Abstract
Recent evidence suggests a potential role for honeys in mediating clinical inflammation and tissue damage. Here, we investigated the anti-inflammatory activity of a selection of previously untested indigenous New Zealand (NZ) honeys. We found that several, but not all, New Zealand rewarewa, manuka and kanuka honey samples exhibited potent, dose-dependent reduction of human neutrophil superoxide production in vitro. This inhibitory activity did not correlate with levels of known phenolic-based free radical scavengers. Furthermore, the active honeys did not scavenge superoxide generated in a cell-free xanthine/xanthine oxidase assay. In C57BL/6 J mice, topical application of manuka and rewarewa honey samples with the highest in vitro activity suppressed arachidonic acid-induced ear oedema, and rewarewa honey suppressed both oedema and leukocyte (monocyte and neutrophil) infiltration
Keywords
Introduction
The use of honey as a therapeutic agent can be traced back more than 4000 years to some of the earliest reported methods of wound-care. 1 Recently, there has been a resurgent interest in the use of honey in predominantly antimicrobial applications and wound healing. New Zealand (NZ) manuka honey in particular has beenassociated with growth inhibition and apoptosis of many bacterial pathogens, 2 – 6 including antibiotic-resistant isolates from clinical wounds,7,8 severe burns,diabetic leg-ulcers, infected surgical sites 9 – 11 and chronic wounds colonised with methicillin-resistant Staphylococcus aureus. 12 The antibacterial activity of manuka honey has been attributed to the presence of high levels of methylglyoxal (MGO)13,14 a compound known to inhibit DNA synthesis.
There is a growing body of evidence to indicate that honey also exhibits beneficial immunomodulatory activities for the treatment of inflammation. 15 – 17 Acute inflammation caused by oxidative stress is associated with the production of reactive oxygen species (ROS) by cells such as neutrophils. 18 Honeys have been shown to exhibit antioxidant, or ‘ROS-scavenging’, properties that have been predominantly attributed to the presence of phenolic compounds.16,19,20 Clinically, the oral application of manuka honey has been shown to reduce inflammation in rodent models of inflammatory bowel disease21,22 and indomethacin-induced gastritis. 23 Recently, clinical trials have demonstrated that oral administration of undiluted honeys can significantly decrease the extent of radiation-induced oralmucositis in head and neck cancer patients. 24 – 27 Together, thesefindings indicate that honeys have thepotential to exhibit multiple anti-inflammatory effectsthat could be exploited for the treatment of inflammation.
The unique NZ flora lends itself to the production ofhoneys containing components with new anti-inflammatory properties. The aim of this study was, therefore, to investigate the ability of different florally-defined indigenous NZ honey samples to suppress: 1) superoxide production by human neutrophils in vitro; and, 2) topical inflammation in vivo. The results of this study showed that different, florally-defined indigenous NZ honey samples were able to suppress neutrophil superoxide production of independent of ROS-scavenging activity. Furthermore, topical application of specific samples of manuka and rewarewa honeys resulted in abrogated ear oedema and leukocyte infiltration in vivo.
Materials and methods
Reagents and chemicals
Polymorphprep (Axis-Shield, Oslo, Norway) was obtained through Medica Pacifica Ltd (Auckland, New Zealand). 2-(4-Iodophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2 H-tetrazolium monosodium salt (WST-1) was obtained from Dojindo Laboratories (Kumamoto, Japan). Annexin V-FITC, PI, PerCP Streptavidin, APC anti-mouse Gr-1, biotinylated anti-mouse CD45.2, FITC anti-mouse CD11b and PE anti-mouse Ly6G were from BD Bioscience (San Jose, CA, USA). Anti-mouse CD16/32 (2.4G2) was provided by Thomas Backstrom (Malaghan Institute, Wellington, NZ). Cell culture reagents were from Invitrogen (Auckland, NZ). Honey samples were provided by Jonathon Stephens (Comvita Ltd, Hamilton, NZ). Honey samples were stored at 4°C in the dark and diluted in respective assay media immediately prior to the addition of cells/active compounds. Each honey sample was assessed atthe following concentrations unless otherwise specified: 50, 10, 5, 2, 0.4, 0.08, 0.016 mg/ml. All other reagents were from Sigma-Aldrich (Auckland, NZ).
Description of honey samples
Neutrophil superoxide inhibition by NZ indigenous honey samples

L. scoparium (Manuka), K. ericoides (Kanuka), K. excelsa (Rewarewa), Trifolium spp. (Clover), var. = variation. OMT, oral mucositis trial honey.
Human neutrophil superoxide assay
Peripheral blood was collected from healthy volunteers with informed written consent under New Zealand Central Ethics Committee Approval CEN/08/11/062. Human neutrophils were isolated from peripheral blood by sedimentation over a Polymorphprep density gradient, as described by the manufacturer. Neutrophil superoxide production was measured in 96-well plates as previously described. 29 Briefly, neutrophils (106/ml) were stimulated with phorbol 12-myristate 13-acetate (PMA, 0.2 µg/ml) in the presence of different concentrations of honey and the colorimetric dye WST-1 (0.25 mg/ml) in Hank's balanced salt solution. The WST-1 reduction to formazan dye by superoxide was measured in real time by absorbance at 450 nm at 37°C using a Versamax spectrophotometer (Molecular Devices, Surrey, VIC, Australia). Superoxide dismutase (SOD, 0.01-50 µg/ml) was used as a positive control.
Xanthine/xanthine oxidase superoxide assay
Superoxide was generated enzymatically as described previously.29,30 Briefly, 25 µl of 1.521 mg/ml xanthine solution (15.21 mg/ml in 1 N sodium hydroxide in Tris-HCl buffer, pH 7.4) and 25 µl of 60 mU/ml xanthine oxidase (in Tris-HCl buffer, pH 7.4) were sequentially added to 50 µl of different concentrations of honey samples (diluted in Tris-HCl buffer, pH 7.4) to a total volume of 100 µl. SOD was included as a positive control and WST-1 reduction was measured as described above.
Neutrophil viability assay
We used the early apoptosis marker, annexin V (AV) and the necrosis marker, propidium iodide (PI) to assess cell viability, which was determined as the AV−/PI− cell population. Neutrophils (106/ml) in the presence or absence of PMA were treated with different honey samples (concentrations = IC50 inhibition of superoxide, see Table 1). After 30 min, neutrophils were collected, washed, and stained with FITC-AV and PI in AV binding buffer (10 mM HEPES, 140 mM NaCl, 2.5 mM CaCl2, in Milli-Q H2O) for 15 min in the dark at 4°C and analysed by flow cytometry. 31 Cell viability was calculated as the percentage of live AV−/PI− neutrophils in each cell sample.
Arachidonic acid-induced ear oedema model
C57BL/6 male mice (aged 10–12 wk) were bred and housed within the animal facility of the Malaghan Institute of Medical Research, Wellington, NZ. All in vivo experiments were approved by the Victoria University Animal Ethics Committee and carried out in accordance with their guidelines for care of animals.
In vivo inflammation was investigated using a modified version of the arachidonic acid-induced ear inflammation model. 32 Briefly, 20 µl of 100 mg/ml arachidonic acid (AA) in acetone was applied to one ear of each mouse and 20 µl of vehicle (acetone) alone to the other. A total of 30 mice were divided into six groups of 5 mice [a positive control group: dexamethasone (20 µl of 250 mg/ml in acetone), a negative control group without any honey and 4 different honey sample groups]. Mice were rested for 30 min to allow inflammation to become established and 50 µl of undiluted sample honey was applied, sufficient to cover the entire surface of the ear. After 4 h, mice were sacrificed and the ears washed to remove residual honey. A 6 mm diameter tissue biopsy was taken from each ear and weighed. The entire ear, including biopsy, was then digested for 2 h (120 µg/ml DNAse I and 2.4 mg/ml collagenase I, 37°C, 5% CO2) in complete Iscove's modified Dulbecco's medium (cIMDM, with 10% heat-inactivated FBS, 1% pen-strep). The digest was passed through a 40 nm filter, washed with PBS buffer containing DNAse I (120 µg/ml), BSA (10 mg/ml) and EDTA (1.86 mg/ml). Cells were resuspended in cIMDM, stained for different cell surface markers and analysed by flow cytometry.
Cell surface expression of CD45.2/CD11b/Ly6G/Gr-1 by flow cytometry
Cell homogenates from the ears of each treatment group were washed in FACS buffer. Cells were incubated with anti-mouse CD16/32 monoclonal antibody (2.4G2) in FACS buffer (10 min, 18–20°C) to block non-specific binding. Cells were then washed in FACS buffer, resuspended in 50 µl antibody preparation containing 1:500 biotinylated CD45.2, 1:800 CD11b FITC, 1:800 Gr-1 APC and 1:800 Ly6G PE in FACS buffer and incubated in the dark (15 min, 4°C). Cells were washed and resuspended in FACS buffer containing 1:2000 streptavidin PerCP and incubated in the dark (15 min, 4°C). Single-stained samples of negative control cells (AA without treatment) were included for each fluorophore. Cells were washed, resuspended in FACS buffer and passed through a 40 nm filter prior to analysis by flow cytometry. Leukocytes were distinguished from non-haematological cells by their CD45+ expression. Neutrophils were identified as CD45+/Ly6G+/CD11b+/Gr-1high and monocytes as CD45+/Ly6G-/CD11b+/Gr-1low, as previously described. 33
Statistical analysis
All honey samples were tested in triplicate and each assay performed at least twice. Statistical significance was determined by a one way ANOVA or student t-test using Prism v5.0 c (GraphPad Prism Software Inc., La Jolla, CA, USA).
Results
Inhibition of neutrophil-produced superoxide in vitro
All 21 honey samples inhibited superoxide production by PMA-stimulated neutrophils with IC50 values between 3.1 and 44.4 mg/ml (Table 1). The four kanuka honeys (H15–H18) and the single rewarewa honey (H21) exhibited the highest activities (6.1, 5.1, 3.1, 4.6 and 4.3 mg/ml respectively), whereas general pasture honey (H20) and commercially-produced UMF30+ manuka (H19) honeys exhibited low anti-inflammatory activity (20.8 and 44.4 mg/ml respectively). Manuka honeys showed variable activities (4.2–37.9 mg/ml) with both regional and inter-regional variability (data not shown).
To determine whether higher anti-inflammatory activity correlated with honey components of known bioactivity, IC50 values for neutrophil superoxide inhibition were plotted against phenolic (ROS scavenging) and methylglyoxyl (MGO; antimicrobial) content as reported by Stephens et al.
28
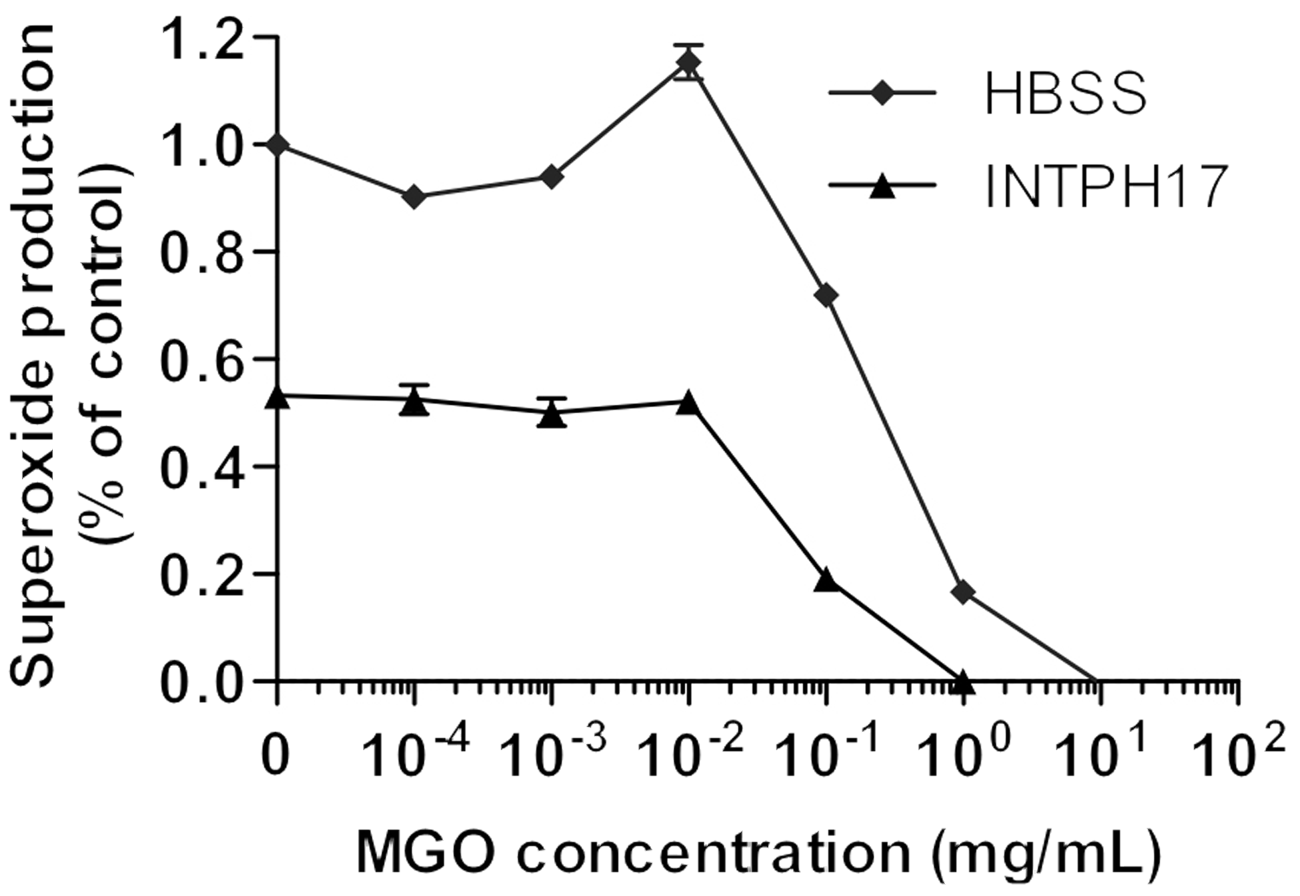
No correlation between superoxide inhibition and phenolic content was observed (Figure 1D). Surprisingly, a significant inverse correlation was observed between the anti-inflammatory activity and MGO content (Figure 1D). To determine if MGO could be blocking the anti-inflammatory activity, neutrophils were treated with H17 (low MGO) at the IC50 for superoxide suppression and spiked with increasing amounts of MGO. As shown in Figure 2, MGO concentrations up to 0.01 mg/ml did not alter the inhibitory effect of H17. At higher MGO concentrations, decreased superoxide production by MGO- and H17/MGO-treated neutrophils was observed for MGO in association with neutrophil death. These data indicated that the inverse correlation of MGO content and suppression of neutrophil superoxide production by the honeys was not caused by MGO-dependent inhibition of the anti-inflammatory activity. It should be noted that different manuka honey samples varied markedly in their MGO content. This may have been as a result of contamination with nectar from other flower sources.
Correlation of neutrophil superoxide inhibition with phenolic compounds and MGO. Superoxide inhibition Neutrophil superoxide inhibition by honey samples is independent of MGO. Human neutrophils were activated with PMA and superoxide production measured in the presence of PH17 (3 mg/ml) and spiked with different concentrations of MGO. Values are mean ± SEM of triplicate measurements. Results are representative of two independent experiments.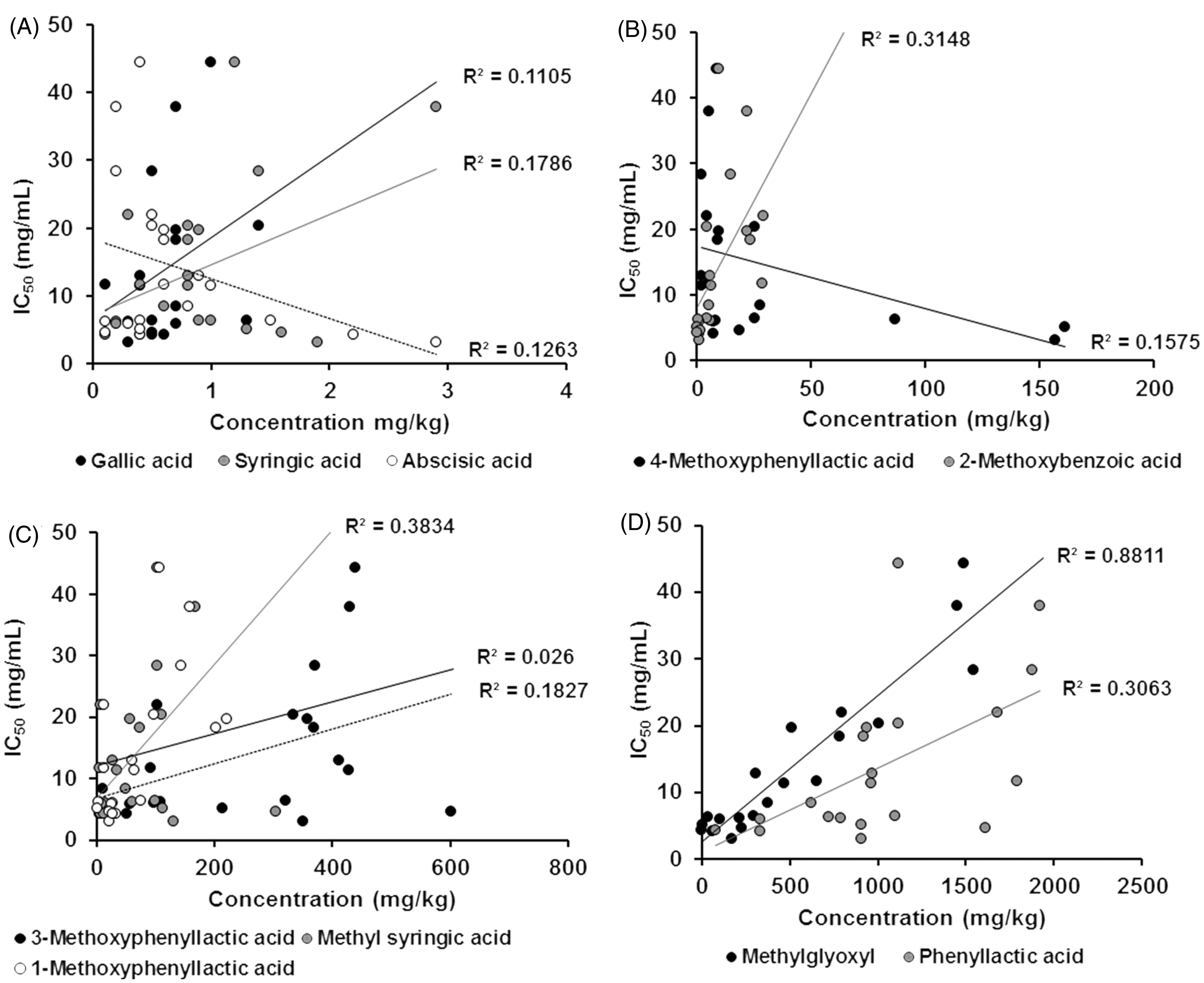
Effect of honeys on neutrophil viability
Next, we investigated whether the observed decrease in neutrophil superoxide production following low dose honey treatment was the result of cell death. The three most active honey samples (H10, H17, H21) and one comparatively inactive honey sample (H20), independent of floral origin or MGO content, were selected for further study. Neutrophils were treated with H10, H17, H21 and H20 at concentrations equivalent to their respective IC50 values for superoxide suppression (Table 1). Cell survival was assessed by flow cytometry based on the expression of the apoptosis marker AV and necrosis/cell death marker PI. The viability (AV-/PI-) of unstimulated and PMA-stimulated neutrophils was not affected by treatment with H17 and H21; however, a small loss in the viability of activated neutrophils was observed with H10 and H20 treatment (Figure 3A). Cell death was evident in all samples at higher honey concentrations (>50 mg/ml).
Honey samples do not induce cell death or scavenge ROS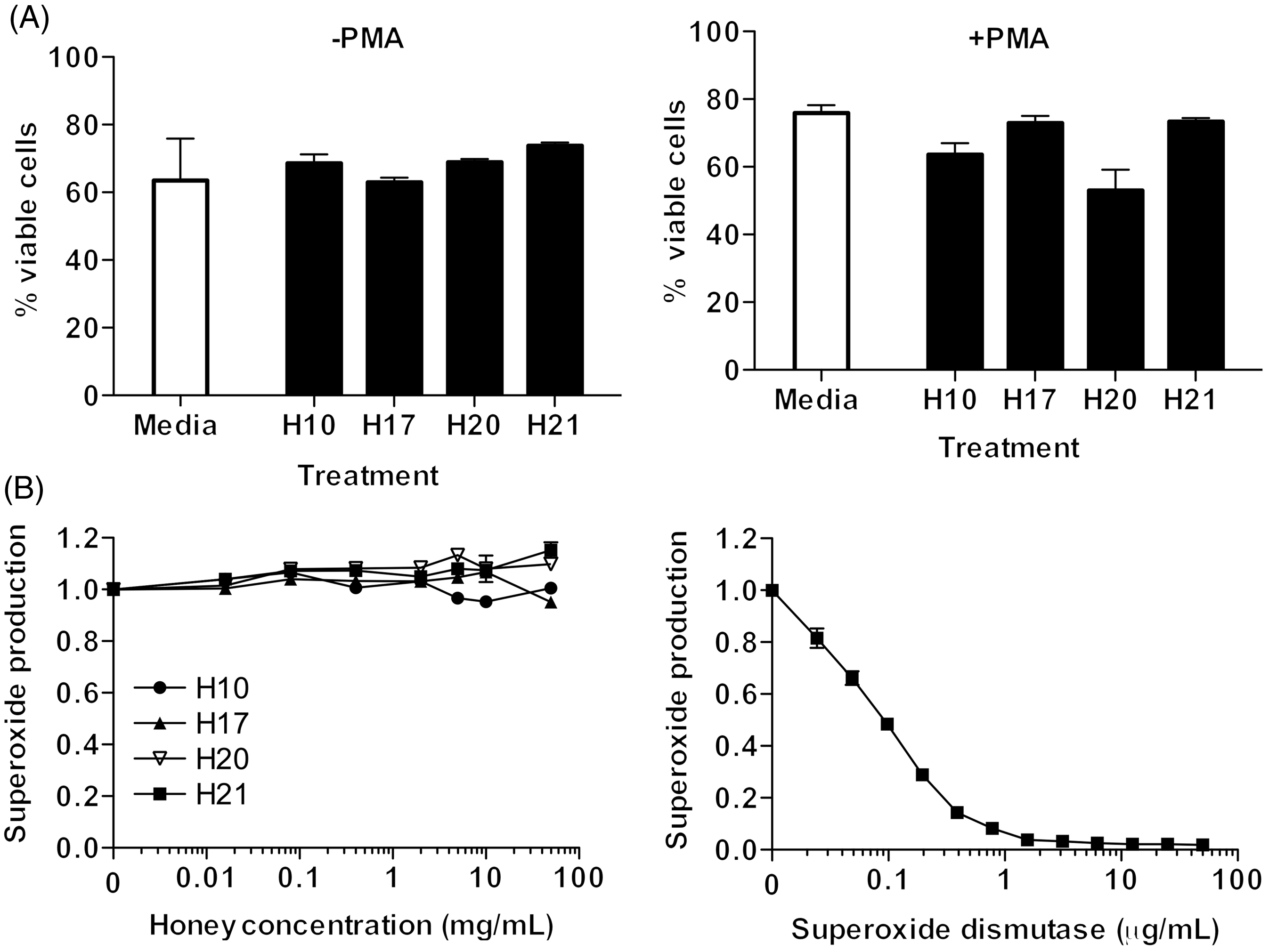
Non-cellular free radical scavenging by honeys
Previous literature has shown that the ability of manuka honeys to lower superoxide levels is associated with freeradical scavenging.16,19,20 To test for free radical scavenging, the more active honeys, H10, H17, H21, and the less active pasture honey, H20, were tested fortheir ability to scavenge superoxide in a cell-free xanthine/xanthine oxidase assay. None of the honeys tested reduced superoxide levels in this assay (Figure 3B), indicating that free radical scavenging was not responsible for the honey-dependent reduction in neutrophil superoxide production.
Effect of honeys on AA-induced ear oedema
Based on the neutrophil superoxide screen described above, three high activity honeys (H10, H17 and H21) and the low activity pasture honey (H20) were tested for additional anti-inflammatory activities in a mouse model of acute local inflammation. Applied topically to the ear, AA induces a well-characterised in vivo model of acute inflammation involving oedema and infiltration of both neutrophils and monocytes to the inflammatory site in 1–4 h.
32
To investigate the therapeutic potential of the honeys to treat established acute inflammation, 30 mice were divided into 6 groups of 5 mice each. One ear of each mouse was exposed to AA for 30 min, the other was exposed to acetone; then, both ears were treated with either nothing (negative control; group 1), H10 (manuka; group 2), H17 (kanuka; group 3), H21 (rewarewa; group 4), H20 (pasture control; group 5) or dexamethasone (positive control; group 6). Tissue samples were harvested at 4 h post-AA administration and oedema was measured. Out of the three most effective anti-inflammatory honeys identified in vitro only H21 significantly reduced AA-induced oedema compared with the untreated negative control group; this effect was similar to that of the positive control, dexamethasone (Figure 4). Application of honeys to acetone-treated uninflamed ears showed no significant induction of oedema.
Honey samples suppress oedema in vivo. Arachidonic acid or vehicle was administered to the ears of C57Bl/6 J mice in six groups (n = 5 per group). After 30 min undiluted honey samples were applied. Four hours post-AA treatment, ear biopsies were collected from each mouse and weighed. Dexamethasone (Dex) = positive control. Values represent mean ± SEM (n = 5 per group). Results are representative of two independent experiments. * P < 0.05.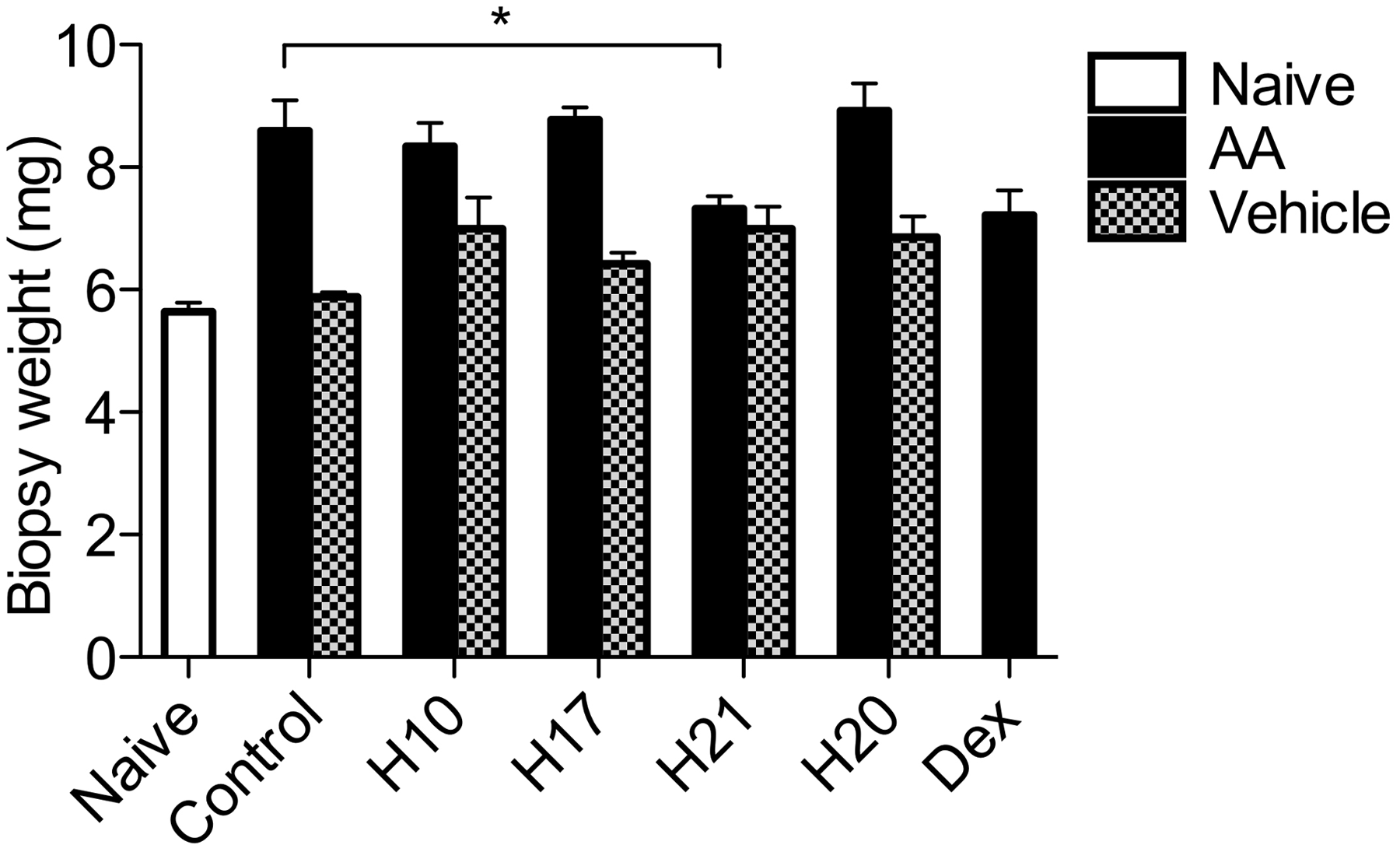
Inflammatory cell infiltration in the AA ear oedema model
To determine the impact of honey treatments on AA-induced cellular infiltration, all ear tissue, including biopsy material, from each treatment group were pooled, digested and the infiltration of monocytes (Ly6G-/CD11b+/Gr-1low) and neutrophils (Ly6G+/CD11b+/Gr-1high) into the tissues was analysed by flow cytometry (Figure 5A). Although all four honey treatments reduced total infiltration of CD45.2+ haematopoietic cells (Figure 5B) compared with the untreated negative control group, H10 and H21 were the most effective, lowering the levels of infiltrating cells below that of the dexamethasone treatment group. The reduction in total cell infiltration was mainly caused by a decrease in neutrophil infiltration (Figure 5C). Conversely, pasture honey (H20) and dexamethasone treatment enhanced neutrophil recruitment, whereas H17 had no effect on neutrophil infiltration. All honeysamples reduced the proportion of infiltrating monocytes to some extent; however, the greatest suppression was observed with dexamethasone treatment (Figure 5D).
Honey samples suppress leukocyte infiltration in vivo. Arachidonic acid or vehicle was administered to the ears of C57Bl/6 J mice in six groups (n = 5 per group). After 30 min undiluted honey samples were applied. Four hours post-AA treatment, ears were harvested, pooled, digested and cells were analysed per group by flow cytometry (A). (B) Gating strategy. (C)Total CD45+ cells. (D) Neutrophils (CD45+/Ly6G+/CD11b+/Gr-1high). Monocytes (CD45+/Ly6G-/CD11b+/Gr-1low). Dexamethasone (Dex) = positive control. Data are representative of two independent experiments.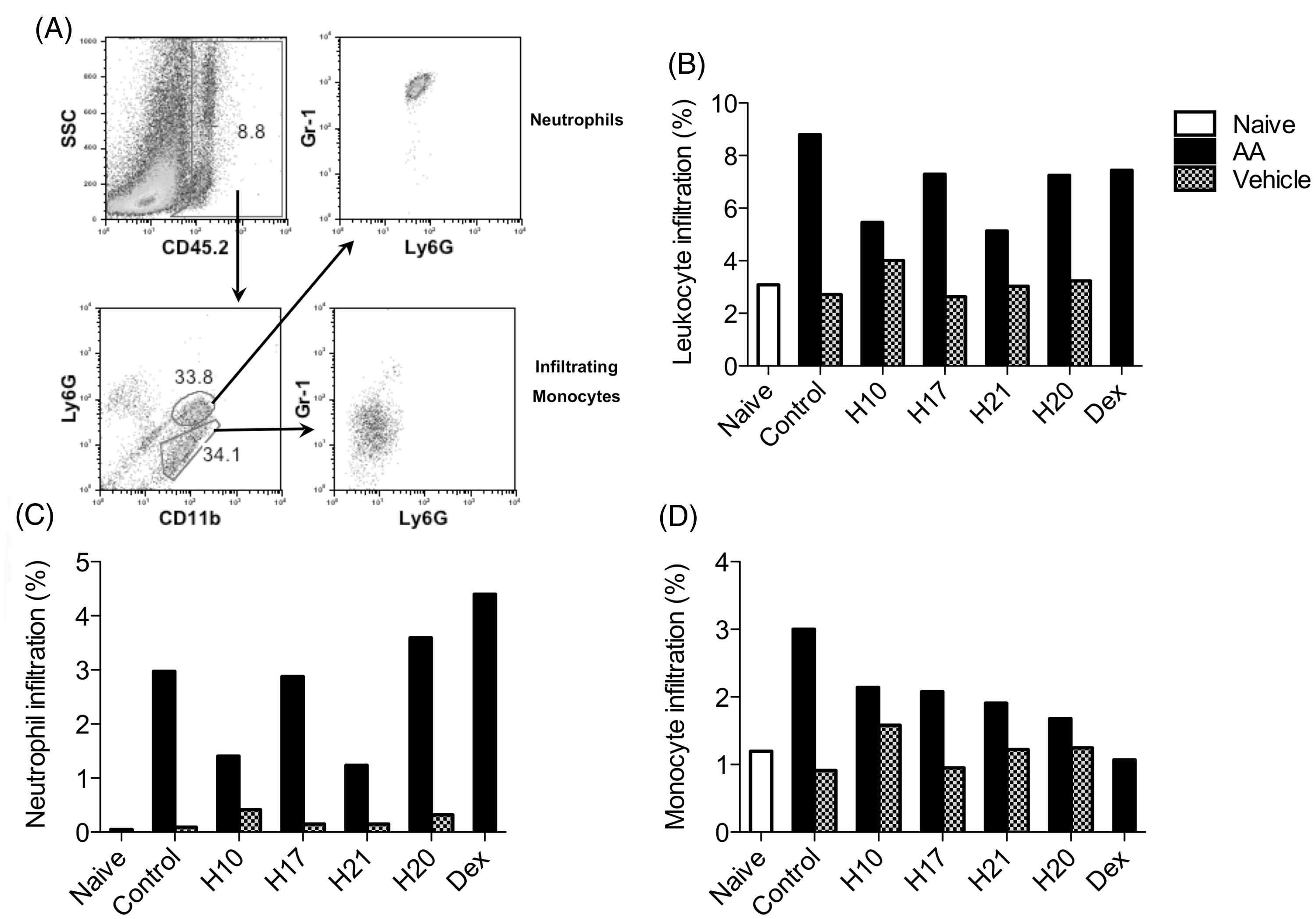
Discussion
Our results identify a new subset of indigenous NZ honeys that exhibit anti-inflammatory activity on human neutrophils in vitro and can suppress oedema and leukocyte infiltration in a mouse model of topical acute neutrophilic inflammation.
A decrease in neutrophil superoxide production by honeys in vitro has been reported previously whereby the activity of other honey samples was attributed to the inhibition of ROS formation, either by inhibitingthe respiratory burst of neutrophils16,17 or by direct ROS scavenging.16,19,20 Inoue et al. 20 have reported that free radical scavenging in their honey samples was mediated by methyl syringate. However, methyl syringate levels in our honey samples did not correlate with anti-inflammatory activity. In fact, the inhibition of neutrophil superoxide production by the NZ indigenous honeys in thisstudy did not correlate at all with free radical scavenging activities nor was honey activity linked with neutrophil death. Together, these data indicate that the anti-inflammatory activity of the indigenous NZ honeys was likely caused by the inhibition of the neutrophil respiratory burst.
MGO is the primary component associated with the antimicrobial activity of manuka honey but it did not appear to contribute to the anti-inflammatory activity observed in this study. The inverse correlation between honey anti-inflammatory activity and the MGO content of the samples tested suggested that a high MGO content could be blocking anti-inflammatory activity. However, the addition of MGO to low MGO-containing kanuka honey with high anti-inflammatory activity did not alter the suppression of neutrophil superoxide production. This demonstrates that high MGO levels do not suppress anti-inflammatory activity of the honeys. The inverse correlation between anti-inflammatory activity and MGO was not an artefact of MGO and anti-inflammatory agents being present in the nectar at the same time as honeys with high anti-inflammatory activity exhibited low MGO-dependent antibacterial activity, regardless of floral origin.
Leukocyte infiltration is a strong correlate of inflammation in vivo. In this study, manuka and rewarewa samples, H10 and H21, were each able to suppress established AA-induced leukocyte infiltration in vivo, with the majority of this suppressive effect caused by decreased neutrophil infiltration. Whether honey treatment abrogates neutrophil trafficking by shutting down the resident cells responsible for initiating the inflammatory response, or by inhibiting infiltrating neutrophil activation and self-recruitment in vivo, is worthy of future study.
Interestingly, of the honeys tested in vivo, only the rewarewa honey reduced both inflammatory leukocyte infiltration and AA-induced oedema. This result indicates that rewarewa honey has the potential to abrogate inflammation by hitting multiple inflammatory targets: neutrophil respiratory burst, neutrophil recruitment and swelling. However, more rewarewa honey samples fromdifferent regions in NZ need to be tested before any claims can be made about the superiority of honey of rewarewa origin with respect to anti-inflammatory activity.
To date, the component(s) responsible for the anti-inflammatory honey activities observed in this study remain to be determined. As such, it is not clear whether a single molecule within the honey samples is responsible for both the in vitro and in vivo activities, or if the anti-inflammatory activities arise from different compounds. However, diacyl compounds, found to be high in rewarewa honeys 34 may provide a logical starting point for further anti-inflammatory testing.
In summary, our findings demonstrate that a subset of indigenous NZ honeys exhibit novel and therapeutically-relevant anti-inflammatory activities that may have benefit as a topical treatment for inflammation. Further investigation into the anti-inflammatory effects of indigenous honeys is warranted to identify the component(s) and mechanisms involved in the inhibition ofneutrophil superoxide production in vitro and the inhibition of oedema and leukocyte infiltration in vivo.
Conflict of interest
The authors declare no conflict of interest.
Footnotes
Acknowledgements
The authors thank Stefanie Steiger and Dr Willy-John Martin for technical assistance with the assays. We also thank Jonathan Stephens, Comvita NZ Ltd for supplying the honey samples tested in this study. This work was funded, in part, by Foundation for Research, Science &Technology and Comvita NZ Ltd.
