Abstract
We investigated the impact of two nights of total sleep deprivation (SD) or four nights of rapid eye movement (REM) SD on immunological parameters in healthy men. Thirty-two volunteers were randomly assigned to three protocols (control, total SD or REM SD). Both SD protocols were followed by three nights of sleep recovery. The control and REM SD groups had regular nights of sleep monitored by polysomnography. Circulating white blood cells (WBCs), T- (CD4/CD8) and B-lymphocytes, Ig classes, complement and cytokine levels were assessed daily. Two nights of total SD increased the numbers of leukocytes and neutrophils compared with baseline levels, and these levels returned to baseline after 24 h of sleep recovery. The CD4+ T-cells increased during the total SD period (one and two nights) and IgA levels decreased during the entire period of REM SD. These levels did not return to baseline after three nights of sleep recovery. Levels of monocytes, eosinophils, basophils and cytokines (IL-1β, IL-2, IL-4, IL-6, IL-10, TNF-α and IFN-γ) remained unchanged by both protocols of SD. Our findings suggest that both protocols affected the human immune profile, although in different parameters, and that CD4+ T-cells and IgA levels were not re-established after sleep recovery.
Introduction
Despite the fact that sleep is essential for good health and quality of life, according to Bonnet and Arand, 1 one-third or more of normal adults suffer from significant sleep loss. Sleep deprivation (SD) is a consequence of societal pressure experienced by individuals, with repercussions for autonomic function, the neuro-endocrine system and the immune system.2,3 It is a commonly held belief that sleep loss increases susceptibility to diseases and conversely that sleep is important for recovery from illness. The brain and host defense systems are capable of bidirectional communication, which provides mechanisms for changes in sleep-wake behavior to impact the immune system and vice versa.4,5 In this sense, one hypothesis that has been proposed is that sleep is essential for the proper functioning of the immune system, thus rendering a sleep-deprived organism more susceptible to pathogens.6,7 Several studies have demonstrated that sleep loss leads to significant alterations in many immune components in animals and humans.3,8– 11
Supporting this scenario, studies have shown that SD increases susceptibility to many diseases, indicating impairment of the surface barriers because of sleep loss. 12 – 14 Everson and Toth 15 demonstrated an increased rate of bacteremia in rats chronically deprived of sleep. Recently, this increase in disease susceptibility was observed in sleep-deprived humans. Cohen et al. 16 demonstrated that poorer sleep efficiency and shorter sleep duration in the weeks preceding exposure to a rhinovirus were associated with lower resistance to the common cold. The influence of SD on host defense relates to more than just the surface barriers. There are several lines of evidence that SD affects both cellular innate immunity and the adaptive immune response. After two or three nights of SD, there is increased circulating leukocytosis 11 and decreased neutrophil and lymphocyte function. 17 – 19
Regarding the humoral immune response, Lange et al. 20 demonstrated a link between poor sleep and clinically significant decreases in immunoresponsiveness to vaccines. One night of SD after vaccination suppressed the antibody response to a hepatitis A vaccine by about 50% in their study. This result suggests that sleep is an essential factor contributing to the success of vaccination. Indeed, sleep and sleep loss have been reported to be associated with alterations in the production of inflammatory markers. 21 Cytokines such as IFN, IL-1 and TNF are well known for their roles as immune response modifiers, and these substances are affected by SD, as reviewed by Obal and Krueger. 22
Despite its undoubted importance, the consequences of extended periods of sleep loss or rapid eye movement (REM) SD on immune function in humans remain to be fully understood. In animal models, our group has shown that different SD protocols (REM SD vs total SD vs sleep restriction) yielded distinct outcomes upon immune function.9,10 Of note, the effects of both total and REM SD and subsequent recovery periods have not yet been examined together in human immune profile. The methodology employed in our study is justified, once such total as selective REM SD are routinely observed in modern society, although in different situations. In addiction, the REM SD is observed, for example, in cases of obstructive sleep apnea (OSA). These sleep-disordered breathing events are more incidents during REM sleep and cause micro-arousals, leading to sleep fragmentation and consequent inability to reach regular REM sleep parameters. Thus, patients with OSA, a highly prevalent sleep disorder, are chronically REM sleep-deprived. On the other hand, individuals who experience daily SD due to bedtime restriction lose a considerable percentage of this stage of sleep. Frequently, this sleep debt is alleviated during the weekends by obtaining extra sleep over and above the daily requirement, a scenario that was replicated in this investigation by the REM sleep-deprived group. When employing both protocols, we aimed to understand which of these sleep manipulations (both common and prevalent, as aforementioned) lead to more significant immunological damages or impairments.
Volunteers were deprived of sleep for two consecutive nights to simulate people who spend long periods without sleeping as a consequence of professional or personal burden. These individuals must to remain active and awake during the night, the period in which sleep pressure is usually higher. However, during the day, period in which they theoretically would sleep, they are subjected to social, domestic, and cultural patterns of the rest of society. This way, this population has his sleep quality impaired chronically, and frequently are exposed to total SD.
In this sense and based on the current literature, our hypothesis was that, not only total SD but also selective REM SD would alter immune components in humans and would have a negative impact on the immune function. To investigate this hypothesis, we evaluated blood cell counts, T- (CD4/CD8) and B-lymphocytes, Ig classes, complement and cytokine measurements, comparing subjects remaining awake for two nights, subjects who underwent four nights of selective REM SD or subjects with a normal sleep-wake schedule in relation to a baseline period. We also examined whether sleep recovery would reverse the resulting alterations in the immune profile.
Subjects and methods
Subjects
The study was conducted at the Sleep Laboratory at the Universidade Federal de São Paulo (UNIFESP) after the approval of the local ethics committee (#1676/06). The methods and laboratory techniques of polysomnography have been described in detail elsewhere. 23 Thirty-two healthy male volunteers (ranging from 19–29 years old) were randomly assigned to one of three experimental groups (11 non-SD, 11 total SD and 10 REM SD) after providing written informed consent before their participation in this investigation and after the nature of the procedures had been fully explained to them. Volunteers were carefully screened by obtaining a detailed medical history. Physical and neurological examination, routine blood tests and urine toxicology for psychotropic drugs were performed to exclude any acute or chronic illness. Volunteers underwent polysomnographic recording to verify that they had no type of sleep disorder. Exclusion criteria were shift work, sleep disorders, extreme morningness or eveningness, history of neurological disease, psychiatric diseases or medical conditions and tobacco, alcohol or substance abuse. All participants had normal results on validated questionnaires, including the Pittsburgh Sleep Quality Index, Epworth Sleepiness Scale and Beck Depression Inventory. The polysomnography screening night also helped the volunteers to adapt to the recording equipment and the study environment.
Experimental protocol
The investigation was performed on nine consecutive days, including one adaptation night, one baseline night, two nights of total SD, four nights of REM SD and three nights of sleep recovery for both groups. The control group was also maintained in the laboratory during the entire experimental protocol and had regular nights of sleep monitored by polysomnography. The choice of these experimental periods was made based on a pilot study which established that more than two nights of total SD is extremely fatiguing for the volunteers, representing a possibly bias for our results. On the other hand, short REM SD periods (less than three nights) are not enough to exert its effects on sleep disruption.
For all groups, naps were not permitted. During the week preceding the study as well as the study period itself, participants were asked to abstain from alcohol, chocolate and caffeinated beverages. A standardized bedtime schedule and average sleep durations (7–9 h sleep per night, with morning waking time between 06:00 and 09:00) were verified by sleep diary and actigraphy for a period of one week before participation in the study. Participants received four meals per day (08:30, 12:00, 16:00 and 20:00), plus a late evening snack at midnight for the total SD group during the SD period. The volunteers wore a wrist activity monitor (Actiwatch, MiniMitter, USA) to verify the compliance of the subjects with scheduled bedtimes one week before and during all of the experimental protocols. All subjects underwent an adaptation night followed by baseline polysomnography at the Sleep Institute. In the laboratory, subjects were instructed to be in bed between 23:00 and 08:00, and sleep was recorded each night. Fasting blood samples were collected once each morning. The schematic representation of the experimental groups is depicted in Figure 1.
Protocol figure. Data are plotted in relative clock hours. Volunteers in the total SD group underwent a baseline night (B) followed by 2 nights (N1–N2) of total SD (black boxes). Volunteers in the REM SD group underwent a baseline night (B) followed by 4 nights (N1–N4) of REM SD (gray boxes). Both SD protocols were followed by 3 nights of sleep recovery (R1–R3). The control group remained in the laboratory during the entire experimental period. Blood samples were collected daily at 08:00. A: adaptation night.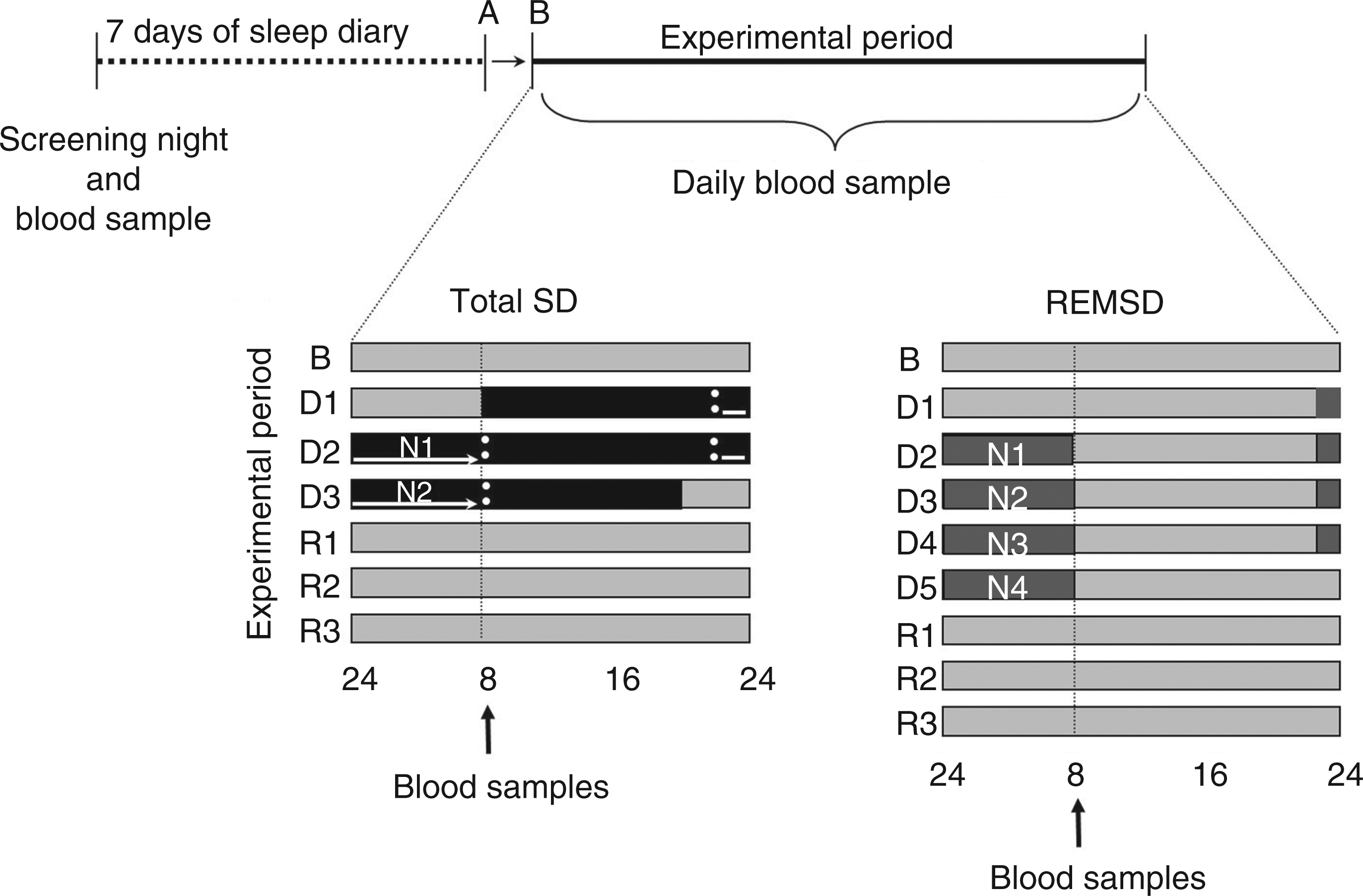
Polysomnography recording
Sleep recordings were performed with a digital electroencephalogram (EEG) acquisition system, Embla Digital A10 recording/amplifier with Somnologica software (Flaga h.f., Requijavique, Iceland). Surface electrodes were used to record EEG signals, including two central electrodes (C3-A2 and C4-A1) and two occipital electrodes (O1-A2 and O2-A1), along with bilateral electroculograms and submental electromyograms. Recordings included the oronasal airflow signal as measured by a thermocouple, airway pressure measurements evaluated by a cannula, respiratory effort signal measured by thoracic and abdominal piezoelectric belts, leg movements measured by tibial electromyograms, and arterial oxygen saturation levels as assessed by pulse oximetry. Sleep recordings were visually scored at 30-s intervals as the stages wake, REM, or 1, 2 and non-rapid eye movement sleep. 24 During the acquisition, the EEG signals were filtered (0.3–70 Hz) and sampled at 200 Hz with 16-bit resolution.
Sleep deprivation and rested wakefulness (non-SD) procedures
REM SD subjects were awakened by having their names called over an intercom at the first sign of REM sleep. If there was no response, the experimenter entered the room and gently shook the subject until receiving a response. This stage was determined by visual polysomnographic inspection in an on-line recording interval. The criteria for interventions were desynchronized EEG without spindles or K complexes and the concomitant reduction of the tonic electromyogram amplitude and occurrence of REM sleep. The volunteers were kept awake for a sufficient time to avoid an immediate relapse into REM sleep while keeping the waking episodes short enough to allow frequent interventions. Throughout the protocol, total SD volunteers could read, play games, watch television or ambulate within the building to help them stay awake. The subjects were also required to abstain from jogging, running, jumping and any form of resistance exercise. To ensure that subjects would not fall asleep during the study, investigators were continuously present to monitor wakefulness. All subjects remained in the research center throughout the study period.
Blood analyses
Blood samples were obtained via the antecubital vein every morning (08:00) using vacutainer tubes from each volunteer during the experimental protocol (baseline, two nights of total SD, four nights of REM SD and three nights of recovery). Blood samples were centrifuged immediately at 4°C, and then the serum and plasma were stored at –80°C until the assays were conducted.
Hemogram
White blood cell (WBC) subset counts were determined by an automatic blood cell counter (Advia 120 Hematology System, NY, USA) on the same day that the blood was collected.
Ig and complement measurements
Serum concentrations of Ig (mg/dl), including IgG, IgA and IgM, and complement proteins (mg/dl), including C3 and C4, were determined by nephelometry (Immage Beckman Coulter, Brea, CA, USA).
Lymphocyte subpopulations
Percentages of lymphocyte subsets were determined by flow cytometry using monoclonal antibodies. Briefly, 100 µl of an undiluted blood sample was immunostained with anti-CD3/FITC, anti-CD4/APC, anti-CD8/PE, and anti-CD19/PerCP in Falcon tubes (BD Biosciences, USA). The solutions were incubated with 2 ml of lysis reagent for 10 min. After this step, the solutions were centrifuged for 5 min at 4°C at 300 g. The supernatant was discarded, and the pellet was resuspended with 2 ml of PBS (2% FCS) and again centrifuged in the above mentioned manner. The pellet was then mixed with 500 µl of sheath fluid. Finally, samples were mixed gently, and at least 10,000 events were acquired on a FACSCalibur (BD, Biosciences, USA) using CellQuest Software (BD Biosciences, EUA).
Circulating cytokine levels
The plasmatic concentration of cytokines was measured using Luminex™ xMAP technology. Briefly, microbeads with defined spectral properties were conjugated to protein-specific antibodies and added to samples (samples included protein standards in a known concentration, control samples, and test samples) in the wells of a filter-bottom microplate. This mixture was incubated for two hours to allow antibody and protein binding. After washing the beads, protein-specific biotinylated detector antibodies were added and incubated with the beads for one hour. After removal of excess biotinylated antibodies, streptavidin conjugated to the fluorescent protein R-phycoerythrin was added and incubated for 30 min. After washing off unbound R-phycoerythrin streptavidin, the beads were analyzed with the Luminex® 100 (Luminex, USA). By monitoring the spectral properties of the beads and the amount of associated R-phycoerythrin streptavidin fluorescence, the concentration(s) of one or more proteins could be determined. The cytokine levels were measured by the MILLIPLEX MAP Human Cytokine/Chemokine Panel MPXHCYTO-60 K with the following targets: IL-1β, IL-2, IL-4, IL-6, IL-10, TNF-α and IFN-γ. The standard curve range was 3.2–10,000 pg/ml, with an accuracy of 100 ± 25%, intra-assay precision of 4.6–13.8% and inter-assay precision of 3.7–17.2%.
Statistical analysis
Average values are given as mean ± SE. Statistical significance of values for leukocytes subsets, complement proteins, Ig, TNF-α and IL-10 was assessed by a repeated-measures analysis of variance (ANOVA), including the treatment (non-SD, total SD and REM SD) and time factor. If the day-by-day group interaction had significantly different values (P < 0.05), a post hoc Duncan test was used for comparison of the values. The Friedman test was used to analyze differences in IL-1β, IL-2, IL-4, IL-6 and IFN-γ levels. Volunteers with immunological parameters exceeding reference values were excluded from analysis. 25
Results
Basal complete blood counts were not significantly different between controls and both SD groups.
Polysomnographic analysis has been described in detail in a previous study by our research group. 23 Briefly, no significant differences in the percentage of sleep time spent in slow-wave sleep were induced by four nights of REM SD. Relative to the baseline, four nights of REM SD promoted a pronounced decrease (∼70%) in the amount of REM sleep during the four experimental SD nights.
Circulating WBC count
White blood cells in baseline (B), 2 nights (N1–N2) of total SD, 4 nights of REM SD (N1–N4) and 3 nights of recovery (R) sleep
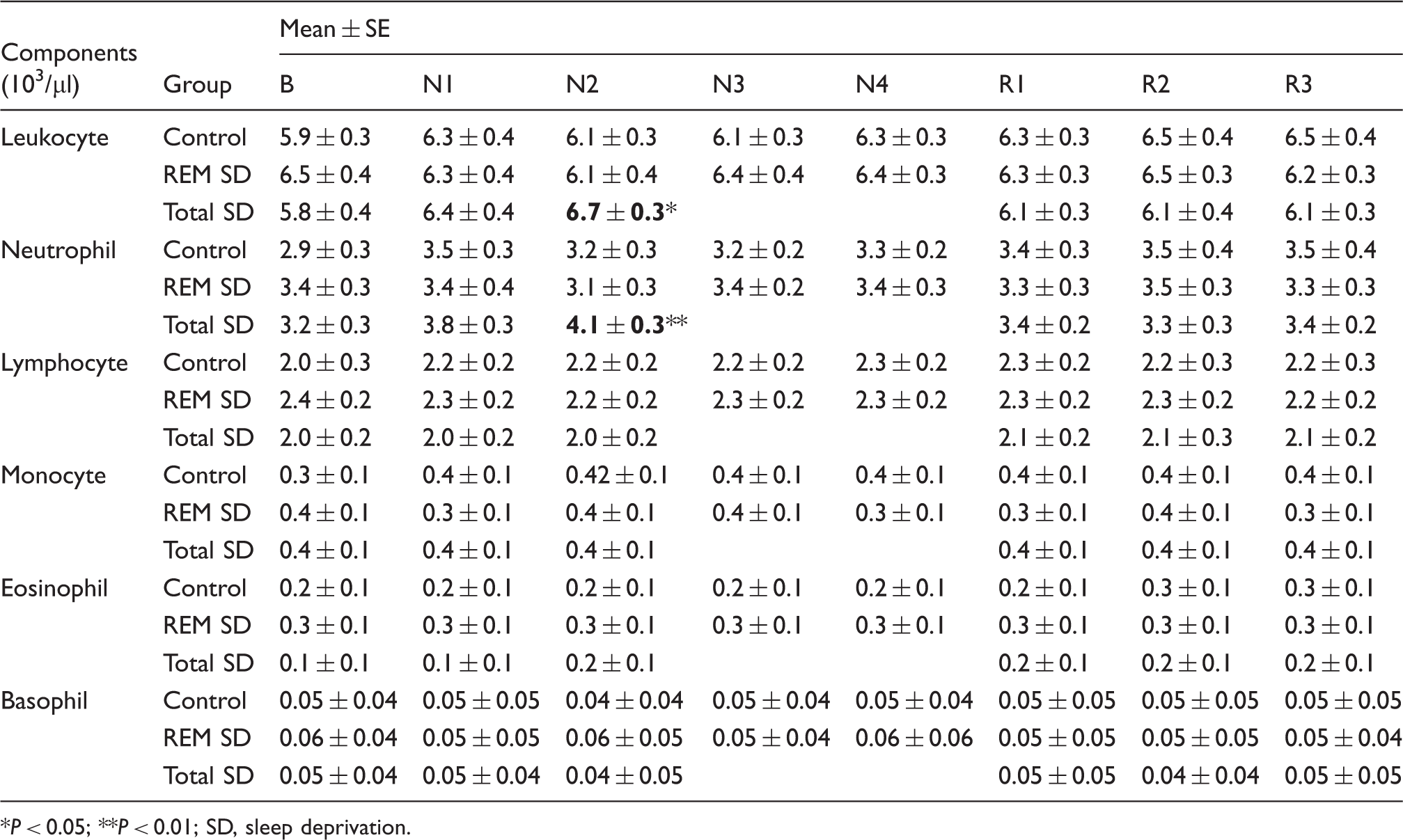
P < 0.05; **P < 0.01; SD, sleep deprivation.
Percentage lymphocyte subset subpopulations
The analysis of lymphocyte subpopulations indicated significant effects of SD [F(7,196) = 3.23; P < 0.01] and interaction effects [F(14,196) = 2.05; P < 0.01]. CD4+ T-cell counts increased after the first night of total SD (P < 0.01) and remained elevated after the second night when compared to baseline values (P < 0.01), corresponding to elevations of 12.8% and 13.5%, respectively (Fig. 2). Furthermore, this CD4+ T-cell subpopulation did not return to baseline levels (P < 0.01) after three nights of sleep recovery. CD4+ T-cell numbers recovered to baseline after the second night of recovery and then increased again after the third night of recovery. Unlike CD4+ T-cells, CD8+ T-cell, total lymphocyte (CD3+ T-cells) and B-cell (CD19+) counts were not significantly different (Fig. 2).
Percentagesof lymphocyte subsets at baseline (B) and during SD (N) and sleep recovery (R). Mean values ± SE of CD3+ T-cells (A), CD19+ B-cells (B), CD4+ T-cells (C), and CD8+ T-cells (D). Control group: n = 11; total SD group: n = 11; REM SD group: n = 10. **P < 0.01: different from respective baseline.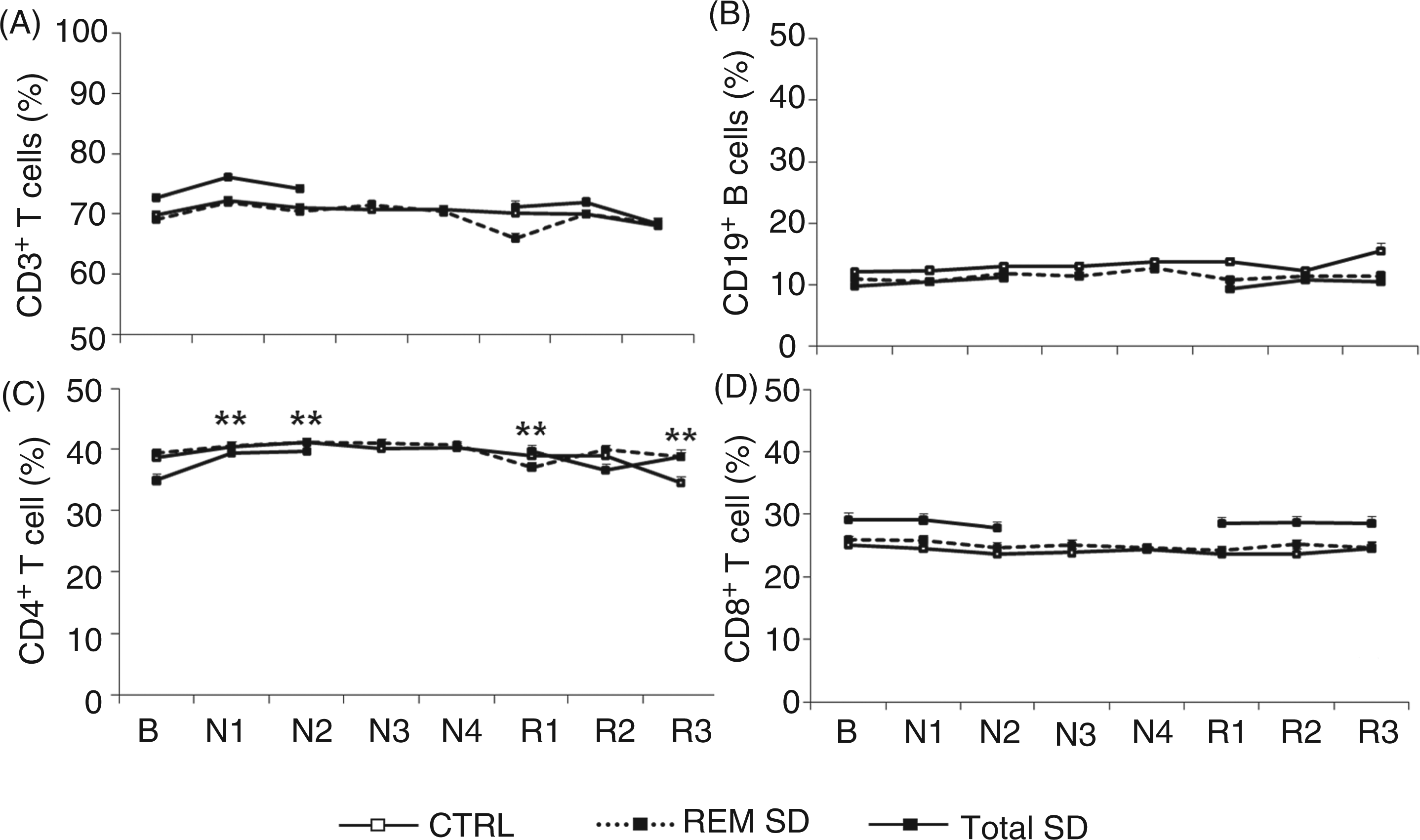
Ig measurements
Serum IgG and IgM levels in the total SD and REM SD groups did not show any significant differences. Although the baseline levels of IgA were ∼160 mg/dl in the control group and ∼220 mg/dl in the REM SD group, these levels were not significantly different. This difference can be explained by an aleatory greater variability regarding the IgA levels in the REM SD group. Although the normal IgA concentration in humans was reported to be ∼200 mg/dl, 25 according to Butler and Rossen, 26 the IgA levels in healthy adult males ranges from 150–350 mg/dl.
The analysis of IgA levels indicated significant effects of SD [F(7,140) = 3.02; P < 0.01] and interaction effects [F(14,140) = 1.9; P < 0.03]. IgA levels decreased by 6.6%, 6.9%, 4.9% and 4.6% each night during the four day period of REM SD when compared to baseline levels (P < 0.01; P < 0.01; P < 0.03; P < 0.04, respectively). Furthermore, similar to T-helper cells, IgA levels recovered after the first night of recovery and declined again after the second and third nights of recovery (P < 0.01), as depicted in Figure 3.
Ig levels at baseline (B) and during SD (N) and sleep recovery (R). Mean values ± SE of IgG (A), IgM (B) and IgA (C). Control group: n = 8; Total SD group: n = 7; REM SD group: n = 8. *P < 0.05; **P < 0.01: different from respective baseline.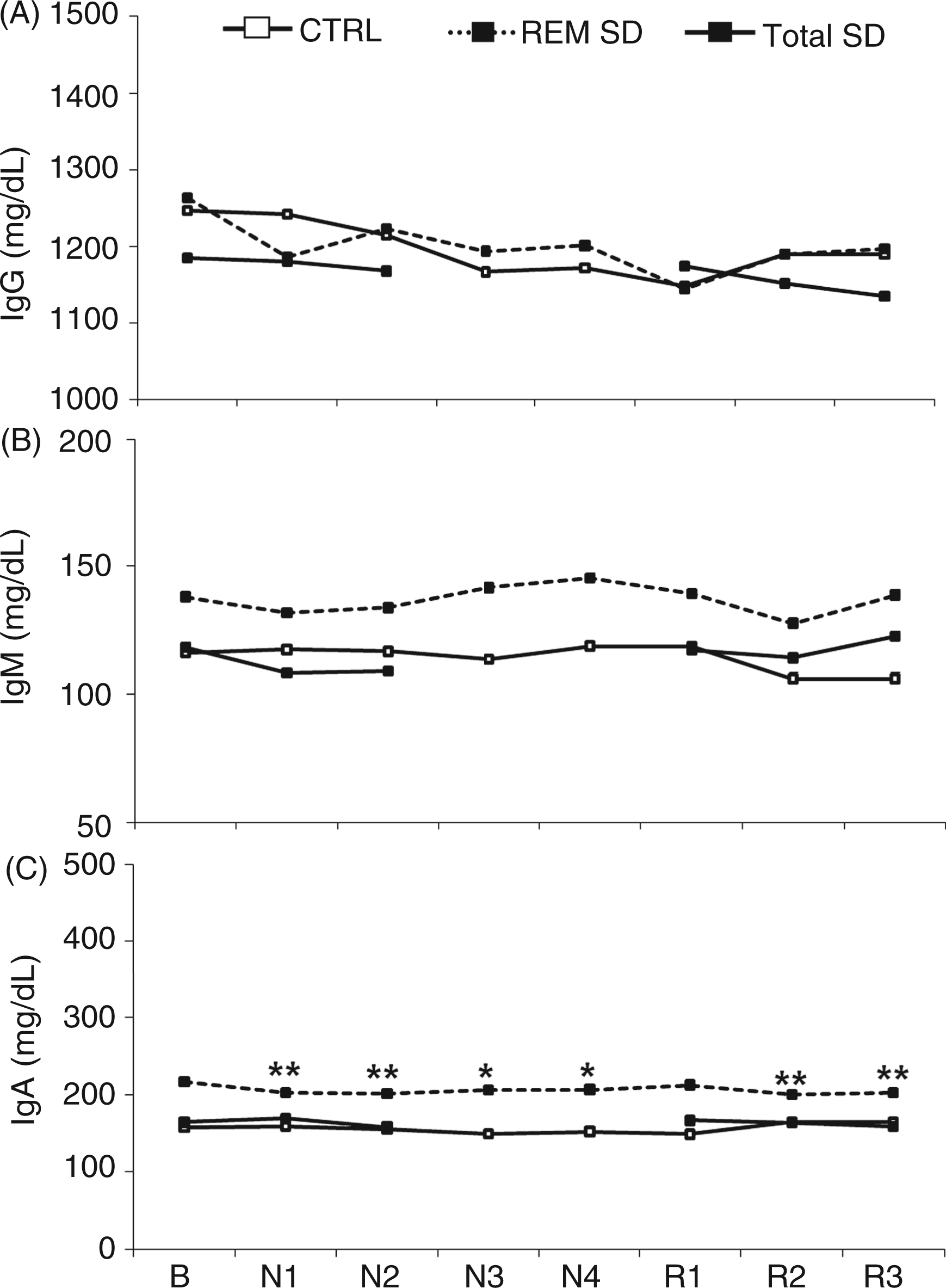
Complement measurements
Complement levels in baseline (B), 2 nights (N1–N2) of total SD, 4 nights of REM SD (N1–N4) and 3 nights of recovery (R) sleep
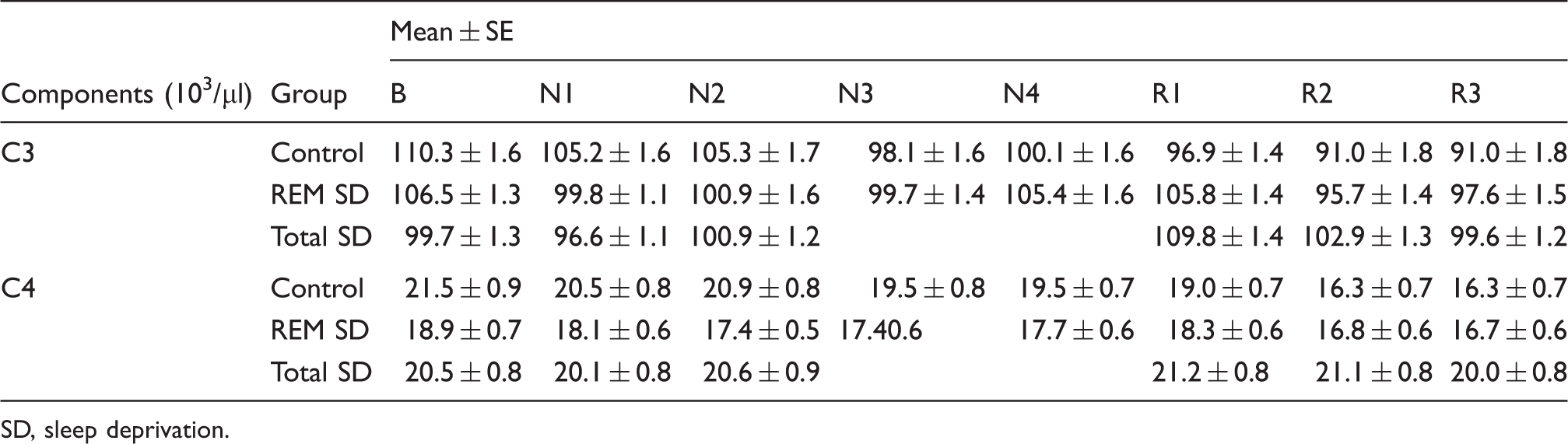
SD, sleep deprivation.
Cytokine measurements
No significant effects on circulating cytokine levels were observed during either SD protocol (Fig. 4). Three volunteers with multiple immunological parameters exceeding reference values in the baseline assessment were excluded from analysis (two from the control group and one from the total SD group). In addition, three volunteers were excluded from IL-1β, IL-10 and IFN-γ analysis for the same reason. Approximately 75% of the IL-1β, IL-2, IL-4, IL-6 and IFN-γ samples were below the lower limit of detection, and these samples were assigned the lower limit of detection value (3.2 pg/ml).
Cytokine levels at baseline (B) and during SD (N) and sleep recovery (R). Mean values ± SE of IL-1β (A), IL-2 (B), IL-4 (C), IL-6 (D), IL-10 (E), TNF-α (F) and IFN-γ (G). Control group: n = 9; total SD group: n = 7; REM SD group: n = 8.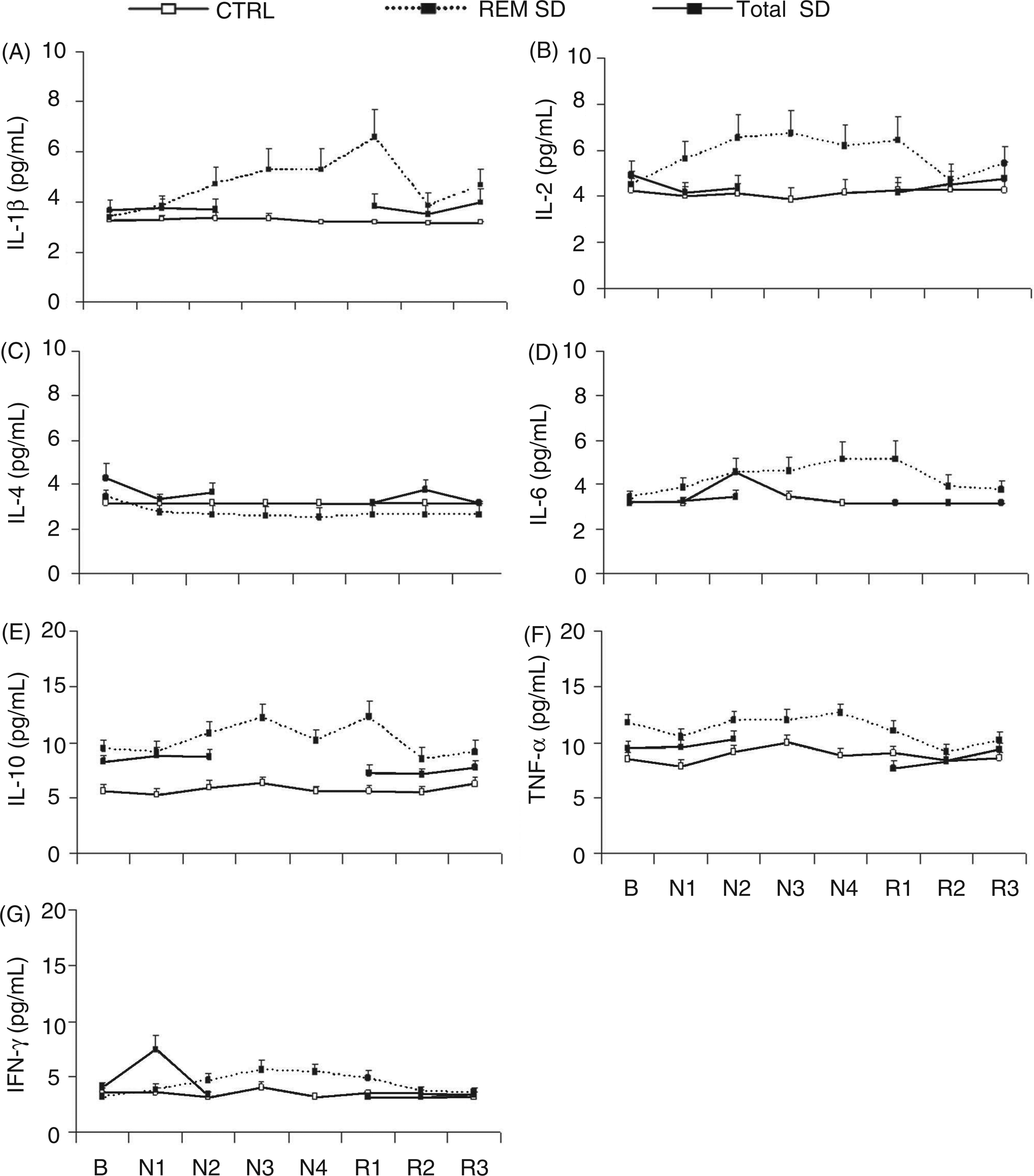
Discussion
The results indicate that both protocols of SD caused marked alterations in immune parameters in healthy young men. Total SD led to increased WBC counts compared with baseline that returned to basal levels after one night of recovery. This alteration was characterized by an increase in neutrophils. The most prominent alteration was the increase in CD4+ T-cells after two nights of total SD compared to baseline, although we did not observe significant alterations in the total numbers of T- and B-lymphocytes or in circulating cytokine levels in either SD protocol. Moreover, CD4+ T-cell percentages did not return to basal levels after three nights of recovery sleep. In the REM SD group, IgA levels decreased during the entire period of SD.
Because sleep is an important factor for maintaining health, identifying the hematological parameters that change during SD is essential for understanding the cellular mechanisms underlying sleep regulation and the functional consequences of sleep loss. Several experiments of total SD have demonstrated alterations in the cellular immune response, although studies performed in humans have not yielded consistent results. In agreement with our results, other studies have shown that circulating levels of WBCs, particularly neutrophils, are elevated during SD.8,11,27– 29 In contrast, other studies3,30,31 did not find such changes. Similar to our findings, Dinges et al. 11 reported that the number of WBCs returned to baseline values after 24 h of sleep recovery.
The SD protocols used herein did not alter the numbers of total B- and T-lymphocytes in agreement with previous reports.3,11,30 Otherwise, when assessing lymphocyte subtypes, we observed increased levels of auxiliary T-lymphocytes (CD4+) after one and two SD nights.
The sleep–wake cycle is thought to be regulated mainly by a combination of homeostatic and circadian mechanisms. 32 Under regular sleep–wake conditions, leukocytes show robust diurnal rhythms with peak counts at night or during the day, depending on the cell type.2,27,33 In the current study, blood was harvested once a day at the same time to minimize the influences of circadian rhythm. However, we did not exclude the possibility that the controversial results in the WBC counts could be related to possible interactions between the effects of SD and circadian rhythm. 27
White blood cell count in the bloodstream is recognized as an important cellular marker of systemic inflammation. The increased number of leukocytes in the peripheral circulation after total SD showed an imbalance in the immune homeostasis suggesting that a state of inflammation settled during SD, an aspect that has been observed by others.29,34,35 In agreement with a previous study, 27 our results indicate a specific regulatory influence of sleep on immune system homeostasis in healthy humans, as sleep was essential for maintaining normal levels of immune parameters, mainly in the total SD group.
Sleep loss has been reported to cause increases in other inflammatory markers, 21 for example, C-reactive protein 36 and inflammatory gene expression. 34 Because a fundamental function of peripheral blood mononuclear cells (PBMCs) is the production and release of cytokines, studies have often measured blood cytokine levels to assess the influence of sleep loss on immune function. According to our results, both total SD and REM SD did not affect the circulating levels of any cytokines.
Findings involving SD and cytokines have yielded controversial results. Some studies have found that patients with nocturnal sleep disturbance show altered circulating concentrations of TNF-α and IL-6,37,38 whereas other studies failed to detect changes in IL-6 as a result of SD.27,39 Plasma IL-1β concentrations were not statistically different between sleep disorder patients and controls, whereas IL-6 was markedly and significantly elevated in sleep apneics. 38 Moreover, SD increased plasma IL-1-like and IL-2-like activity 40 but did not change plasma concentrations of TNF-α, IL-1β, IL-2, IL-6, and IFN-γ. 39 These discrepancies could be related to the fact that in humans, most cytokines, unlike hormones, are released locally and hence show very low systemic concentrations under normal physiological conditions. 41 In this sense, it is important to stress that approximately 75% of our cytokine samples were below the lower limit of detection, which may explain the lack of reliability of the findings. It is necessary to mention that substantial circadian variations have been demonstrated for the production of various cytokines, 42 – 44 which could be a confounding factor in studies on the effects of SD on cytokine levels.
Immunoglobulin and complement proteins are important inflammatory mediators in the assessment of the integrity of immunological response. Our results demonstrated that SD in both protocols did not alter the complement proteins C3 and C4 or affect IgG and IgM, which is in agreement with a previous investigation. 45 Recently, Hui et al. 46 assessed the effects of a night of total SD on men and women and observed increased levels of C3, C4, IgA, IgG and IgM. Such discrepancies can be attributed to differences between genders. Indeed, our previous study showed that SD promoted distinct effects according to gender.9,47
Our results point to a decline in IgA levels during the entire SD REM protocol in relation to baseline, although IgM and IgG levels were unchanged. This decrease in IgA levels during REM SD could reflect the differential homeostatic drive for sleep between the total SD and REM SD conditions. In addition, this decrease could be related to an increased susceptibility to antigenic challenges by means of the mucosae, a fact that is extensively suggested in the literature.15,48 Secretory IgA (SIgA) constitutes the predominant immunoglobulin isotype in secretions, and it is considered the first line of defense of the host against pathogens. So far, no other study evaluated the effects of SD on SIgA levels.
Deprivation of sleep is a stressful situation that has multiple effects on neuro-endocrine function. 45 Studies have shown that SD and sleep restriction lead to increased cortisol levels in the afternoon and evening. 49 The differences observed in our study were not likely to have been the result of other stressors because they occurred without significant increases in 24-h mean levels of cortisol between the conditions. 23 These results suggest that the immune alterations triggered by our SD protocols are independent of hormones, particularly cortisol and adrenocorticotropic hormone (ACTH).3,11,30,50,51
Collectively, our results reinforce the existing hypothesis of the importance of sleep in the integrity of immunity. It is possible to distinguish the distinct effects of total SD in relation to SD REM, since SD has been proven more influential on the immunological response because it alters a higher number of parameters, and such aspects are just as related to innate immunity (WBCs and neutrophils) as to adaptive immunity. Although the immediate effects of total SD on WBCs are robust, we can highlight that these alterations are transient and return to basal levels following a night of recovery sleep. However, the alterations in the adaptive immune response, such as CD4+ T-cells and IgA triggered by total and REM SD consecutively, were not re-established after sleep recovery. These results suggest that adaptive immunity may be more vulnerable to the effects wrought by SD as more than three recovery nights would be needed to re-establish basal levels.
To our knowledge, this is the first study addressing the effects of such total as REM SD protocols in human immune function. In addition, this approach was previously investigated by our group in different animal models of sleep loss (acute vs chronic SD), leading to the conclusion that SD duration, as well as deprivation of specific sleep stages had distinct impact on immunity.9,10
Studies evaluating the effects of sleep loss on immunity are important given the documented decrease in the mean duration of sleep and an increased shift work in recent decades. 52 Although it is becoming widely recognized the negative effects of extended periods of sleep loss (in our study represented by total SD), it is not clear the real impact of extended sleep restriction on health. Our results indicate that both SD protocols triggered marked and different alterations in immune parameters, since less than one week (four-nights) of specific REM SD decreased the IgA levels. The choice of a four nights long protocol was due to the possibility of masking effects in more extensive protocols since we completely suppressed the REM sleep. In this sense, future studies, including prolonged periods of decreased (instead of suppressed) REM SD, are needed to investigate the real impact of prolonged REM sleep restriction on immune profile.
The current study did not compare directly total and REM SD groups because we assumed that both protocols reflect different professional or personal burden. However, future studies are essential in order to access the differences between these two approaches on volunteers randomized in both protocols in a unique experiment. Our results have shown that both protocols affected the human immune profile, although in different parameters. The reason behind the distinct impact of total and REM SD remains unclear. It may have been related to the amount of awake extended and sleep stages affected particularly by each one. Both sleep restriction and extended periods of SD have been related to impairments in immune competence, including an increase in infections53,54 and susceptibility to chronic diseases. 20 How highlighted for one of our results, complementary studies evaluating the effects of SD on SIgA are warranted to provide more evidence about the interface between susceptibility to immune challenges and SD.
Conclusions
Neural systems implicated in the control of sleep also impact on the functioning of host defenses. The challenge for future research is to determine the ultimate meanings of the sleep loss effects in molecular terms in order to clarify the mechanistic processes involved in the impairment of cellular functional activity and the impact of SD on other essential inflammatory markers for immune function.
Footnotes
Acknowledgements
The authors are grateful to Magda Bignotto and Renata Ananias for technical assistance. This work was supported by Associação Fundo de Incentivo à Psicofarmacologia (AFIP); and Fundação de Amparo à Pesquisa do Estado de São Paulo (CEPID #98/14303-3 to ST; #07/55445-6 to FSR; #06/1676 to RCSM; #06/58275-1 to AZ). MLA, JDL and ST are recipients of CNPq fellowships.
