Abstract
Eritoran, a synthetic analogue of lipid A, has been shown to bind to TLR4/MD-2 complex and thereby block the interaction of endotoxins with TLR4. We report here the results of a study conducted to assess the single-dose safety and tolerability, as well as the pharmacokinetics and pharmacodynamics, of eritoran infusion in Japanese and Caucasian healthy adult men. Sixty-four men (aged 20–45 years; body mass index 18–30 kg/m2) were randomized into four groups: 4-mg total dose (six Japanese and six Caucasian men); 12-mg total dose (12 Japanese and 12 Caucasian men); 28-mg total dose (six Japanese and six Caucasian men); and placebo (eight Japanese and eight Caucasian men). Eritoran in single doses up to 28 mg over 4 h was well tolerated, with no apparent ethnic differences noted. Plasma concentrations were slightly higher in Japanese versus Caucasian men; these differences were not significant after adjustment for differences in body mass (clearance: approximately 1.2 ml/h/kg; volume of distribution at steady state: approximately 0.07 l/kg). The ex vivo endotoxin inhibitory activity of eritoran was similar in Japanese and Caucasian men. The data do not indicate any need for clinical dose adjustment for possible ethnic-based differences in drug distribution or metabolism.
Introduction
Eritoran, a synthetic analogue of lipid A (Figure 1A, B),
1
antagonizes the immune-activating effects of endotoxin from Gram-negative bacteria by binding to TLR4/MD2 and blocking its interaction with endotoxin.
2
The structures of endotoxin with the TLR4/MD-2 complex have been elucidated and reveal that these molecular associations are involved in endotoxin recognition.3,4 TLR4 activation, an essential component of the innate immune response system in healthy individuals, results in the release of cytokines and pro-inflammatory mediators.
5
Activation of the TLR4 signaling pathway by endotoxin is the primary mediator of sepsis and septic shock via excessive production of cytokines and pro-inflammatory mediators.
5
Structure of (A) eritoran; (B) lipid A; and (C) identified eritoran metabolites (1-dephospho-eritoran [M1]; 4′-dephospho-eritoran [M2]; 1,4′-di-dephospho-eritoran [M3]).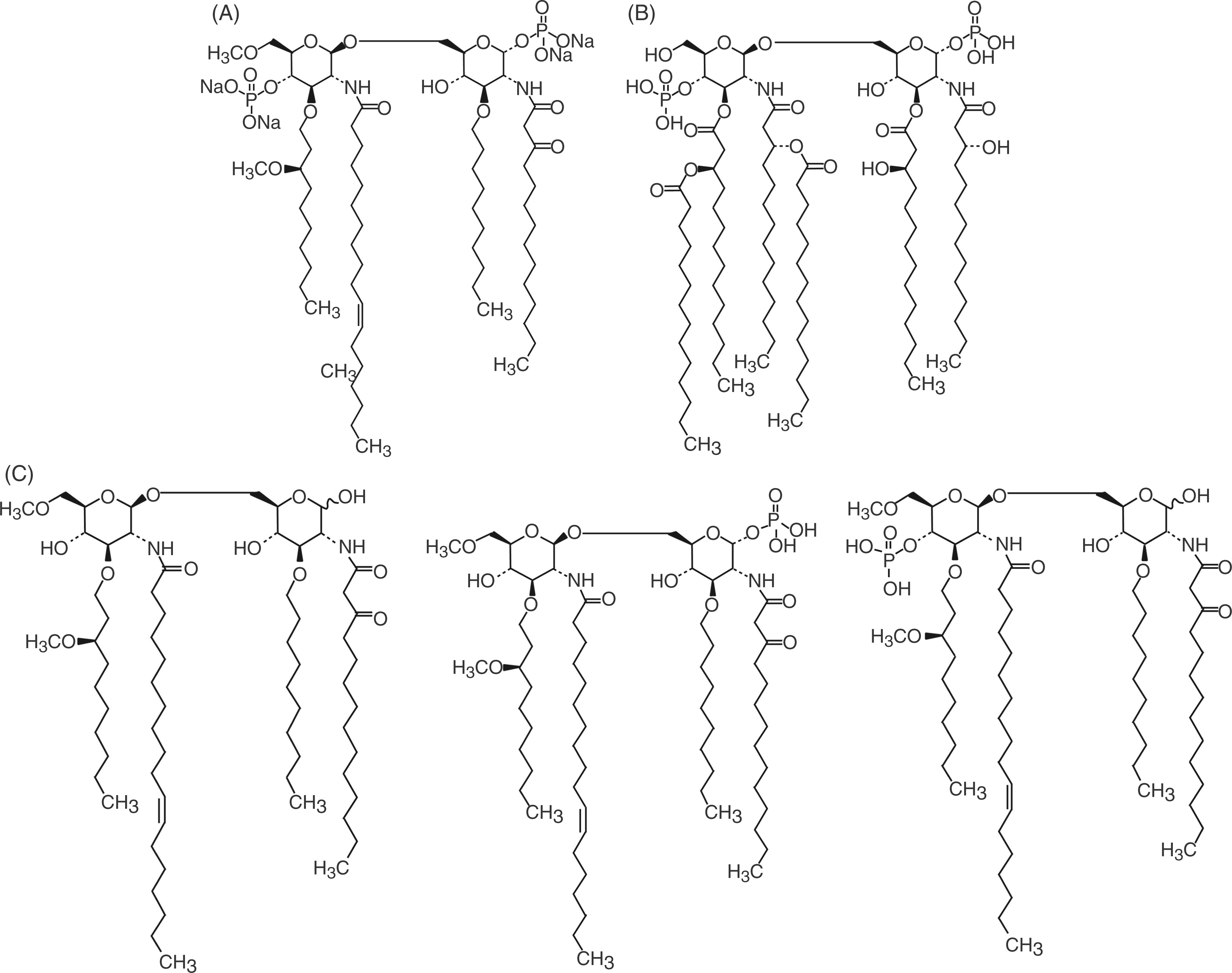
Eritoran has been shown to be well tolerated when administered by continuous infusion at total doses up to 252 mg in healthy volunteers 6 and by twice-daily infusion at total doses of up to 105 mg in healthy volunteers and in patients with severe sepsis.7,8 Unless otherwise noted, all doses and pharmacokinetic values described for eritoran in this article are uniformly expressed as the tetrasodium salt form of eritoran. In a phase II trial, eritoran treatment demonstrated a trend towards a lower mortality rate at the 105 mg dose in patients with severe sepsis and high predicted risk for mortality. 8 Eritoran was under investigation for the treatment of severe sepsis in a phase III trial being conducted at 159 study sites worldwide, including 28 sites in Japan; however, the study did not meet its primary endpoint of reduction in 28-d all-cause mortality in patients with severe sepsis. 9 Previous studies of other drugs have identified differences between Asian and Caucasian patients that are potentially significant for drug therapy. 10 Therefore, it is important to evaluate differences in the safety, pharmacokinetic and pharmacodynamic profiles between Japanese and Caucasian populations.
Pharmacokinetic studies in healthy volunteers have shown that eritoran has a small volume of distribution (approximately 3–6 l) and slow clearance (clearance (CL); <0.1 l/h).6,7,11 In plasma, the majority of eritoran (55%) is bound to high-density lipoprotein (HDL). 6 The binding of eritoran to HDL, but not to other lipoprotein fractions, is associated with rapid loss of eritoran activity. 6 Three metabolites of eritoran were identified in animal studies in dogs and rats. These were characterized as dephosphorylated moieties, 1-dephospho-eritoran (M1), 4′-dephospho-eritoran (M2), and 1,4′-di-dephospho-eritoran (M3) (Figure 1C). The metabolites do not bind to TLR4; metabolites M1 and M2 are 17–230-fold less active than eritoran and M3 is inactive (data not shown).
Human studies in healthy volunteers have used an ex vivo pharmacodynamic assay to assess the ability of eritoran to block the activity of endotoxin. These studies have shown dose-dependent activity for both eritoran and endotoxin, with plasma levels of approximately ≥3 µg/ml eritoran demonstrating ≥85% inhibition of the activity of up to 1 ng/ml endotoxin.6,7 However, it is not known whether there are differences in the safety, pharmacokinetics or pharmacodynamics of eritoran between different ethnic groups. Potential ethnic differences may be related to a number of variables, including differences in weight, plasma lipoprotein profiles 12 or hepatic metabolism. 10
The primary objective of this study was to assess the safety and tolerability of eritoran when administered as a single, 4-h i.v. infusion (4, 12, 28 mg or placebo) to Japanese and Caucasian healthy adult men. The dosing regimens in this study were being developed for severe sepsis and were chosen based on previous work carried out to establish a dose response. 7 Secondary objectives included assessment of the pharmacokinetics of eritoran and its primary metabolites M1, M2 and M3, assessment of the pharmacodynamics of eritoran and determination of the plasma lipoprotein distribution of eritoran after a single, 4-h i.v. infusion of eritoran 28 mg to Japanese and Caucasian healthy adult men.
Materials and methods
Study design
This was a double-blind, randomized, single-center, placebo-controlled, ascending single-dose, sequential-group study. Patients were randomized on d 1 and remained as inpatients at the study site for the duration of the dosing and sampling periods (until d 8). The study protocols were conducted in accordance with the Declaration of Helsinki, reviewed by an independent Institutional Review Board (California IRB, Inc., Pasadena, CA, USA), and approved by Eisai Medical Research Inc. (EMR, Ridgefield Park, NJ, USA). All men participating in the study provided informed consent forms. They also all met the inclusion criteria but none of the exclusion criteria listed below.
Treatment groups consisted of the following: group 1 : 12 men administered 4 mg total dose of eritoran tetrasodium (1 mg/h); group 2 : 24 men administered 12 mg total dose of eritoran tetrasodium (3 mg/h); group 3 : 12 men administered 28 mg total dose of eritoran tetrasodium (7 mg/h); group 4 : 16 men administered placebo in 5% dextrose water (4-h infusion). In this article, eritoran tetrasodium is represented as eritoran and the values were based on eritoran tetrasodium. Eritoran 1 mg (free acid) is equivalent to eritoran tetrasodium 1.067 mg.
Study subjects
Japanese and Caucasian men between the ages of 20 and 45 years with body mass indices (BMIs) from 18 to 30 kg/m2, in good health and with no clinically significant abnormalities were enrolled in the study from a single center in California, with ethnicity verified from a detailed family history and after providing written consent to the study procedures. Japanese men in this study were defined as first-generation Japanese (with Japanese parents and grandparents), and must have had a Japanese passport, lived for no more than five years outside of Japan and had retained their Japanese lifestyle or habits, including diet. Exclusion criteria included significant health abnormalities, known or suspected history of significant alcohol or drug misuse, positive hepatitis B surface antigen or hepatitis C virus screen, or a positive HIV test at screening, recent mass loss or gain >10%, recent blood donation or receipt, smoking more than 10 cigarettes a day and unable to refrain from smoking during the study, and use of any medications, supplements or herbal preparations within the previous week.
A total of 64 men were randomized: 12 to group 1 (eritoran 4 mg) and group 3 (eritoran 28 mg); 24 to group 2 (eritoran 12 mg); and 16 to group 4 (placebo). Sixteen men were randomized to group 2 (eritoran 12 mg) or group 4 (placebo) and received the incorrect infusion rate of eritoran (12 men) or placebo (4 men). One of the mis-dosed men was re-enrolled in group 3 (eritoran 28 mg). Data from the 16 mis-dosed men in group 2 or group 4 and the 1 man who was re-enrolled were excluded from all analyses. The Japanese man in group 3, who was dosed twice, was excluded from the final pharmacokinetic parameter summaries in accordance with criteria set in the statistical analysis plan for the study. Therefore, the safety and pharmacodynamic populations included all remaining 47 men, with 6 men in each group with the exception of 5 Japanese men in group 3. The pharmacokinetic population included the 35 men infused with eritoran. The lipoprotein population included 11 men in group 3 (eritoran 28 mg). All randomized men completed the study.
Patient demographics and baseline characteristics.
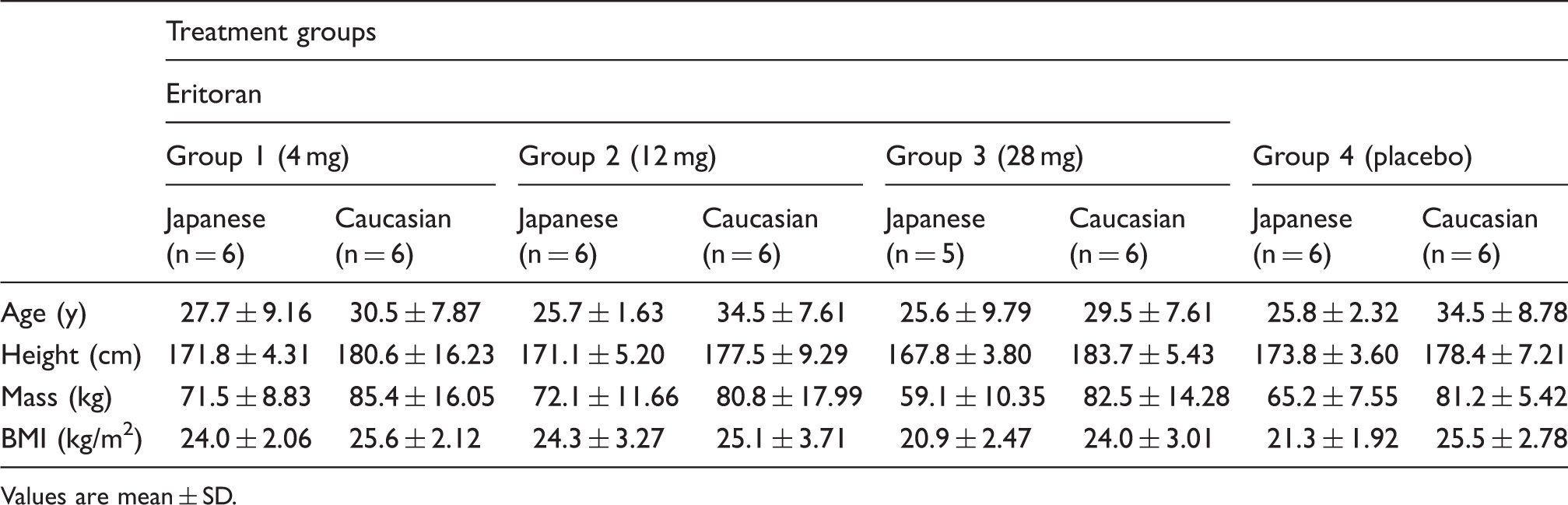
Values are mean ± SD.
Safety evaluation
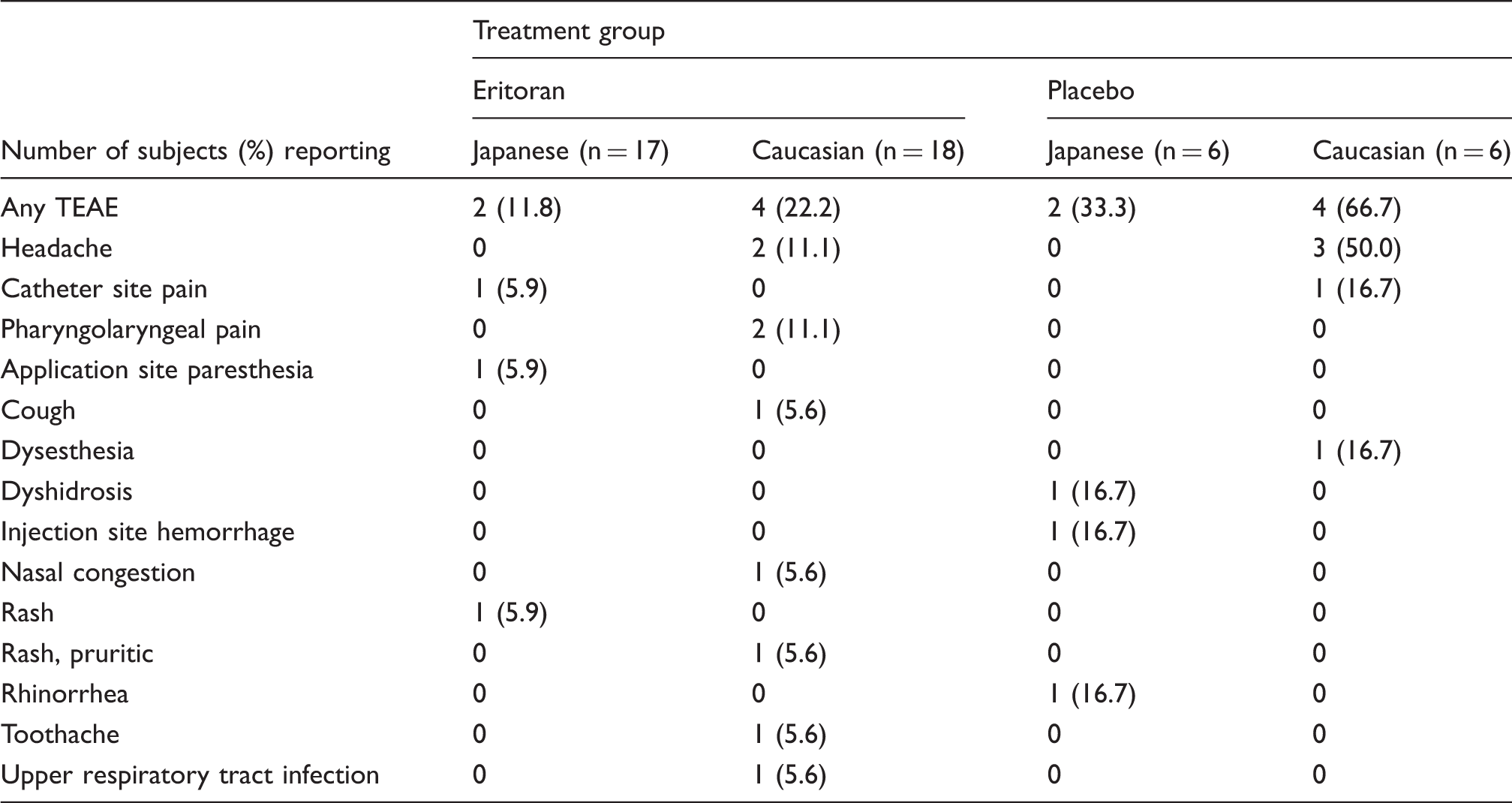
Treatment-emergent adverse events (TEAEs).
Pharmacokinetic assessments
For the analysis of plasma concentrations of eritoran and metabolites, 5-ml blood samples were collected immediately before infusion and at 1, 2, 3, 4, 5, 6, 8, 12, 24, 32, 48, 56, 72, 80, 120 and 168 h after the start of the infusion. The sampling period of 168 h was consistent with previous experiments where elimination half-life in humans ranged from ∼35 to 55 h. 7 Plasma was harvested within 30 min of collection and stored at approximately −70°C until analysis. Plasma concentrations of eritoran and metabolites M1, M2 and M3 (group 3 only) were determined using a validated liquid chromatography/mass spectrometry/mass spectrometry (LC/MS/MS) method with a quantifiable range of 5 to 2000 ng/ml. 13
Pharmacokinetic parameters of eritoran and metabolites M1, M2 and M3 were estimated by non-compartmental analysis (NCA) using WinNonlin, Version 5.01 (Pharsight; Mountain View, CA, USA). The maximum plasma concentration (Cmax) of eritoran, M1, M2 and M3, and the time to reach Cmax (tmax), were obtained through visual inspection of the plasma concentration-versus-time profiles. The observed area under the plasma concentration-versus time-curve from time zero to the last measured time point (AUC0–t) and to infinity (AUC0–∞) were calculated using the trapezoidal method. The elimination half-life (t1/2) was calculated as t1/2 = 0.693/λz. Lambda z (λz), the slope of the concentration-versus-time-curve during the terminal phase, was determined by linear regression. For eritoran, CL was calculated as CL = dose/AUC0–∞. The volume of distribution at steady state (Vdss) was calculated as Vdss = CL · MRT, where MRT is the mean residence time from time zero (pre-dose) to infinity and was calculated as MRT = AUMC/AUC0–∞. The AUMC represents the estimated area under the first moment plasma concentration versus time curve from time zero (pre-dose) to infinity, calculated as AUMC0–∞ = AUC0–t +(Clast · tlast)/λz + Clast/λz2.
Assessment of plasma lipoprotein distribution of eritoran
In group 3 only, three blood samples were collected at 4 h (corresponding to the end of the infusion), 24 h and 72 h after the start of infusion of eritoran to measure the distribution of eritoran into plasma protein and lipoprotein in Japanese (n = 5) and Caucasian (n = 6) men. The plasma was separated into its HDL, low-density lipoprotein (LDL), very-low-density lipoprotein (VLDL) and lipoprotein-deficient plasma (LPDP) fractions by single-spin density gradient ultracentrifugation. 14 The plasma fraction was frozen neat while the lipoprotein fractions were frozen in cesium bromide. The plasma and lipoprotein cholesterol concentrations were determined in each fraction by an enzymatic assay (Sigma Diagnostics Inc., St. Louis, MO, USA) utilizing established colorimetric techniques15,16 and were correlated to the percentage of eritoran recovered in each fraction. After fractionation, the lipoprotein fractions were mistakenly stored for up to 85 d at 4°C and then stored for up to 18 additional d at −70°C prior to quantification of eritoran by LC/MS/MS. This extended storage exceeded the time period (69 h) during which eritoran was shown to be stable in lipoprotein factions at 4°C. When eritoran recovery was determined based on the total amounts found in each lipoprotein/protein fraction and compared with the known amounts recovery in the plasma fraction before lipoprotein separation, very little degradation was observed. The concentration of eritoran in whole plasma and in each fraction was determined by validated extraction and LC/MS/MS assays. 13
Pharmacodynamic assessments
Ex vivo activity of eritoran was assessed as previously described. 11 Human studies in healthy volunteers used an ex vivo pharmacodynamic assay to assess the inhibitory activity of eritoran on endotoxin-stimulated tumor necrosis factor (TNF)-α production in whole blood collected from all men pre-dose and at 4, 8, 12, 24, 32, 48, 56, 72, 80, 120 and 168 h after the start of the infusion. Briefly, endotoxin from Escherichia coli 01111:B4 (List Biological Laboratories Inc. (Campbell, CA, USA), concentrations: 0, 0.05, 1 or 10 ng/ml) was added to whole blood drawn from the men before and after administration of placebo or eritoran; cellular activation was assessed by measuring release of TNF-α.6,7 Mean TNF-α levels were calculated for each time point; percent response with respect to baseline (pre-dose) values were calculated for each endotoxin concentration, for each time point and for each dose level and ethnic group. All endotoxin stimulation assays were performed in triplicate per time point and endotoxin concentration, and the means are expressed as percent response and inhibition of baseline (pre-dose) endotoxin response.
Statistical analysis
Descriptive summary statistics were calculated for demographic, pharmacokinetic and pharmacodynamic parameters, and for plasma lipoprotein distribution of eritoran. For demographic parameters, statistics include number, mean, SD, median, minimum and maximum for continuous variables, and number and percentage of patients for categorical variables. Plasma concentrations and pharmacokinetic parameters are expressed as mean ± SD except for tmax, where medians were calculated. For pharmacodynamic parameters, mean, SD, median, minimum and maximum were calculated for each time point, dose and ethnic group. For evaluation of dose-linearity, the dose-normalized Cmax (Cmax/D) and AUC0–∞ (AUC0–∞/D) were calculated for each dose and ethnic group. For safety parameters, tabular summaries were prepared that compiled the number and percentage of patients with TEAEs; in addition, TEAEs were classified by system organ class/preferred term.
Results
Safety
There were no serious AEs reported and eritoran was well tolerated in single doses tested up to 28 mg infused over 4 h. Among the 12 men who reported TEAEs, all reported only 1 or 2 TEAEs (Table 2). Eritoran administration was not associated with changes in vital signs or AEs that would suggest endotoxin-like agonistic activity, such as fever, chills or myalgia. 17 Some infusion site pain and inflammation was observed, which resolved spontaneously, as noted in follow-up clinic visits. With the exception of headache, catheter site pain and pharyngolaryngeal pain, no TEAE was reported in more than one man. All TEAEs were mild and only two were considered by the blinded investigator to be possibly related to administration of study drug: application site paresthesia in a 25-year-old Japanese man who received 12 mg eritoran and headache in a 27-year-old Caucasian man who received placebo.
Abnormal laboratory values were reported for two Japanese men and two Caucasian men, but none was considered clinically significant. No clinically significant abnormality was noted in physical examinations and there were no trends noted in the evaluations of vital signs. Some abnormalities (mostly sinus bradycardia) were noted in post-baseline electrocardiogram, but none was considered clinically significant. Results of a complete QTc study reported elsewhere indicate that there is no indication of clinically significant heart rhythm changes. 7 No patient had study drug discontinued or interrupted because of AEs. Although TEAEs were reported at a greater frequency in Caucasian men compared with Japanese men, the same pattern was observed whether patients received placebo or eritoran. Single doses of 4, 12 and 28 mg eritoran over 4 h were considered generally safe and well tolerated in healthy Japanese and Caucasian men, with no apparent ethnic differences noticed.
Pharmacokinetics
Pharmacokinetic parameters after single-dose administration.
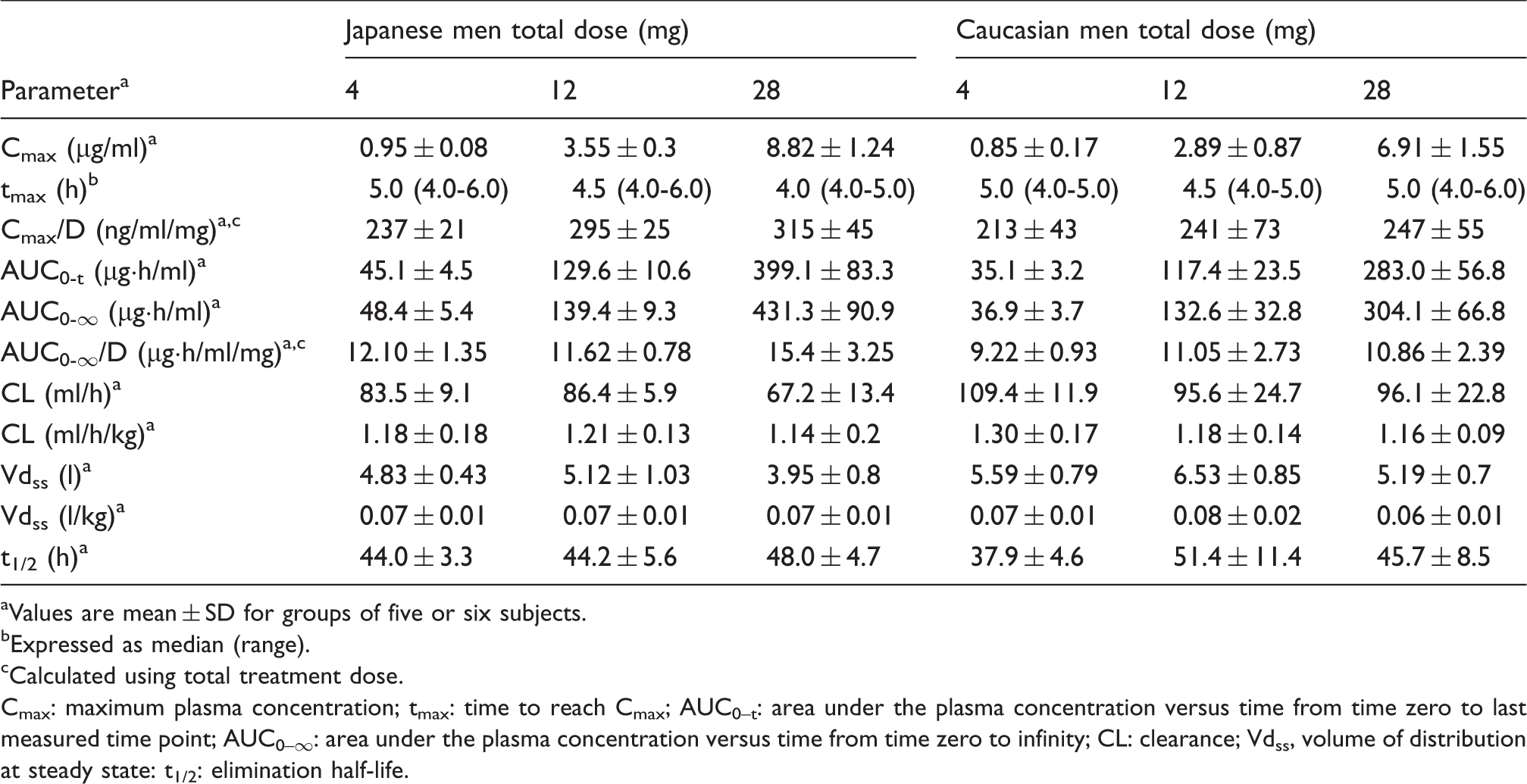
Values are mean ± SD for groups of five or six subjects.
Expressed as median (range).
Calculated using total treatment dose.
Cmax: maximum plasma concentration; tmax: time to reach Cmax; AUC0–t: area under the plasma concentration versus time from time zero to last measured time point; AUC0–∞: area under the plasma concentration versus time from time zero to infinity; CL: clearance; Vdss, volume of distribution at steady state: t1/2: elimination half-life.
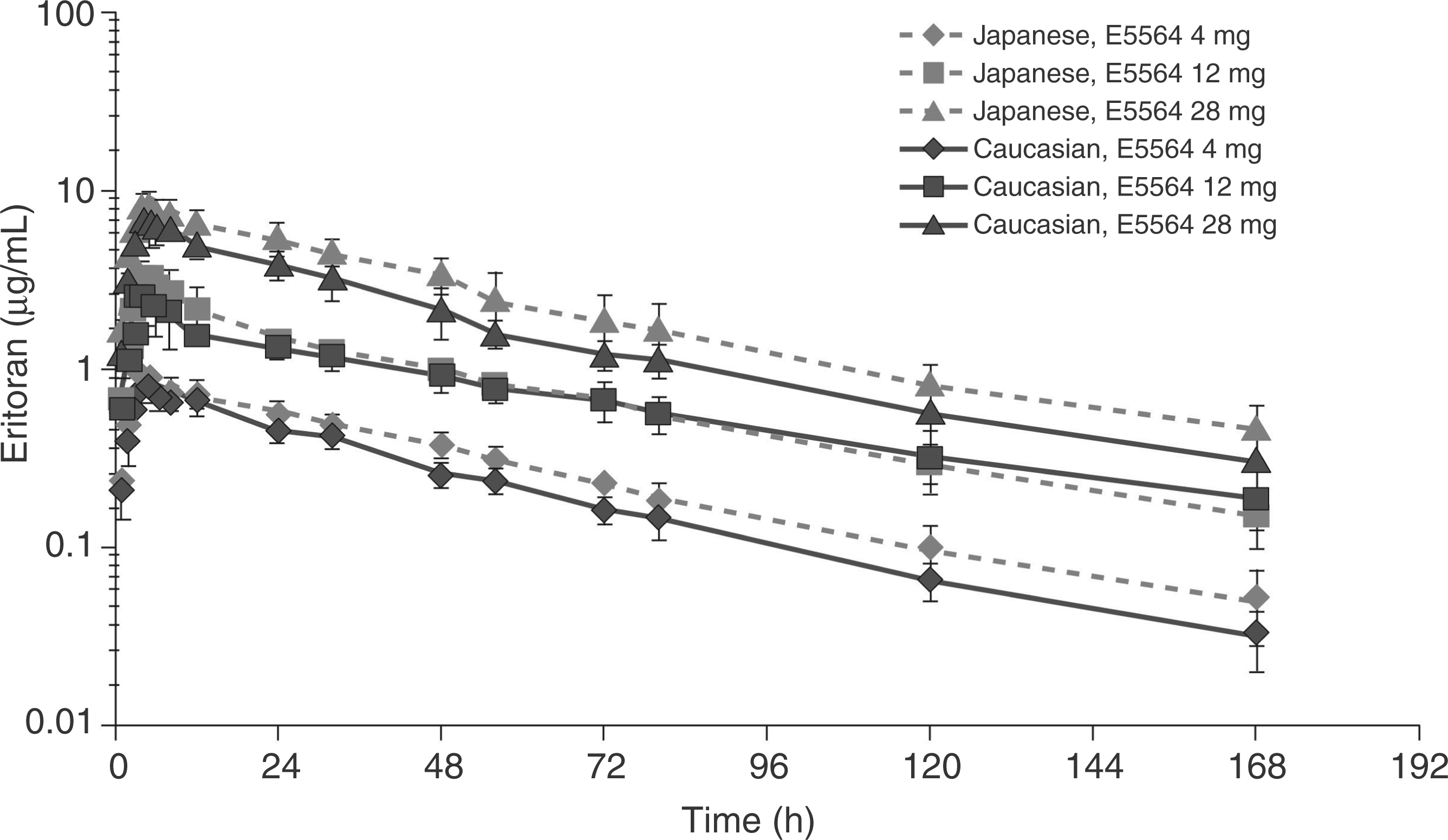
Plasma concentration-time profile of eritoran after single administration (mean ± SD, n = 6 per group except n = 5 for Japanese 28 mg).
Consistent with previous studies, the pharmacokinetic profile of eritoran was characterized by small Vdss, slow CL and slow elimination rate.6,7,11,18 The volume of distribution of eritoran observed is consistent with the prior clinical studies and suggests that eritoran is predominantly maintained in vascular compartment. Exposure to eritoran was slightly higher in Japanese men compared with Caucasian men (Table 3). When normalized by mass, Cmax and AUC0–∞ were comparable for Japanese and Caucasian men (data not shown). While mean CL and Vdss were significantly lower in Japanese men compared with Caucasian men, they were equivalent across all treatment groups when normalized by body mass (CL: approximately 1.2 ml/h/kg; Vdss: approximately 0.07 l/kg). Dose-normalized Cmax and AUC0–∞ (Cmax/D and AUC0–∞/D) in Japanese and Caucasian men increased with increasing dose. Mean Vdss parameters differed among Caucasian dose groups; however, no dose-related differences in Vdss parameters were detected for Japanese men. A mean eritoran half-life of approximately 38 h was shorter for Caucasian men given 4 mg compared with higher doses with mean half-life of 51 and 46 h, while no dose-related differences were noted in Japanese men where mean half-life ranged from 44 to 48 h among dose levels (Table 3).
Pharmacokinetic parameters of eritoran metabolites in healthy Japanese and Caucasian men following eritoran infusion (28 mg).
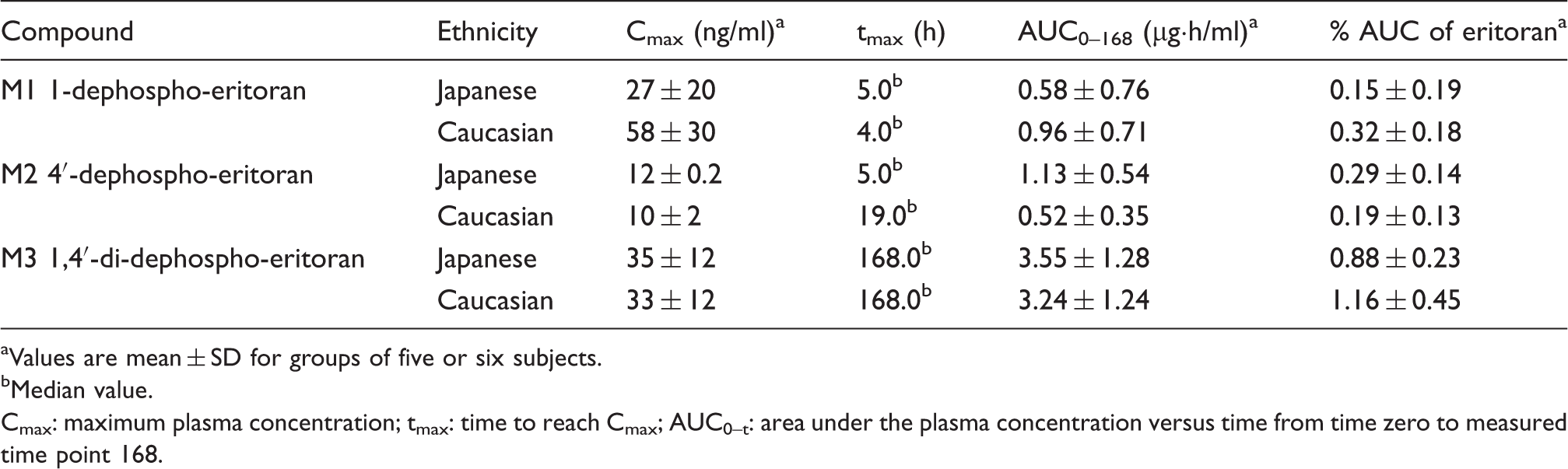
Values are mean ± SD for groups of five or six subjects.
Median value.
Cmax: maximum plasma concentration; tmax: time to reach Cmax; AUC0–t: area under the plasma concentration versus time from time zero to measured time point 168.
Plasma lipoprotein distribution of eritoran
Overall, recovery of eritoran in the plasma components ranged from 66.9% to 84.8% of total, with a trend for higher recovery in Japanese men. As shown in Table 4, the following plasma distribution of eritoran was observed in both Japanese and Caucasian men: 47.4% to 67.5% was recovered in the HDL fraction; 5.1% to 8.0% was recovered in the LDL fraction; 0.7% to 5.5% was recovered in the VLDL fraction; and 6.9% to 11.6% was recovered in the LPDP fraction.
Plasma distribution of eritoran and eritoran concentration by lipoprotein fraction for healthy Japanese and Caucasian men following eritoran infusion (28 mg).
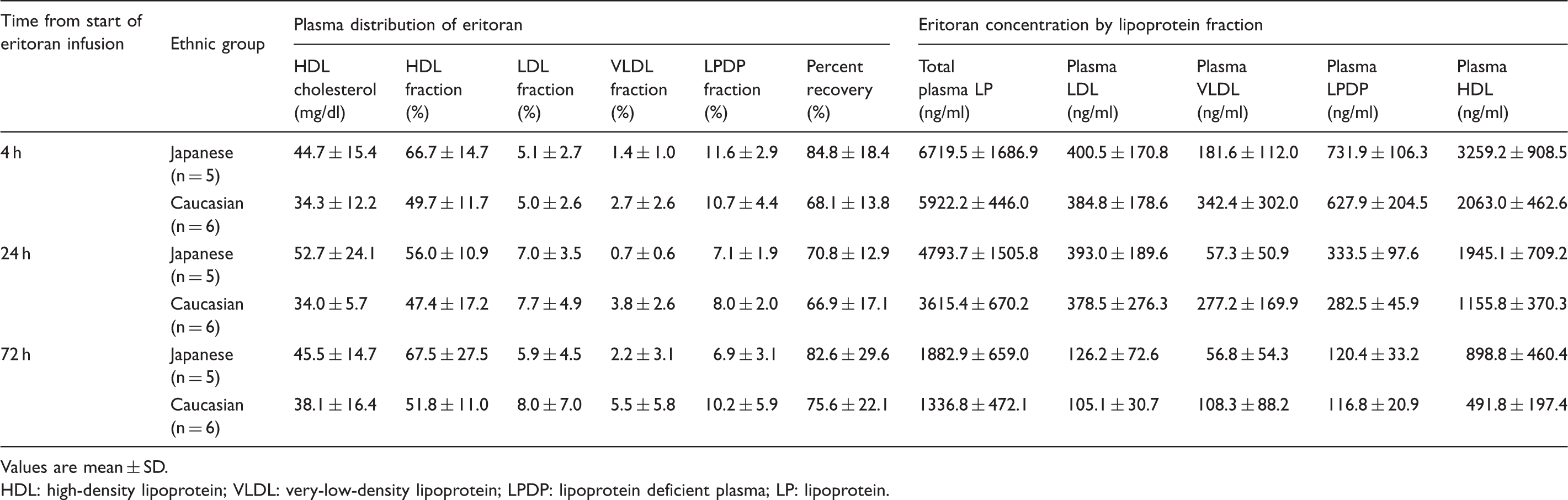
Values are mean ± SD.
HDL: high-density lipoprotein; VLDL: very-low-density lipoprotein; LPDP: lipoprotein deficient plasma; LP: lipoprotein.
Pharmacodynamics
The primary pharmacodynamic variable was the comparison of the ex vivo activity of eritoran at inhibiting endotoxin-stimulated TNF-α production in whole blood samples collected from patients before and following the 4-h infusion of eritoran 4, 12 or 28 mg or placebo. No differences in baseline endotoxin-stimulated TNF-α levels were observed between healthy male Caucasian and Japanese men. Results obtained with the 1 ng/ml endotoxin challenge dose indicated that inhibition of endotoxin-stimulated TNF-α release by eritoran was similar in Caucasian and Japanese men (Figure 3), and the endotoxin antagonistic activity of eritoran was greatest immediately after the end of the infusion of eritoran.
Inhibition of LPS (endotoxin)-induced (1 ng/ml) TNF-α concentrations versus time in healthy Caucasian and Japanese men administered eritoran (mean + SD). (A) Eritoran 4 mg (n = 6 per group). (B) Eritoran 12 mg (n = 6 per group). (C) Eritoran 28 mg (n = 6 Caucasians and n = 5 Japanese).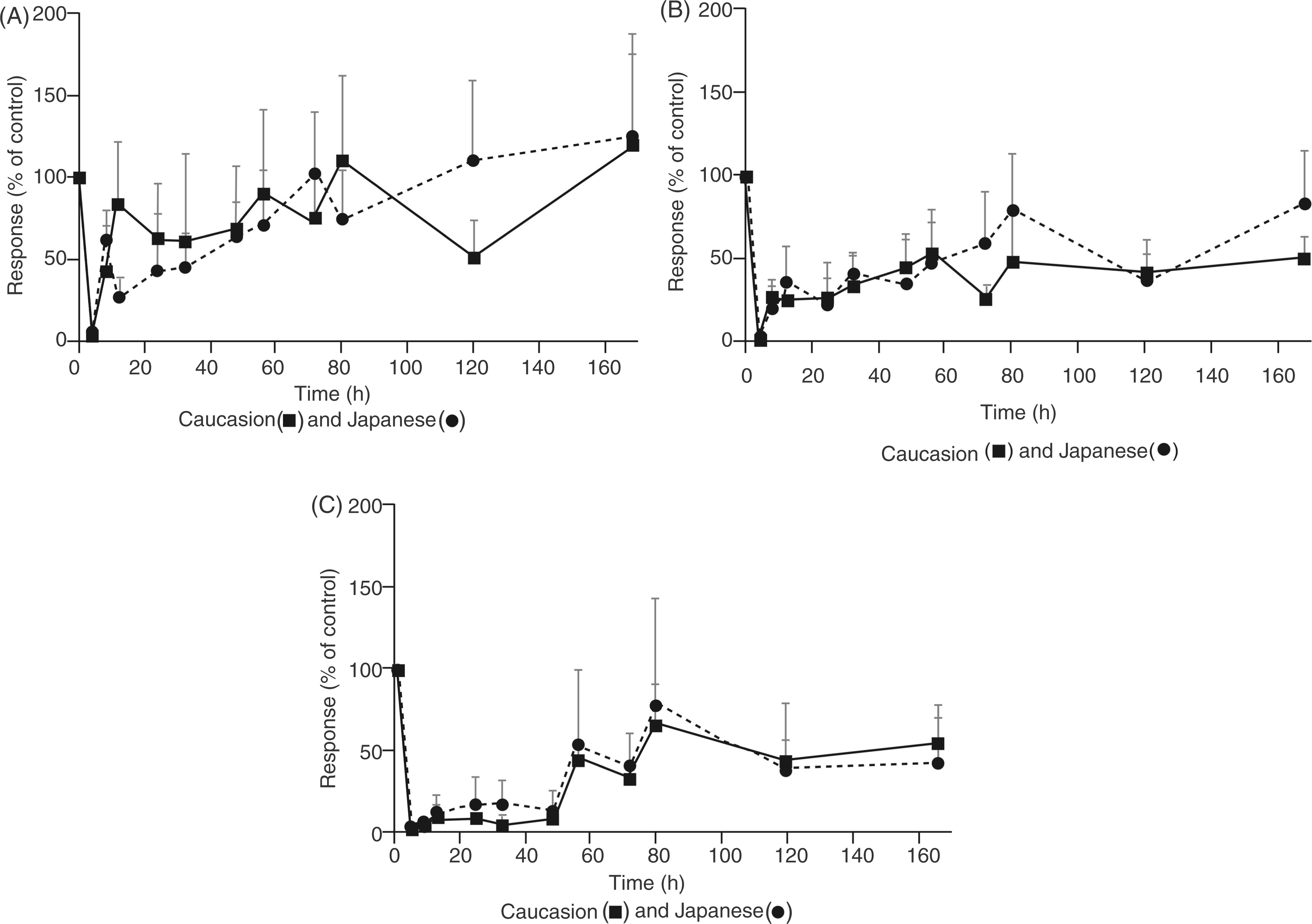
In an exploratory analysis, mean percent inhibition of endotoxin response was plotted as a function of the eritoran plasma concentration obtained at the same time point. Analysis was run without regard to dosing regimen and was based only on challenge dose of endotoxin and measured plasma concentration of eritoran versus percent inhibition of response. Thus, results from all eritoran dose levels (4, 12 and 28 mg) were pooled for this analysis. Analysis of the antagonistic activity of eritoran measured in plasma during the elimination phase demonstrated little or no differences between Caucasian and Japanese men (Figure 4). Figure 4 shows the plot for mean percent inhibition of 1 ng/ml endotoxin. Results using 0.05 ng/ml endotoxin and 10 ng/ml endotoxin were similar in that they did not reveal ethnic differences (data not shown).
Inhibition of mean change in endotoxin-induced TNF-α levels from baseline versus mean plasma eritoran concentrations: comparison of healthy Japanese and Caucasian men at 1 ng/ml endotoxin.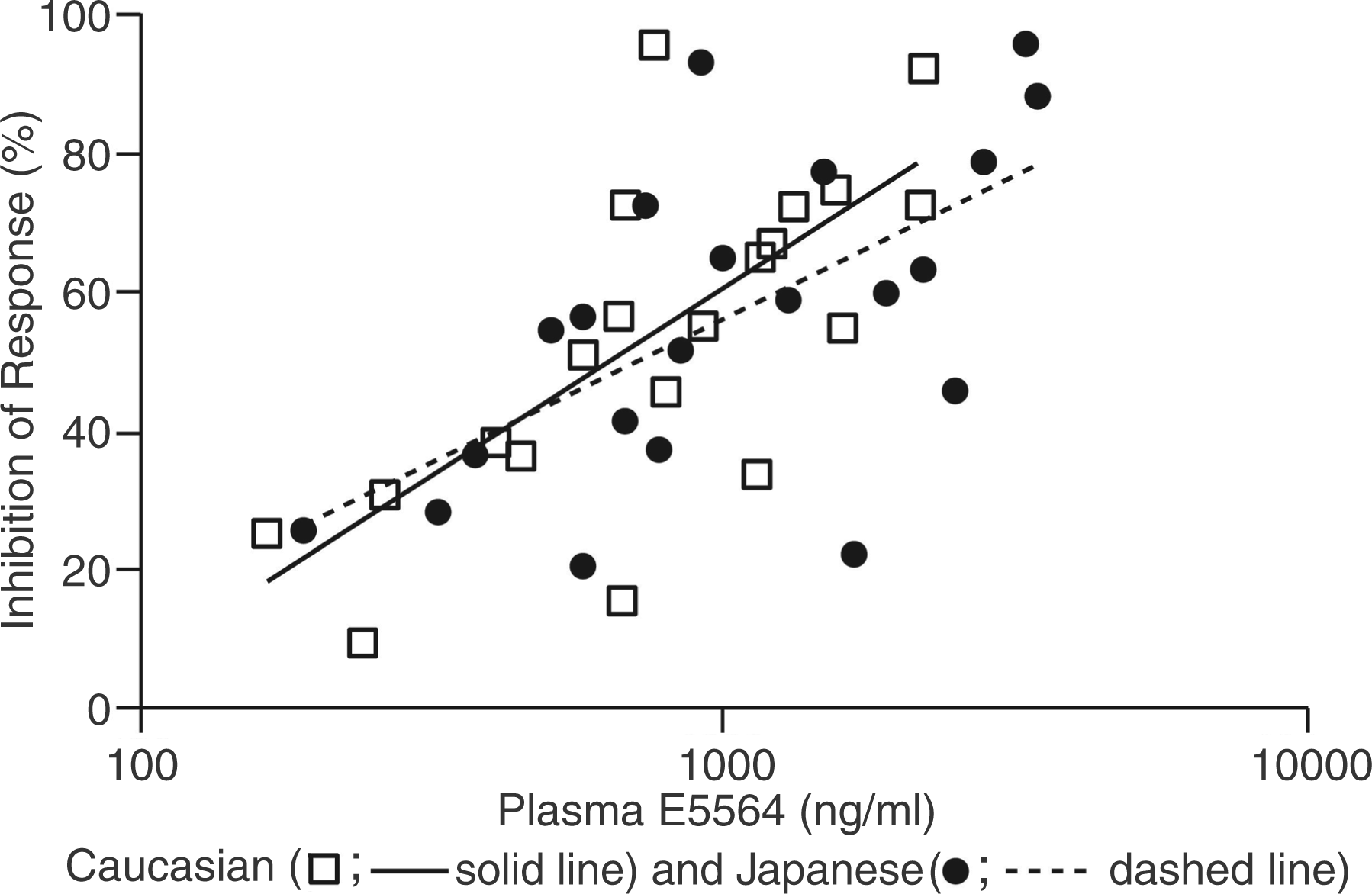
Discussion
These data provide additional evidence that eritoran, a TLR4 antagonist, is generally well tolerated in healthy patients and the pharmacokinetic disposition may be characterized by small Vdss and slow CL.6,7,11,18 Overall, a 4-h i.v. infusion of eritoran at total doses of 4, 12 and 28 mg in healthy Japanese and Caucasian men was generally safe and well tolerated.
The pharmacokinetic profile of eritoran observed in this study was consistent with that of previously reported studies (Table 3). Although in general, the pharmacokinetic profile of eritoran in Japanese men was similar to that of Caucasian men, some differences were observed. Eritoran plasma concentrations and total plasma exposure of eritoran were slightly higher, while CL and Vdss were lower in Japanese men compared with Caucasian men. However, it is difficult to relate the antagonistic activity of eritoran to plasma concentration alone, as the level of association with plasma lipoproteins (especially HDL) is likely to affect activity. In both Japanese and Caucasian men, eritoran was distributed mainly to the HDL fraction; Japanese men had increased levels of HDL with a concomitant increase in the amount of eritoran in the HDL fraction. However, pharmacodynamic analysis of eritoran measured during the elimination phase demonstrated little or no difference between Caucasian and Japanese men in the activity of eritoran as a function of plasma concentration (Figure 4). It is possible that the two effects observed by pharmacokinetic analysis—an increased Cmax in Japanese men and increased eritoran in the HDL compartment (which inactivates the drug)—may have counteracted one another to result in similar pharmacodynamic activity in the two populations.
Metabolism of eritoran appears resistant to minor changes in hepatic function or physiology, including ethnic differences. Hepatic impairment does not affect the pharmacokinetic profiles of eritoran. A comparison study with hepatically-impaired and healthy volunteers identified no difference in Cmax, AUC0–12, AUC0–last and AUC0–∞ of eritoran between the groups. 18 Studies that have identified differences between ethnic groups that are potentially significant for drug therapy most commonly implicate cytochrome P450 (CYP) enzymes, especially lower activity of CYP2D6 and CYP2C19 in an Asian population than in a Caucasian one. 10 These enzymes are not involved in eritoran metabolism as evidenced from in vitro hepatic enzyme studies, as well as mass balance and disposition studies in rats and dogs. 19 Instead, pre-clinical evaluation of eritoran metabolism and disposition demonstrated that dephosphorylated metabolites M1, M2 and M3 present in the plasma and liver of dogs and rats are slowly excreted predominantly in the feces. 19 Similar metabolic pathways have been reported for the lipid A moiety of endotoxin that is dephosphorylated at identical residues in vivo.19–21
This is the first report to characterize eritoran metabolites in humans. The gradual and prolonged increase in M3 identified in both Japanese and Caucasian men in this study is consistent with pre-clinical disposition and toxicity studies implicating hepatic macrophages or Kupffer cells in eritoran clearance. 19 Plasma profiles of M3 after single dosing indicate that extremely long time periods may be required to reach M3 steady state and that prolonged dosing may result in continuously increasing levels of the metabolite. Furthermore, these metabolites are only poorly soluble in water, and metabolites M1 and M2 are estimated to be 17–230-fold less active than eritoran, while M3 is inactive (data not shown).
Extrapolation of these study results to sepsis patients may be limited. This study examined healthy patients rather than patients with sepsis and there may be differences in safety, pharmacokinetics or pharmacodynamics in patients with sepsis. 22 Importantly, HDL is known to be qualitatively and quantitatively altered in sepsis. 23
Although 16 men in groups 2 and 4 received the incorrect infusion rate of eritoran and 1 of the mis-dosed men re-enrolled in group 3, these men were excluded from all analyses and did not affect the final results and interpretation. In addition, lipoprotein fractions were mistakenly stored for up to 85 d at 4°C prior to quantification of eritoran. This extended storage exceeded the time period (69 h) during which eritoran was shown to be stable in lipoprotein factions at this temperature. However, when eritoran recovery was determined based on the total amounts found in each lipoprotein/protein fraction and compared with the known amounts in the plasma fraction before lipoprotein separation, very little degradation was observed. In addition, it is important to note that the lipoprotein fractions from Caucasian and Japanese men were stored under similar conditions until analysis. Therefore, the relative comparison of the eritoran concentrations in the lipoprotein fractions from the two ethnic groups should remain valid.
In conclusion, eritoran was found to be generally safe and well tolerated in healthy Japanese and Caucasian volunteers. Safety and pharmacokinetic/pharmacodynamic profiles did not appear to indicate that a dose adjustment for differing ethnic variables is required for adult Japanese or Caucasian patients within normal ranges of body mass. Eritoran exposure parameters increased with increasing dose administered as i.v. infusions. Metabolic conversion to dephosphorylated metabolites was low, based on relative plasma exposures of M1, M2 and M3 measured to eritoran in plasma. Although pharmacokinetic evaluation of eritoran and its major metabolites after a single infusion identified some differences between Japanese and Caucasian men, these did not translate into differences in pharmacodynamics. Inhibition of endotoxin by eritoran was equivalent in healthy Japanese and Caucasian volunteers.
Footnotes
Funding
Editorial assistance was provided by Phase Five Communications Inc. and was financially supported by the Eisai Corporation of America. Dr N. Aikawa received financial support from Eisai Co., Ltd to publish the results of this study; Dr O. Sivak and Dr K.M. Wasan received financial support for lipoprotein analyses for this study. All other authors are employees of Eisai.
