Abstract
The pharmacokinetics of antimigraine drugs zolmitriptan and sumatriptan varied considerably with a fourfold to 10-fold variation in plasma levels. In addition, the pharmacodynamics of triptans as investigated in vitro also varied considerably. In theory, there should probably be a 10-fold variation in doses available, but in clinical practice a fourfold difference in doses will probably cover the needs of most patients.
Introduction
It is well known that migraine patients, like any other, vary in their response to drugs concerning both efficacy and tolerability. Thus, the dose required in therapy can vary among patients and within patients from migraine attack to migraine attack. This variabily is in most cases due to pharmacokinetic variability, pharmacodynamic variability and variability of the migraine attack treated.
Pharmacokinetic variability may cause some of the interindividual variability, especially in drugs with a low oral bioavailability such as sumatriptan (14%) (1) and in situations where there is delayed emptying of the stomach, as has been observed repeatedly during migraine attacks (2–5). Generally, pharmacokinetic variability has been better investigated because it is easier to measure than pharmacodynamic variability (6).
Pharmacokinetic variability in migraine
In one study a gender difference was found for the oral bioavailability of zolmitriptan with 60% in female and 38% in male healthy volunteers (7). There was a large between-subject variability in kinetic parameters (7). Thus, the C max was 16 ± 8 (SD) in males and 25 ± 9 in females (both n = 6) (7). In a study in which migraine patients were investigated for pharmacokinetic parameters both during and outside migraine attacks the plasma zolmitriptan levels varied considerably among subjects (4). Thus, during migraine attacks the 2-h plasma levels after 10 mg zolmitriptan orally varied from 0 to 15 ng/ml, whereas outside attacks the 2-h plasma levels varied from 3 to 27 ng/ml (4). Similarly, the C max after an oral dose of 200 mg sumatriptan varied from 52 to 227 ng/ml in healthy volunteers (8).
There is thus a considerable between-subject variability in pharmacokinetic parameters. In addition, there is the general influence of the migraine attack with delayed gastric emptying and resulting delayed absorption during attacks (2–5). If the triptans were administered parenterally, the variability would probably be less.
Possible pharmacodynamic variability in migraine
One way of studying dynamic variability is in isolated vascular tissues under well-defined conditions, such as using the myograph technique. When the results of these in vitro studies are presented it is normally with means and SEM (see Fig. 1) and from this way of presentation it is not obvious that there is often a considerable dynamic variability among the different tissues (see Fig. 2).

The effect of eletriptan, 10−11−10−5
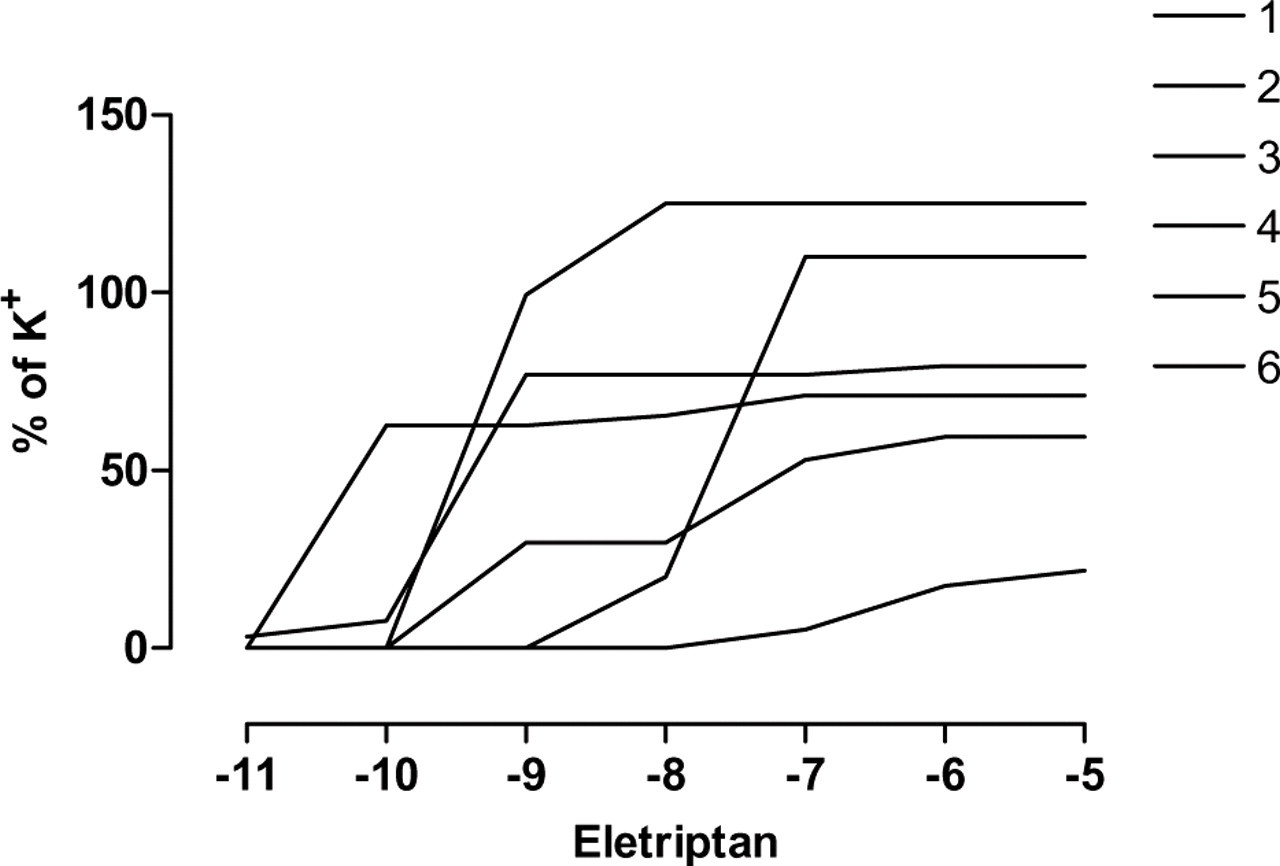
The effect of eletriptan 10−11−10−5 M on six human cerebral arteries in vitro. From (13).
Previously, we have examined the variability for the naturally occurring receptor agonists, noradrenaline and 5-hydroxytryptamine(5-HT) (9–12). Variability for the EC50 for 5-HT on the temporal artery varied 30-fold (8) and the EC50 for the effect of noradrenaline in omental arteries varied 10-fold (11).
The variability of the vasoconstrictor effect on human cerebral arteries of the 5-HT1B/1D receptor agonists sumatriptan, rizatriptan and eletriptan was recently investigated (13). For sumatriptan the EC50 varied 51-fold, for rizatriptan 21-fold and for eletriptan 69-fold (13). The variability of triptans is thus in the same range as that for the naturally occurring receptor agonists.
The dose–response curves for the natural agonists and the triptans in vitro are constructed with a 10 000-fold variation in agonist concentrations. What of the dose–response curve for the vascular effect of triptans in man? The mean results show that the C max/EC50 ratio for the triptans is not different from 1 (13), indicating that with the usual C max obtained in man one is near the 50% of the vascular effect of the triptans sumatriptan, rizatriptan, zolmitriptan and eletriptan.
The best described dose–response curve in man, although in relatively small groups of patients, is that for subcutaneous naratriptan (see Fig. 3) (14). With a 10-fold increase in dose a 29% increase in headache relief from 65% (0.5 mg) to 94% (5 mg) was observed. This is a mean curve for the response to the different doses and considerable variability can underlie such a mean curve. The mere fact, however, that a dose–response curve can be obtained for subcutaneous naratriptan despite the possible pharmacodynamic variability (see above) demonstrates that there is a dose–response curve for the antimigraine effect.
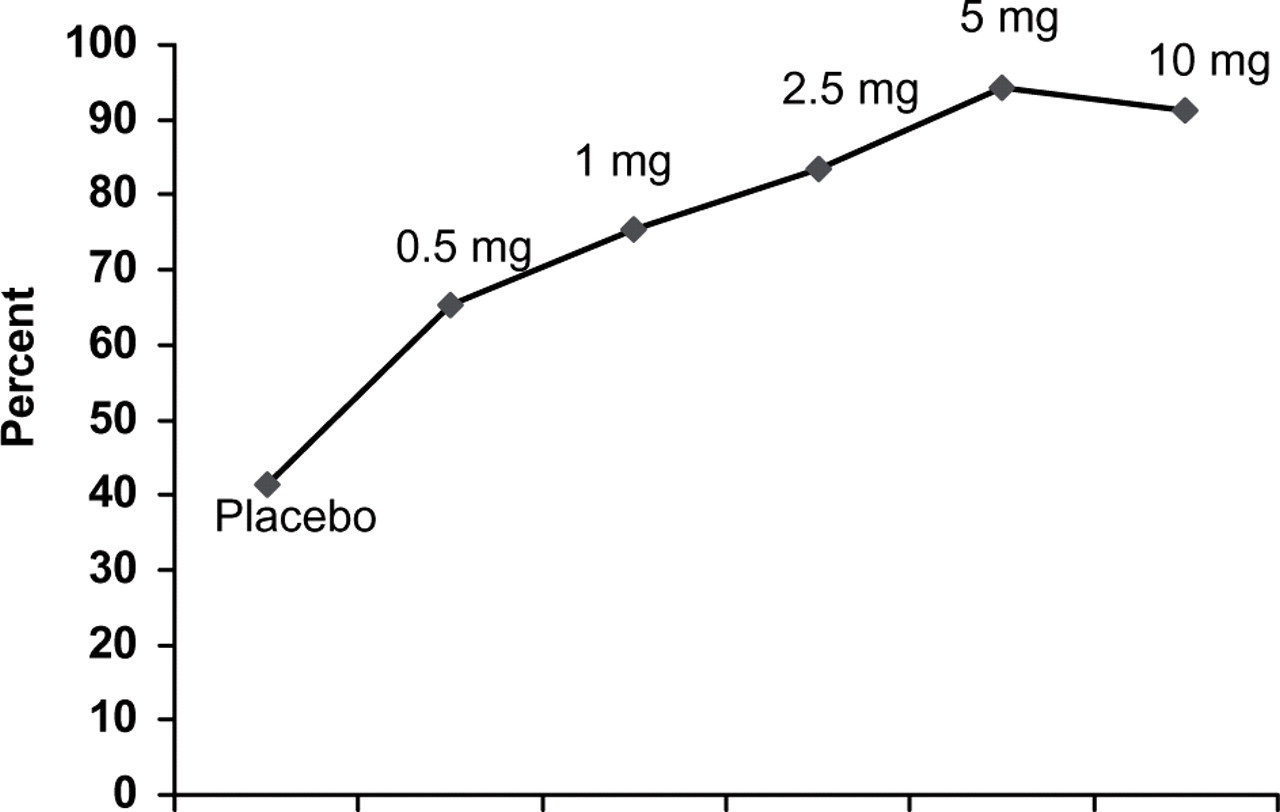
Headache relief after subcutaneous naratriptan, 0.5–5 mg, in one randomized, placebo-controlled, double-blind study (n = 34–60). From (14).
Possible clinical implications
In addition to the inherent variability of the migraine attack, there is thus a sound, both pharmacokinetic and pharmacodynamic, basis for the clinical experience that the patients' need for antimigraine drugs varies considerably. In theory, there should probably be a 10-fold variation in doses available, but in clinical practice a fourfold difference in doses will probably cover the needs of most patients.
This shows that changing doses in patients and possibly triptan, with differences in lipid solubility and bioavailability, can benefit individual patients.
