Abstract
Activation of NF-κB has been reported to play a key role in causing endotoxin-induced hepatic damage through enhanced production of reactive oxygen species and pro-inflammatory mediators. In this context, the potential of polyphenolic phytochemicals in preventing endotoxin-induced liver damage remains unclear. Here, we demonstrate that catechin and quercetin have the potential to down-regulate the initial signalling molecule NF-κB which may further inhibit the downstream cascade including TNF-α and NO. These results were confirmed using N-nitro-
Keywords
Introduction
Endotoxin/LPS acts as a potent signalling molecule and is critically involved in the development of a broad spectrum of pathophysiological reactions like fever, hypoxia, intravascular coagulation, tachycardia and tachypnea, ultimately leading to sepsis with consequent multiple organ failure.1,2 Lipopolysaccharide, a major structural and functional component of the outer membrane of Gram-negative bacteria, is considered the primary inciting mediator of the inflammatory response in the host. 3 Lipopolysaccharide binding to host cells induces a receptor-mediated signalling cascade that leads to activation of NF-κB which may happen directly through interleukin-1 receptor-associated kinase (IRAK1) followed by TNF receptor-activated factor 6 (TRAF6), NF-κB inducing kinase (NIK), IκB kinases (IKK), IκB and NF-κB or indirectly through MAPKs including JNK, p38 and ERK pathway. Following activation, NF-κB gets translocated to the nucleus and causes rapid gene induction resulting in the expression of inflammatory mediators, including cytokines (particularly TNF-α and IL-6), chemokines, lipid mediators, inducible nitric oxide synthase (iNOS), enzymes such as cyclooxygenase 2 (COX-2) and adhesion molecules. 1 – 3 Tumor necrosis factor-α further stimulates the production of reactive oxygen species (ROS) and reactive nitrogen intermediates (RNIs) by activated cells causing liver damage due to the oxidative stress. Therefore, NF-κB activated directly or indirectly appears to be the key molecule playing a pivotal role in LPS-induced inflammatory responses. In this context, the inhibition of NF-κB by antioxidants may provide a pharmacological basis for manipulating these acute processes.4,5
Flavonoids are phenolic phytochemicals that represent substantial constituents of the non-energetic part of the human diet and are thought to promote optimal health, partly via their antioxidant effects in protecting cellular components against reactive oxygen species. 6 Flavonoids have been reported to be chain-breaking inhibitors of the peroxidation process, scavenging intermediate peroxyl and alkoxyl radicals.7,8
Amongst these, Quercetin (3,5,7,3′,4′-pentahydroxyflavone) is a widely distributed flavonoid, present in fruit, vegetables, and many other dietary sources. 9 This compound has been reported to protect against oxidative stress in various models of tissue injury. 10 – 15 In vitro studies have shown that quercetin has anti-inflammatory effects regulating nitric oxide, IL-6, and TNF-α release by lipopolysaccharide-stimulated immune cells. 16 – 19 Another subclass of the flavonoids is the flavanols, commonly known as green tea catechins. 20 Tea polyphenols have been reported to display potent antioxidant, anti-inflammatory, antibacterial, hypocholesterolemic and anticarcinogenic properties. 21 Recently, tea polyphenols have been shown to protect against liver injuries including carbon-tetrachloride-induced liver injury and hepatic ischemia-reperfusion injury. 22 – 26 However, the effect of these polyphenols on the signalling cascade of free radical generation culminating into oxidative stress-mediated liver damage has not been evaluated. Although, catechin and quercetin have been shown to inhibit NF-κB activation and NO production in cell culture, their effect on LPS-mediated oxidative stress and NF-κB activation in animal models has not been analyzed in detail. The present study, was, therefore, designed to evaluate the potential of catechin and quercetin as natural polyphenolic inhibitors against endotoxin-induced liver injury in a rat model.
Materials and methods
Agents
Lipopolysaccharide (LPS from Escherichia coli serotype O111:B4), catechin hydrate and quercetin were obtained from Sigma Aldrich Chemicals (St Louis, MO, USA). The preparations were made fresh every time before the commencement of the experiment. Lipopolysaccharide, dissolved in water for injection, was administered as single intraperitoneal injection. Catechin hydrate and quercetin were dissolved in warm distilled water and administered by oral gavage. N-Nitro-
Animals
Female Wistar rats (200–250 g) were procured from Central Animal House, Panjab University, Chandigarh, India. The animals were housed under standard laboratory conditions, maintained on a 12-h light and dark cycle and had free access to food (Ashirwad Industries Pvt Ltd, Punjab, India) and water ad libitum. The experimental protocols were approved by the Institutional Animals Ethics Committee of the Panjab University, Chandigarh, and conducted according to the Indian National Science Academy Guidelines for the use and care of experimental animals.
Experimental design
Rats were divided into following groups each comprising of at least 6–8 rats:
The dose of catechin and quercetin was selected on the basis of preliminary studies done in our laboratory.
Since nitric oxide plays a pivotal role in liver injury, in addition to the above mentioned groups, additional experimental groups were also put up to assess the modulatory effect of catechin and quercetin in the presence of L-NAME (nitric oxide synthase inhibitor) against endotoxin-induced liver injury. Rats were divided into following groups, each consisting of 6–8 rats:
Animals in all these groups were sacrificed 8 h post-endotoxin challenge by cervical dislocation. Livers were removed quickly, rinsed in cold phosphate buffer saline (0.05
Assessment of liver function
Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) enzyme activities in serum were determined using ERBA test kits (ERBA Diagnostics, Mannheim, Germany). Alkaline phosphatase (ALP) was estimated using Enzopak Diagnostic kit (Reckon Diagnostics, India).
Histological studies
Liver tissues removed aseptically from all the groups were cut into small pieces and fixed in 10% buffered formalin. Samples were processed, stained with hematoxylin-eosin and examined under the light microscope.
Mechanistic studies
Liver removed aseptically from all the groups were rinsed in 0.05
Extent of peroxidative liver damage
The quantitative measurement of lipid peroxidation in liver was performed as described by us earlier. 27
Antioxidative enzymes
Superoxide dismutase (SOD) and catalase activities in the liver homogenates were estimated as described earlier. 28
Estimation of hepatic nitrite levels
The amount of nitric oxide was determined by Griess reaction, as described by Green et al. 29 The assay is based on the propensity of nitric oxide to be oxidized to nitrate and nitrite under physiological conditions. For this, 100 µl aliquots of PMS were mixed with 100 µl of Griess reagent (0.1% naphthylethylene diamine dihydrochloric acid and 1% sulphanilamide in 5% phosphoric acid) and incubated at room temperature (28–30°C) for 10 min. Absorbance was measured at 546 nm. Nitrite levels in all the samples were quantified according to the standard graph of sodium nitrite.
Liver TNF-α assay
Assay for TNF-α was performed by ELISA in the liver homogenate in all the groups by commercially available cytokine assay kit (R&D Systems, USA) according to the manufacturer’s instructions. Briefly, standards for TNF-α were dispensed in the 96-well microtitre plates pre-coated with monoclonal antibody specific for rat TNF-α. To each of the designated wells, 50 µl of each test sample and 50 µl of assay diluent was added, the plates were sealed with acetate plate sealer and incubated at room temperature for 2 h. Plates were then washed five times with the wash buffer and 100 µl of rat TNF-α conjugate was dispensed into each well. Plates were again sealed and incubated at room temperature for 2 h, after which they were washed five times with the wash buffer and 100 µl of substrate solution was dispensed into each well. Plates were finally incubated at room temperature (in dark) for 30 min. Stop solution (100 µl) was added into each well and absorbance was read at 450 nm. The results were expressed as picogram/ml of the TNF-α released. The ELISA was sensitive to 5 picogram/ml of the TNF-α released.
Assay for NF-κB p50 subunit
Assay for NF-κB p50 subunit in the nuclear extracts was performed in all the groups by commercially available Transcription Factor Assay kit (Upstate Biotechnology, NY, USA) according to the manufacturer’s instructions. This assay combines the principle of the electrophoretic mobility shift assay (EMSA) with the 96-well based enzyme linked immunosorbent assay (ELISA). Briefly, nuclear extracts from all the groups were prepared using Chemicon’s Nuclear Extraction Kit. During the assay, the capture probe, a double stranded biotinylated oligonucleotide containing the consensus sequence for NF-κB was mixed with the nuclear extract in the transcription factor assay (TFA) buffer provided directly in the streptavidin coated plate and incubated in plate well for 2 h at room temperature. Plates were then washed to remove the unbound material. The bound NF-κB transcription factor subunit p50 was detected with specific primary antibody, rabbit anti-NF-κB p50. Horse radish peroxidase conjugated secondary antibody was then used for detection using TMB/E (3,3′,5,5′-tetramethybenzidine) as the substrate and absorbance was read at 450 nm. Positive and negative controls were also run simultaneously.
RT-PCR analysis of liver iNOS mRNA expression
Primers used
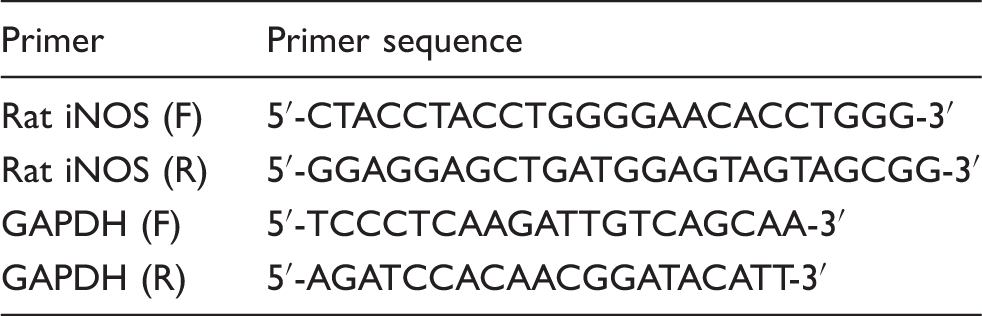
Statistical analysis
Results were expressed as mean ± SD. The inter-group variation was measured by one-way analysis of variance (ANOVA) followed by Fisher’s least significant difference test. The statistical analysis was done using Jandel Sigma Stat Statistical Software v2.0. Statistical significance of the results were calculated at least at P < 0.05.
Results
For all the parameters mentioned below, the effect of catechin and quercetin supplementation was assessed at two different doses, i.e. 25 and 50 mg/kg body weight. It was found that both the phytochemicals were more effective at a dose of 50 mg/kg body weight as compared to 25 mg/kg body weight (data not shown).
Clinical chemistry
Challenge with endotoxin caused nearly 2.7-fold rise in serum levels of ALT, 2.4-fold increase in the serum levels of AST and 6-fold increase in the serum ALP levels as compared to the levels in the control group (P < 0.05). The activities of liver enzymes were decreased significantly (P < 0.05) on supplementation with catechin and quercetin. Catechin and quercetin per se had no effect on liver enzyme levels (Figure 1).
Effect of catechin and quercetin on ALT, AST and ALP levels in serum of LPS-challenged rats compared to control groups. Values are expressed as mean ± SD of six different observations. *P < 0.05 vs control, catechin and quercetin per se; #P < 0.05 vs LPS.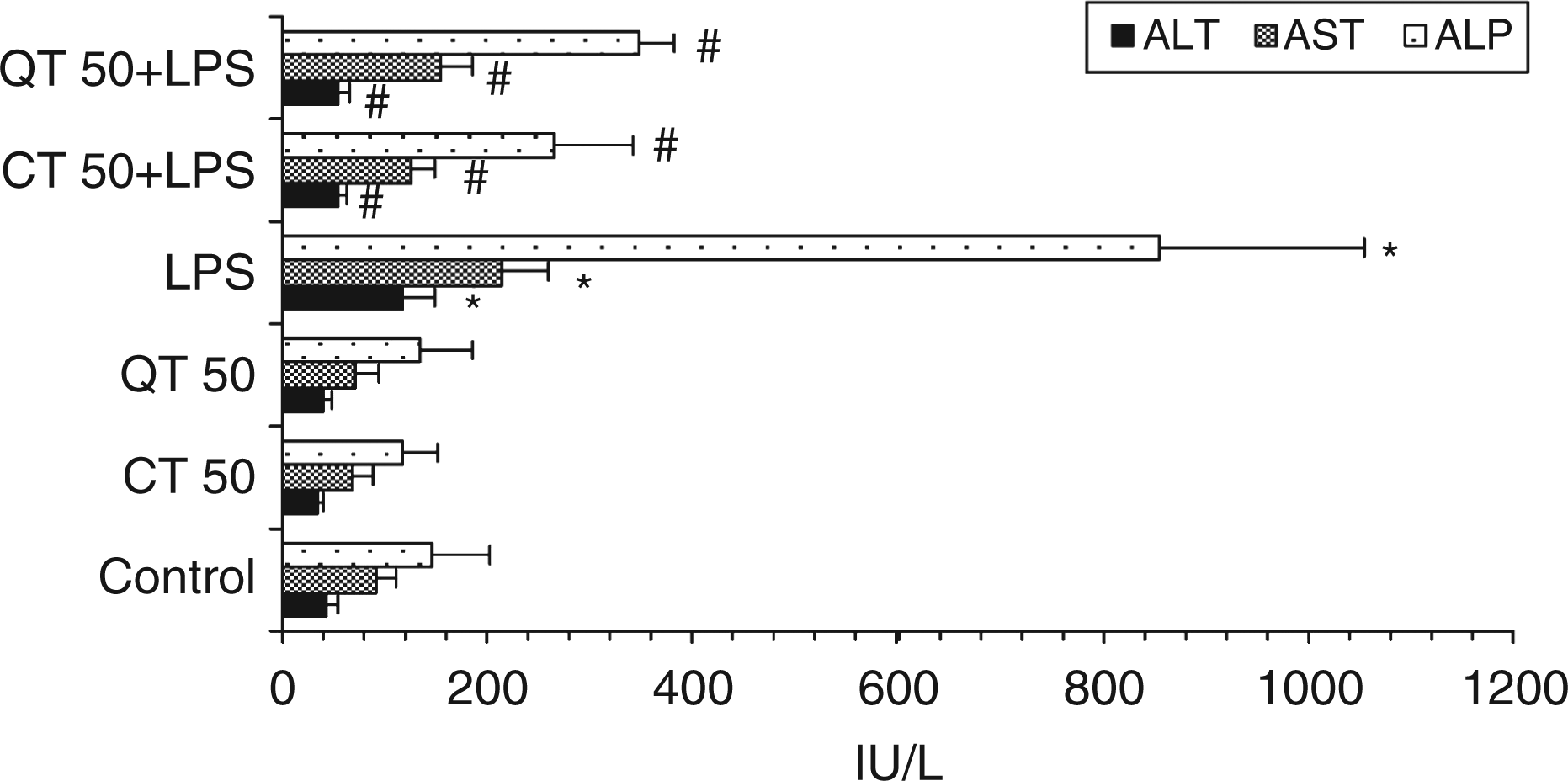
Hepatic histo-architecture
Histological evaluation of liver tissues did not reveal any morphological alterations in the control group (Figure 2A), catechin (Figure 2B) and quercetin per se group (Figure 2C). In contrast, livers of LPS-challenged rats showed marked morphological disruption such as portal triaditis, Kupffer cell hyperplasia, necrosis and lymphocytic infiltration (Figure 2D,E). Supplementation with catechin (Figure 2F) and quercetin (Figure 2G) resulted in significant morphological protection in terms of marked reduction in inflammation and hepatocyte damage in LPS challenged rats.
(A) Photomicrograph of the normal/control rat liver showing normal liver morphology. (B) Photomicrograph of rat liver of catechin per se group showing normal liver morphology. (C) Photomicrograph of rat liver of quercetin per se group showing normal liver morphology. (D,E) Photomicrographs of liver 8 h after LPS challenge to rats showing portal triaditis (portal tract distended with mononuclear cell infiltration) with some breach of lamina limitence indicating development of chronic hepatitis, surrounding liver showing significant Kupffer cell hyperplasia. (F) Photomicrograph of rat livers supplemented with catechin (50 mg/kg) for 15 d before LPS challenge showing mild non-specific changes, but is normal. The portal tracts are intact and hepatocytes are normal. (G) Photomicrograph of rat livers supplemented with quercetin (50 mg/kg) for 15 d before LPS challenge showing mild edema of portal tracts but very little inflammation.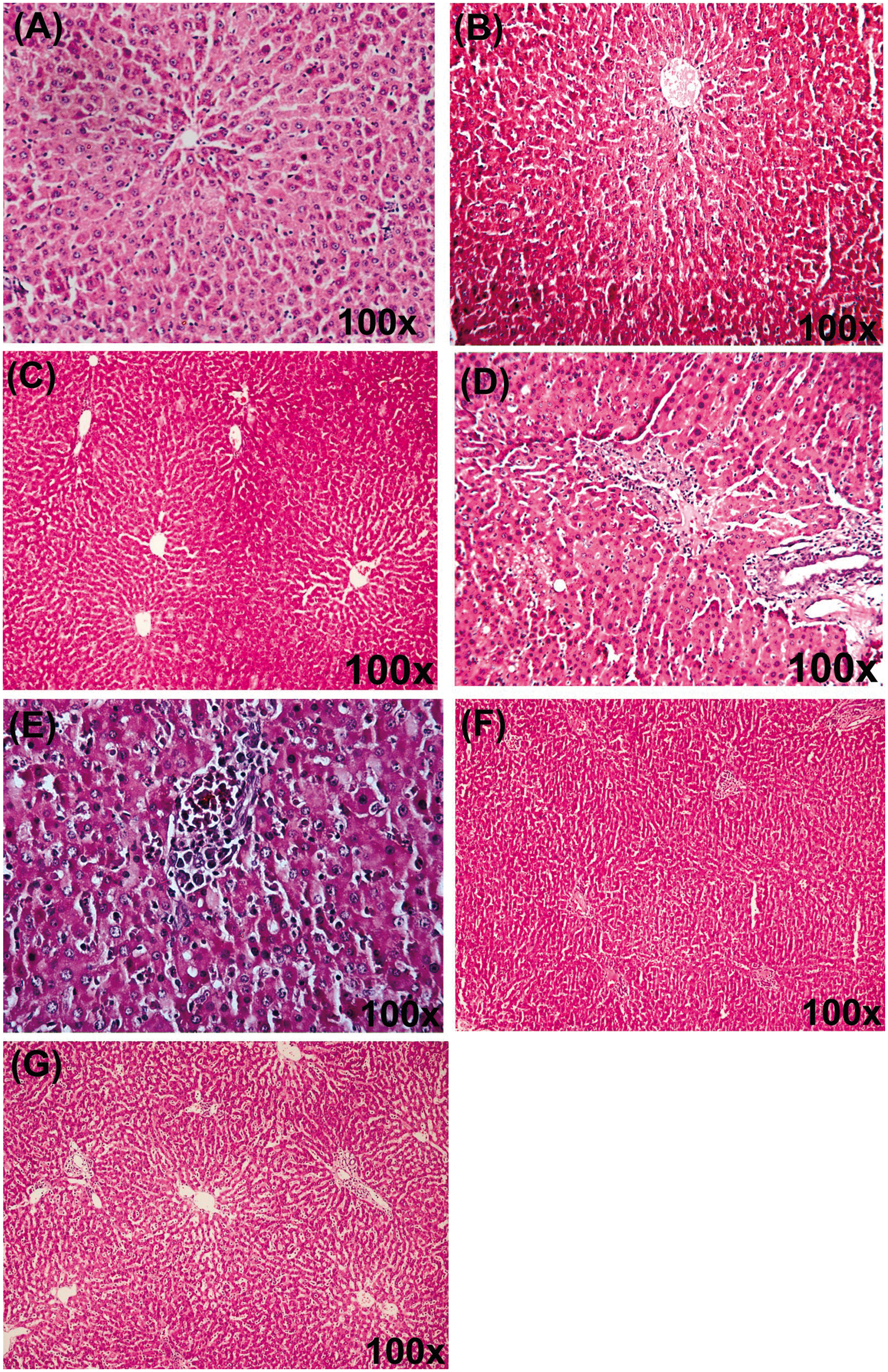
Activation of transcription factor NF-κB
Nuclear factor-κB p50 subunit was significantly (P < 0.01) elevated in LPS-challenged rats as compared to the control and per se groups (Figure 3). Supplementation with catechin and quercetin significantly attenuated the activation of NF-κB induced by endotoxin. However, the effect was more pronounced in case of supplementation with catechin. The levels of NF-κB p50 subunit were further reduced significantly (P < 0.001) by the administration of L-NAME, the inhibitor of iNOS, to the LPS-challenged groups with or without supplementation.
Effect of catechin and quercetin on LPS-induced activation of NF-κB in liver. Values are expressed as mean ± SD of five different observations. *P < 0.05 vs control, catechin and quercetin per se; #P < 0.05 vs LPS; †P < 0.05 vs QT 50 + LPS; §P < 0.05 vs LPS + Inh. Positive control (+control) refers to the TNF-α treated Hela whole cell extract; negative control (–control) refers to the biotinylated double-stranded non-specific competitor oligonucleotide probe which does not contain the NF-κB consensus sequence.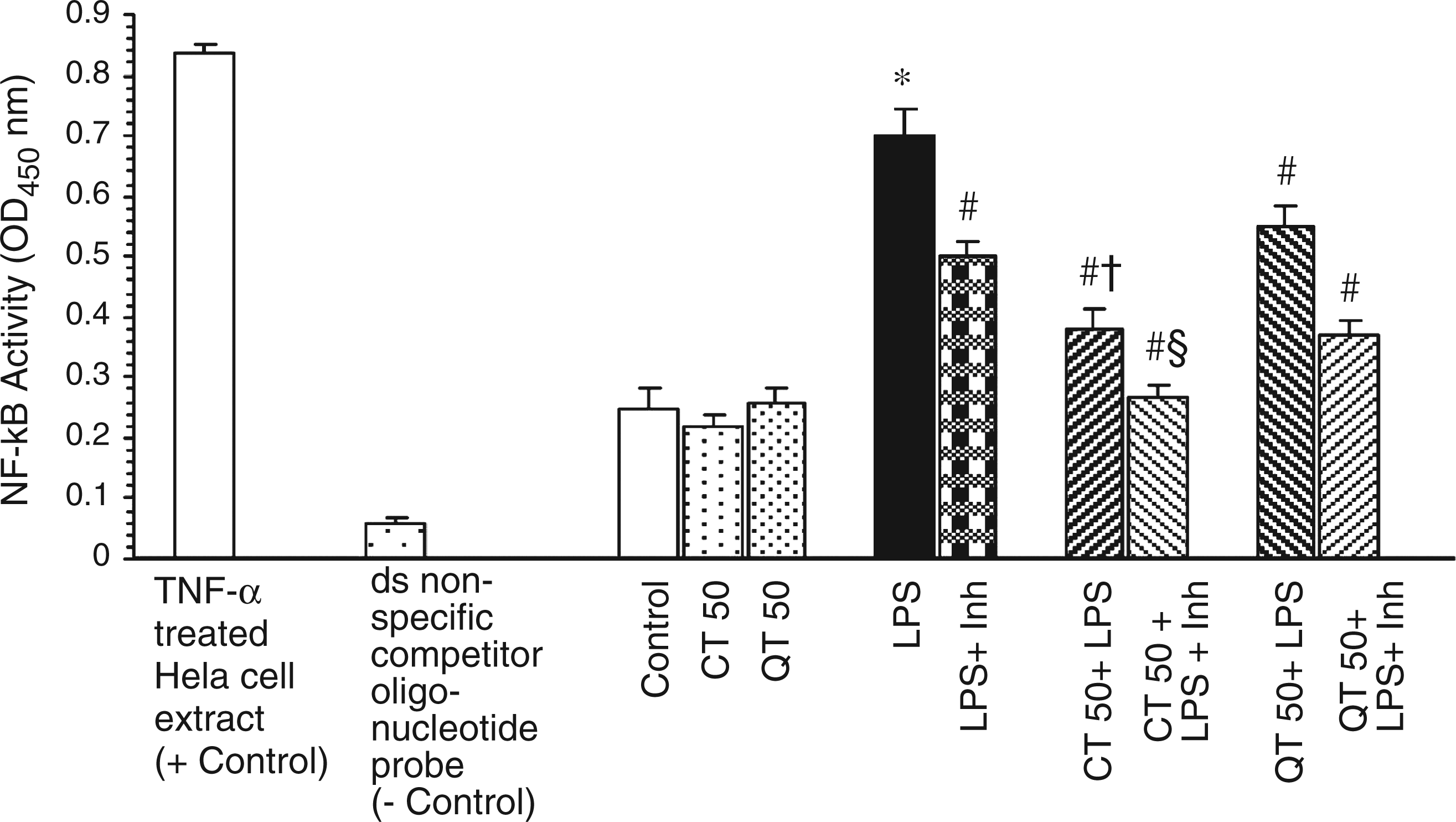
Hepatic TNF-α levels
Lipopolysaccharide challenge caused a marked rise in the levels of TNF-α compared to the control group (240.86 ± 20.3 pg/ml vs 119.5 ± 10.2 pg/ml in control). Administration of catechin and quercetin pre-LPS challenge significantly decreased the levels of TNF-α by 1.59- (at P < 0.01) and 1.35-fold (at P < 0.01), respectively (Figure 4). However, catechin showed higher protective effect than quercetin (at P < 0.05).
Effect of catechin and quercetin on hepatic TNF-α levels in LPS-challenged rats. Values are expressed as mean ± SD of four different observations. *P < 0.05 vs control, catechin and quercetin per se; #P < 0.05 vs LPS; †P < 0.05 vs QT 50 + LPS; ‡P < 0.05 vs LPS + Inh.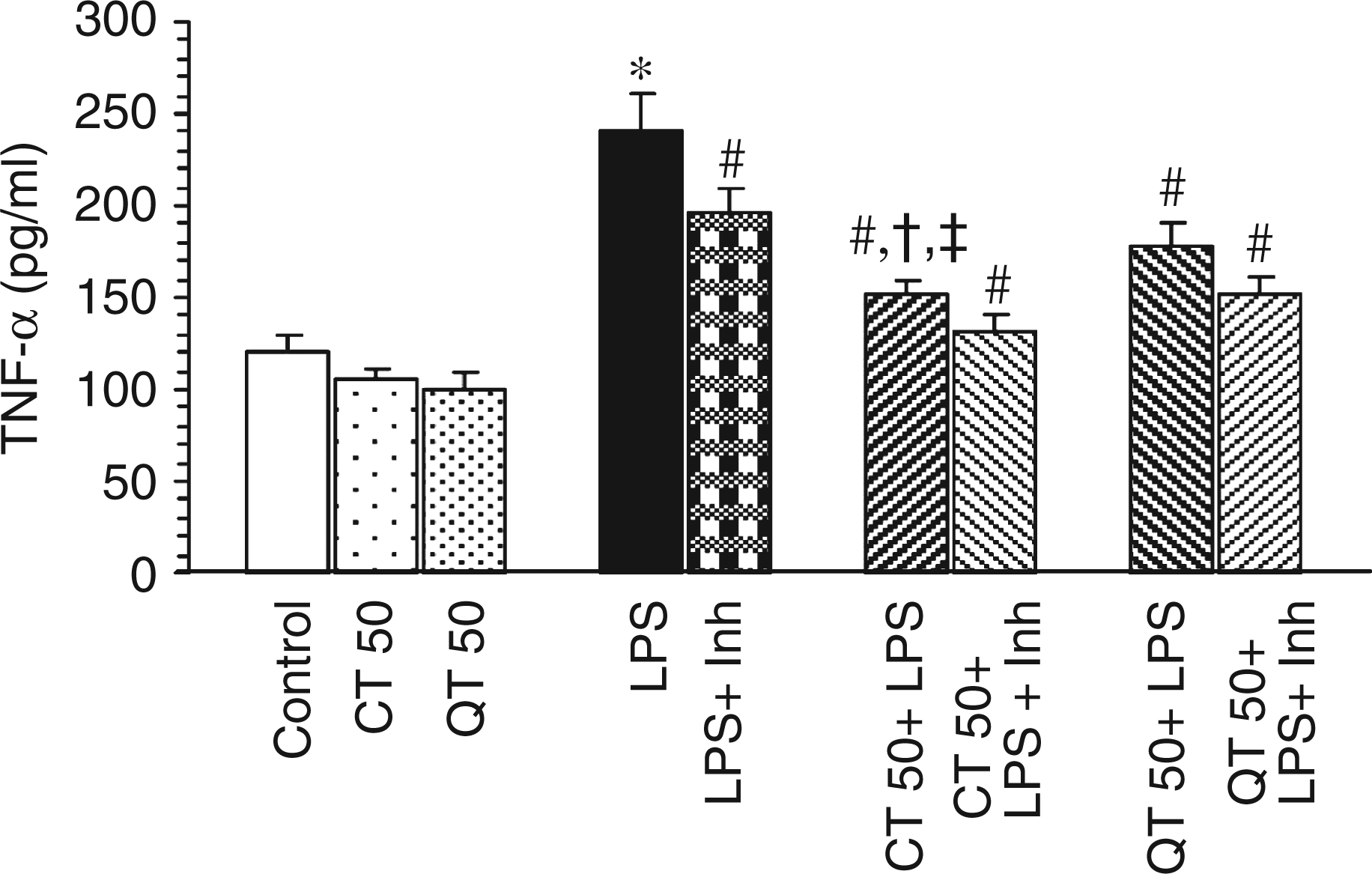
Extent of lipid peroxidation
Modulatory effects of catechin and quercetin on status of oxidative stress and antioxidant defense enzymes in the LPS-challenged rat liver
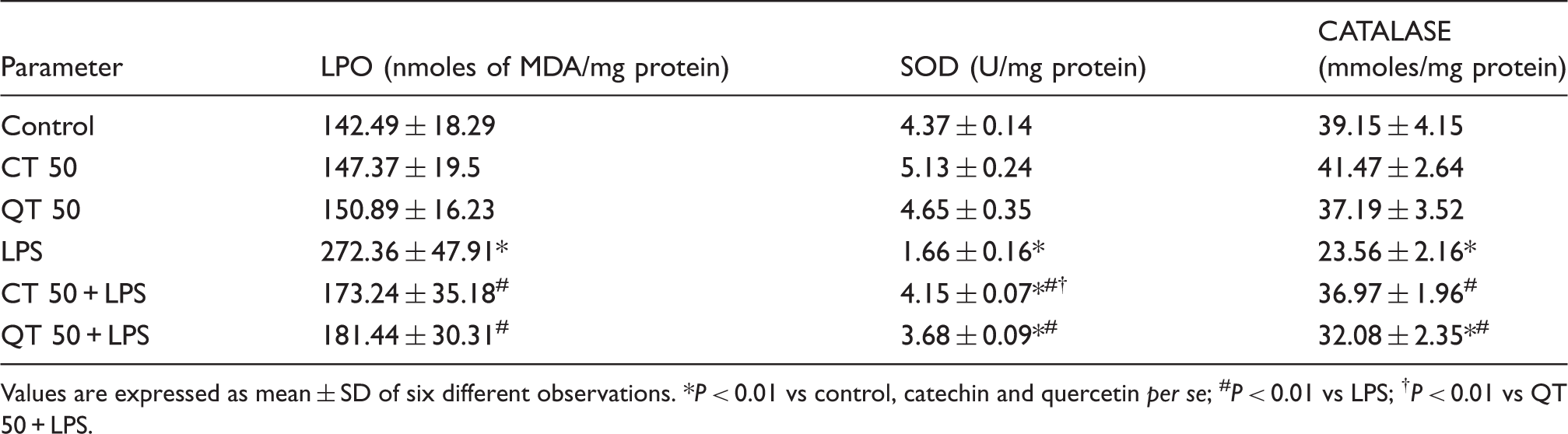
Values are expressed as mean ± SD of six different observations. *P < 0.01 vs control, catechin and quercetin per se; #P < 0.01 vs LPS; †P < 0.01 vs QT 50 + LPS.
Hepatic antioxidative enzymes
Lipopolysaccharide significantly reduced the levels of liver SOD and catalase as compared to the control group (2.6- and 1.7-fold, respectively). Supplementation with catechin and quercetin increased the SOD levels in LPS challenged groups by 2.5- and 2.2-fold, respectively, at P < 0.01. However, the increase in SOD activity was more pronounced in case of catechin supplementation than quercetin (P < 0.01). Catalase activity was also significantly increased at P < 0.01 in LPS challenged groups which were supplemented with catechin and quercetin by 1.57- and 1.36-fold, respectively (Table 2). Per se groups did not show any significant change in the antioxidant enzyme levels.
Hepatic nitrite levels
Nitrite levels in the liver homogenates were found to be significantly higher in LPS challenged groups as compared to the control counterpart (13.43 ± 2.08 µmol/mg protein and 3.55 ± 0.71 µmol/mg protein, respectively). Supplementation with polyphenols significantly decreased the hepatic nitrite levels (P < 0.05). Catechin was found to be more effective than quercetin in inhibiting the LPS-induced increase in nitrite levels (P < 0.05). It was also observed that LPS-induced rise in nitrite level was further significantly decreased in the presence of inhibitor, L-NAME (P < 0.05) in both supplemented and non-supplemented groups (Figure 5). Per se groups did not show any effect.
Estimation of hepatic nitrite levels of different experimental groups. Values are expressed as mean ± SD of six different observations. *P < 0.05 vs. control, catechin and quercetin per se; #P < 0.05 vs LPS; †P < 0.05 vs QT 50 + LPS; §P < 0.05 vs LPS + Inh.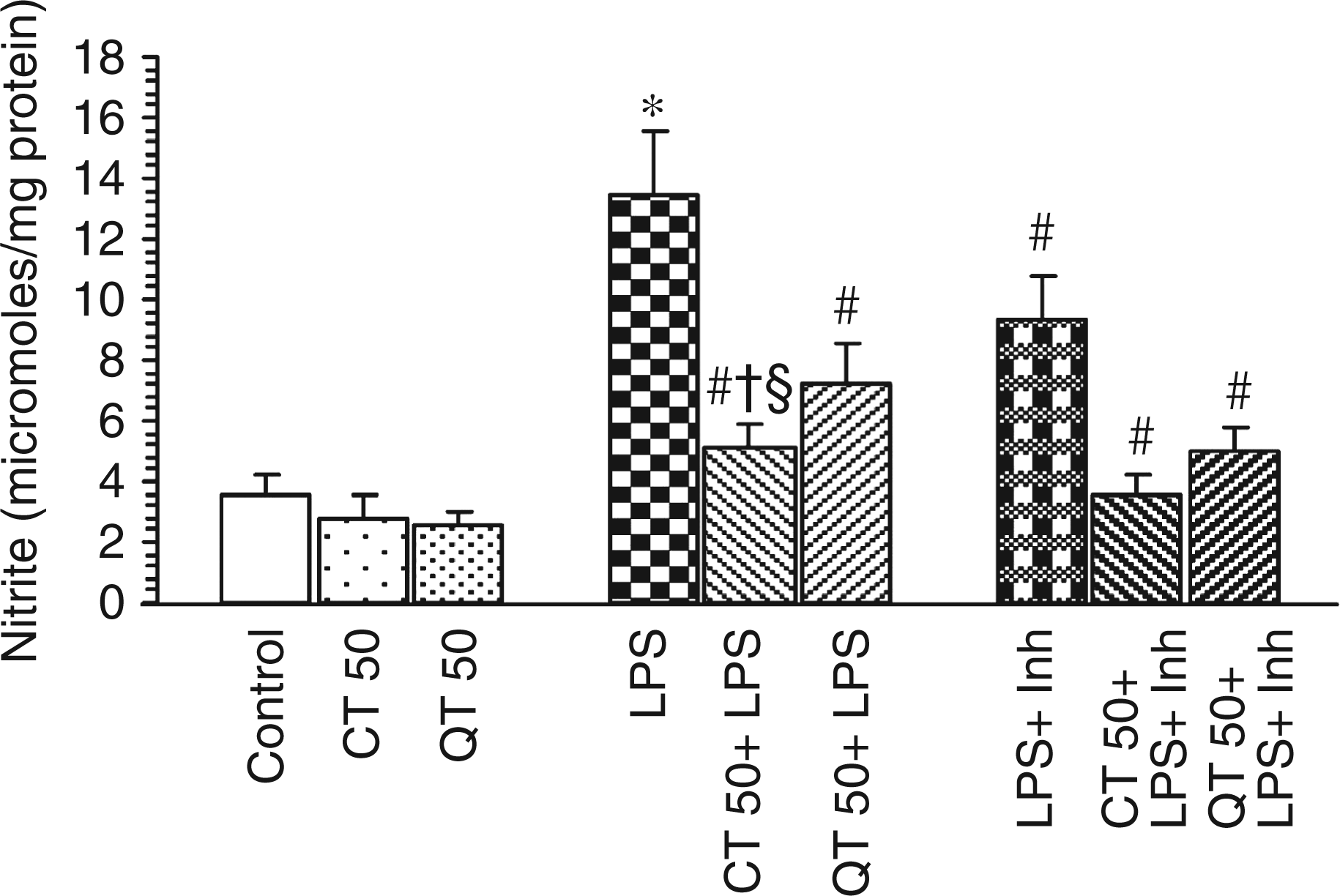
Hepatic iNOS expression
As observed from Figure 6, mRNA expression levels encoding GAPDH were identical in all the groups. In contrast, iNOS mRNA was strongly expressed in LPS-challenged tissue compared with the control tissue. Densitometric analysis of PCR bands showed nearly 20% inhibition of the expression of iNOS mRNA after the administration of iNOS inhibitor in comparison to the LPS-challenged group. However, no significant reduction was observed with the polyphenols used.
RT-PCR showing expression of iNOS in different experimental groups. Total RNA was isolated by using the RNeasy Mini Kit (Qiagen) from liver tissues of the rats subjected to the indicated treatments. RNA was then reverse transcribed and subjected to PCR using iNOS- and GAPDH-specific primers. GAPDH mRNA was used as an internal control.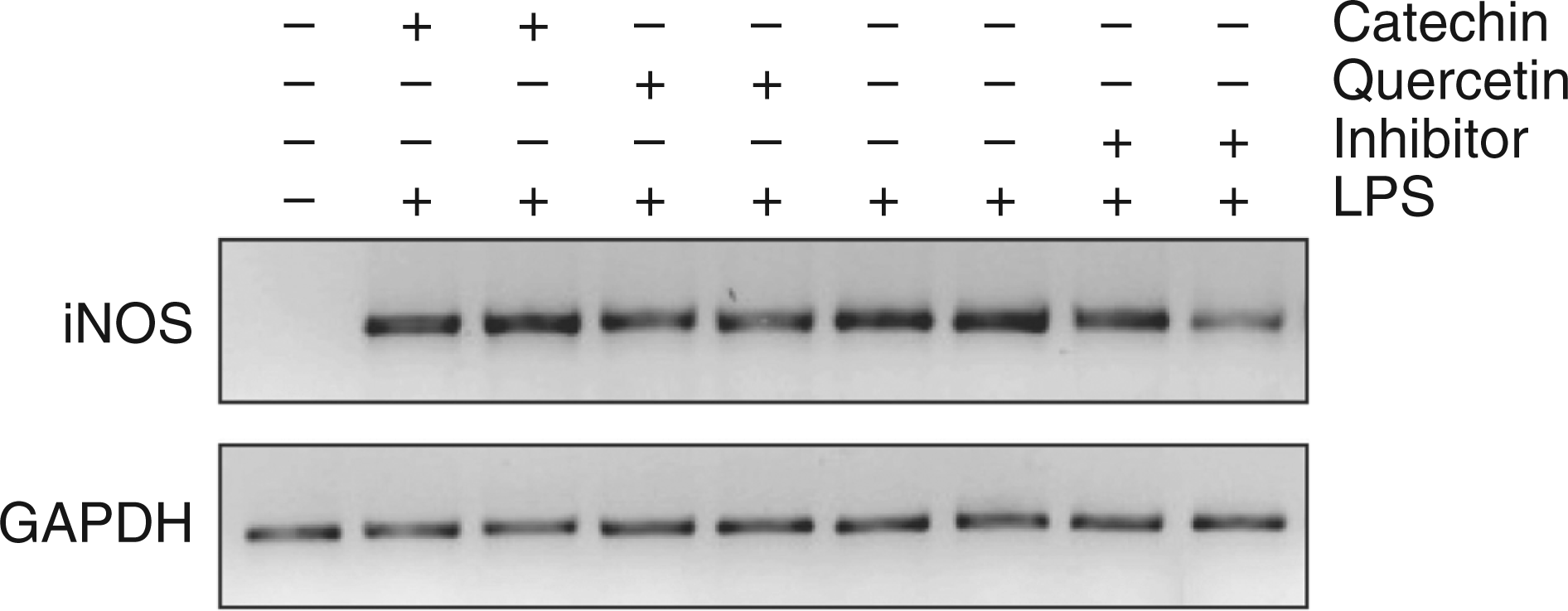
Discussion
Nuclear factor-κB is functional as hetero- or homo-dimeric form of proteins in the Rel family, such as RelA (p65), RelB, cRel, p50 and p52.30,31 Under normal conditions, NF-κB is sequestered in the cytoplasm as an inactive complex, bound to the inhibitory κB (IκB) proteins. Following any kind of stress/insult, IκB phosphorylated by IKK undergoes degradation. 32 The liberated NF-κB translocates to the nucleus, binds to a 10 base pair (bp) consensus sequence (κB) in the promoter region of target genes, thereby allowing rapid gene induction and the expression of inflammatory mediators, including cytokines (particularly TNF-α and IL-6), chemokines, lipid mediators, inducible nitric oxide synthase (iNOS), enzymes and adhesion molecules.33,34 Thus, NF-κB plays a key role during inflammation and proliferation.
In the present study, we observed that both catechin and quercetin inhibited the activation of NF-κB in LPS challenged rat livers which might have suppressed the liver injury (Figs 1 and 2). This observation is consistent with previous reports wherein green tea polyphenols and quercetin have been shown to inhibit NF-κB activation in ischemia-reperfusion liver injury 35 and streptozotocin-diabetic rats, 14 respectively. These polyphenols might have induced suppression of the release of NF-κB by preventing the degradation of IκBα. 14 Blocking of NF-κB activation may further down regulate TNF-α, as has been reported earlier. 36 In the present study also, catechin and quercetin significantly decreased the levels of TNF-α in LPS challenged animals. Decreased levels of TNF-α may thus be attributed to the anti-inflammatory mechanism of these polyphenolic compounds. In agreement with the present data, Yang and colleagues 37 have shown that green tea polyphenol, epigallocatechin gallate inhibits LPS-induced TNF-α gene and protein production in mouse macrophage cell line. Cellular sensitivity or resistance to TNF-α is also correlated with decreased or increased levels of SOD, respectively.27,38 In the present study also, increased levels of TNF-α after LPS injection correlated with the increased level of peroxidation and decreased activities of SOD and catalase. In the absence of an appropriate compensatory response from the endogenous antioxidant network, the system may become overwhelmed (redox imbalance), leading to the activation of stress-sensitive signalling pathways, such as NF-κB, and others. Furthermore, polyphenols attenuated the rise of liver malondialdehyde levels, increased the levels of hepatic antioxidants after LPS-challenge, and the liver injury was ameliorated (Table 2). These effects might have been due to the antioxidative property of these polyphenols in the supplemented rats. It has been suggested that flavonoids such as quercetin and catechin could localize near the membrane surface, trapping directly any radical generated in the lipid environment of the membranes as well as ROS generated in the aqueous phase. 39 This might block the superoxide radical in the tissues and decrease SOD consumption thus enhancing SOD activity.
One major consequence of the activation of stress-sensitive signalling pathways is the generation of gene products such as nitric oxide that cause cellular damage. In the liver, it is produced by at least two different isoforms of NOS, e.g. eNOS and iNOS. The eNOS is constitutively expressed and generates relatively small amounts of NO, but plays an important role in vasorelaxation. 40 Overproduction of NO following acute endotoxemia is known to be mediated by increased iNOS expression. 41 Nitric oxide in conjunction with superoxide radical has been reported to form a potent and versatile oxidant peroxynitrite, which may lead to the stimulation of TNF-α production in Kupffer cells. 42 Enhanced expression of iNOS and hence increased NO levels observed in the endotoxin-challenged animals might have been associated with TNF-α as it is known for its potent stimulatory activity of iNOS which increases the NO levels. 43 – 45 The present study showed that catechin and quercetin significantly inhibited the LPS-induced increased hepatic nitrite levels, thus correlating well with the decreased TNF-α levels indicating an interplay of these molecules in determining the outcome of a clinical manifestation.
Inhibition of NF-κB activation is considered to be important when designing iNOS inhibitors because NF-κB activation is the primary regulatory step involved in iNOS expression. 46 It was observed that administration of L-NAME not only decreased the expression of liver iNOS thereby resulting in decreased hepatic nitrite levels but also suppressed the LPS-induced activation of NF-κB which also highlights the role of NO in the injury. The effect was more pronounced in case where flavonoids were supplemented before LPS challenge. Therefore, in this model also, inhibition of nitric oxide is presumably associated with the hepatoprotective effect of catechin and quercetin. In support of our findings, in vitro studies have shown that green tea polyphenol, EGCG and quercetin decreases LPS-induced iNOS expression and activity in murine peritoneal macrophages via preventing NF-κB activation. 47 – 49 It is also indicated from the present study that these polyphenols may not be directly inhibiting the iNOS expression at the transcription level. However, a decrease in nitrite levels might be due to the inhibition of NF-κB induced by the compounds.
Conclusions
Both catechin and quercetin have the potential to attenuate endotoxin-induced liver injury by down-regulating NF-κB activation and further going downstream the signalling cascade including TNF-α, NO and ROS and by enhancing the antioxidant profile of the challenged host. However, catechin was found to be more effective than quercetin against endotoxin-induced liver injury. Our findings suggest that polyphenols may block the production of noxious mediators involved in liver injury by targeting the IKK/NF-κB signal transduction pathway and therefore, may be of importance in making strategies for management of the disease.
Footnotes
Acknowledgement
The authors are grateful to the Indian Council of Medical Research (ICMR), New Delhi, India, for providing financial assistance to carry out this research work.
