Abstract
Endotoxin research in recent years at the molecular level has required chemically synthesized lipid A without contamination by other bioactive components. Total synthesis of Escherichia coli-type lipid A was achieved in the 1980s by the challenging spirits of the scientists at Osaka University, Japan. They clarified the role of lipid A in the immunological activities of endotoxin in collaboration with Japanese and German researchers, based on the friendships that existed between them. This article introduces the great contributions made by three generations of professors, Tetsuo Shiba, Shoichi Kusumoto, and Koichi Fukase, at the Laboratory of Natural Product Chemistry at Osaka University, to the study over four decades of endotoxin.
Introduction
Collaboration in scientific research is sometimes not easy because most scientists aggressively pursue their own results, and have a tendency not to work with scientists in other fields. However, in competitive investigations performed in the 1980s, searching for the immunoactive principle of endotoxin, productive collaboration between research groups was achieved, which was based on the friendships between individual researchers. Professors of three generations, Tetsuo Shiba, Shoichi Kusumoto, and Koichi Fukase at the Laboratory of Natural Product Chemistry, Faculty of Science, Osaka University, have always been open-minded and communicative with scientists outside of their field, irrespective of whether they were domestic or foreign research groups. Such attitudes attracted people and cultivated friendships between scientists. This article mainly focuses on the friendships between the researchers of Osaka University and those in Japan and in Germany. Their collaboration clarified the precise chemical structure of lipid A and solved the long-lasting question on the endotoxic principle by means of synthetic organic chemistry.
Inspired by peptidoglycan
Tetsuo Shiba (Figure 1), born in 1924, started his career as a scientist just after World War II, and worked for more than 40 years at Osaka University until his retirement. As described in this article, he established the greatest achievements in the chemistry of endotoxin, but at the start of his career his main subject of research was peptide chemistry, in particular peptide antibiotics. One of his evaluated works at that time was the structural determination and synthesis of tuberactinomycin. 1 , 2 When he was promoted to professor in 1971, he was driven by the idea that he should not take an easy path, and had to start a new challenge and seek out a new research area within the field of chemistry. What he did first for this purpose was to look for a younger collaborator to take on this challenge, and invited Shoichi Kusumoto back to his laboratory. Shoichi Kusumoto was born in 1940, studied as a graduate school student in the same laboratory as Shiba, and had research experience in the laboratory of Erich Hecker in Heidelberg, Germany. There he worked on phorbol ester and diterpene compounds, during which time he learned how chemistry could contribute to solve biological problems.

Tetsuo Shiba (1924–2010) in 1988.
After Kusumoto joined the Shiba laboratory, they started to visit various medical and microbiological laboratories at Osaka University as well as those at other institutions to search for a new theme for their challenge. Among them was the laboratory of Shozo Kotani. Kotani was only two years older than Shiba but was already a well-known professor of microbiology and oral microbiology at Osaka University. Kotani had worked for a long time on cell walls and peptidoglycan of bacteria, and was competing with the French group of Edgar Lederer and Louis Chedid. Shiba became interested in Kotani’s research projects because Shiba had the idea of synthesizing antibacterial agents that could inhibit peptidoglycan biosynthesis more efficiently than penicillin derivatives. It is not difficult to imagine how the discussions among them proceeded, because Kotani was of cheerful and friendly personality and always welcomed visitors (Figure 2). One day during a conversation with Shiba, Kotani mentioned that a major research subject regarding peptidoglycan was the inhibition of its synthesis by antibiotics, but there was an alternative aspect of peptidoglycan study, namely, research on its immunological activity. From that day onward, Kotani and Shiba maintained a close collaboration until and even after they retired from Osaka University.

Shozo Kotani (1922–2004) (right) and Otto Westphal (1913–2004) (left) at the international symposium on endotoxin in 1982 in Susono City, Japan.
At the end of 1973, Kotani told Shiba that a relatively small molecule derived from peptidoglycan might exhibit adjuvanticity. These words strongly captivated Shiba and Kusumoto because such a small molecule could be the target of chemical synthesis with relatively little difficulty; thus they felt that they had finally found the target for their new study. For an experienced synthetic chemist like Shoichi Kusumoto, the synthesis of N-acetylmuramyl-mono-, -di-, -tri-, and -tetrapeptides was not a troublesome work. They soon reported that muramyl-dipeptide (MDP) is the smallest molecular unit of peptidoglycan, 3 , 4 independently from, but shortly after, the group of Lederer. 5 , 6 Kusumoto received an award from the International Society for Immunopharmacology, mainly for his work on the synthesis of MDP; however, this was of course only a prologue to the synthesis of a more complicated molecule, lipid A.
Charmed by endotoxin
The success of the work on MDP was the peak of the collaboration among scientists at Osaka University, and many other pharmacological fruits that they could harvest also came within reach. 7 , 8 Kusumoto felt, however, that he should not keep working only on the derivatization of MDP, which was required and highly regarded in the field of pharmacology. Instead, he felt that he should try to synthesize more difficult molecules, for which he needed advanced theories and methods of synthetic chemistry.
By this time Shiba and Kusumoto had become acquainted with many microbiologists and immunologists through their study of MDP, and received information on various microbial products and components; what charmed them most among those cell components was endotoxin (LPS). Although many studies had been performed over decades on the carbohydrate portion of endotoxin, little was known at that time about the structure and biosynthesis of lipid A, the lipid portion of endotoxin that acts as an anchor for the whole molecule at the surface of Gram-negative bacterial cells. 9 , 10 At this time, the most precise and suggestive information on endotoxin was provided also by their friend, Shozo Kotani.
At the start of the synthetic study in 1979, the structure of the lipid A Kdo region had been reported, as shown in Figure 3.11–14 As would be readily noticed by current researchers on endotoxin, the linkage position of Kdo was improperly placed at the 3′-O-position of non-reducing end (distal) glucosamine (GlcN) instead of at the 6′-O-position. This presumed linkage position arose from the experiment of periodate oxidation on the Kdo-GlcN oligosaccharide reported by Gmeiner et al. in 1971. 15 This misidentification of the position later led the study to a dramatic ending of the structural identification and the total synthesis of lipid A.
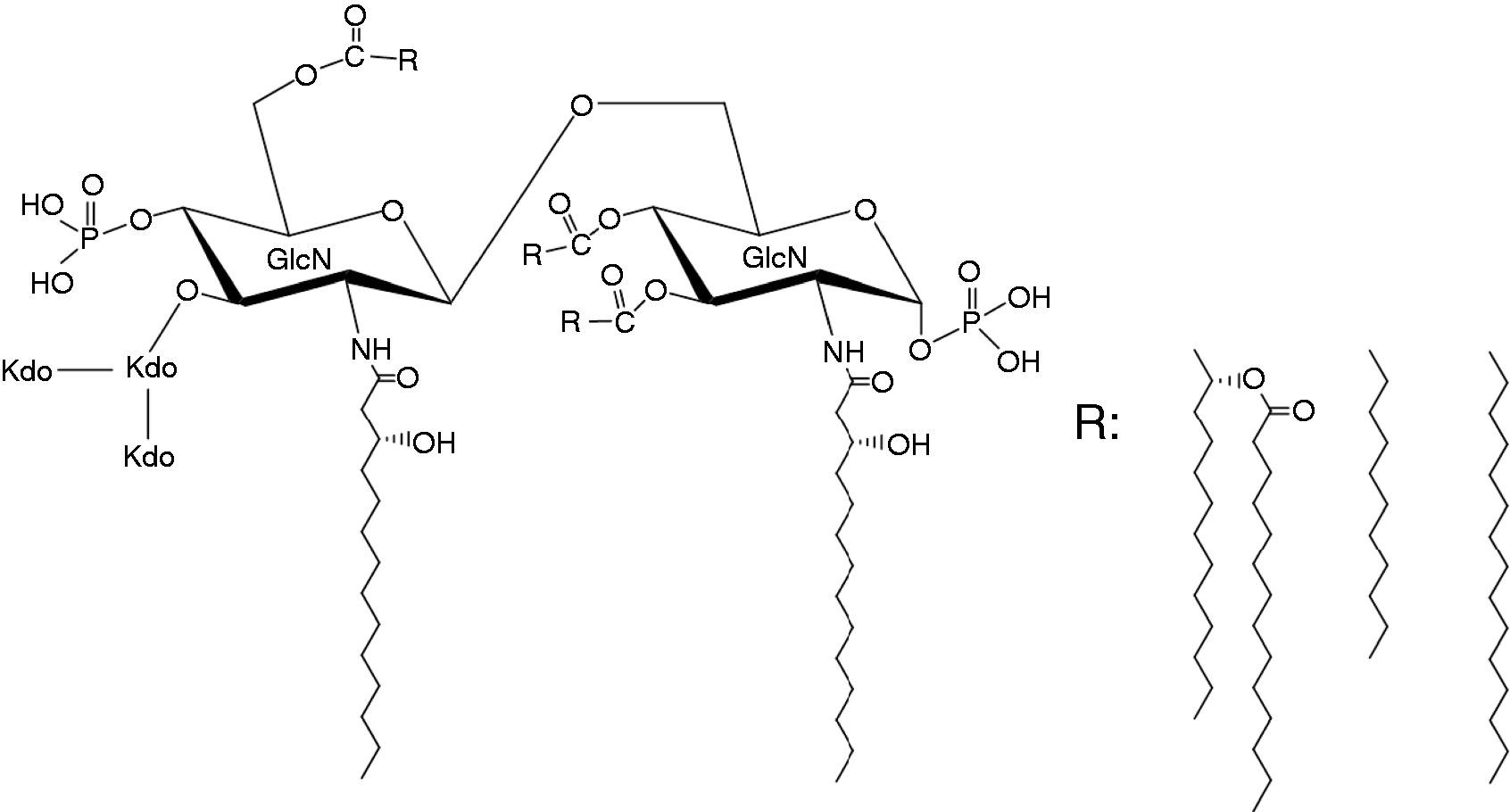
Presumed structure of the lipid A Kdo region of Salmonella as of 1979. The linkage position of Kdo to the GlcN backbone in Escherichia coli LPS had been deduced to be the same as that of Salmonella (Kdo was later proved to be linked at the 6′ position of distal glucosamine, as indicated in Figures 5 and 7).
Kotani and his coworkers remained good advisers also for the lipid A synthesis, but Kusumoto thought that they had to make contact with the most highly regarded and experienced group of endotoxin researchers to complete the project successfully. Therefore, he asked Shiba to visit the Max Planck Institute for Immunobiology in Freiburg, Germany while attending a meeting on peptide synthesis in Europe. Shiba visited Freiburg in September 1980, and was warmly welcomed by many German researchers of endotoxin, including Otto Lüderitz, Chris Galanos, and Ernst Th. Rietschel (Otto Westphal was absent on that day, but kindly called Shiba the next morning). That was the first day of a successful collaboration and friendship between the Osaka and Freiburg groups. As a matter of fact, the collaboration had already started when Sumihiro Hase, a colleague of Kusumoto at Osaka University, worked with Rietschel on the GlcN backbone of lipid A, 16 , 17 and this made the basis of smooth start of the collaboration. When Kusumoto himself visited Research Institute Borstel located in the northern part of Germany in 1983 to discuss the project with Rietschel, they soon became close friends, and this friendship (Figure 4) led the collaboration to a great success.

Shoichi Kusumoto and Ernst Th. Rietschel (front row, third and fourth from the right) with their colleagues of Osaka University and Research Center Borstel (formerly Research Institute Borstel) together with Tetsuo Shiba (front row, furthest on the right), Koichi Fukase (second row, third from the right), and Sumihiro Hase (second row, second from the left). They celebrated the 60th birthday of Kusumoto and Rietschel in 2000 on the occasion of the international symposium on lipid A in Osaka, Japan.
Disappointment and final success
As mentioned above, Shiba, Kusumoto, and their young associates started the synthesis according to the structure shown in Figure 3. For the synthesis, they had to develop methods for preparing pure (R)-3-hydroxymyristic acid, efficient formation of glycosyl phosphates, and selective protection and deprotection of hydroxyl groups. They solved all of these problems and finally obtained the synthetic compounds with various phosphate and fatty acid substitutions, and sent them to Freiburg. 18 , 19 However, they received disappointing news that all of the synthetic compounds were much less active, or even inactive, compared with natural lipid A prepared from S. Minnesota Re mutant. 20 , 21 At about the same time, they passed the synthetic compounds also to Japanese scientists, Shozo Kotani and his associate, Haruhiko Takada, at Osaka University, Yuzuru Homma at Kitasato Institute, Nobuhiko Kasai at Showa University, and Shiro Kanegasaki at the University of Tokyo, whose colleague Kenichi Tanamoto was working on this project in Freiburg at exactly that time. However, no clear activity was reported by them, either.
All of the collaborators on this project were disappointed by the results, but Shoichi Kusumoto thought differently when he received those negative results. He thought that there were only two possibilities: (1) the proposed structure was wrong or (2) lipid A was not the center of endotoxic activities. He had the impression that the proposed structure (Figure 3) was incorrect but not very far from the true structure. Thus, he decided to determine the structure before starting the synthesis again, using large amounts of lipid A preparation from Escherichia coli provided by German collaborators. After chemical modification and purification to reduce the diversity of the structure, Kusumoto and coworkers succeeded in determining the linkage positions of Kdo (6′-O-position of distal GlcN) and all fatty acids, and reached the structure shown in Figure 5. 22 , 23
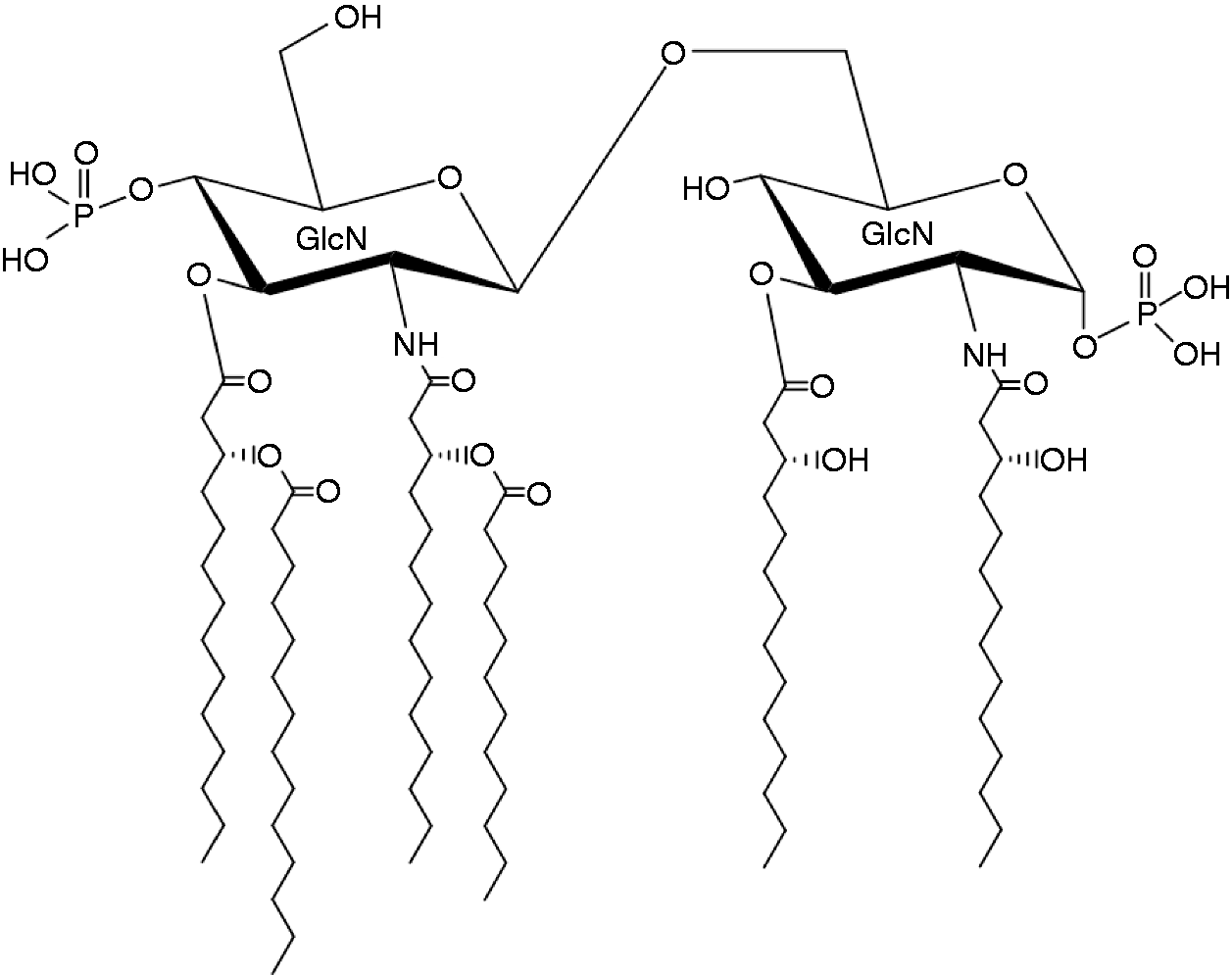
Chemical structure of Escherichia coli lipid A.
Structural studies always depend on analytical instruments based on new technologies. For the analysis of natural products at that time, technologies of NMR spectroscopy and MS with improved ionization methods had been developed: two-dimensional (2D)-1H- and 13C-NMR, fast atom bombardment MS, or MALDI-TOF MS. By using these technologies other groups were also approaching the true structure of lipid A. Strain et al. reported earliest on Escherichia coli, 24 Takayama et al. on S. Typhimurium, 25 and Sidorczyk et al. on Proteus mirabilis, 26 nearly at the same time as Kusumoto’s group; these new results were summarized by Rietschel et al. 27 The newly proposed structure was also confirmed from a different aspect by intensive biosynthetic study by Christian R. H. Raetz at University of Wisconsin, USA, and his collaborators, including Masahiro Nishijima.28–30 Compared with the structure proposed earlier, the finally proved structure of lipid A (Figure 5) clearly suggested its function in the outer membrane of bacteria.
After the structure of lipid A had been established, Shiba, Kusumoto, and their associates initially started work on the synthesis of an intermediate compound, precursor Ia (later designated lipid IVa), 31 because the synthesis without an acyloxyacyl structure was easier than the acyloxyacylated structure. For this synthesis, they could use the knowledge accumulated during the synthesis of the old model lipid A and its derivatives, and only within a year they could synthesize that intermediate compound and its derivatives, 32 and sent them immediately to Germany. Shortly after they sent the compounds, a happiest telegram arrived in Osaka (the quickest means of international communication at the time) telling them that the synthetic precursor Ia expressed lethal toxicity toward galactosamine-sensitized mice as well as B-lymphocyte mitogenicity and other endotoxic activities, and those activities were comparable to those for the natural precursor prepared from bacteria. 33 Kotani also obtained the same results by using these synthetic compounds. 34 These data convinced Kusumoto and coworkers that they were moving in the right direction, and surely accelerated the synthesis of the complete structure.
For the synthesis of the complete structure of lipid A, they had to introduce acyloxyacyl groups to the distal GlcN, and therefore elaborated the synthetic route indicated in Figure 6. 35 Lauroyloxymyristic acid was introduced to the 2′-N-position of the distal GlcN after the coupling of two GlcN molecules to avoid a β-elimination reaction during the coupling, and all of the protective groups were removed by hydrogenolysis in the last step of the total synthesis. The fully synthesized lipid A (compound 506) and its derivatives were sent to Freiburg, to Research Institute Borstel, and also to all Japanese collaborators mentioned above. Finally, all of the experimental results, not only from German groups but also Japanese teams,36–39 indicated that synthetic compound 506 exhibited endotoxic activities identical to those of natural Escherichia coli lipid A on a weight basis (Table 1). 36 , 40 All of these reports clearly proved that lipid A is the principle of the endotoxic activity.
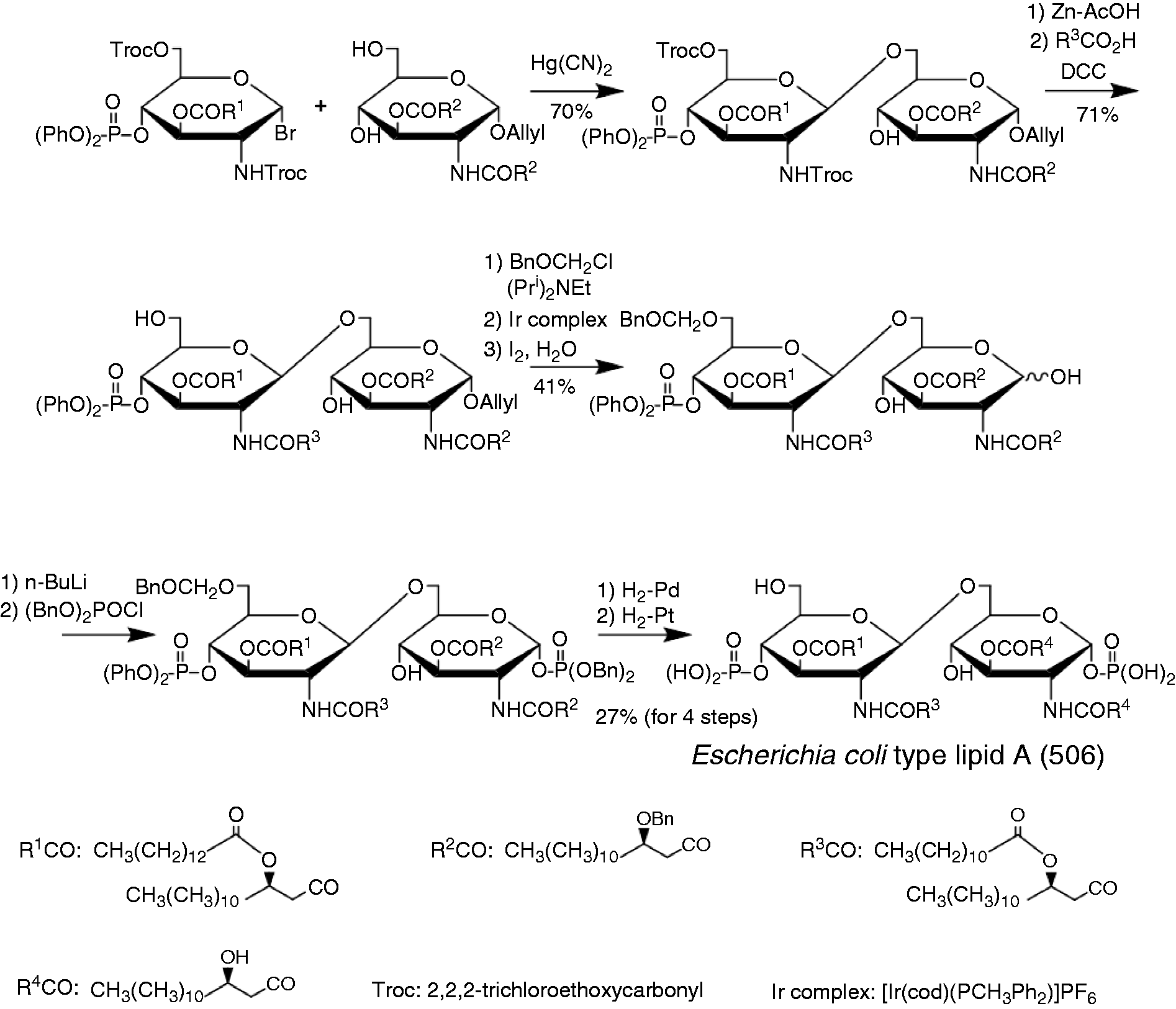
The first chemical synthesis of Escherichia coli lipid A.
Each lipid A preparation was injected intravenously to galactosamine-sensitized mice, and the dose required for 50% lethality was determined.
Each lipid A preparation was injected intravenously to rabbits, and fever responses were measured. The dose that caused more than a 1°C rise at 3 h is indicated.
Degree of skin necrosis reaction of rabbits was measured after pretreatment by intradermal injection and second intravenous injection of each lipid A preparation. The dose required for maximum reaction (+++) is indicated.
Mice were pretreated with each lipid A preparation, and challenged with Salmonella LPS after 4 d. The dose required for more than 90% survival is indicated.
Mouse spleen cells were incubated with each lipid A preparation and 3H-labeled thymidine. The dose required for 15-fold increase of thymidine incorporation is indicated.
Mouse peritoneal macrophages were incubated with each lipid A preparation, and prostaglandin E2 produced in the supernatants was measured. The dose required for more than 10 ng/ml induction of prostaglandin E2 is indicated.
Total synthesis of Re LPS
Even after the great success of the total synthesis of lipid A, Kusumoto and coworkers were not satisfied and continued the challenge of more difficult synthesis of lipid A derivatives. The next, and hardest, target was Re LPS of Escherichia coli (Figure 7). Kdo disaccharide might be considered as an additional and perhaps unnecessary part of Re LPS, but in nature, lipid A never exists without Kdo molecule. At least in the case of enterobacterial LPS, the transfer of Kdo to the 6′-O-position of the intermediate lipid IVa occurs before the acylation by nonpolar fatty acids to form the complete structure of lipid A. 31 Furthermore, Re LPS and other rough-type LPS with core oligosaccharides are present as major components of LPS in all bacterial cells that have LPS, whether they are smooth- or rough-type strains. Therefore, the molecule that functions in the outer membrane to form a membrane structure, or that is recognized by the receptor to stimulate immunity, is Re LPS, or the LPS with a longer carbohydrate portion, and not lipid A. In this regard, synthetic Re LPS was highly required for the study of LPS–receptor interactions.
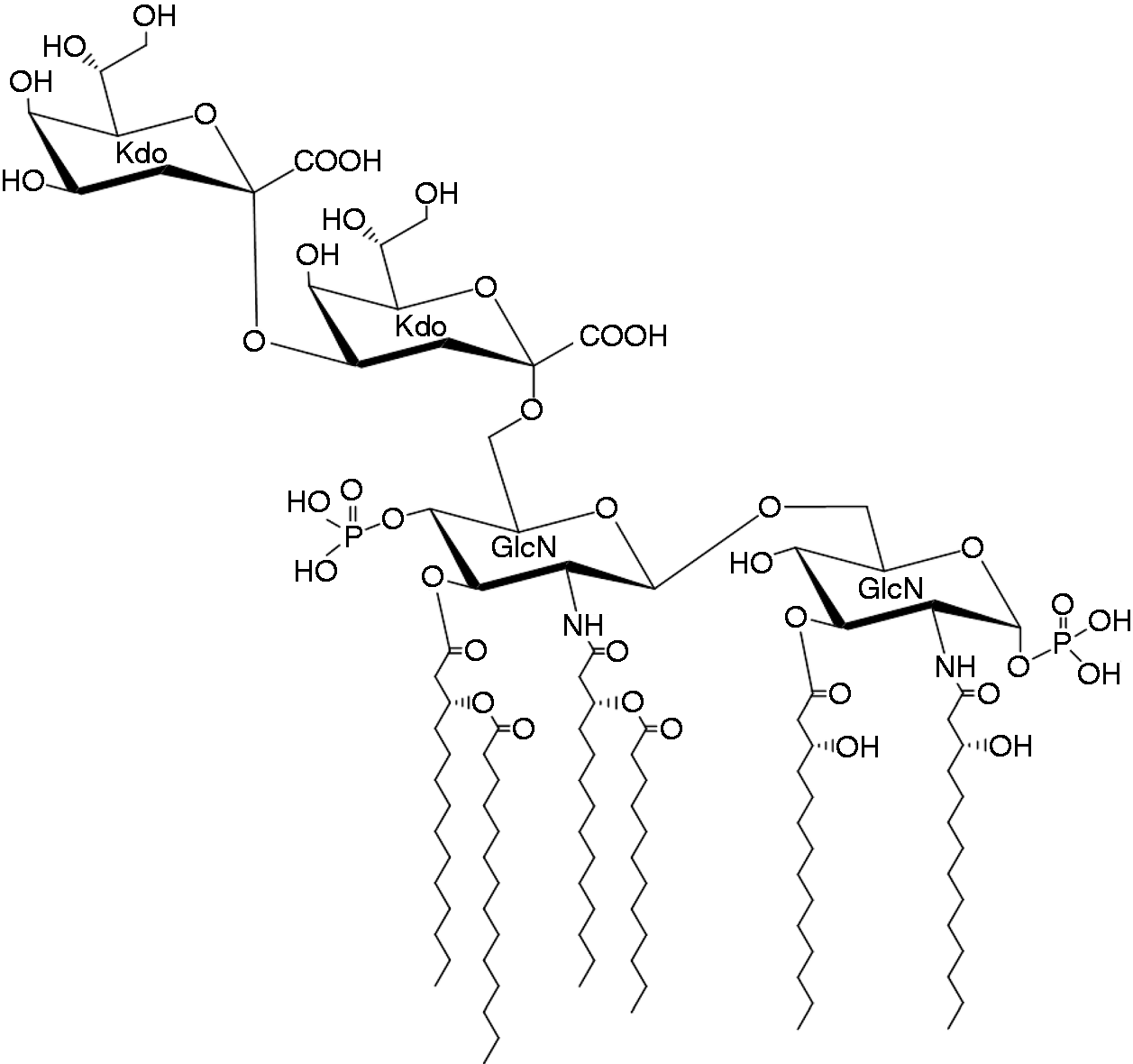
Chemical structure of E. coli Re LPS.
Kusumoto and younger coworkers, including Koichi Fukase (20 years younger than Kusumoto) and Masato Oikawa, finally completed the total synthesis of Re LPS. 41 , 42 An outline of the synthetic steps was as follows: (1) site-specific and stereospecific coupling of GlcN to form a partially acylated GlcN disaccharide; (2) introduction of acyl and acyloxyacyl groups at the 3-O and 2′-N-positions of the GlcN disaccharide, respectively; (3) stereospecific coupling of a first Kdo (Kdo-fluoride) to the 6′-O-position of the GlcN disaccharide; (4) introduction of phosphate at the 4′-O-position; (5) stereospecific coupling of a second Kdo (Kdo-fluoride) to the 4-O-position of the first Kdo; (6) removal of the protective group and phosphorylation at the glycosidic position of reducing GlcN; and (7) removal of all protective groups by hydrogenolysis. The overall yield of the 23 steps of the synthesis was 0.92%. The synthetic Re LPS showed significantly higher IL-6 inducing activity and other endotoxic activities than the synthetic lipid A molecule without Kdo. 42 This synthetic Re LPS, together with synthetic lipid A will be and should be used as standard endotoxins, which are completely free of other cellular component contamination, in various endotoxin studies.
Synthetic lipid A: an essential and ideal tool for recognition studies
There is no doubt that synthetic lipid A and its derivatives are powerful tools for endotoxin research at the molecular level, and their value was truly noticed when the specific receptor for endotoxin, TLR4, was identified at the end of the last century. 43 Kusumoto and Fukase attempted to synthesize 3H-labeled lipid A even before TLR4 had been identified because it would be very useful for the study of receptor and receptor–ligand interactions. Synthetic radiolabeled lipid A is much better than those prepared by bacteria because labeled compounds with higher specific activity can be produced. They found that 1-O-phosphorooxyethyl (PE) derivatives of lipid A and lipid IVa containing 3H in the PE group were suitable for that purpose. 44 , 45 PE derivatives were stable and, most importantly, maintained all their endotoxic activities. These radiolabeled lipid A and lipid IVa were supplied to the group of Kensuke Miyake at the University of Tokyo, and used to investigate the interaction of lipid A with MD-2 with or without TLR4.46–50 Synthetic lipid A analogs were also used to elucidate the crystal structural study of the TLR4/MD-2/LPS complex. 51 , 52 Also in these studies, the personal relationships and trust between these two groups in Osaka and Tokyo, especially between Fukase and Miyake, have been the basis of the fruitful collaboration.
Final remarks
The concept of lipid A structure and its role in immunological activities were established by competitive and collaborative investigations of German, Japanese, and American research groups as described here. Additionally, biosynthetic studies contributed greatly to confirming the proposed structure. Among these researchers, Shiba and Kusumoto played a central role, not only for determining the structure but also for unveiling the mysterious principle of endotoxic activity. This could be achieved through the spirit of Shiba and Kusumoto, which advanced their research of organic chemistry to a new and higher level, as Shiba had originally intended. Their collaboration crossed borders of research fields and of countries. They always maintained a friendly atmosphere with their collaborators and also with endotoxin researchers from Germany, Austria, Russia, and many other countries. These friendships stemmed from their honest and open-minded talent. Koichi Fukase has inherited that talent and has been developing a network of collaboration with researchers from many fields of endotoxin research. Synthetic materials always bring clear-cut results in research, especially in the field of endotoxin, because natural endotoxins are always a mixture of molecules with various kinds of fatty acids, various lengths of carbohydrates, and may contain additional immunoactive contaminants such as lipoproteins. For the future of endotoxin research, the precious knowledge and technologies of the Osaka School for the synthesis of lipid A, Re LPS, and their derivatives are highly desired to be maintained and further developed.
Footnotes
Acknowledgements
The author thanks Dr. Otto Holst for providing the opportunity to prepare this manuscript, and also thanks Drs. Haruhiko Takada, Koichi Fukase, and Shoichi Kusumoto for providing necessary materials, especially the commemorative books published at the retirement of Profs. Shozo Kotani, Tetsuo Shiba, and Shoichi Kusumoto, and for the warm and useful advice on this manuscript.
Declaration of conflicting interests
The author(s) declares no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

