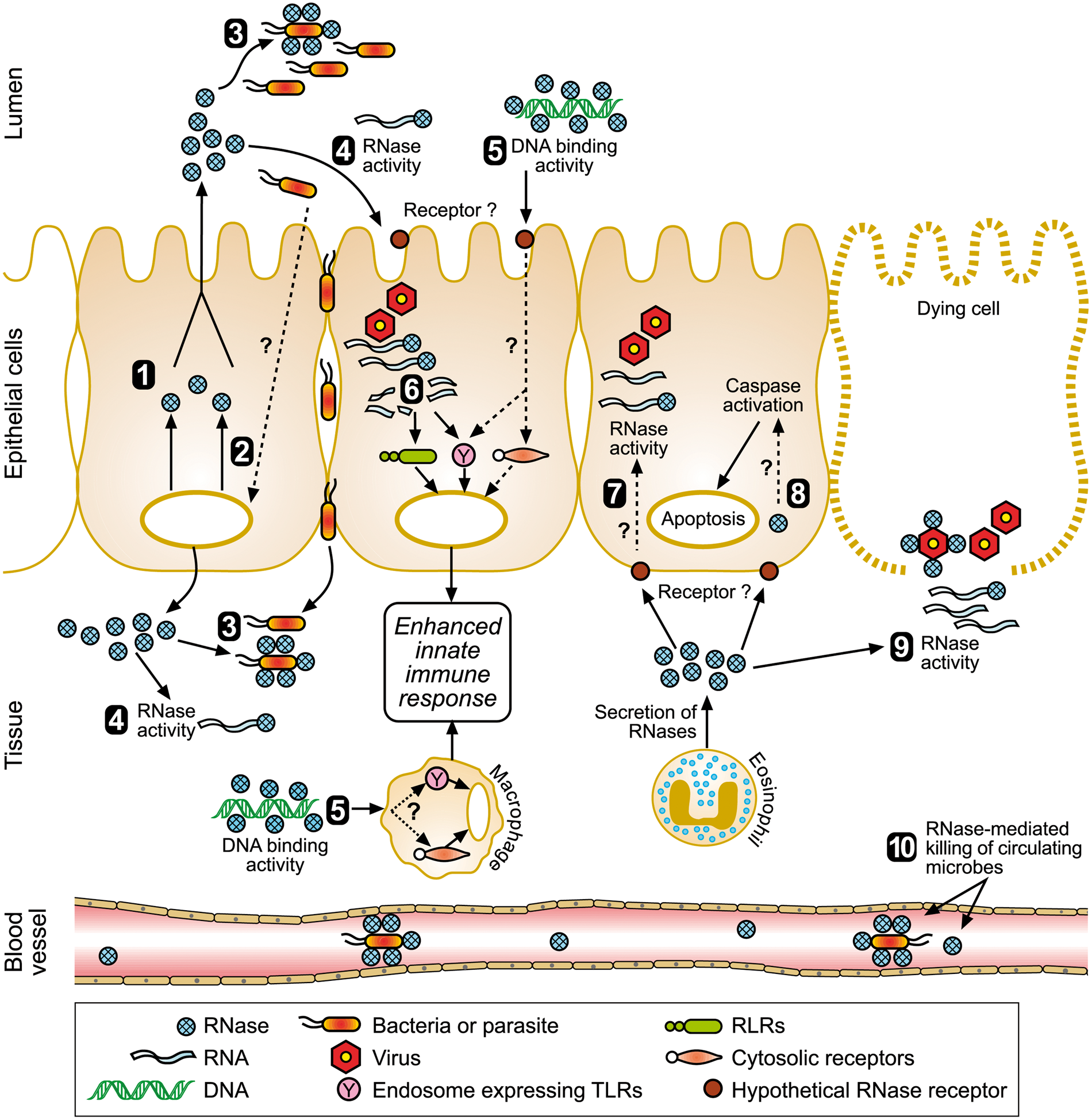
Footnotes
Funding
The authors wish to acknowledge the following financial support: PhD scholarships from the University of Otago and AgResearch awarded to SG, and a Ministry of Science and Innovation research contract (C10X0707) ‘Host Defence Proteins in Milk’ awarded to TW.
Acknowledgements
The figure was prepared by Ms Pauline Hunt.
