Abstract
Use of norepinephrine to increase blood pressure in septic animals has been associated with increased efficiency of hepatic mitochondrial respiration. The aim of this study was to evaluate whether the same effect could be reproduced in isolated hepatic mitochondria after prolonged in vivo exposure to faecal peritonitis. Eighteen pigs were randomized to 27 h of faecal peritonitis and to a control condition (n = 9 each group). At the end, hepatic mitochondria were isolated and incubated for one hour with either norepinephrine or placebo, with and without pretreatment with the specific receptor antagonists prazosin and yohimbine. Mitochondrial state 3 and state 4 respiration were measured for respiratory chain complexes I and II, and state 3 for complex IV using high-resolution respirometry, and respiratory control ratios were calculated. Additionally, skeletal muscle mitochondrial respiration was evaluated after incubation with norepinephrine and dobutamine with and without the respective antagonists (atenolol, propranolol and phentolamine for dobutamine). Faecal peritonitis was characterized by decreasing blood pressure and stroke volume, and maintained systemic oxygen consumption. Neither faecal peritonitis nor any of the drugs or drug combinations had measurable effects on hepatic or skeletal muscle mitochondrial respiration. Norepinephrine did not improve the efficiency of complex I- and complex II-dependent isolated hepatic mitochondrial respiration [respiratory control ratio (RCR) complex I: 5.6 ± 5.3 (placebo) vs. 5.4 ± 4.6 (norepinephrine) in controls and 2.7 ± 2.1 (placebo) vs. 2.9 ± 1.5 (norepinephrine) in septic animals; RCR complex II: 3.5 ± 2.0 (placebo) vs. 3.5 ± 1.8 (norepinephrine) in controls; 2.3 ± 1.6 (placebo) vs. 2.2 ± 1.1 (norepinephrine) in septic animals]. Prolonged faecal peritonitis did not affect either hepatic or skeletal muscle mitochondrial respiration. Subsequent incubation of isolated mitochondria with norepinephrine and dobutamine did not significantly influence their respiration.
Introduction
The contribution of mitochondrial dysfunction to sepsis-related morbidity is controversial. While the presence of mitochondrial dysfunction has been demonstrated in skeletal muscle biopsies collected from septic patients within 24 h of admission to intensive care, 1 and in skeletal muscle and liver specimens in rats in a long-term, fluid-resuscitated, faecal peritonitis model, 2 others found normal skeletal muscle and hepatic mitochondrial complex functions in rat endotoxaemia and faecal peritonitis models lasting 16–24 h.3,4 Even more controversially, hepatic, but not skeletal, muscle mitochondrial dysfunction has been reported in a 24 h porcine endotoxaemia model. 5
While some of the controversies may be species-related, type and severity of sepsis model, and specific mitochondrial functions assessed, concomitant treatment may also have an influence. 6 – 10 For example, mitochondrial alterations have been found in muscle biopsies of patients on statin therapy; 7 the volatile anaesthetics halothane, isoflurane and sevoflurane have been shown to inhibit complex I of cardiac mitochondria; 8 the sedative drug propofol seems to impair mitochondrial respiration in isolated perfused guinea pig hearts; 9 and levosimendan, a relatively new inotropic and vasodilatory drug, has been associated with decreased mitochondrial membrane potential in rat liver mitochondria. 10
Catecholamines such as norepinephrine, dopamine and dobutamine are commonly used to support haemodynamics in patients with sepsis. Whilst norepinephrine increases cardiac output and oxygen delivery during sepsis, 11 this drug also has a direct effect on isolated hepatocytes by activating alpha-adrenergic receptors, increasing cytosol and intra-mitochondrial calcium levels, and stimulating dehydrogenases of the citrate cycle.12,13 In endotoxaemic pigs, hepatic mitochondrial respiration was more efficient when norepinephrine was infused to increase blood pressure as compared to endotoxaemic animals without norepinephrine. 14 While this occurred in the presence of similar hepatic oxygen delivery and consumption rates in the two groups, an effect of norepinephrine-induced improvement in oxygen availability at the microcirculatory level could not be ruled out. 14
The aim of the present study was to evaluate whether norepinephrine has a direct effect on isolated porcine hepatic mitochondria previously exposed to prolonged faecal peritonitis. We hypothesized that norepinephrine improves mitochondrial respiration under these circumstances. To address whether such effects are specific for hepatic mitochondria or the drug, we also measured skeletal muscle mitochondrial function under the same conditions and after incubation with dobutamine.
Materials and methods
The study was performed in accordance with the National Institutes of Health guidelines for the care and use of experimental animals and with the approval of the Animal Care Committee of the Canton of Bern, Switzerland.
Norepinephrine, dobutamine, yohimbine, prazosin, atenolol, propranolol and phentolamine were purchased from Sigma (Sigma Chemical Co., St Louis, MO, USA). All other chemicals used were of analytical grade.
Animal preparation
Eighteen domestic pigs weighing 41 (38–44) kg (median [range]) were fasted overnight and intramuscularly premedicated with ketamine hydrochloride 100 mg (Narketan, Vetoquinol AG, Ittigen, Switzerland), azaperone 200 mg (Stresnil, Biokema, Crissier-Lausanne, Switzerland) and atropine 0.5 mg (Atropin Sintetica S.A., Mendresino, Switzerland) before induction of anaesthesia and orotracheal intubation. All animals were ventilated (Servo i™, Maquet Critical Care, Solna, Sweden) with a time-cycled, volume-targeted mode using a tidal volume of 6–8 ml/kg, a fraction of inspired oxygen of 0.4 and a positive end-expiratory pressure of 5 cm H2O. Anaesthesia was maintained throughout the experiment with continuous intravenous infusions of ketamine hydrochloride (200–300 mg/h; Ketalar, Pfizer AG, Zurich, Switzerland), midazolam (20–30 mg/h; Roche Pharma AG, Reinach, Switzerland), and fentanyl (25–75 mcg/h; Janssen-Cilac, Baar, Switzerland). Neuromuscular blocking agents were not used. The animals were equally randomized into a non-septic control group and a faecal peritonitis group (n = 9 each group). In the peritonitis group, 1 g/kg of autologous faeces dissolved in 200 ml glucose 5% (glucose 5%, Sintetica-Bioren S.A., Couvet, Switzerland) was instilled into the abdominal cavity. Twenty-seven hours after induction of peritonitis, 14–16 g of the left liver lobe was excised before sacrificing the animals with an overdose of potassium chloride. Samples for the study of muscle mitochondria were taken in the last 14 experiments only (control, n = 7; faecal peritonitis, n = 7). Skeletal muscle samples (8–10 g) were taken at baseline and at the end of the experiment from the sternocleidomastoid muscle in 14 animals and the mitochondria were immediately isolated in order to test the respiratory activity.
Haemodynamic monitoring and fluid management
Mean arterial blood pressure (MAP), cardiac stroke volume, and mixed venous oxygen saturation (SvO2) (Vigilance II, Edwards Lifesciences, Irvine, CA, USA) were monitored continuously. Global oxygen consumption (VO2) was calculated using standard formulas.
A combination of Ringer’s lactated and glucose 50% solutions was administered continuously throughout the experiment at a combined total rate of 5 ml/kg h−1. Glucose infusion was adjusted to maintain blood glucose levels between 3.5 and 6 mmol/l.
Additional fluid boluses and vasoactive drugs were administered according to a haemodynamic protocol slightly modified from one described previously. 15 – 18 Briefly, a bolus of 50-100 ml Ringer’s lactated solution was administered when MAP decreased <50 mmHg. The procedure was repeated maximally twice per hour if MAP remained <50 mmHg and if the first bolus had resulted in an increase in stroke volume. If stroke volume did not increase, a continuous norepinephrine (Noradrenaline, Sintetica S.A., Mendrisio, Switzerland) infusion was started and increased to a maximum dose of 600 mcg/h.
Measurement of creatine kinase activity and arterial lactate concentrations
Blood samples for the measurement of lactate were taken at baseline and at the end of the experiment from the carotid artery (ABL 835 Radiometer®, Copenhagen, Denmark), and creatine kinase was measured from arterial samples at the end of the experiment (Modular P800, Roche Diagnostics, Mannheim, Germany).
Liver mitochondrial isolation
Isolation of liver mitochondria was performed immediately after tissue harvesting at 4°C using a standard procedure based on differential centrifugation. 19 The samples of liver (15 g) excised at the end of the experiment were rapidly immersed in ice-cold liver isolation buffer (mannitol 220 mmol/l, sucrose 70 mmol/l, morpholinopropane sulphonic acid 5 mmol/l, pH 7.4), minced with scissors and homogenized with an additional 10 ml/g of homogenization media (liver isolation buffer plus EDTA 2 mmol/l) in a Potter Elvehjem homogenizer with a loose-fitting Teflon® pestle (four strokes) (Schütt Homgen Plus; Schütt Labortechnik GmbH, Göttingen, Germany). The homogenate was then centrifuged for 10 min at 700 g. The supernatant was collected and centrifuged again for 10 min at 7000 g. The supernatant was discarded at this time; the pellet was then resuspended in isolation buffer and centrifuged twice for 10 min at 7000 g for further purification of the mitochondria. The pellets were then suspended in buffer at a final concentration of 50–100 mg mitochondrial protein per millilitre.
Skeletal muscle mitochondrial isolation
Skeletal muscle mitochondria were isolated as described by Hoppel et al. 20 The muscle specimens were rapidly immersed in ice-cold isolation buffer (KCl 100 mmol/l, MgSO4 10 mmol/l, morpholinopropane sulphonic acid 50 mmol/l, ethylenedinitrilotetraacetic acid 1.0 mmol/l, ATP 1.1 mmol/l, pH 7.4), transported to the laboratory and weighed. After several rinses with isolation buffer, the skeletal muscle was minced using scissors and was suspended in 10 volumes (wt/vol) of the same medium and treated with a protease (Sigma-Aldrich, St. Louis, MO, USA) 5 mg/g for 10 min at 4°C with constant stirring. The suspension was diluted with an equal volume of isolation medium supplemented to 0.2% (wt/vol) with defatted bovine serum albumin (BSA) and homogenized in a Potter Elvehjem homogenizer with a loose-fitting Teflon pestle (10 strokes). The supernatant was separated by centrifugation (10 min at 10000 g) and the pellet was resuspended in BSA-supplemented isolation medium (10 ml/g tissue). The suspension was centrifuged for 10 min at 350 g, the supernatant was filtered through two layers of gauze and the mitochondria were sedimented at 7700 g for 10 min. The mitochondria were subjected to two additional washes using 5 ml BSA-supplemented muscle isolation buffer/g tissue and 2.5 ml of 100 mmol/l KCl, 50 mmol/l morpholinopropane sulphonic acid, and 0.5 mmol/l ethylene glycol tetraacetic acid (EGTA), pH 7.4, buffer/g muscle and finally resuspended in ∼1.0 ml of 100 mmol/l KCl, 50 mmol/l morpholinopropane sulphonic acid and 0.5 mmol/l EGTA, pH 7.4.
Experimental protocol for hepatic and muscle mitochondrial respiration
Mitochondria from each liver biopsy sample were divided into six aliquots and incubated on ice under different conditions: the first aliquot was incubated with vehicle; the second aliquot was incubated with norepinephrine (10 µ
Mitochondria from each muscle biopsy sample were divided into 13 groups and incubated on ice. The first group was incubated with vehicle. The second and third groups were incubated with norepinephrine (10 µ
Determination of mitochondrial oxygen consumption by high-resolution respirometry
Protein concentration was determined using the Quant-iT™ protein assay kit (Qubit fluorometer, Invitrogen, Basel, Switzerland) according to the manufacturer’s instructions. Respiratory rates were determined at a final mitochondrial protein concentration of 0.4 mg/ml. Respiration was measured at 37°C in 2 ml glass chambers using the High Resolution Oxygraph (OROBOROS; Oxygraph-2 k, Graz, Austria). The medium used for respiration measurements consisted of 110 m
Statistics
Normal distribution was assessed by the Kolmogorov-Smirnov Test. In case of normal distribution, ANOVA for repeated measurements was applied using one (drug, for hepatic mitochondrial respiration; time, for haemodynamics) or two (drug and time, for muscle mitochondrial respiration) within-group factor(s) and one between-group factor (peritonitis vs. control). When data were not normally distributed (mitochondria), placebo samples at baseline from septic and control animals were compared using the Mann-Whitney test, and placebo samples at baseline and in the end (muscle) in each group separately using Wilcoxon test. Afterwards, the effect of drugs at each time point was assessed using non-parametric ANOVA for repeated measurements (Friedman test). A P- value of <0.05 was considered significant. All analyses were performed using the SPSS® 15.0 software package (SPSS Inc., Chicago, IL, USA).
Results
Anaesthetics and analgesics
Administered cumulative doses of fentanyl (controls 4.06 ± 1.27 mg, septic animals 4.73 ± 2.11 mg; p = 0.404), midazolam (controls 803 ± 136 mg, septic animals 731 ± 168 mg; p = 0.311) and ketamine (controls 9.04 ± 1.34 g, septic animals 8.44 ± 3.13 g; P = 0.585) were not different between the groups.
Systemic haemodynamic, creatine kinase activity and arterial lactate concentrations
During the experiment, MAP and stroke volume decreased and heart rate increased in septic animals, while MAP increased and stroke volume and heart rate remained unchanged in controls (Figure 1a–c). In five septic animals a median (range) total dose of norepinephrine of 883 (490-2913) mcg was administered during the study. Systemic oxygen consumption was maintained in both groups (Figure 1d). Central venous pressure (CVP; Figure 1e) and pulmonary artery occlusion pressure (PAOP; Figure 1f) remained unchanged in both groups. Fluid balance was +1047 ± 1108 ml in controls and +4947 ± 1235 ml in septic animals (P < 0.001).
Changes in (a) mean arterial pressure (MAP), (b) stroke volume, (c) heart rate, (d) systemic oxygen consumption (VO2), (e) central venous pressure (CVP) and (f) pulmonary artery occlusion pressure (PAOP) from before induction of sepsis (baseline) to the end of the protocol in non-septic controls (solid line) and in septic animals (dashed line). P value: MANOVA time x group interaction. Data points are group mean values, error bars indicate ± one standard deviation.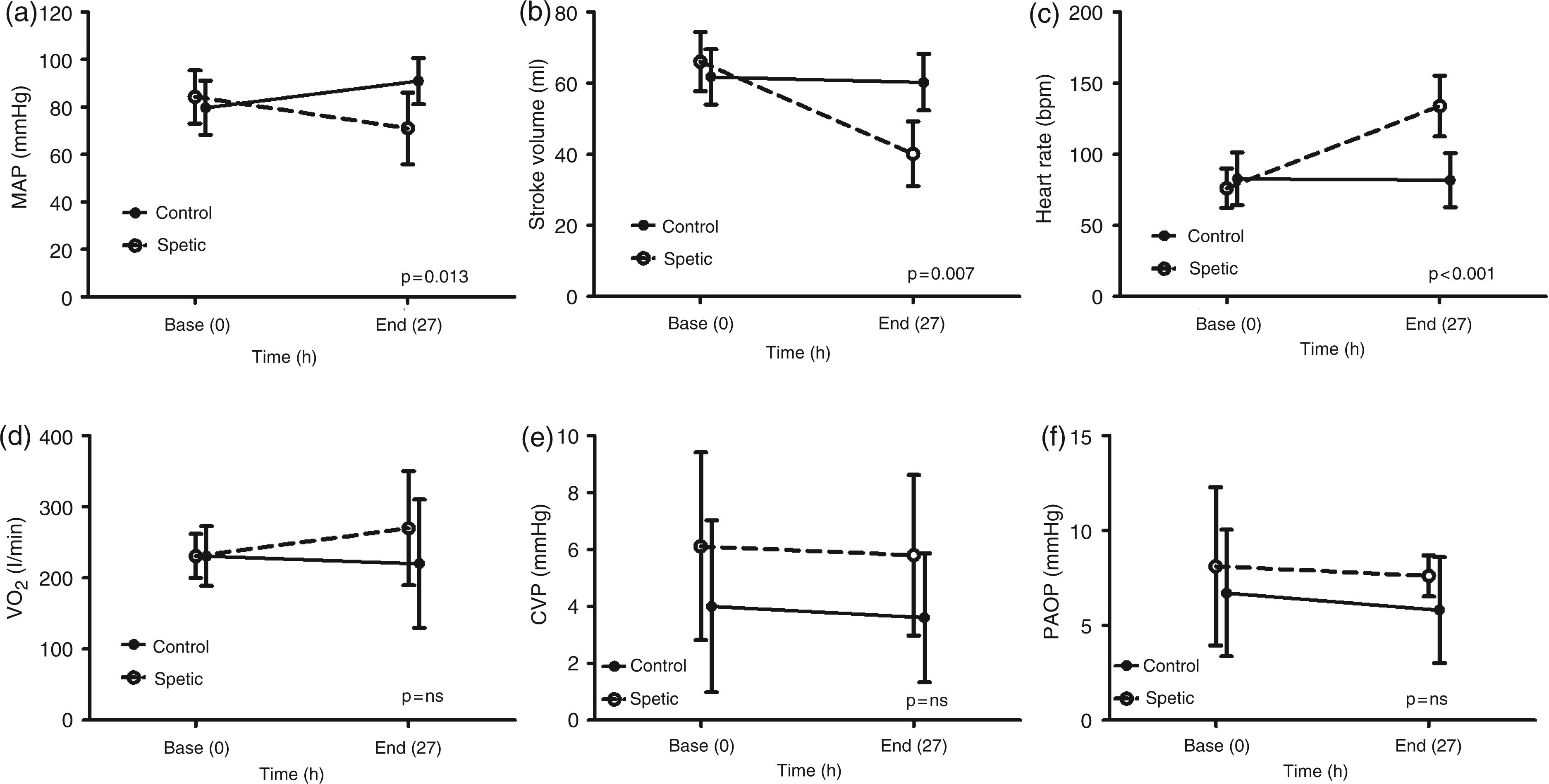
Creatine kinase activities at the end of the experiment were 622 (465;10154) U/l in controls and 808 (245;1167) U/l in septic animals (p = 0.967). Lactate levels in arterial blood changed from 0.76 ± 0.20 mmol/l at baseline to 0.89 ± 0.24 mmol/l at the end of the experiment in control animals, and from 0.90 ± 0.46 mmol/l at baseline to 1.89 ± 1.01 mmol/l at the end of the experiment in septic animals (P = 0.074, time-group interaction MANOVA).
Mitochondrial function
A representative diagram of measurement of respiration rates in isolated hepatic/muscle mitochondria at an experimental density of 0.4 mg of mitochondrial protein/ml using high-resolution respirometry is shown in Figure 2. The oxygen flux (solid line) represents the directly calculated oxygen use and is expressed as pmol/s/mg of mitochondrial protein. Oxygen concentration (dashed line) decreases over time as mitochondria use the available oxygen.
Representative diagram of measurement of respiration rates in isolated liver/muscle mitochondria using high-resolution respirometry (Oxygraph-2 k) and DatLab 4.2® software for data acquisition and analysis. Oxygen consumption is expressed as pmol/second/mg protein. State 3: active respiration after addition of ADP. State 4: respiration after consumption of ADP. The ratio between state 3 and state 4 was calculated as the respiratory control ratio (RCR; state 3/state 4). Oxygen concentration (dashed line) decreases in time as mitochondria use the available oxygen. The oxygen flux (solid line) represents the directly calculated oxygen use (negative time derivative of oxygen concentration).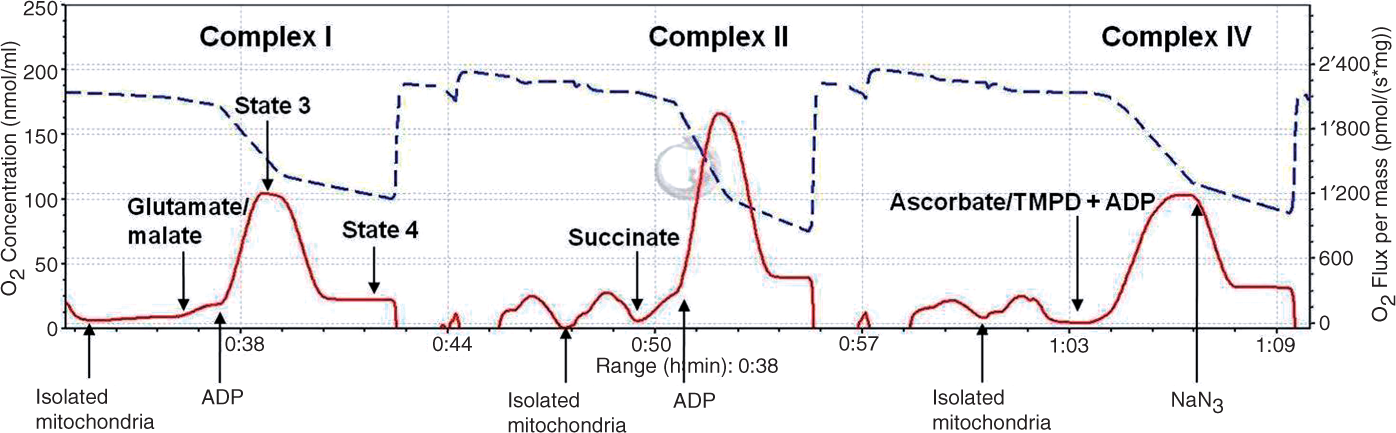
Complex I-dependent hepatic mitochondrial respiration

Values are mean ± SD for state 3 and state 4 and median (range) for respiratory control ratio (RCR).
Comparison of complex I-dependent isolated hepatic mitochondrial respiration from control and septic animals after incubation with norepinephrine and antagonists prazosin and yohimbine. State 3 and 4 oxygen consumption is expressed as pmol/s/mg protein. RCR, respiratory control ratio (oxygen consumption of state 3/state 4). No significant differences were found between the placebo and any of the drugs or drug combinations (P > 0.05 for all comparisons).
Complex II-dependent hepatic mitochondrial respiration
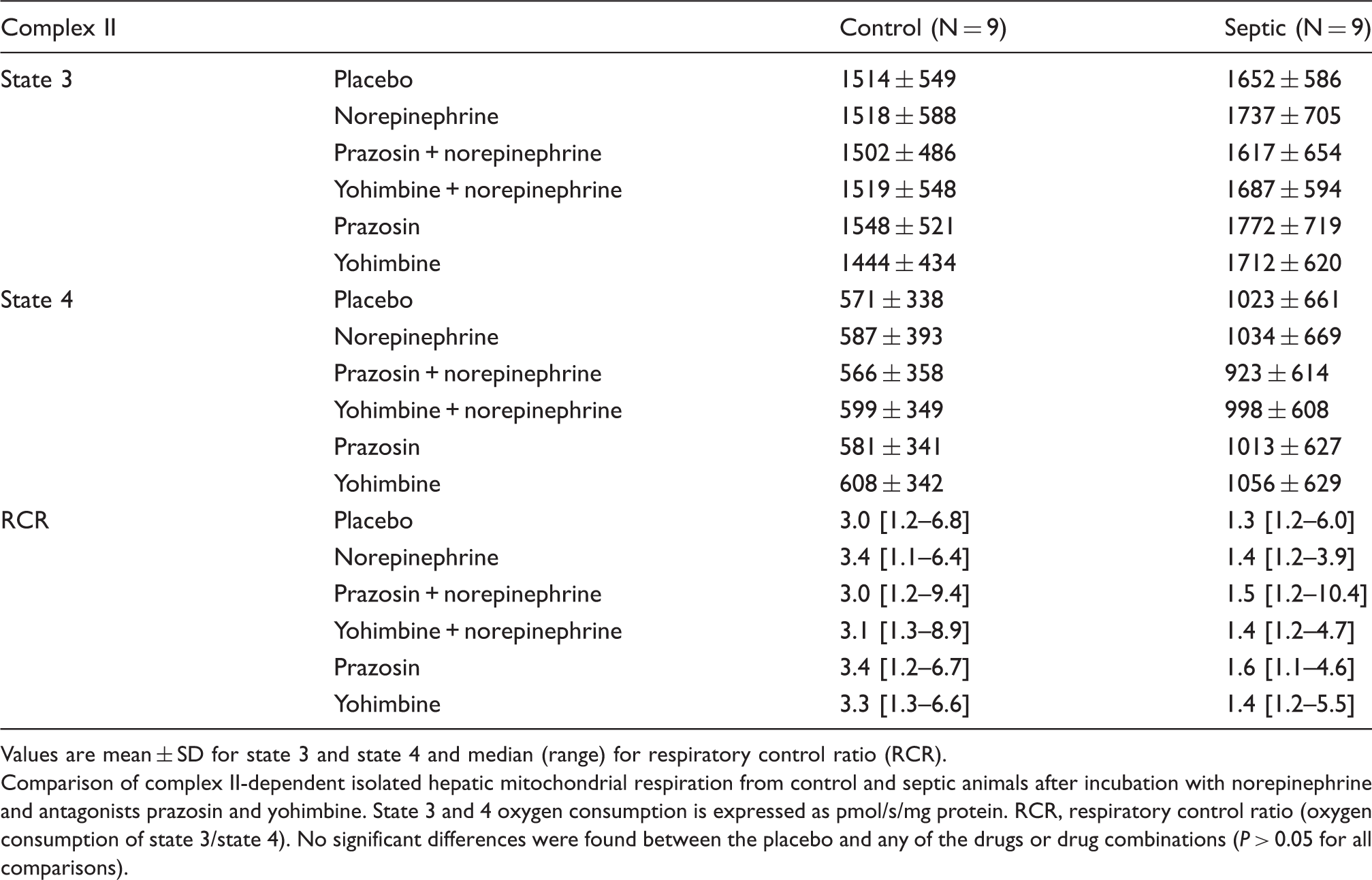
Values are mean ± SD for state 3 and state 4 and median (range) for respiratory control ratio (RCR).
Comparison of complex II-dependent isolated hepatic mitochondrial respiration from control and septic animals after incubation with norepinephrine and antagonists prazosin and yohimbine. State 3 and 4 oxygen consumption is expressed as pmol/s/mg protein. RCR, respiratory control ratio (oxygen consumption of state 3/state 4). No significant differences were found between the placebo and any of the drugs or drug combinations (P > 0.05 for all comparisons).
Complex IV-dependent hepatic mitochondrial respiration
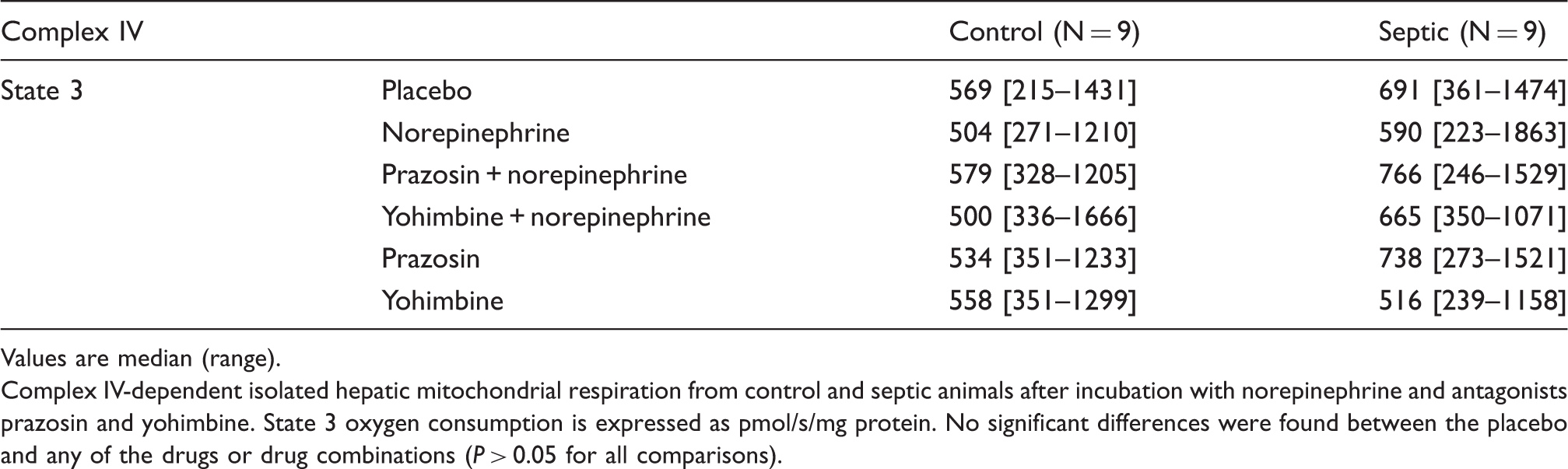
Values are median (range).
Complex IV-dependent isolated hepatic mitochondrial respiration from control and septic animals after incubation with norepinephrine and antagonists prazosin and yohimbine. State 3 oxygen consumption is expressed as pmol/s/mg protein. No significant differences were found between the placebo and any of the drugs or drug combinations (P > 0.05 for all comparisons).
Complex I-dependent muscle mitochondrial respiration
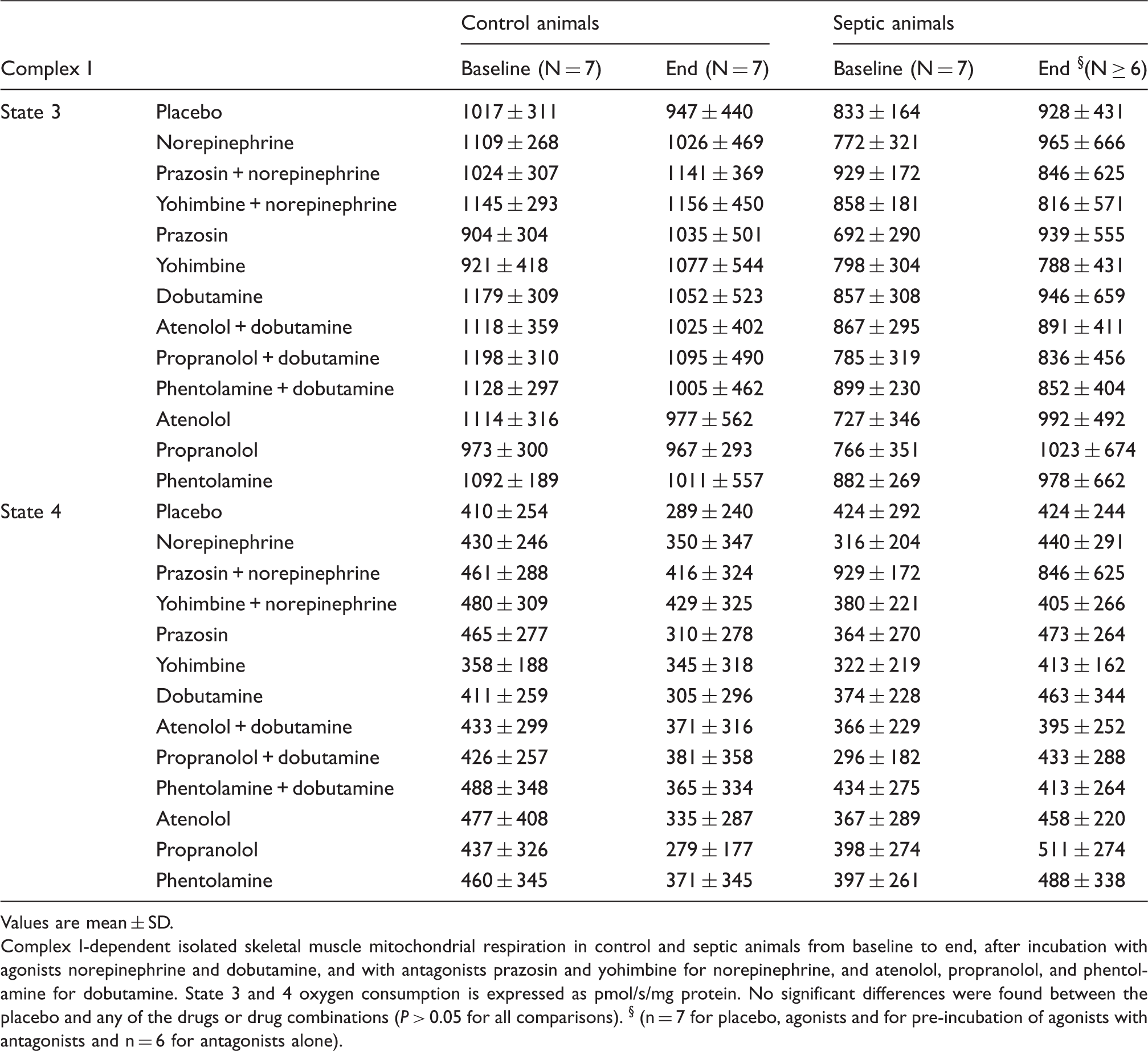
Values are mean ± SD.
Complex I-dependent isolated skeletal muscle mitochondrial respiration in control and septic animals from baseline to end, after incubation with agonists norepinephrine and dobutamine, and with antagonists prazosin and yohimbine for norepinephrine, and atenolol, propranolol, and phentolamine for dobutamine. State 3 and 4 oxygen consumption is expressed as pmol/s/mg protein. No significant differences were found between the placebo and any of the drugs or drug combinations (P > 0.05 for all comparisons). § (n = 7 for placebo, agonists and for pre-incubation of agonists with antagonists and n = 6 for antagonists alone).
Complex II-dependent muscle mitochondrial respiration
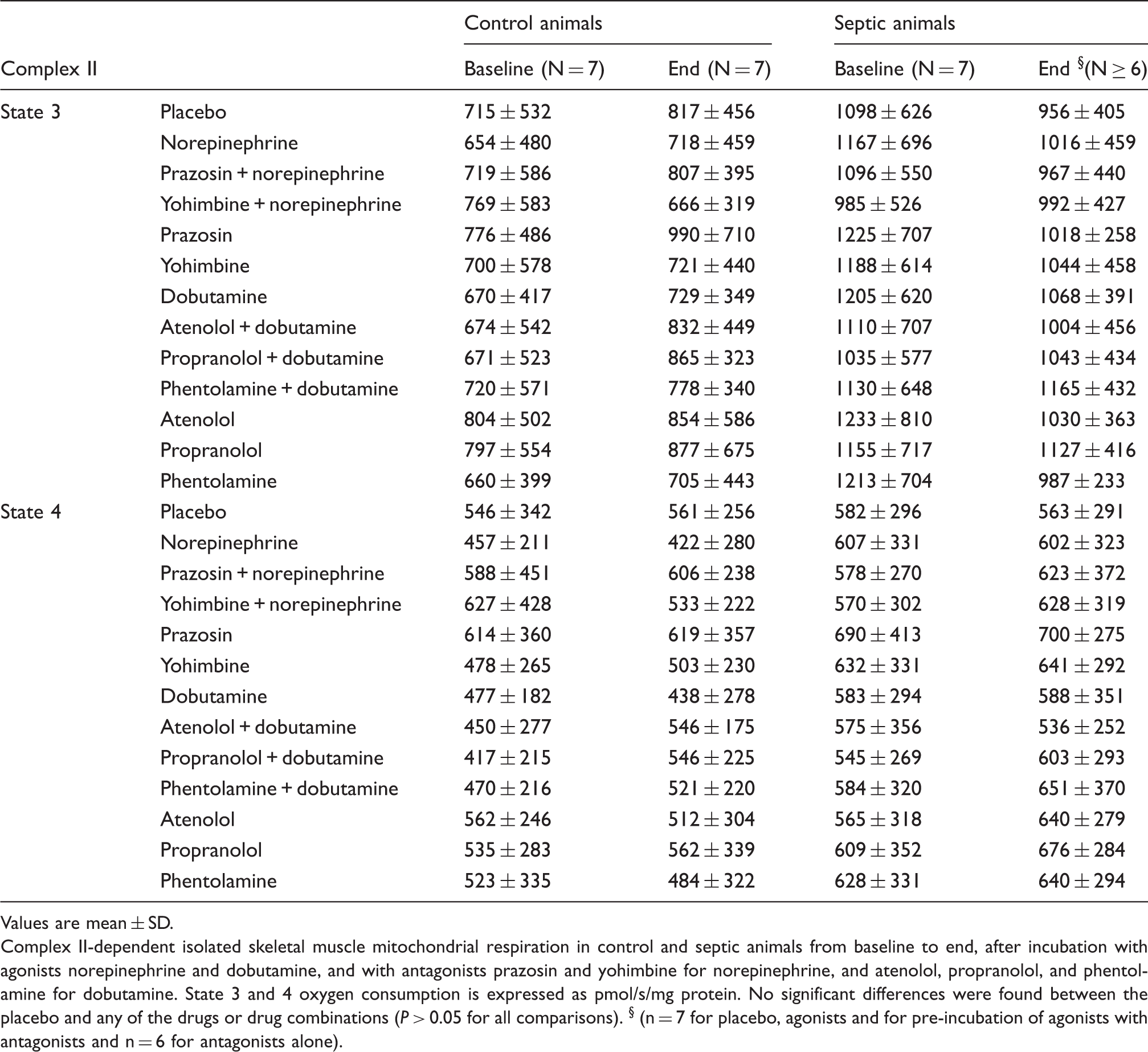
Values are mean ± SD.
Complex II-dependent isolated skeletal muscle mitochondrial respiration in control and septic animals from baseline to end, after incubation with agonists norepinephrine and dobutamine, and with antagonists prazosin and yohimbine for norepinephrine, and atenolol, propranolol, and phentolamine for dobutamine. State 3 and 4 oxygen consumption is expressed as pmol/s/mg protein. No significant differences were found between the placebo and any of the drugs or drug combinations (P > 0.05 for all comparisons). § (n = 7 for placebo, agonists and for pre-incubation of agonists with antagonists and n = 6 for antagonists alone).
Complex IV-dependent muscle mitochondrial respiration
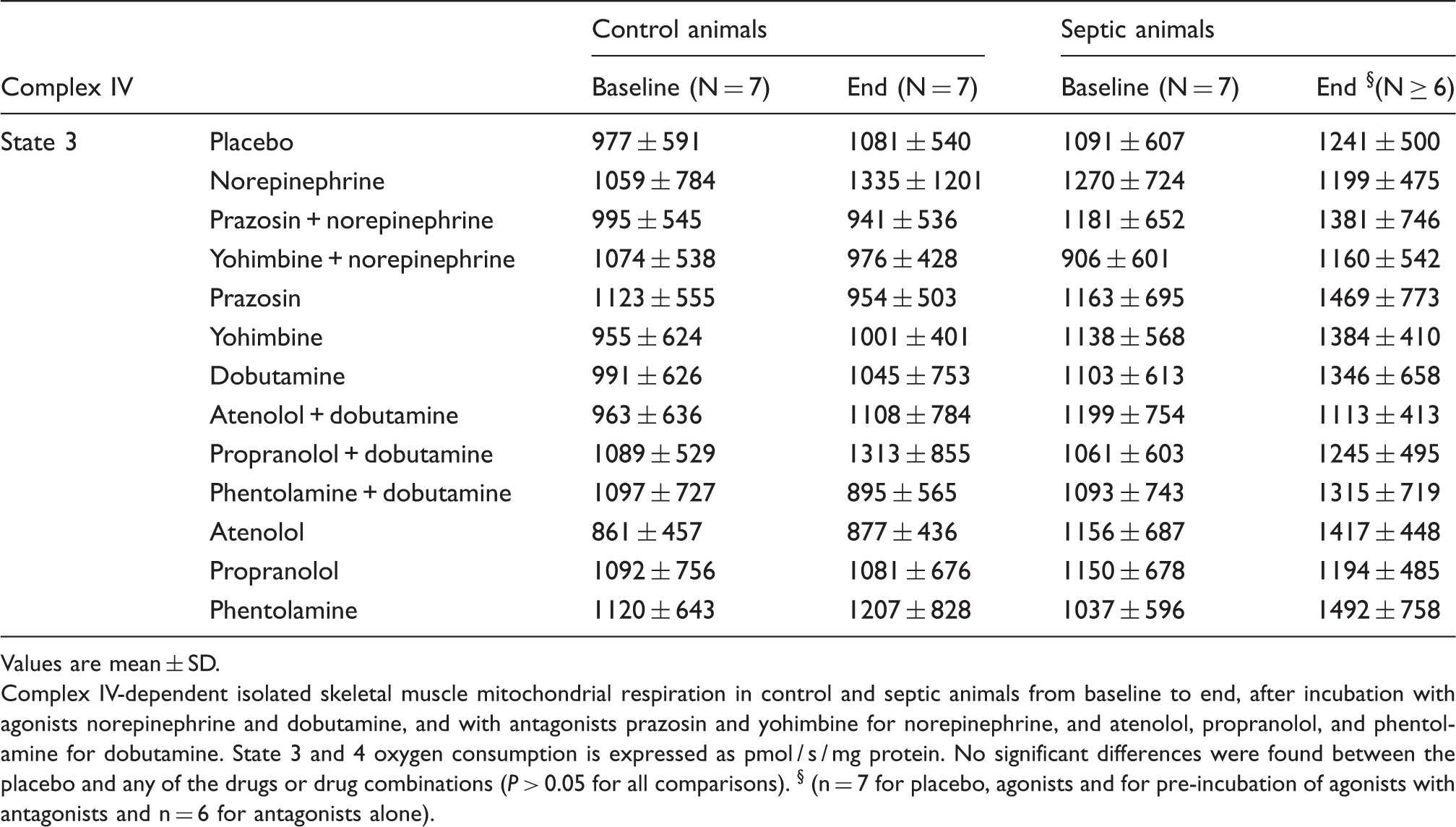
Values are mean ± SD.
Complex IV-dependent isolated skeletal muscle mitochondrial respiration in control and septic animals from baseline to end, after incubation with agonists norepinephrine and dobutamine, and with antagonists prazosin and yohimbine for norepinephrine, and atenolol, propranolol, and phentolamine for dobutamine. State 3 and 4 oxygen consumption is expressed as pmol / s / mg protein. No significant differences were found between the placebo and any of the drugs or drug combinations (P > 0.05 for all comparisons). § (n = 7 for placebo, agonists and for pre-incubation of agonists with antagonists and n = 6 for antagonists alone).
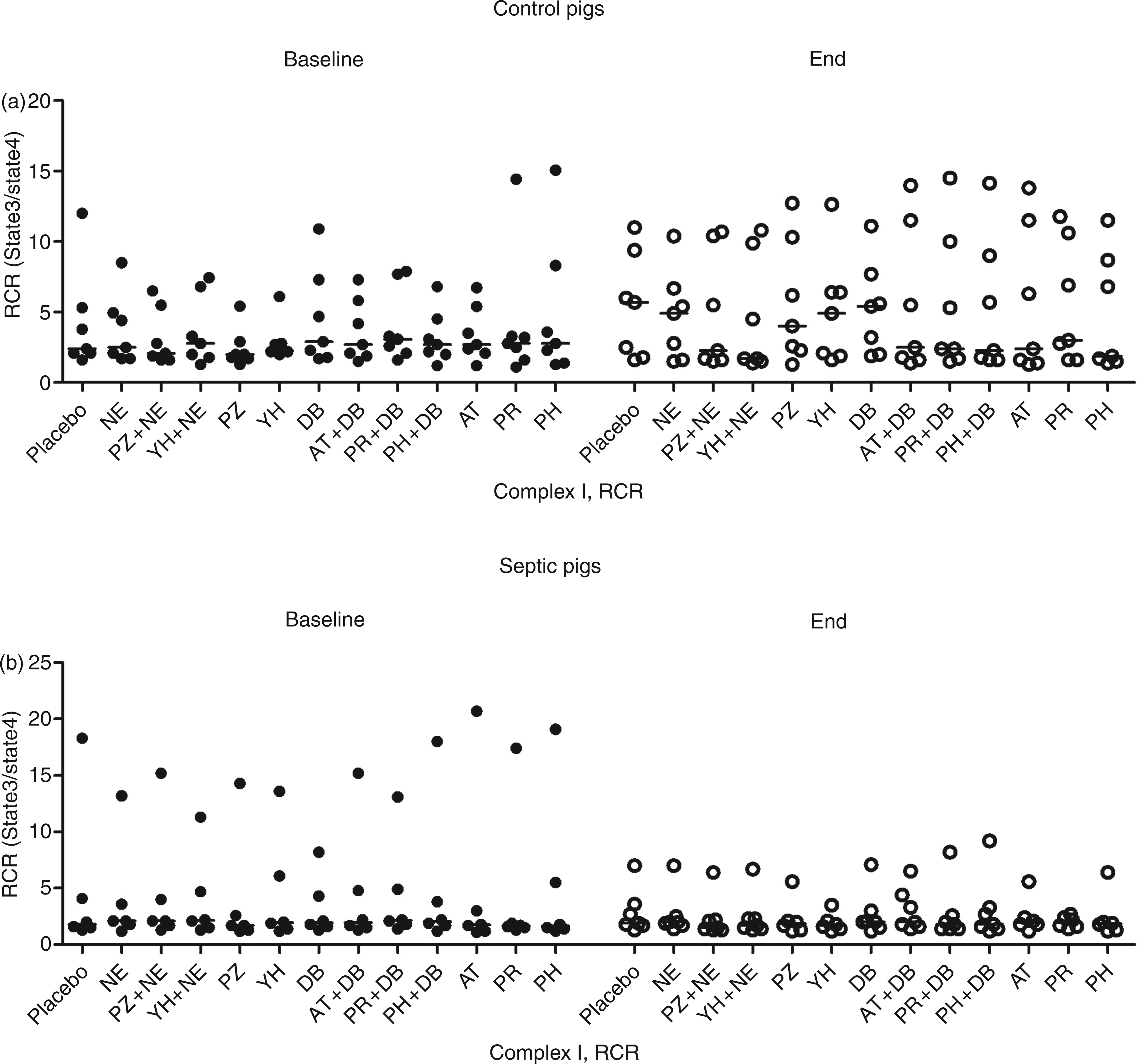
Comparison of complex I-dependent skeletal muscle mitochondrial respiratory control ratio in (a) control and (b) septic animals from baseline (filled circles) to end (open circles), after incubation with agonists norepinephrine and dobutamine, and with antagonists prazosin and yohimbine for norepinephrine and atenolol, propranolol, and phentolamine for dobutamine. No significant differences were found between the placebo and any of the drugs or drug combinations (P > 0.05 for all comparisons). Horizontal lines represent median values. NE, norepinephrine; PZ, prazosin; YH, yohimbine; DB, dobutamine; AT, atenolol; PR, propranolol; PH, phentolamine; RCR, Respiratory control ratio (oxygen consumption of state 3/state 4).
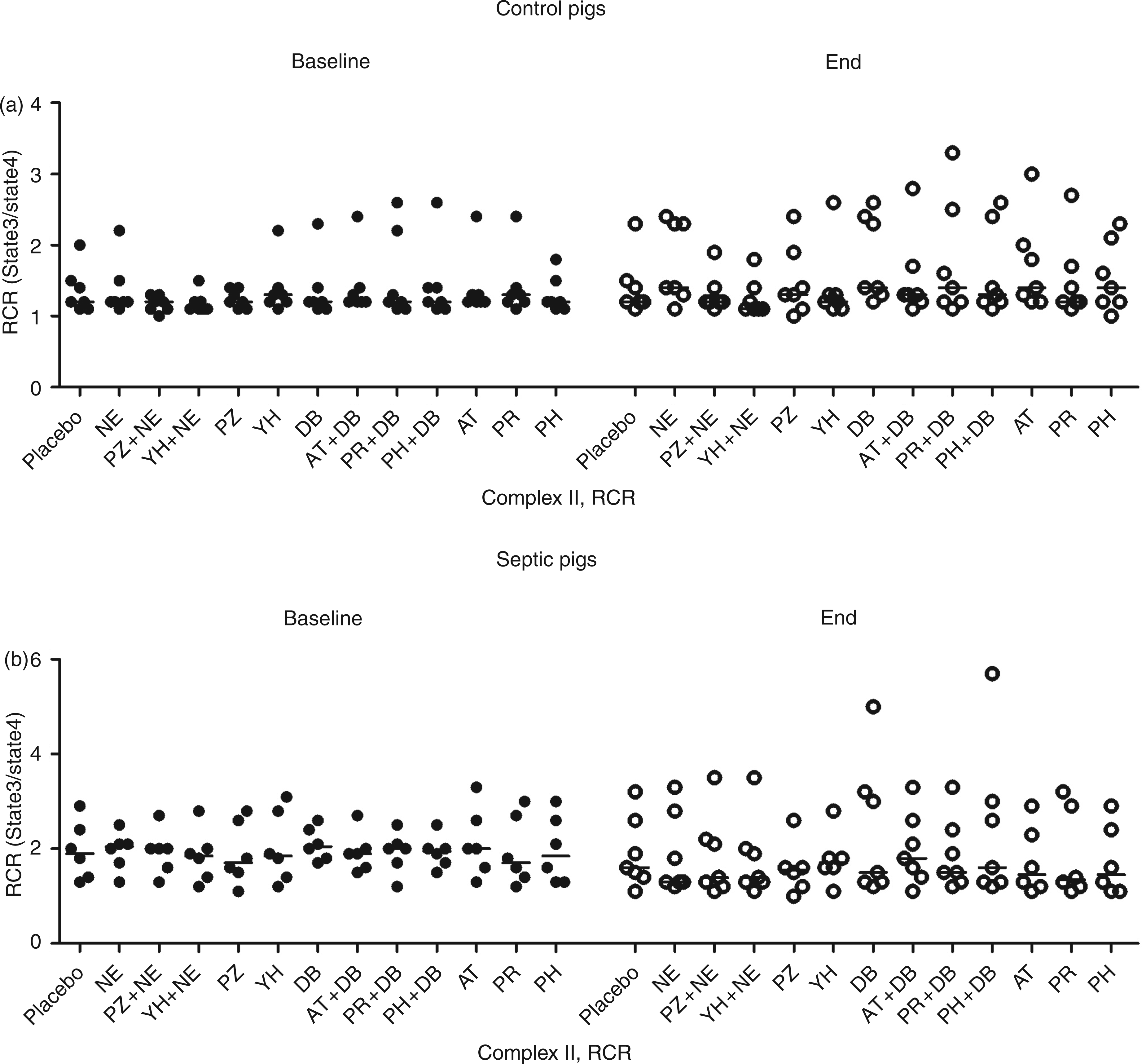
Comparison of complex II-dependent skeletal muscle mitochondrial respiratory control ratio in (a) control and (b) septic animals from baseline (filled circles) to end (open circles), after incubation with agonists norepinephrine and dobutamine, and with antagonists prazosin and yohimbine for norepinephrine and atenolol, propranolol, and phentolamine for dobutamine. No significant differences were found between the placebo and any of the drugs or drug combinations (P > 0.05 for all comparisons). Horizontal lines represent median values. NE, norepinephrine; PZ, prazosin; YH, yohimbine; DB, dobutamine; AT, atenolol; PR, propranolol; PH, phentolamine; RCR, Respiratory control ratio (oxygen consumption of state 3/state 4).
Effect of norepinephrine on isolated liver mitochondrial respiration
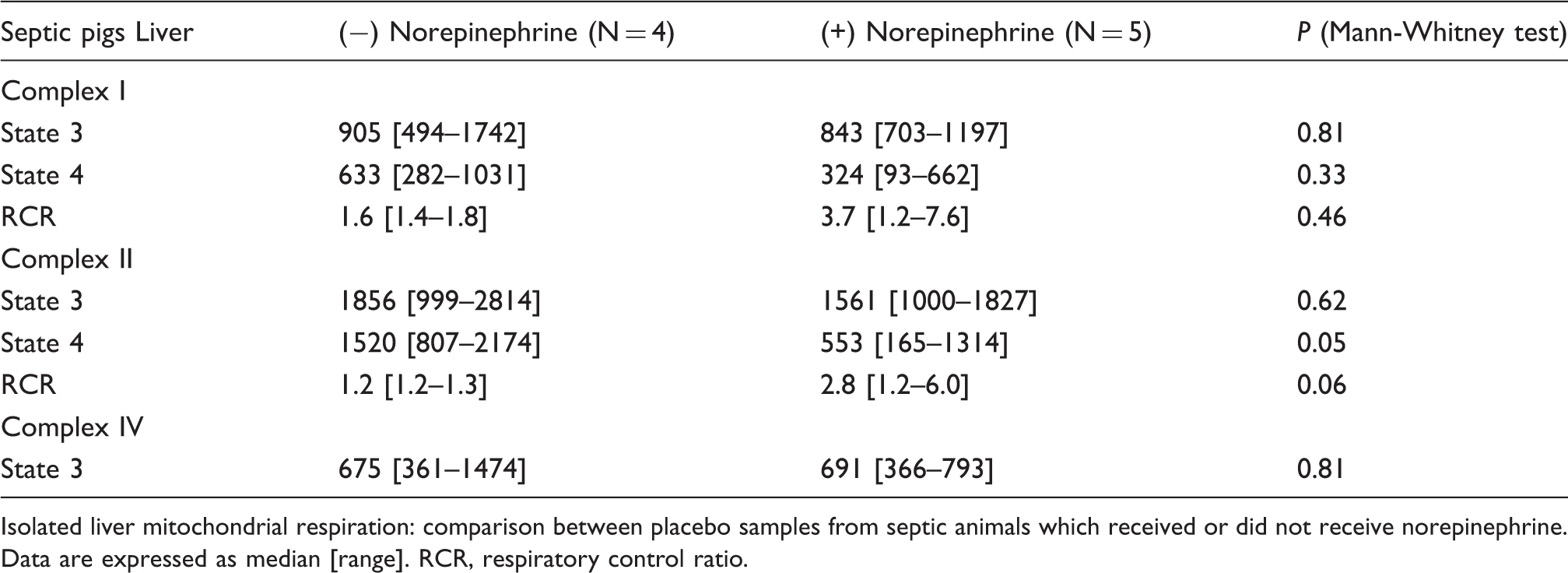
Isolated liver mitochondrial respiration: comparison between placebo samples from septic animals which received or did not receive norepinephrine. Data are expressed as median [range]. RCR, respiratory control ratio.
Effect of norepinephrine on isolated skeletal muscle mitochondrial respiration
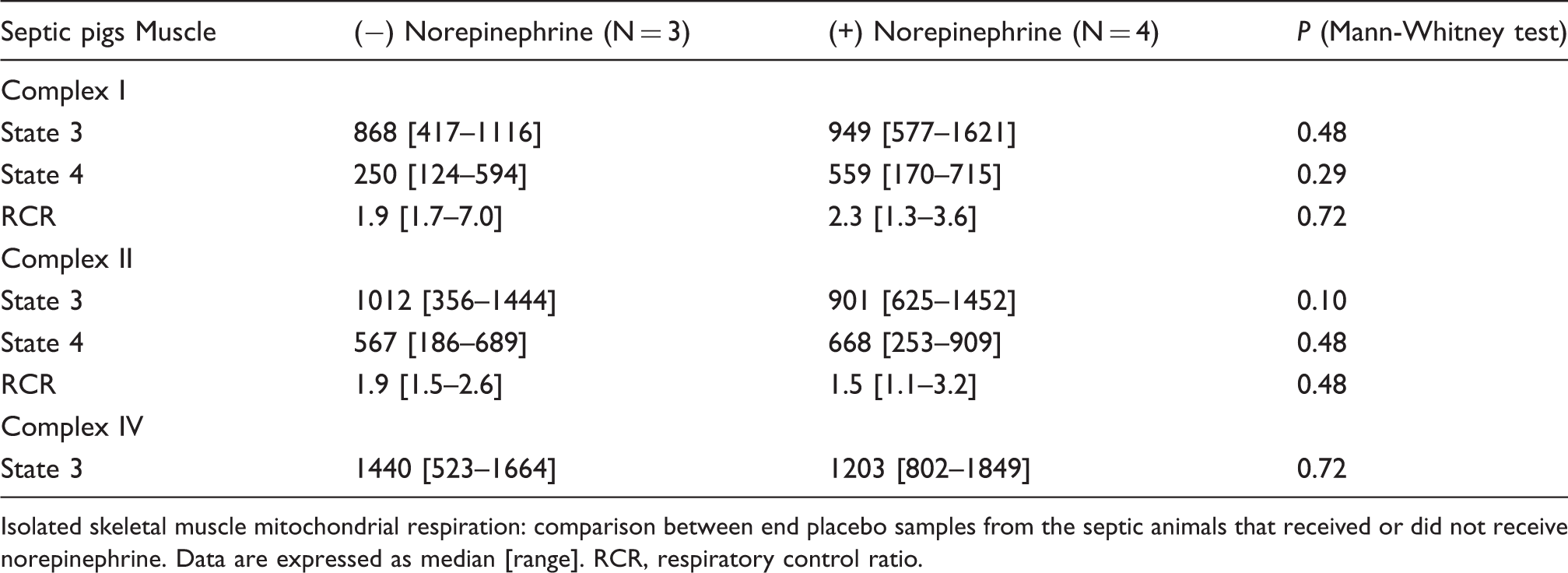
Isolated skeletal muscle mitochondrial respiration: comparison between end placebo samples from the septic animals that received or did not receive norepinephrine. Data are expressed as median [range]. RCR, respiratory control ratio.
Similarly, norepinephrine had no effect on hepatic isolated mitochondrial respiration, either in control animals or in septic animals [complex I-state 3: 917 ±421 pmol/s per mg (placebo) vs. 914 ± 438 pmol/s per mg (norepinephrine) in controls; 978 ± 371 pmol/s per mg (placebo) vs. 1007 ± 410 pmol/s per mg (norepinephrine) in septic animals; complex I-state 4: 302 ±238 pmol/s per mg (placebo) vs. 292 ± 246 pmol/s per mg (norepinephrine) in controls; 510 ± 290 pmol/s per mg (placebo) vs. 446 ± 290 pmol/s per mg (norepinephrine) in septic animals; respiratory control ratio: 5.6 ± 5.3 (placebo) vs. 5.4 ± 4.6 (norepinephrine) in controls; 2.7 ± 2.1(placebo) vs. 2.9 ± 1.5 (norepinephrine) in septic animals (all P > 0.05)] (Table 1).
The maximal hepatic mitochondrial respiration (state 3) for complex II was 1514 ± 549 pmol/s per mg (placebo) vs. 1518 ± 588 pmol/s per mg (norepinephrine) in controls; 1652 ± 586 pmol/s per mg (placebo) vs. 1737 ± 705 pmol/s per mg (norepinephrine) in septic animals, state 4 was 571 ± 338 pmol/s per mg (placebo) vs. 587 ± 393 pmol/s per mg (norepinephrine) in controls; 1023 ± 661 pmol/s per mg (placebo) vs. 1034±669 pmol/s per mg (norepinephrine) in septic animals, and the respiratory control ratio was 3.5 ± 2.0 (placebo) vs. 3.5 ± 1.8 (norepinephrine) in controls; 2.3 ± 1.6 (placebo) vs. 2.2 ± 1.1 (norepinephrine) in septic animals (all P > 0.05) (Table 2).
Complex IV-dependent hepatic mitochondrial respiration was 708 ± 414 pmol/s per mg (placebo) vs. 623 ± 348 pmol/s per mg (norepinephrine) in controls, and 718 ± 324 pmol/s per mg (placebo) vs. 740 ± 468 pmol/s per mg (norepinephrine) in septic animals (P > 0.05) (Table 3).
Neither norepinephrine nor dobutamine, nor any of the drug combinations, had measurable effects on skeletal muscle mitochondrial respiration (Figures 3 and 4, Tables 4–6).
Discussion
This study provided two main findings: 1) 27 h of faecal peritonitis had no effect on respiration of isolated hepatic and skeletal muscle mitochondria; and 2) invitro incubation of isolated hepatic mitochondria with norepinephrine after faecal peritonitis neither improved nor worsened their respiration.
Our results contrast with our previous finding of increased efficiency of hepatic mitochondrial respiration after norepinephrine infusion in endotoxaemic animals, 14 and suggest that norepinephrine has no direct effect on hepatic mitochondrial respiration. This is supported by previous findings on lack of effect of norepinephrine on hepatic mitochondrial respiration in mitochondria previously exposed to endotoxin in vitro. 22 As prolonged exposure to faecal peritonitis had no effect on hepatic or skeletal muscle respiration per se, the potential effect of norepinephrine on sepsis-induced deterioration of mitochondrial respiration cannot be determined from the present results. In the present study, in vivo administration of norepinephrine to five out of nine septic animals was associated with a significant reduction in isolated liver complex II-dependent state 4 respiration and an insignificant increase in complex II-dependent RCR. Regueira et al. previously reported that a continuous infusion of norepinephrine to endotoxaemic pigs was associated with improved complex I- and II-dependent RCRs. 14 These, and other data, 22 suggest that potential effects of norepinephrine on mitochondrial respiration in sepsis are indirect.
Whether, how, and to what extent sepsis may alter mitochondrial function is a matter of debate. 23 – 25 We previously reported well-preserved complex I- and II-dependent mitochondrial respiration in muscle and liver in pigs subjected to 24 h of either endotoxin or faecal peritonitis, 26 despite the presence of organ failure and substantial mortality. Similarly, in a rat caecal ligation and puncture model of sepsis lasting 16 h, the authors found no significant changes in complex I- and complex II-dependent hepatic mitochondrial respiration. 3 Furthermore, hepatic and skeletal muscle mitochondrial function of rats remained unaffected 18–24 h after treatment with either intravenous endotoxin or faecal peritonitis. 4 In contrast, others reported inhibition of complex I-dependent mitochondrial respiration in skeletal muscle biopsies collected from septic patients within 24 hours of intensive care unit admission. 1 The same group demonstrated an association between sepsis and impaired complex I-dependent skeletal muscle and hepatic mitochondrial respiration in a long-term, fluid-resuscitated, faecal peritonitis rat model. 2
In the present study we used a large animal model of sepsis induced by faecal peritonitis that shares many of the features of clinical sepsis, but did not result in organ failure or mortality. As we measured complex I- and II-dependent hepatic and skeletal muscle mitochondrial respiration by adding physiological substrates to the isolated mitochondria, we cannot exclude sepsis-induced impairment of mitochondrial function by mechanisms not tracked by the methods used in the present study. Such mechanisms may include pyruvate dehydrogenase inhibition, 27 poly-(ADP-ribose) polymerase (PARP-1) activation 28 and decreased tissue mitochondrial density. 29
There are several potential reasons for the conflicting findings with regard to mitochondrial function in the pathophysiology of sepsis. These include differences in sepsis models (endotoxaemia,5,30 faecal peritonitis,2,26 caecal ligation and puncture,3,31 length of observation period and severity of sepsis), species investigated (humans, 1 pigs,5,26 rats, 2,3,4 rabbits, 32 guinea pigs 33 ), concomitantly administered medication (e.g. anaesthetics), and methods used to prepare samples and assess mitochondrial function (isolated mitochondria vs. intact tissue samples, mitochondrial isolation procedures—Percoll gradients31,34 vs. differential centrifugation5,26,35—spectrophotometric and reverse-phase HPLC techniques 2 vs. oxygen consumption assessment by Clark-type oxygen electrodes3,22 or high-resolution oxygraph, 14 etc.).
While it has been shown that norepinephrine may activate hepatocellular receptors and increase intra-mitochondrial calcium levels and enzyme activities,12,13 our results do not provide evidence for a direct effect of norepinephrine on (isolated) mitochondria. It should be mentioned that in the present study, the norepinephrine incubation time was short when compared to an infusion of norepinephrine for several hours. We also explored effects of norepinephrine and dobutamine on skeletal muscle mitochondria and found no influence of either drug on any of the complexes of the mitochondrial respiratory chain. However, in a recent in vitro study, catecholamines (dopamine, dobutamine, norepinephrine) were shown to impair the efficiency of glutamate-dependent native liver mitochondrial respiration without significant alterations in state 3 and state 4 respiration rates and any of the other complexes. 22 The use of higher doses of catecholamines and—to a lesser extent—a different technique for the measurement of respiration (Clark-type electrode) may offer some explanations for the different findings. On the other hand, in vitro incubation of catecholamines (dopamine, dobutamine) did not alter native muscle mitochondrial respiration. 36
Ketamine, midazolam and fentanyl may have inhibitory effects on mitochondrial complex activity and oxidative metabolism. 37 – 39 However, in the present study, cumulative administered doses of anaesthetics and analgesics were not different between controls and septic animals.
Limitations of our study are the relatively mild disease we produced in our animals, the lack of dose responses, and the short duration of incubation for norepinephrine and dobutamine. Further, we evaluated complex functions in isolated mitochondria only. It is conceivable that investigating whole cells or muscle fibres and exploring other components of mitochondrial functions would have revealed hidden effects of norepinephrine. In the present study, cardiac stroke volume decreased in septic animals, whereas it remained stable in controls. Myocardial depression and decreased response to volume administration have been described in septic patients.40,41 Although fluid was administered based on the stroke volume response using an established haemodynamic protocol with resulting large positive fluid balance, we cannot exclude that an even more aggressive fluid resuscitation strategy would have resulted in less reduction of stroke volume in septic animals. However, as shown previously, high loading with fluids from the start of experimental sepsis—while preventing a decrease in stroke volume—increases mortality. 26
We conclude that 27 h of faecal peritonitis—resulting in sepsis without shock or organ failure—did not affect hepatic and skeletal muscle respiration of isolated mitochondria and that consecutive incubation with norepinephrine and dobutamine neither improved nor caused deterioration in the efficiency of mitochondrial respiration.
Footnotes
Acknowledgements
The authors are grateful to Jeannie Wurz for editorial support.
