Abstract
Mast cells (MCs) control allergic reactions and contribute to protective innate immune responses through TLR4 activation. The tyrosine kinase Lyn is important to the high affinity IgE receptor (FcεRI) signal transduction system in MCs, but its role on the TLR4 signalling cascade is still elusive. Here, we characterized several TLR4-triggered responses in bone marrow-derived mast cells (BMMCs) from wild-type (WT) and Lyn−/− mice. We found that Lyn−/− MCs secreted lower amounts of TNF-α after LPS challenge when compared with WT cells. Lyn−/− BMMCs showed less MAPK, IκB phosphorylation and NF-κB nuclear translocation after TLR-4 triggering than WT cells. LPS-induced MAPK and inhibitor of IκB kinase (IKK) phosphorylation were importantly reduced in the absence of Lyn. A constitutive interaction between TNF receptor associated factor 6 (TRAF-6) and phosphorylated TGF-β-activated kinase (TAK-1) was observed in Lyn−/− BMMCs and this complex was insensitive to LPS addition. Lyn kinase was activated and associated to TRAF-6 shortly after LPS addition in WT MCs. Analyzing two local MC-dependent innate immune responses in vivo, we found that Lyn positively controls early TNF-α production and immune cell recruitment after an intraperitoneal injection of LPS. Our results indicate that Lyn plays a positive role in TLR4-induced production of TNF-α in MCs controlling the activity of the TRAF-6/TAK-1 protein complex.
Introduction
Mast cells (MCs) are important regulatory elements in the immune system that elicit different responses in both innate and adaptive immunity. 1 These cells have been widely studied because of their participation on IgE-dependent reactions (type I hypersensitivity). 2 In those circumstances, the high affinity IgE receptor (FcεRI) is activated by IgE/Ag complexes and leads to the secretion of preformed and newly synthesized inflammatory mediators. Besides their role in IgE-dependent reactions, MCs are also important for innate immune responses as they can initiate inflammatory reactions against pathogen-associated molecular patterns (PAMPs). 3 TLR4 is expressed on MC-plasma membranes and after activation with Gram-negative bacteria-derived LPS, induces the secretion of multiple pro-inflammatory cytokines, such as TNF-α, IL-1β, IL-6 and several chemokines. 4 The similarity of the effects of FcεRI and TLR4 triggering on cytokine production (although apparently not on degranulation) in MCs has led to the investigation of key common molecules regulating cytokine production after IgE/Ag or LPS stimulation of MCs.
The TLR4 signal transduction system comprises distinct adapters, serine-threonine kinases and ubiquitin ligases that couple receptor activation mainly to NF-κB and AP-1-dependent gene transcription. In myeloid cells, MyD88 and MyD88-adaptor like (Mal) proteins are able to associate to the intracellular tail of TLR4 through a Toll/IL-1R (TIR)-mediated interaction in the so-called MyD88-dependent pathway. TIR-mediated adaptor association induces the recruitment of serine/threonine IL-1R-associated kinases (IRAK)-1 and IRAK-4 to the membrane-associated signalling complex which results in the activation of the E3 ubiquitin ligase TNF receptor-associated factor (TRAF)-6. The complex formed by TRAF-6 and the proteins UBC13 and UEV1A induce K63-dependent ubiquitination on TAK-1 binding protein (TAB)-2, which results in TGF-β-activated kinase (TAK)-1 autophosphorylation. This latter molecular complex and the reactions of ubiquitination and phosphorylation of TAK-1 lead to the important step of MAPK and inhibitor of IκB kinase (IKK) pathway activation. 5 IKK activity controls the phosphorylation/degradation of IκB and the nuclear translocation of NF-κB, whereas the MAPK pathway activates AP-1, which also contributes to the expression of NF-κB-induced pro-inflammatory genes.
On the other side, in the MyD88-independent pathway, TIR-domain containing adapter-inducing interferon-β (TRIF) and TRIF-related adaptor molecule (TRAM) are mainly involved in the internalization of the TLR4 complex after LPS recognition6,7 and induce the interferon regulatory factor (IRF)3/7-dependent type I IFN expression. This arm of the TLR4 signalling pathway is involved in IFN synthesis and constitutes a complementary cascade leading to pro-inflammatory cytokine gene expression mediated by NF-κB activity in myeloid cells, 5 although recent evidence suggests that this pathway is not active in MCs. 8
A number of studies indicate that the main steps of TLR4 activation encompass homotypical interactions, phosphorylation on serine/threonine residues and K63-mediated ubiquitination on distinct substrates. However, some evidence suggests that tyrosine phosphorylation also can be important in modulating TLR4 signalling. In particular, the Src-family kinase Lyn has been involved in TLR4-induced NF-κB activation. 9 Different studies have found that Lyn increases its autophosphorylation and associates with PI3K after LPS treatment in different cell types, 9 – 12 and it has been reported that Lyn associates with CD14 in human monocytes 12 or with TLR4 (when both molecules are over-expressed 9 ) in a LPS-dependent fashion. Mainly recognized for the positive 13 and negative13,14 regulation on FcεRI signalling, the participation of this important regulatory kinase in the TLR4 signal transduction system in MCs is far from being elucidated.
In the present study, we analyzed the effect of the absence of Lyn kinase on TLR-4-triggered responses in MCs. We utilized bone marrow-derived mast cells (BMMCs) from WT and Lyn−/− mice to analyze LPS-induced TNF-α secretion, detecting the activation of key molecules downstream of the TLR4 receptor, such as TRAF-6, TAK-1, MAPK, IKK and NF-κB. In addition, we utilized an in vivo murine model of endotoxaemia in normal and MC-deficient (Kitw/Kitw-sh) animals to analyze the role of Lyn kinase in the canonical MC-dependent inflammatory responses of early TNF-α production and cell recruitment triggered by LPS in peritoneal cavity.
Materials and methods
Mice
Wild-type, Lyn−/− 15 and Kitw/Kitw-sh 16 mice were purchased from Jackson Laboratories (Bar Harbor, ME,USA). As Lyn−/− mice were originally in the 129SvImJ, they were back-crossed with C57BL/6J WT mice (also from Jackson Laboratories), at least five times, to obtain Lyn−/− mice with the genetic background of C57BL/6J. 17 The genotype of each mouse was analyzed by PCR utilizing the protocol suggested by the provider. MC-deficient Kitw/Kitw-sh mice were subjected to intraperitoneal MC reconstitution (described below). All the procedures using mice were approved by the Institutional Committee for Use and Care of Laboratory Animals in Cinvestav (CICUAL, protocols 032/02 and 384/07).
Cell culture
MCs were derived from WT and Lyn−/− C57BL/6J mice aged 4–8 weeks old, as reported
18
. Briefly, mice were euthanized and the femurs were extracted to obtain bone marrow progenitors by flushing them with complete BMMC media (see below) using a syringe with a 30 G × 13 mm needle. Complete BMMC media was composed of RPMI supplemented with 2 mM
Flow cytometry
WT and Lyn−/− BMMCs were incubated with or without IgE during 1 h. After that, cells were washed twice with staining buffer (BSA 2%, sodium azide 0.02% in PBS) and pellets were re-suspended in blocking buffer 1X (from Biogenex, San Ramon, CA, USA) for 5 min at 4°C. Then, FITC-coupled anti-IgE or anti-TLR4 Abs were added. In parallel, anti-isotype Abs were used. One hour after Ab incubation, cells were centrifuged and pellets were re-suspended in staining buffer to be analyzed in a Flow Cytometer FACSort (BD Biosciences, San Jose, CA, USA).
LPS stimulation of BMMCs, Western blotting and immunoprecipitation
WT and Lyn−/− MCs (two million cells in 1 ml) were treated with LPS (500 ng/ml) or a mixture of phorbol 12-myristate-acetate and a calcium ionophore, PMA/A23187 (1 µm/100 nM) at 37°C for different times in complete BMMC culture media. Cells were collected by centrifugation and supernatants were taken for TNF-α determination by ELISA (see following sections). For Western blot, two million cells were re-suspended in 1 ml of Tyrodés-BSA buffer (20 mM HEPES buffer at pH 7.4, 135 mM NaCl, 5 mM KCl, 1.8 mM CaCl2, 1 mM MgCl2, 5.6 mM Glc and 0.05% BSA) and stimulated with LPS (500 ng/ml). After proper times, cells were collected and lysed in Laemmli buffer 2X supplemented with 4 mM ortho-vanadate and 0.28 mM β-ME. For immunoprecipitation assays, 10 million cells per condition, in a total volume of 200 µl of Tyrode’s/BSA were utilized. All samples were stimulated with LPS and centrifuged at 800 × g at 4°C before being re-suspended in a lysis buffer containing 1% NP-40, 60 mM octyl-β-glucoside, 2 mM phenylmethanesulphonylfluoride (PMSF), 10 µg/ml aprotinin, 2 µg/ml leupeptin, 5 mM pepstatin, 5 mM sodium pyrophosphate, 50 mM NaF and 1 mM sodium orthovanadate for 30 min on ice. Lysates were centrifuged for 10 min (4°C) at 15000 g and supernatants were separated from pellets and kept on ice for 1 h before use. Supernatants were incubated overnight with specific Abs pre-bound to Sepharose beads-coupled to G or A protein. After immunoprecipitation, beads were washed once with 1% NP40-containig buffer and once with 0.1% NP-40-containing lysis buffer. Proteins were recovered with an equal volume of Laemmli 2X buffer and separated in 8%, 10% or 12% SDS-PAGE gels. They were then electrophoretically transferred to polyvinylidene fluoride (PVDF) membranes and blocked with either 4% dry milk and/or immunoglobulins cocktail in Tris-buffered saline containing 0.1% Tween-20. Membranes were then probed with the desired primary Ab and an adequate secondary HRP-conjugated Ab before being visualized by chemiluminesence.
Electrophoretic mobility shift assay
NF-κB activation was measured obtaining nuclear extracts from WT and Lyn−/− BMMCs. Briefly, 15 × 106 BMMC were activated with LPS or IgE/Ag for 20 min. After this time, cells were collected and nuclear extraction was performed using the protocol provided with the TransAm Transcription Factor Kit (Active Motif, Carlsbad, CA, USA), supplementing the hypotonic buffer (HB) with leupeptin (10 mg/ml), pepstatin A (5 mg/ml) and aprotinin 4.7 (U/mg prot). Electrophoretic mobility shift assays (EMSAs) were performed using the LightShift EMSA optimization and Control kit (Pierce, Rockford, IL, USA) according the manufacturer’s instructions. The NF-κB biotinylated primers sequences (Invitrogen, Carlsbad, CA, USA) 19 were as follows: sense 5′ biotin-CAA-ATG-TGG-GAT-TTT-CCCP-ATG-AGT-CTC-AAA-ATT-AGA-GAG 3′ and antisense 5′ biotin-CTC-TCT-AAT-TTT-GAG-ACT-CAT-GGG-AAA-ATC-CCA-CAT-TTG 3′. The samples were resolved in a 6% acrylamide/bisacrylamide gel and the bands were developed using the Chemiluminescent Nucleic Acid Detection Module (Pierce), following the instructions provided.
Intraperitoneal reconstitution of Kitw/Kitwsh mice
MC-deficient mice Kitw/Kitw-sh (8–12 weeks old) were selectively reconstituted by an intraperitoneal injection with 2 × 106 BMMCs from 4-week-old WT and Lyn−/− BMMC cultures. In all adoptive transfer assays, cells were concentrated in a total volume of 200 µl of sterile isotonic saline solution and were injected in the peritoneal cavity of mice, as described. 20 Four weeks after injection of BMMCs, mice were used for experiments. Reconstitution of MCs was confirmed by flow cytometric analysis, toluidine blue stain of MCs obtained by peritoneal washes and TNF-α production after LPS triggering, as reported. 21
Murine model of endotoxaemia and peritoneal washes
WT, Lyn−/−, Kitw/Kitw-sh and Kitw/Kitw-sh mice reconstituted with WT or Lyn−/− BMMCs were injected i.p. with LPS (1 mg/kg dissolved in saline solution) or control saline solution. 20 One hour later, peritoneal lavages were performed by euthanizing mice and injecting 2.5 ml PBS (137 mM NaCl, 2.7 mM KCl, 10 mM NaH2PO4, 2 mM K2HPO4, pH 7.4) into the peritoneal cavity. After a gentle massage of the abdominal surface, at least 1 ml of peritoneal lavage was collected from each mouse. Another group of animals were injected i.p. with LPS (1 mg/kg) as described above, and five hours later mice were euthanized and peritoneal lavages were performed as described. Samples were taken to calculate the total number of migrating cells into peritoneal lavages using the Neubauer chamber and, in parallel, total cells were labeled with an Ab against the neutrophil-specific marker, Ly6G, 22 and analyzed by flow cytometry.
ELISA
TNF-α concentration in supernatant of stimulated cells or peritoneal lavages was determined with TNF-α ELISA kits from Peprotech (Rocky Hill, NJ, USA) and Invitrogen. For cell supernatants, two million WT and Lyn−/− BMMCs were incubated with 500–1000 ng/ml LPS for different times in complete cell culture media. Cells were then collected by centrifugation and supernatants were subjected to ELISA analysis. Alternatively, TNF-α secretion of peritoneal washes from WT, Lyn−/−, MC-deficient or MC-deficient reconstituted mice, stimulated or not with LPS, were centrifuged at 350 g for 5 min and supernatants were collected to determine TNF-α by ELISA. All ELISA determinations were performed according to the manufacturer’s instructions with the standard curve provided.
Abs and reagents
Polyclonal Abs against TLR4, Lyn, TRAF-6, pTAK-1, IκB, p65, JNK, p38, ERK1/2, MEK3/6 and ubiquitin were from Santa Cruz Biotechnology (Santa Cruz, CA, USA). FITC-conjugated monoclonal rat anti-mouse IgE, FITC-conjugated rat IgE1 κ monoclonal immunoglobulin isotype standard were from BD Pharmingen (San Diego, CA, USA). Pacific blue-labelled monoclonal anti mouse Ly6G antibody was from Biolegend (San Diego, CA, USA). HRP-conjugated extravidin and monoclonal Abs against mouse and rabbit immunoglobulins were from Amersham Pharmacia Biotech (Little Chalfont, UK). Cell culture medium (RPMI) and BMMC complete media components were from Gibco-BRL-Life Technologies (Gaithersburg, MD, USA). Laemmli buffer, octyl-β-glucoside, pepstatin, leupeptin and aprotinin, 2-(ME), DNP coupled to human seroalbumin (DNP-HSA), monoclonal anti DNP IgE (clone SPE7), A23187, phorbol myristate acetate (PMA) and Escherichia coli LPS (serotype 026:B6) were from Sigma (St Louis, MO, USA). Murine IL-3 were from Peprotech. NP-40 was from Pierce. PVDF was from Perkin Elmer (Boston, MA, USA).
Statistical analysis
Results are expressed as the mean ± S.E.M. of at least three independent experiments performed with distinct cell cultures. Software Prism V4.0c was utilized for the analysis of the data.
Results
TLR4-induced TNF-α secretion is impaired in Lyn−/− BMMCs
With the aim of evaluating the role of Lyn kinase on MC responsiveness to LPS we first determined the expression of TLR4 on the plasma membrane of WT and Lyn−/− BMMCs to discard any possible differences attributable to the amount of TLR4 on the surface of both cell types. WT and Lyn−/− cells expressed equivalent quantities of TLR4 receptor on plasma membranes (Figure 1A). TNF-α secretion following TLR4 triggering was then determined in a time-course experiment. WT and Lyn−/− BMMCs were stimulated with LPS (500 ng/ml) and cell supernatants were collected to determine TNF-α by ELISA (Figure 1B, left panel). TNF-α in the supernatant of WT and Lyn−/− cells reached maximal values after 8–10 h of LPS addition; however, peak levels of TNF-α were lower in Lyn−/− than in WT BMMCs (306 ± 21 pg/ml in WT vs 190 ± 26 pg/ml in Lyn−/−). This deficiency observed in the absence of Lyn was more evident after four hours of stimulation. In order to discard any defect on the secretory machinery of Lyn−/− cells, TNF-α was quantified in the supernatant of cells stimulated with the calcium ionophore A23187 plus the protein kinase C (PKC) activator phorbol mirystate acetate (PMA; Figure 1B, right panel). In those conditions, it was possible to observe that, although Lyn−/− BMMCs secrete slightly less TNF-α in the absence of stimulus, 4 h post-stimulation, both cell types were able to secrete equivalent maximal levels of TNF-α (approx. 1100 pg/million cells).
Lyn positively regulates LPS-induced TNF-α secretion in MCs. (A) Two million WT and Lyn−/− mature BMMCs were labeled with specific, FITC-coupled anti-TLR4 Ab. Isotype control is presented in the left histogram of each graph, while the specific TLR4 signal can be observed in the right histogram of each graph. A representative experiment of at least three performed with different cell cultures is shown. (B) WT and Lyn−/− BMMCs were incubated in the presence of 500 ng/ml LPS (left panel) or A23187/PMA (right panel) as described in the ‘Material and methods’. After distinct times of stimulation, supernatants were collected and TNF-α concentration was determined by ELISA. (C) WT and Lyn−/− BMMCs were stimulated with LPS (500 ng/ml) in the absence or presence of actinomycin D (5 µg/ml added 15 min before LPS). TNF-α concentration in the supernatant was determined by ELISA. Data in (B) and (C) are presented as the mean ± SEM from 3–5 different cultures. *P < 0.05 with respect to non-stimulated cells; &P < 0.01 with respect to WT cells.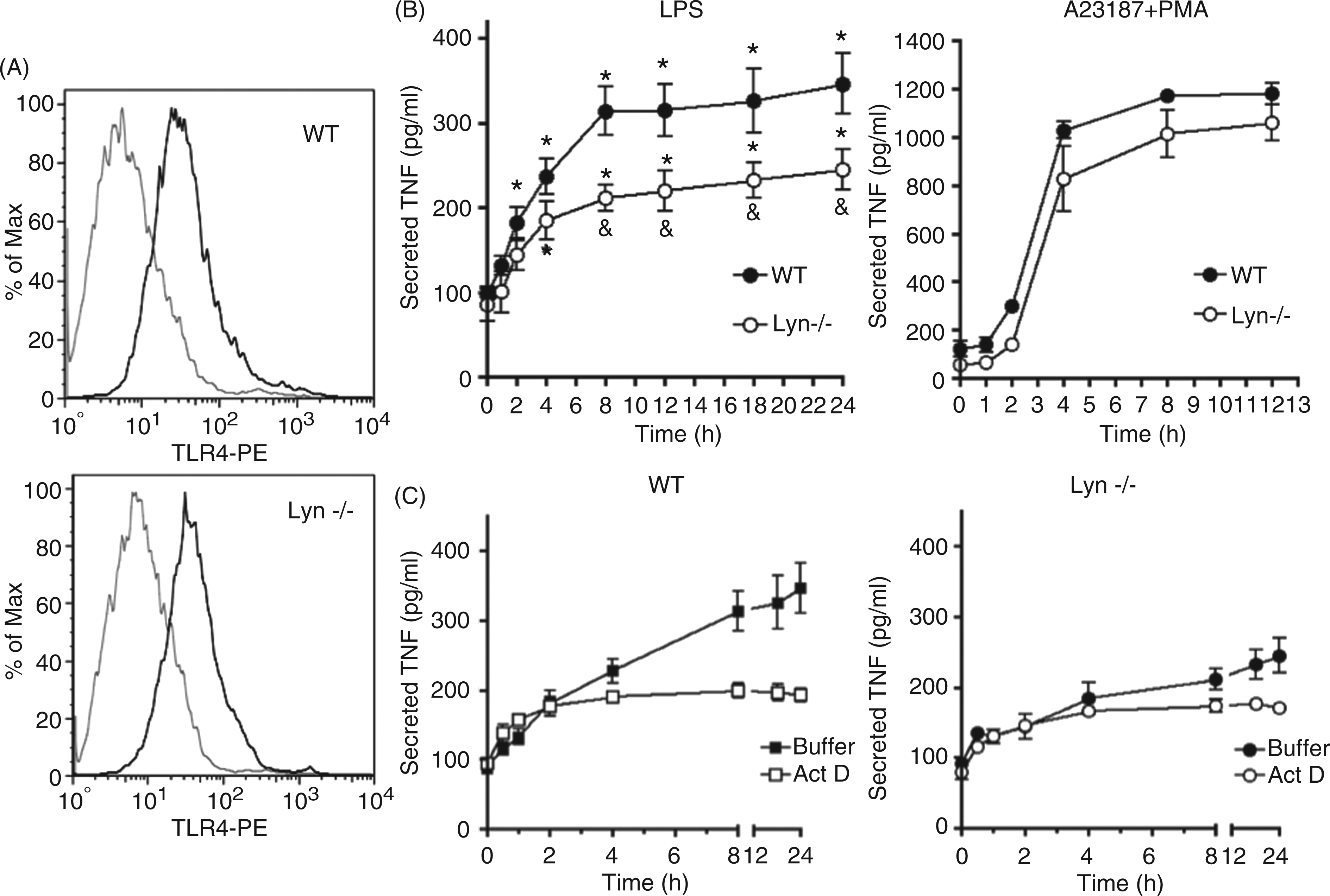
As MCs can secrete TNF-α from pre-formed stores but also can initiate de novo TNF-α synthesis after proper stimulation, 23 we decided to measure LPS-induced TNF-α secretion in the presence of the transcription inhibitor actinomycin D to locate the step on TNF-α production that was affected in Lyn−/− BMMCs. As can be observed in Figure 1C (left panel), in WT BMMCs, an early actinomycin-insensitive TNF-α release was observed after LPS addition and the inhibitor blocked only late TNF-α secretion (that occurred after 4–8 h) in WT cells. In contrast, a lower amount of pre-stored TNF-α was secreted in Lyn−/− cells and the release of de novo synthesized TNF-α obtained after 4–8 h after stimulation was significantly diminished (Figure 1C, right panel).
Defective MAPK pathway activation after LPS treatment is observed in the absence of Lyn
In different myeloid cell lineages, a functional role for NF-κB on TNF-α gene transcription has been clearly established24,25 and, after LPS treatment, an important AP-1 and NF-κB-dependent transcriptional activity leading to TNF-α synthesis is observed in human and murine immune cells.24,26 As this main mechanism of TNF-α regulation is conserved in MCs activated with distinct ligands,
27
WT and Lyn−/− BMMCs were stimulated with LPS for different times and, first, MAPK activation was analyzed. As can be observed in Figure 2A, LPS treatment was able to induce the phosphorylation of MEK3/6 in WT, but not in Lyn−/− BMMCs, where a slightly higher basal phosphorylation of MEK3/6 was observed in some experiments; this increase was not statistically significant (Figure 2B). Accordingly, an important increase on the phosphorylation of the c-Jun N-terminal kinase (JNK), ERK1/2 and p38 MAP kinases was observed in WT, but not in Lyn−/− BMMCs treated with LPS (Figure 2C). Specifically, LPS-induced p38 and JNK phosphorylation was not detected in the absence of Lyn and a lower ERK phosphorylation was observed in Lyn−/− cells compared with WT. As it has been reported that LPS is able to aggregate several receptors on cell surface besides TLR4,28,29 a control experiment stimulating the cells with LPS in the presence of an inhibitory antibody against TLR4 was performed. In those conditions, no LPS-dependent ERK phosphorylation was detected, suggesting that in our cellular model, LPS selectively activates to TLR4 (results not shown).
Lyn positively controls LPS-dependent MAPK activation in MCs. WT and Lyn−/− BMMCs were stimulated with LPS (500 ng/ml) and, at each time, cells were collected and lysed in Laemmli buffer 2X as described in the ‘Materials and methods’. Western blots were performed to detect pMEK 3/6 (A). (B) Densitometric analysis of pMEK signal in WT and Lyn−/− cells. Data are shown as the mean ± SEM from at least three experiments performed with different cell cultures. *P < 0.05. (C) Phosphorylation of ERK, JNK and p38 after LPS (500 ng/ml) stimulation of WT and Lyn−/− BMMCs. A representative experiment, of at least four performed with different cell cultures, is shown.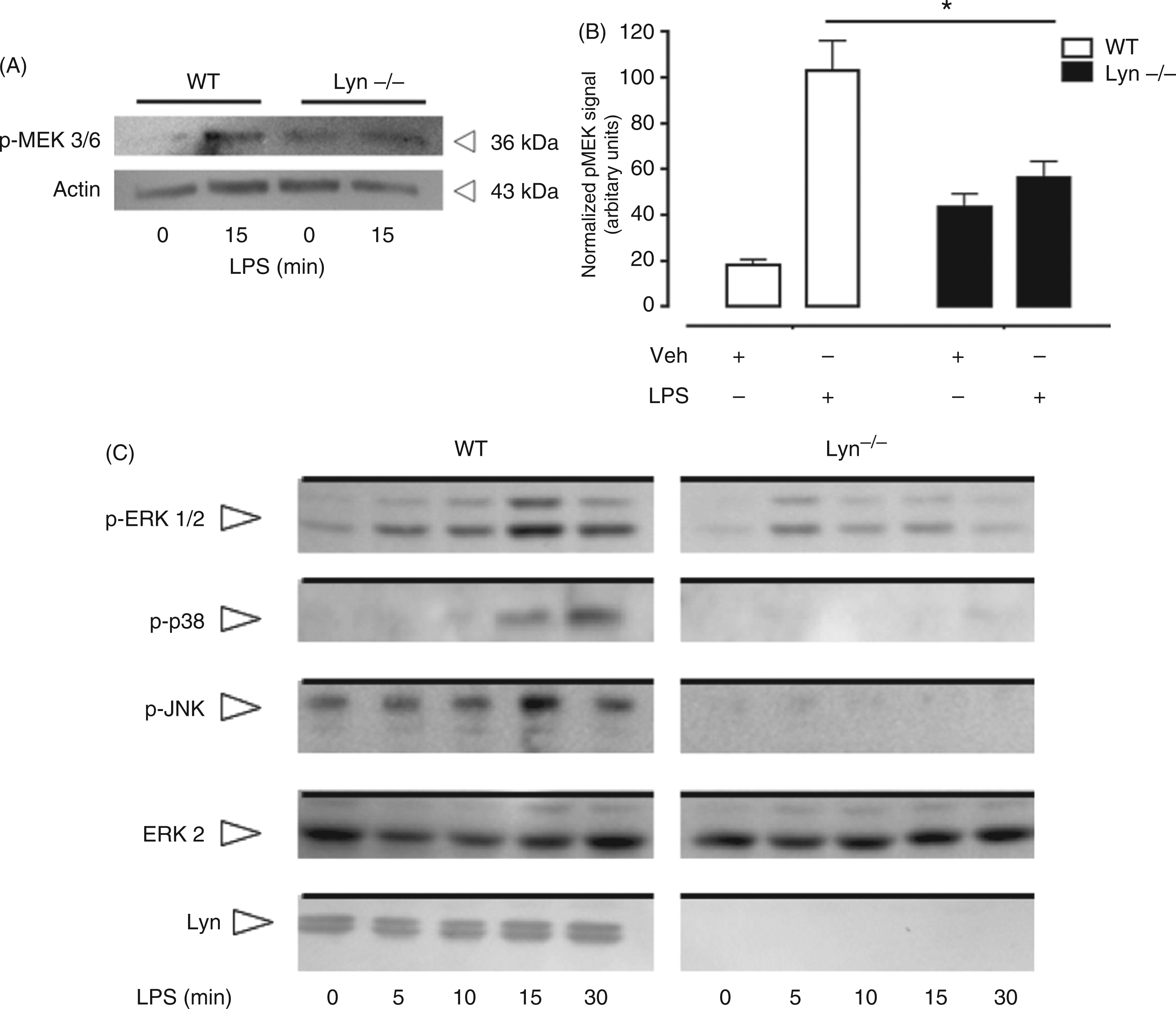
Impaired IKK-NF-κB pathway activation after TLR4 triggering is observed in Lyn−/− BMMCs
To ascertain the role of Lyn on the activation of the IKK-NF-κB pathway, WT and Lyn−/− BMMCs were stimulated with LPS and IKK, IκB and NF-κB activation was analyzed. Figure 3A shows that IKK was phosphorylated in the Thr180/181 residues in response to LPS in WT but not in Lyn−/− BMMCs. Accordingly, in WT MCs, IκB phosphorylation was increased at 15 min after LPS addition—this state was sustained for at least 30 min after stimulation (Figure 3B). Despite that, IκB maximal phosphorylation was severely affected in Lyn−/− BMMCs, IκB phosphorylation was detected as soon as 5 min after LPS addition in this cell type (Figure 3B). To analyze the role of Lyn kinase on NF-κB activity, WT and Lyn−/− BMMCs were treated with LPS and EMSA assays utilizing canonical κB binding sites were performed (Figure 3C). LPS-induced NF-κB (p50p65) binding activity was detected at 15 min of stimulation in WT cells; this response was maintained after 30–60 min of LPS treatment. In contrast, Lyn−/− BMMCs showed a severely diminished response of p50p65 binding activity in response to LPS. NF-κB binding activity after 30 min of IgE/antigen-dependent FcεRI crosslinking was determined as a positive control.
Lyn kinase regulates LPS-activated NF-κB activity in MCs. WT and Lyn−/− BMMCs were activated with 500 ng/ml LPS and, at the indicated times, cells were isolated and lysed to analyze IKK αβ (A) and IκB (B) phosphorylation by Western blot. (C) EMSA essays performed with nuclear extracts from 10 million WT and Lyn−/− BMMCs stimulated with LPS, as described in the ‘Material and methods’. A representative experiment (from four independent assays performed with distinct cellular cultures) is shown. Specific p50/p65 complex migration was previously determined utilizing specific Abs (data not shown). Positive controls of NF-κB activation were obtained by stimulating cells with IgE/Ag.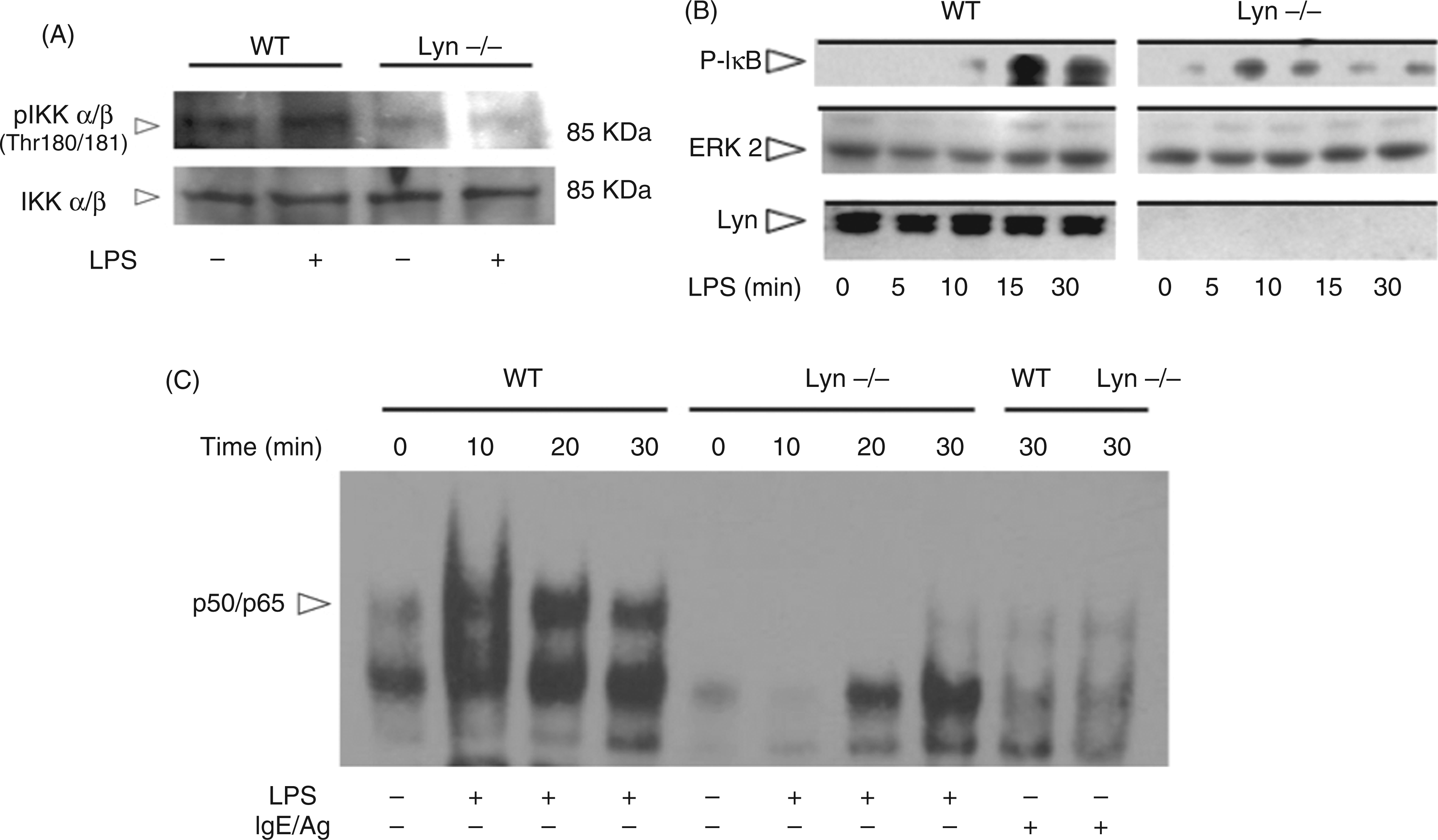
Increased basal TRAF-6/TAK-1 association and TAK-1 phosphorylation in Lyn−/− BMMCs
As defects on MAPK and IKK activation were detected in Lyn−/− BMMCs, we decided to explore the participation of Lyn kinase on the signalling events upstream of IKK phosphorylation. The complex formed by UBC13/UEV1A/TAB2/TRAF-6 and TAK-1 has been involved in the important step of TLR4-dependent MEK and IKK phosphorylation in distinct myeloid immune cells.30,31 Co-immunoprecipitation experiments were performed to detect the interaction between TAK-1 and TRAF-6 in WT and Lyn−/− cells. Figure 4A shows that the association between those proteins increased in WT cells after LPS addition. In contrast, in Lyn−/− BMMCs, TRAF-6/TAK-1 association was detected in basal conditions and, interestingly, it did not increase after LPS stimulation. TRAF-6-associated TAK-1 was found to be phosphorylated in the regulatory Thr 184/187 residues
32
in WT cells after LPS stimulation, but, in the absence of Lyn, TAK-1 was phosphorylated—even in basal conditions—and this state did not change with short or long-lasting treatments with LPS (Figure 4B).
Lyn kinase modulates TLR4-induced formation and function of the TRAF-6/TAK-1 protein complex in MCs. Thirty million WT and Lyn−/− BMMCs (per time point) were stimulated with LPS (500 ng/ml) and, after distinct times, cells were collected by centrifugation and re-suspended in the lysis buffer, as described in the ‘Materials and methods’. Cell extracts were incubated with sepharose/protein A spheres coupled to Abs recognizing TAK-1 (A) or TRAF-6 (B, C). Protein samples were separated in acrylamide gels and Western blot was performed using anti TRAF-6 (A), phospho-TAK-1 (p-Thr 184/187) (B) or ubiquitin (C). A representative experiment (from at least four independent assays utilizing different cell cultures) is shown.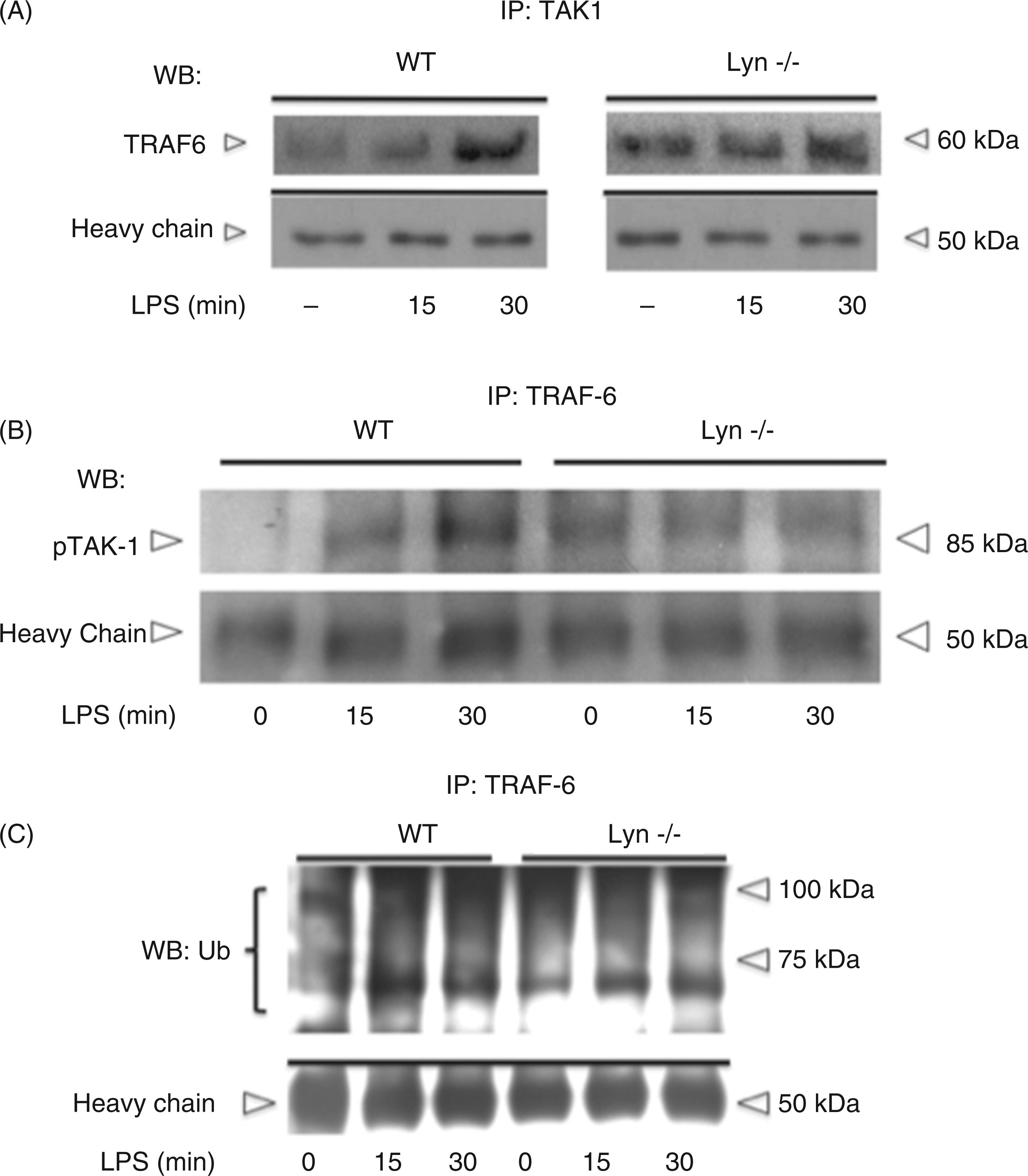
As it has been shown that TAK-1 phosphorylation depends on the ubiquitin ligase activity of TRAF-6, WT and Lyn−/− were stimulated with LPS and TRAF-6 was immunoprecipitated to detect its ubiquitination profile. Figure 4C shows that an increase on the signal of the anti-ubiquitin Ab occurred 15 min after LPS stimulation of WT BMMCs and it was sustained to 30 min after challenge. In Lyn−/− BMMCs, the change on TRAF-6 ubiquitination profile after LPS addition was slightly lower than in WT cells and did not vary with the treatment of LPS.
Lyn kinase is activated and associates with TRAF-6 after LPS stimulation
Lyn activation was evaluated after TLR4 triggering in BMMCs. WT cells were stimulated with LPS at different times, Lyn kinase was immunoprecipitated with specific anti-Lyn Abs and a Western blot was performed utilizing an Ab raised against phosphorylated Tyr 416 of Src kinase. This residue is located in the catalytic domain of Src and it has been widely utilized as a marker of kinase activity of that protein.
33
We used this Ab as that Tyr residue is conserved among all the members of the Src-family kinases. Figure 5A shows that phosphorylation of the canonical Tyr residue located in the catalytic domain of Lyn was detected 5 min after TLR4 triggering and was still observed at 30 min after LPS addition in WT cells. This result indicates that Lyn kinase is rapidly activated after TLR4 triggering in BMMCs.
Lyn is activated and associates to TRAF-6 in response to LPS in MCs. (A) Lyn kinase was immunoprecipitated from LPS-treated WT BMMCs, as described in the ‘Materials and methods’ and a Western blot utilizing an Ab recognizing the conserved phosphorylated Tyr 416 residue of Src was performed. (B) TRAF-6 was immunoprecipitated from LPS-treated WT and Lyn−/− BMMCs and a Western blot against Lyn kinase was performed. A sample of whole cell lysate (WCL) was included as a control for the Western blot. A representative experiment from five independent assays utilizing different cell cultures is shown.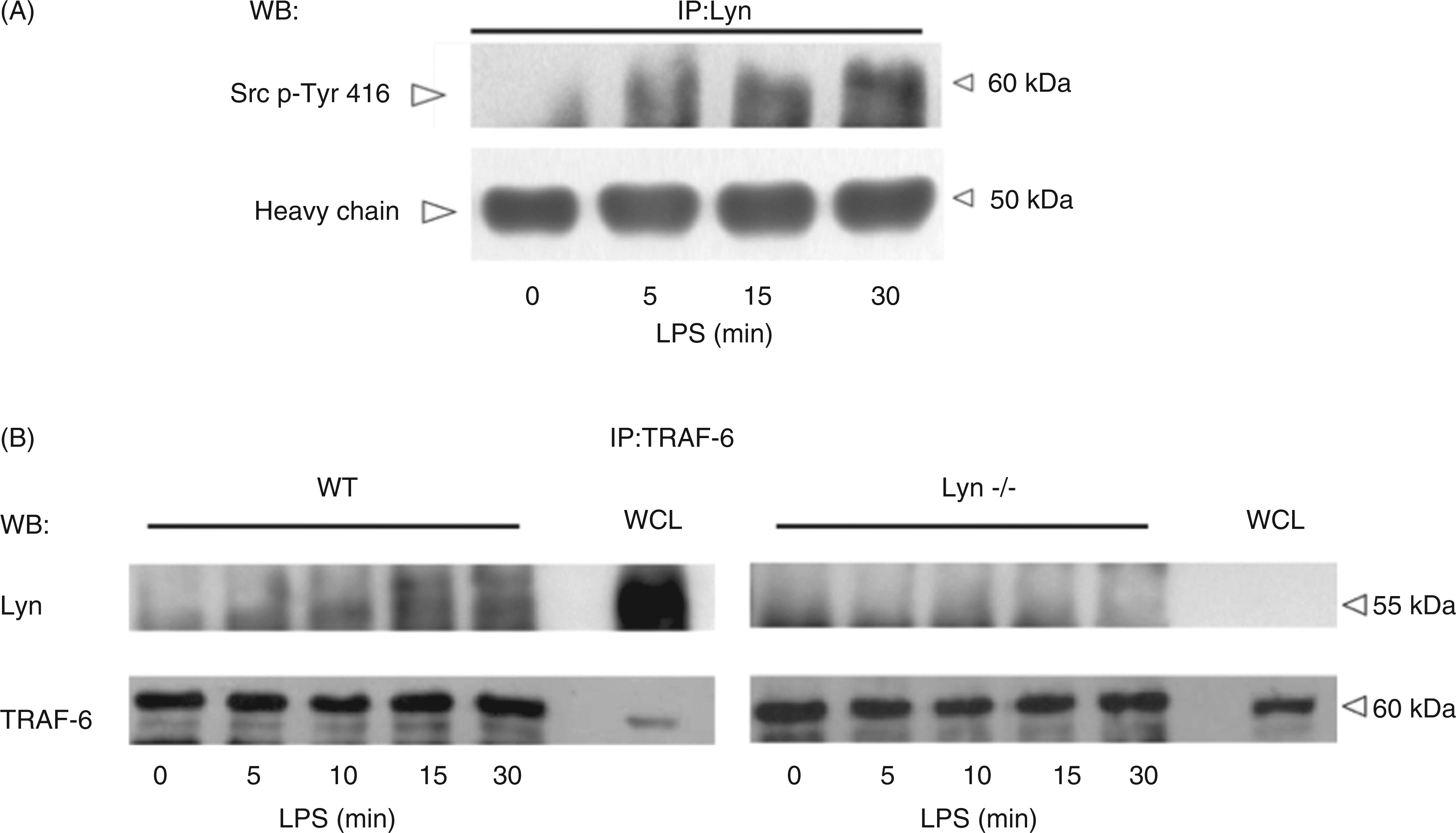
The participation of Lyn kinase in the conformation of the TRAF-6/TAK-1 protein complex was evaluated by exploring a direct interaction between TRAF-6 and Lyn. As can be observed in Figure 5B, Lyn kinase co-immunoprecipitated with TRAF-6 in WT but not in Lyn−/− BMMCs after LPS treatment. Maximal TRAF-6-Lyn co-immunoprecipitation was observed 30 min after TLR4 triggering in WT BMMCs.
Lyn kinase is an important element in MC-dependent innate immune responses in vivo
Utilizing different paradigms of infection with Gram-negative bacteria and the cecal ligation and puncture model, it has been demonstrated that MCs are responsible for the early release of TNF-α in the peritoneal cavity and also for the recruitment of neutrophils to the site of inoculation, shaping the local inflammatory reaction.4,20,21,34–
36
MC-dependent TNF-α production was found to be protective against low intensity peritoneal infections.
21
In order to evaluate the role of Lyn on MC TLR4 signaling in vivo, we injected mice i.p. with LPS and determined two canonical MC-mediated responses: the early synthesis of TNF-α20,34,35 and the neutrophil recruitment to the peritoneum after challenge.
35
–
38
WT, Lyn−/−, MC-deficient (Kitw/Kitw-sh, Wsh) and MC-reconstituted (Wsh Rec) mice were injected i.p. with LPS (1 mg/kg) and, after 1 h, peritoneal lavages were obtained in order to determine TNF-α concentration by ELISA. Figure 6A shows that, as reported,20,34 WT mice were able to secrete TNF-α early after an i.p. LPS injection (400 ± 70 pg/p.w). However, Lyn−/− mice did not secrete intraperitoneal TNF-α in the same conditions. MC-deficient Wsh mice showed the described defect on TNF-α production; reconstitution of those animals with WT MCs was able to restore early TNF-α secretion in response to LPS. When Kitw/Kitw-sh mice were reconstituted with Lyn−/− BMMCs, one-hour levels of TNF-α were not different from non-reconstituted animals. Equal amounts of TNF-α were secreted when a mixture of the calcium ionophore A23187 plus the PKC activator PMA was injected in animals reconstituted with WT or Lyn−/− cells (data not shown). Figure 6B shows that i.p. recruitment of Ly6G-positive cells after LPS injection was affected in the absence of Lyn. WT or Lyn−/− mice were injected with LPS (1 mg/kg) and 5 h later, peritoneal washes were performed and Ly6G
22
positive cells in those washes were evaluated by flow cytometry. The number of recruited Ly6G-positive cells per peritoneal wash was found to be close to 200 × 103 in WT animals stimulated with LPS, whereas recruited cells were only around 50 × 103 in LPS-stimulated Lyn−/−.
Lyn kinase positively controls early MC-dependent TNF-α secretion induced by LPS in the peritoneal cavity. (A) Twelve-week-old WT, Lyn−/− and MC-deficient Kit
W
/Kit
Wsh
(Wsh) mice were injected i.p. with LPS (1 mg/kg) or saline solution and 1 later, peritoneal lavages were collected to determine TNF-α secretion by ELISA, as described in the ‘Materials and methods’. MC-deficient mice were reconstituted with BMMCs derived from WT or Lyn−/− animals (Wsh Rec WT or Lyn−/− respectively). Four weeks after reconstitution, LPS challenge (1 mg/kg) was administered and peritoneal lavages were obtained after 1 h. Results are presented as the mean ± SEM of at least six animals.*P < 0.001 with respect to saline solution injection in the respective group. (B) WT and Lyn−/− mice were stimulated i.p. with LPS (1 mg/Kkg) and 5 h later, peritoneal lavages were performed to determine Ly6G positive neutrophils in each p.w. by flow cytometry, as described in the ‘Materials and methods’. Results are presented as the mean ± SEM of at least three animals. *P < 0.05 with respect to indicated values.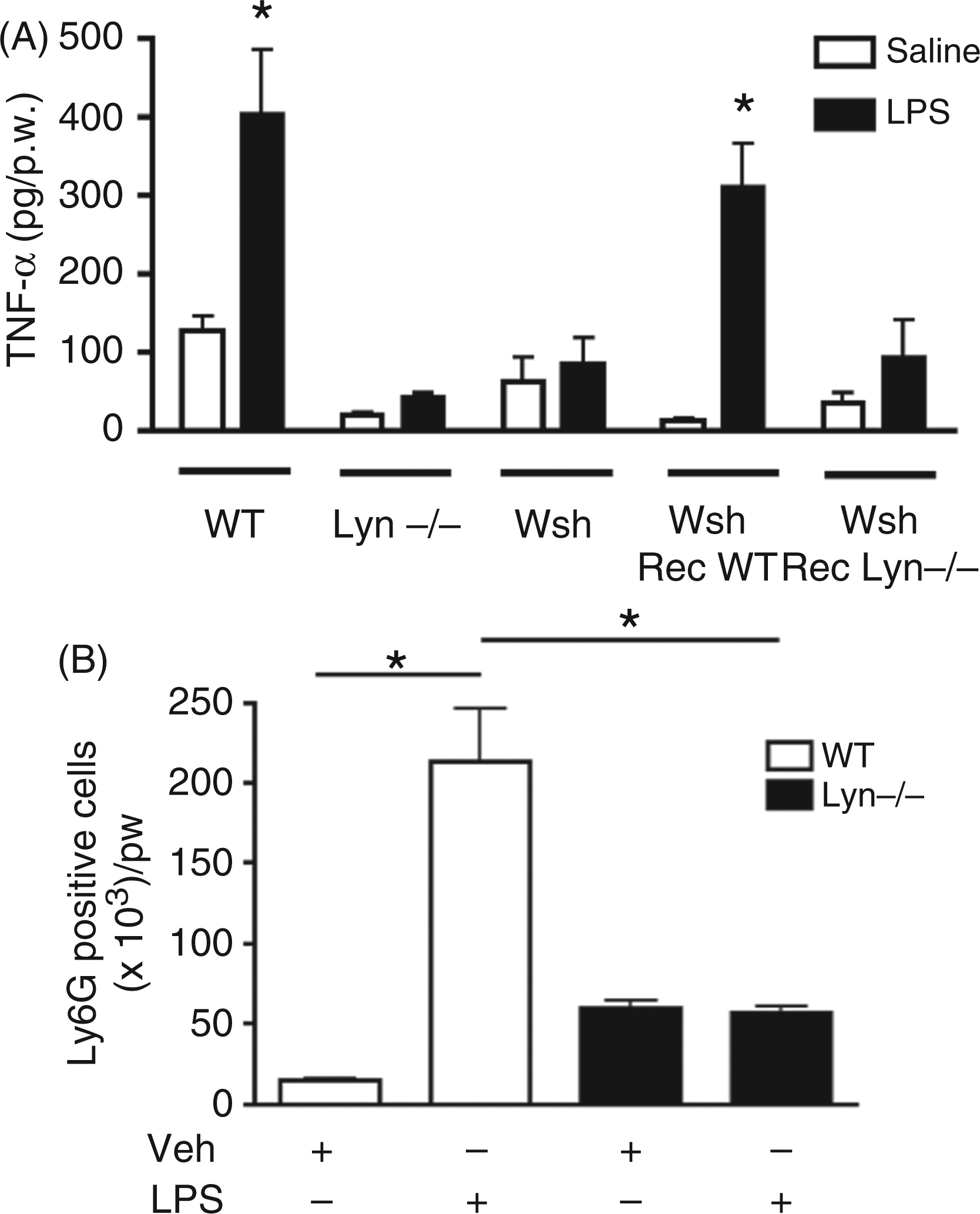
Discussion
In this work we report that protein kinase Lyn plays a positive role in TLR4-induced production of TNF-α controlling the activity of the TRAF-6/TAK-1 protein complex in MCs. Our finding is quite relevant as, to our knowledge, this is the first time that the participation of a tyrosine kinase in TLR4 signalling system and an interaction between a Src-family kinase and a TRAF-family member are reported in this important cell type.
The TLR4 signal transduction system in MCs has been explored because of their importance on distinct innate and adaptive immune responses. 39 The pathway leading to TNF-α production after TLR4 activation has been characterized using different MC models, i.e. utilizing the immortalized rat cell line RBL-2H3 transfected with different dominant-negative mutant proteins, it has been shown that LPS-induced TNF-α production requires the participation of MyD88 and TRAF6 proteins, 40 and, by utilizing BMMCs, it was shown that LPS induces a rapid and intense IκB phosphorylation. 4 Each cellular model has given specific data about the complex response of MCs against LPS; however, as RBL-2H3 cells do not express adequate levels of MyD88 and CD14 co-receptor,41,42 we decided to utilize BMMCs to further analyze the signalling cascade of TLR4 and, specifically, the role of the regulatory kinase Lyn on TLR4-dependent cytokine production in MCs.
Pre-formed and neo-synthesized TNF-α production by MCs occurs after IgE/Ag stimulation. 23 Transcription-dependent (actinomycin D-sensitive) synthesis of TNF-α has been associated with long-term secretion involved in extended regulation of immune reactions, whereas early (actinomycin D-insensitive) secretion of granule-stored TNF-α seems to be involved in acute protective responses. 23 As in the case of FcεRI-dependent stimulation, we found that TLR4-triggering induces the release of pre-stored TNF-α and also the long-term synthesis of the newly produced cytokine. Although we cannot discard a role of Lyn on the secretion of pre-formed TNF-α, the results obtained utilizing actinomycin D strongly suggest Lyn to have a role mainly in the production of the transcription-dependent TNF-α in BMMCs.
When upstream regulators of AP-1 and NF-κB activity were analyzed in LPS-activated cells, we observed an important impairment of the events responsible for MAPK and IKK activation in Lyn−/− MCs. IKK was not phosphorylated, MEK 3/6 was found to be unresponsive to TLR4 triggering and ERK, JNK and p38 MAP kinases were not phosphorylated in the absence of functional Lyn. As TAK-1 has been shown to phosphorylate MEK3/6 43 and IKK, a possible defect on the activation of TAK-1 was explored; we found a constitutive, LPS-independent association between TRAF-6 and TAK-1 in the absence of Lyn.
In TLR4-dependent signalling, the formation of a molecular complex composed of UBC13/UEV1A/TAB2/TRAF-6 and TAK-1 is a fundamental step in bifurcating the signal to the IKK/NF-κB and the MAPK/AP-1 activation pathways. The molecular basis of the interaction between TRAF-6 and TAK-1 has been explored in distinct cellular models. Recently, it has been proposed that an activating mechanism for TAK-1 in which K63-mediated TAB2 ubiquitination (induced by the TRAF-6/UBC13/UEV1A complex) leads to the autophosphorylation of TAK-1 on residues 187/184, resulting in TAK-1 activation 32 and dissociation of the complex. Both K63-mediated TRAF-6 ubiquitination and phosphorylation of TAK-1 on threonine residues 184 and 187 are considered as critical steps for TAK-1 activation; 32 biochemical data indicate that TAK-1 phosphorylation occurs on the membrane; however, active TAK-1 is found only in the cytosol. 44 Our results indicate that Lyn modulates the TRAF-6/TAK-1 engagement/dissociation cycle, as an increased association between those proteins was detected in Lyn−/− BMMCs and no change after LPS treatment was observed in that cell type. Accordingly, with Lyn having a role in the activity of the complex, increased phosphorylation of TAK-1 associated with TRAF-6 was detected, but a defective MAPK and IKK activity was observed in Lyn−/− cells. The paradoxical increase on TAK-1 phosphorylation and the lower activity of downstream elements observed in the absence of Lyn could be explained if TAK-1 dissociation from the membrane TRAF-6-containing complex was required for downstream element activation and Lyn was necessary for active TAK-1 dissociation from the TRAF-6-containing complex. Accordingly, we observed, in some experiments, an increase in basal MEK3/6 phosphorylation and it is possible to speculate that, under our experimental conditions, MEK3/6 could form a part of the TAK-1-containing complex at the plasma membrane and that this complex should be dissociated in a Lyn-dependent manner to allow the signal to proceed. In fact, a p-MEK3/6 positive signal can be observed by Western blot in LPS-induced TRAF-6/TAK-1-containing aggregates (results not shown).
However, an indirect role of Lyn in the phosphorylation of accessory residues on TAK-1 could be possible, as it has been reported that other phosphorylation sites (i.e. positions 178 and 192) are also important for TAK-1 activation. 45 Using transfected 293 cells with distinct TAK-1 constructs, it has been shown that both Thr residues 178 and 184 can regulate AP-1 and NF-κB activation pathways. 45 As we did not evaluate the phosphorylation of all the residues possibly involved in TAK-1 activity, or the association of Lyn with an intermediate Ser-Thr kinase able to phosphorylate them, we cannot involve Lyn kinase in the induction of full TAK-1 activity. Although our results suggest a positive role of Lyn on TAK-1-dependent downstream signalling, a discrete, but detectable, increase on IκB phosphorylation was observed as early as 5 min post-LPS stimulation in Lyn−/− cells, suggesting that, probably, TAK-1 constitutive phosphorylation in the absence of Lyn leaks to continuous, but low-level, activation of NF-κB.
The detailed mechanism by which Lyn could modulate the activity of the TRAF-6/TAK-1 complex remains to be further analyzed, although some possibilities can be hypothesized based on recently reported functions and interactions of that kinase. Thymocyte-expressed molecule involved in selection 2 (Themis2) has been found to be a signalling scaffold that participates in the TLR4 signal transduction system. 46 Depletion of Themis 2 protein inhibited LPS-induced TNF-α production in human macrophages. Unexpectedly, Themis 2 was shown to be tyrosine-phosphorylated after LPS challenge and interacted with Lyn kinase, the Rho guanine nucleotide exchange factor, Vav and the adapter protein Grb2. 46 Interestingly, LPS-induced ERK and p38 was enhanced in cells over-expressing Themis 2, although no change on JNK or p65 activity was detected under those conditions. Analysis of Lyn-dependent phosphorylation of Themis 2 and the consequences of this event on TRAF-6/TAK1 complex function remain to be evaluated.
On the other hand, in MCs, Lyn kinase has been found to associate with, and activate, sphingosine kinase type I (Sphk1), leading to the production of sphingosine 1 phosphate (S1P), which is necessary for initial calcium released from intracellular stores leading to degranulation. 47 In different cell lines, Sphk1 and the production of S1P was found to be necessary for Lys-63-linked polyubiquitination of receptor-interacting protein 1 (RIP1), phosphorylation of IKK, IκB degradation and NF-κB activation after TNF-α receptor triggering. Interestingly, it was found that S1P specifically binds TRAF2 at the amino-terminal RING domain and stimulates its E3 ubiquitin ligase activity. 48 The conserved transductional mechanisms between TNFR and TLR4 allow us to hypothesize a possible participation of Lyn kinase on TLR4-dependent Shpk1 activation. This could lead to the production of S1P and to TRAF-6 activation. This possibility is now under investigation in our laboratory.
Tyrosine phosphorylation of TLR4 and Lyn association with the receptor and CD14 after LPS treatment has been reported in distinct cellular systems. Utilizing peripheral human monocytes, an important increase in the activity of different tyrosine kinases, such as Lyn, Hck and Fgr, and direct interaction of Lyn with the CD14 co-receptor was observed. 12 In a detailed series of experiments, where HEK293 cells were transfected with different TLR4 and Lyn constructions, it was shown that TLR4 is tyrosine phosphorylated and that an important fraction of Lyn kinase co-immunoprecipitates with the receptor. 9 Our results show that in MCs, Lyn becomes activated upon LPS stimulation and, interestingly, Lyn clearly interacts with TRAF-6. Our efforts to identify Lyn in TLR4 immunoprecipitates from stimulated cells gave only a very slight signal that was barely detectable in our cell system (data not shown). An interaction between Src and TRAF-6 has been proposed, in other cell types as an important interaction leading to Akt 49 and AP-1 activation 50 after stimulation. Some reports indicate that the SH3 domain of Src is important for its association to TRAF-6 and for the activation of downstream targets, 49 but the role of specific domains of Lyn in the interaction with TRAF-6 in MCs remains to be analyzed. The TRAF-6/TAK-1 complex is a central regulatory module controlling the synthesis of numerous cytokines after distinct TLRs. 51 Whether the activation of Lyn kinase in regulating TRAF-6/TAK-1 is a general step on the action of other TLRs is an attractive question that remains to be explored, as preliminary data also indicate lower TNF-α production after a TLR1/2 ligand addition in Lyn−/− BMMCs (data not shown).
In MCs, it is known that Src-family kinases are involved in the initial steps of FcεRI activation and, particularly, it has been reported that Lyn contributes to signalling phosphorylating receptor immunoreceptor tyrosine-based activation motifs (ITAMs), Syk kinase and the adaptor protein linker of activated T cells (LAT), inducing the formation of membrane-associated molecular aggregates, which leads to calcium mobilization, PKC activation, degranulation and cytokine synthesis.52,53 However, Lyn can not only induce positive signals upon FcεRI crosslinking, as it also generates a negative control loop phosphorylating the Csk-binding protein (Cbp), which allows to the recruitment of Csk to the membrane and causes the silencing of different pathways dependent on Src-related tyrosine kinases. 14 Interestingly, the positive or negative role of Lyn kinase on the FcεRI signalling system has been associated with the intensity of the stimulus, having a positive role in low-intensity stimulation and a negative role under a high level stimulation. 13 Regarding TLR4-dependent responses, a positive role of CD14-associated Lyn 9 and Lyn-dependent tyrosine phosphorylation of TLR49,12 has been described in human monocytes. In our case, results show that Lyn also exerts this positive role on LPS-induced TNF-α production in BMMCs. However, a negative role of this kinase on TLR-induced responses was found in bone marrow-derived macrophages. 11 Taken together, recent research indicates a complex regulatory function that Lyn can exert on stimulus-specific cytokine production;33,54 systematic studies comparing low versus high stimulation of TLR4 receptors with the same ligand and in the same cell types will help to characterize the pathways for positive and negative regulation of innate immunity receptors.
The results reported here extend the observations on the role of Lyn in MCs by involving it in the process of NF-κB activation after TLR4 triggering, indicating that this kinase could mediate a crosstalk between FcεRI and TLR4 receptors, as TRAF-6 is necessary for p38 and JNK activation and the synthesis of TNF-α after FcεRI triggering in mouse liver-derived MCs. 55 However, the fact that Lyn−/− BMMCs are able to produce significant amounts of TNF-α over longer periods, suggests the participation of other signalling pathways that are active for longer in the absence of Lyn. The participation of the Bcl-10/Malt-1 complex (that has been implied in the FcεRI-triggered synthesis of TNF-α 56 ) on LPS-dependent TNF-α synthesis is now under exploration in our laboratory.
Finally, it has well been documented that MCs are able to synthesize, store and secrete TNF-α in response bacterial insults into the peritoneal cavity.20,21,34– 36 This early TNF-α production has been associated with the defence against low-intensity infections with Gram-negative bacteria. 21 In particular, the protective role of TNF-α seems to depend on the attraction of inflammatory cells to the peritoneal cavity of mice. 34 – 36 Our in vivo results indicate that Lyn acts as a positive regulator of peritoneal MC-dependent TNF-α secretion in response to a low dose of LPS and to the recruitment of inflammatory cells to the site of bacterial infection. Our finding is of particular interest as TNF-α secreted by MCs early on is not only synthesized de novo, but is stored in granular compartments. These results strongly suggest that, in vivo, Lyn could be also involved in the secretory mechanism leading to MC-derived TNF-α release triggered by LPS.
As Lyn−/− BMMCs synthesized TNF-α at longer period of time, and the systemic increase of TNF-α during endotoxaemia is mediated by other cell types 57 (i.e. macrophages), the specific relevance of intraperitoneal MC-derived TNF-α production and the role of Lyn kinase on the whole animal response to sepsis remains to be elucidated.
MCs are important regulators of the innate and adaptive immune responses. However, in contrast with the knowledge about the machinery of cytokine synthesis and release after FcεRI-dependent signalling, the description of the molecules controlling cytokine production after TLR4 is far from complete. The TLR4 signal transduction system, at least in myeloid immune cells, has been defined as the pathway connecting the formation of the complex LPS/CD14/TLR4/MD-2 in the plasma membrane 5 and the recruitment of adapters (MyD88/Mal or TRIF/TRAM), with NF-κB, AP-1 and IRF nuclear translocation to induce cytokine gene expression. This signalling system has been described as utilizing intermediate serine-threonine kinases such as IRAKs, TAK-1 and IKK. 5 The participation of tyrosine kinases, although reported, 9 has not been considered a critical step on this signalling cascade, but the complex machinery leading to the release of pre-formed and neo-synthesized inflammatory mediators in MCs strongly suggests that new pathways connecting TLR receptors to cytokine production remain to be discovered, or are differentially wired in this remarkable cell type.
Footnotes
Funding
This project was supported by the National Council for Science and Technology of Mexico (Conacyt) (grant no. 83079 to CGE), and scholarships no. 208218 to MA and no. 102830 to AMJ.
Acknowledgements
The authors wish to thank to Hector Espinosa for the bibliography search and Armando Resendiz for technical assistance. MA is student of the Biomedical Sciences (PDCB) PhD program of the National Autonomous University of Mexico (UNAM), Mexico City, Mexico.
