Abstract
Introduction:
Trapeziometacarpal joint stability during pinch depends on the balance between the opponens pollicis and the first dorsal interosseous muscles, which control pronation and supination of the first metacarpal, respectively. Disruption of this muscular balance may contribute to joint instability in patients with trapeziometacarpal osteoarthritis, although the mechanisms underlying movement pattern changes in these patients remain unclear. This study aimed to quantitatively compare thumb rotation patterns during tip pinch between patients with trapeziometacarpal osteoarthtits and healthy individuals.
Methods:
The thumb rotation angle during tip pinch was measured in 36 hands of 27 patients with trapeziometacarpal osteoarthritis and 19 hands of 11 healthy controls using a three-axis gyroscope equipped with a three-axis accelerometer fixed to the dorsum of the thumb metacarpal. The pronation/supination angle was recorded as the rotation around the long axis of the gyroscope.
Results:
The trapeziometacarpal osteoarthritis group demonstrated a lower median change in pronation angle during pinch (2.6°) compared with the control group (3.7°) (p = 0.014). The frequency of the thumb supination during pinch was higher in the trapeziometacarpal osteoarthritis group (25%) than in the control group (8.8 %) (p = 0.013). Pronation angles were moderately and negatively associated with the Eaton–Littler and Kellgren–Lawrence classifications for osteoarthritis.
Conclusions:
Altered thumb rotation patterns during pinch may contribute to joint misalignment and the development of osteoarthritis. The findings may influence targeted rehabilitation interventions to improve dynamic stabilization of trapeziometacarpal joint.
Level of Evidence:
III
Keywords
Introduction
Trapeziometacarpal (TMC) osteoarthritis (OA) has a multifactorial pathology, involving abnormal joint loading, repetitive hand use, hormonal changes associated with the menopause, genetic predisposition, and muscle weakness around the joint (Halilaj et al., 2014a; Koff et al., 2003; Ladd et al., 2014; Mobargha et al., 2016). However, the most widely supported theory suggests that ligamentous laxity or disruption increases TMC joint instability, resulting in joint incongruence during thumb movement (Goto et al., 2014; Lin et al., 2014). The TMC joint is primarily stabilized by the dorsal radial ligament and the anterior oblique ligament (Colman et al., 2007; Edmunds, 2011), whereas its secondary stability depends on the function of the first dorsal interosseous (1DI) and the opponens pollicis (OP) muscles (Adams et al., 2018; Boutan, 2000). Previously, an anatomical study revealed that the dorsal radial ligament forms a part of the joint capsule and 1DI aponeurosis, whereas anterior oblique ligament forms a thin capsule beneath the OP muscle (Norose et al., 2022). Therefore, the functional interaction between 1DI and OP muscles may be involved in the early pathology of TMC OA. To understand TMC OA pathology, an assessment of the kinematic functions of these muscles is warranted.
The 1DI supinates the first metacarpal to resist subluxation forces exerted by the adductor pollicis and flexor pollicis brevis muscles (Boutan, 2000; Brand and Hollister, 1993; McGee et al., 2015), while OP pronates the first metacarpal (O’Brien et al., 2022). Both the 1DI and OP contract antagonistically to stabilize the TMC joint during pinch (Adams et al., 2018; Boutan, 2000). Sahrmann (2002) proposed that musculoskeletal pain is associated with directional susceptibility to movement, where the joints show increased mobility in specific directions, contributing to the pathology. Regarding TMC OA, directional susceptibility to movement of TMC joint – driven by both 1DI and OP – during pinch has been rarely discussed. We hypothesized that patients with TMC OA exhibit some changes in thumb rotation patterns during pinch, compared with healthy individuals.
In this study, we aimed to quantitatively compare thumb rotation patterns during tip pinch between patients with TMC OA and healthy individuals using a three-axis gyroscope (Kuroiwa et al., 2018). A deeper understanding of these biomechanical changes may provide novel insights into TMC OA pathology.
Methods
Study design and participants
This cross-sectional study compared patients with TMC OA with a healthy control group. The TMC OA group included 36 hands from 27 consecutive patients with TMC OA who visited the outpatient department of the Institute of Science Tokyo Hospital between July 2022 and December 2023. The participants included in the present study were a subset of the participants reported in a previous study (Tsukamoto et al., 2025). TMC OA was diagnosed by hand surgeons based on pain at the base of the thumb, physical examination findings, and radiography results. Trapeziometacarpal osteoarthritis was classified according to the Eaton–Littler and Kellgren–Lawrence classifications (Eaton et al., 1984; Kellgren et al., 1957), and patients of all stages were included in the TMC OA group. The control group included 19 hands from 11 volunteers who had undergone total hip arthroplasty at the same institution. The inclusion criteria for the control group were as follows: (i) no hand symptoms and (ii) negative TMC OA findings based on physical examination and plain radiography results. Patients scheduled for total hip arthroplasty at Institute of Science Tokyo Hospital routinely undergo preoperative evaluation using frontal hand radiography to assess the effect of T-cane use. Exclusion criteria for both groups were as follows: (i) a history of or specific symptoms of thumb fracture; (ii) inflammatory hand disease; (iii) peripheral neuropathy; and (iv) central nervous system disorders. Age, sex, and hand dominance did not differ between the TMC OA and control groups (Table 1). As there are limited reports investigating altered pronation angles during pinch in patients with TMC OA, data from previous measurements on 16 cases in the TMC OA group and 15 cases in the control group were used, and an effect size of 0.87 was calculated. Using this effect size, a power analysis was performed with an assumed power of 0.8 and a type I error of 0.05. As a result, the required sample size was determined to be 52 hands (35 with and 17 without TMC OA). Three additional patients were analysed in case of unexpected failure during the study. All participants provided written informed consent, and the Ethics Committee of Institute of Science Tokyo approved the study protocol (no. M2021-264).
Demographic characteristics of study participants.
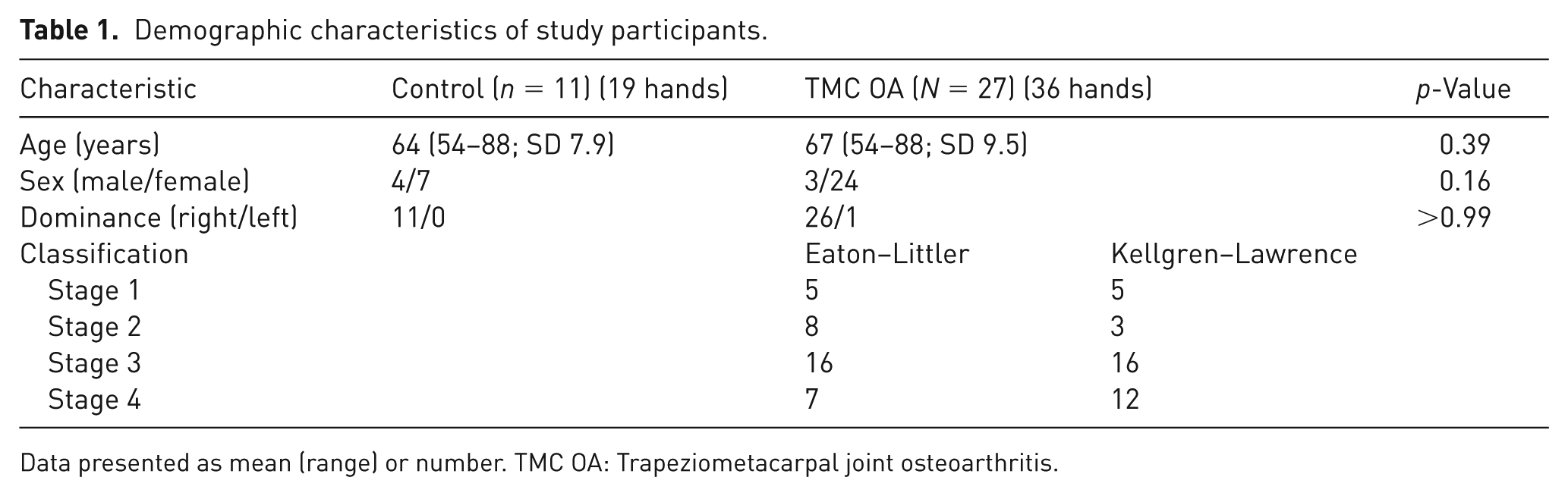
Data presented as mean (range) or number. TMC OA: Trapeziometacarpal joint osteoarthritis.
Measuring the pronation angle during pinch
The thumb rotation angle was measured as described previously (Kuroiwa et al., 2018; Tsukamoto et al., 2025). A three-axis gyroscope equipped with a three-axis accelerometer (MP-M6-02/500C; MicroStone, Nagano, Japan) was used for the measurements. We presented the measured angles with one decimal as previously performed studies using the same equipment (Kuroiwa et al., 2018; Tsukamoto et al., 2025). The gyroscope was fixed to the dorsal aspect of the first metacarpal region. Participants were instructed to make fingertip contact between their thumb and index finger and pinch strongly while maintaining the fingertip contact. During measurements, the wrist and the second to fifth metacarpal heads were secured using straps (Figure 1). Each participant conducted five sequential pinching trials. The rotational angle around the long axis of the gyroscope was considered the pronation/supination angle. The first fingertip contact position was considered the baseline pronation angle. The change in (Δ) pronation angle was defined as the difference between the pronation angle at the start of the pinch and the maximum or minimum angle during strong pinch (Figure 2). Of the five trials, the mean of the second to fourth movements was used. A pinch was categorized as a pronation pattern if the pronation angle was positive (i.e. positive pronation angle) (Figure 2a). In contrast, a pinch was categorized as a supination pattern if the pronation angle was negative (i.e., negative pronation angles) (Figure 2b). The proportions of supination-pattern pinches for the second to fourth pinches were calculated for both the control and TMC OA groups.
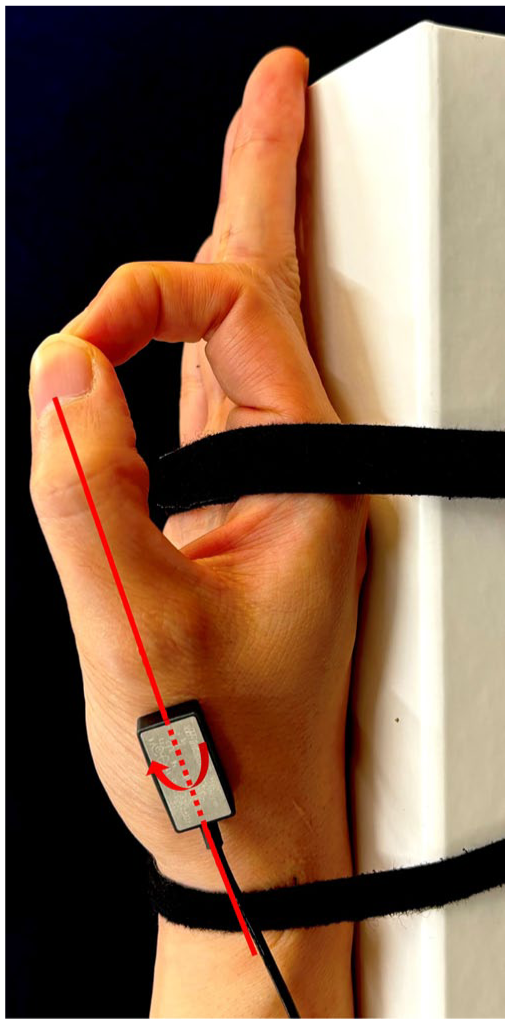
The start position of the pinch. The wrist and the second to fifth metacarpal heads are secured. Pronation angle (red arrow) is defined as the rotational angle around the gyroscope’s longitudinal axis fixed on the dorsal first metacarpal (red line).
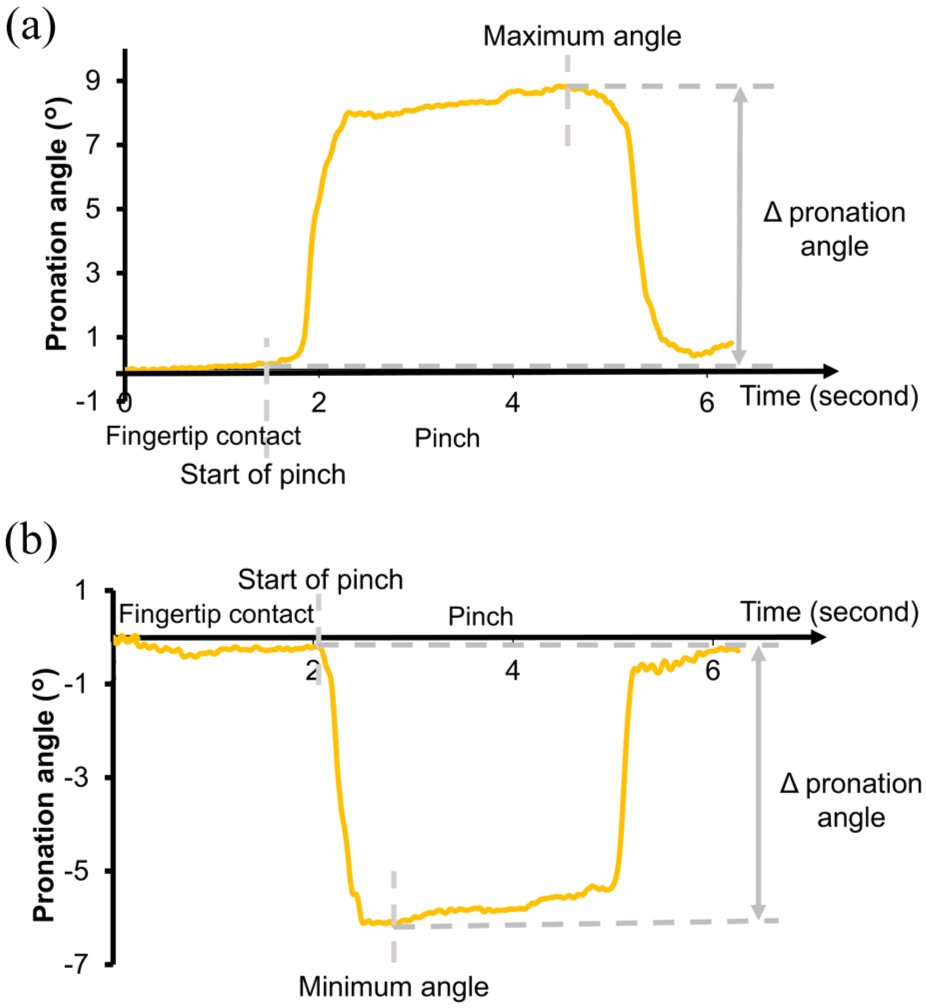
Representative pronation angle changes during pinch. The Δ pronation angle represents the difference in pronation angle between the start of pinch and the maximum or minimum angle. (a) Pronation pattern and (b) supination pattern (positive and negative Δ pronation angle, respectively).
Statistical analysis
Fisher’s exact test was used to analyse differences in sex and hand dominance. Data on age and pronation angle were examined for normal distribution using the Shapiro–Wilk test. Consequently, both datasets were shown not to be normally distributed. Therefore, the Mann–Whitney U-test was used to compare age and pronation angles between the groups. Statistical significance was set at p < 0.05. Spearman’s rank correlation coefficients were used to evaluate the association between pronation angle and disease severity, as evidenced by the Eaton–Littler and Kellgren–Lawrence classifications. The proportions of negative pronation angles in the control and TMC OA groups were compared using Fisher’s exact tests.
Results
Age, sex, and hand dominance did not differ between the TMC OA and control groups (Table 1). The median change in (Δ) pronation angle during pinch in the TMC OA group (2.6° (IQR 0.5 to 3)) was lower than that in the control group (3.7° (IQR 2.6 to 6.0); p = 0.014) (Figure 3). The change in pronation angle demonstrated a moderate negative correlation with the Eaton–Littler classification (r = −0.42, p = 0.0013) and with the Kellgren–Lawrence classification (r = −0.46, p = 0.00037) (Figure 4).
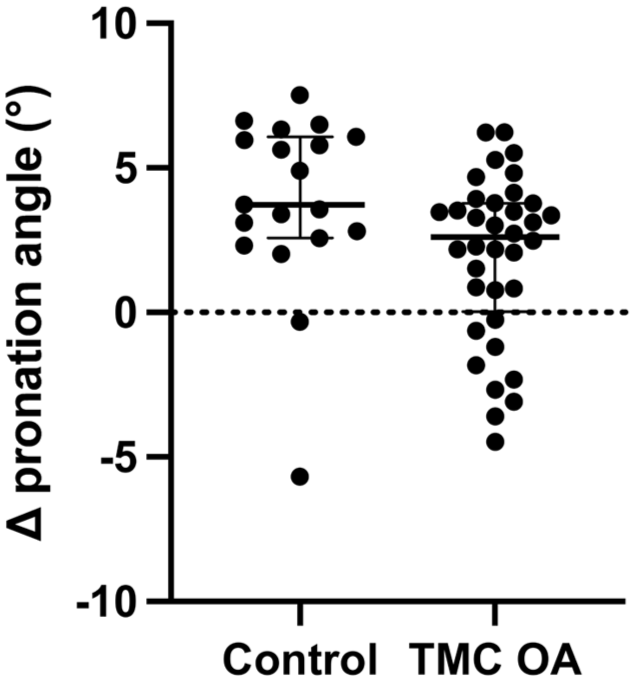
Scatterplot of Δ pronation angles in the control and TMC OA groups. Δ pronation angles are presented as medians and interquartile ranges. Each dot denotes the mean of the Δ pronation angle for each participant. TMC OA: Trapeziometacarpal joint osteoarthritis.
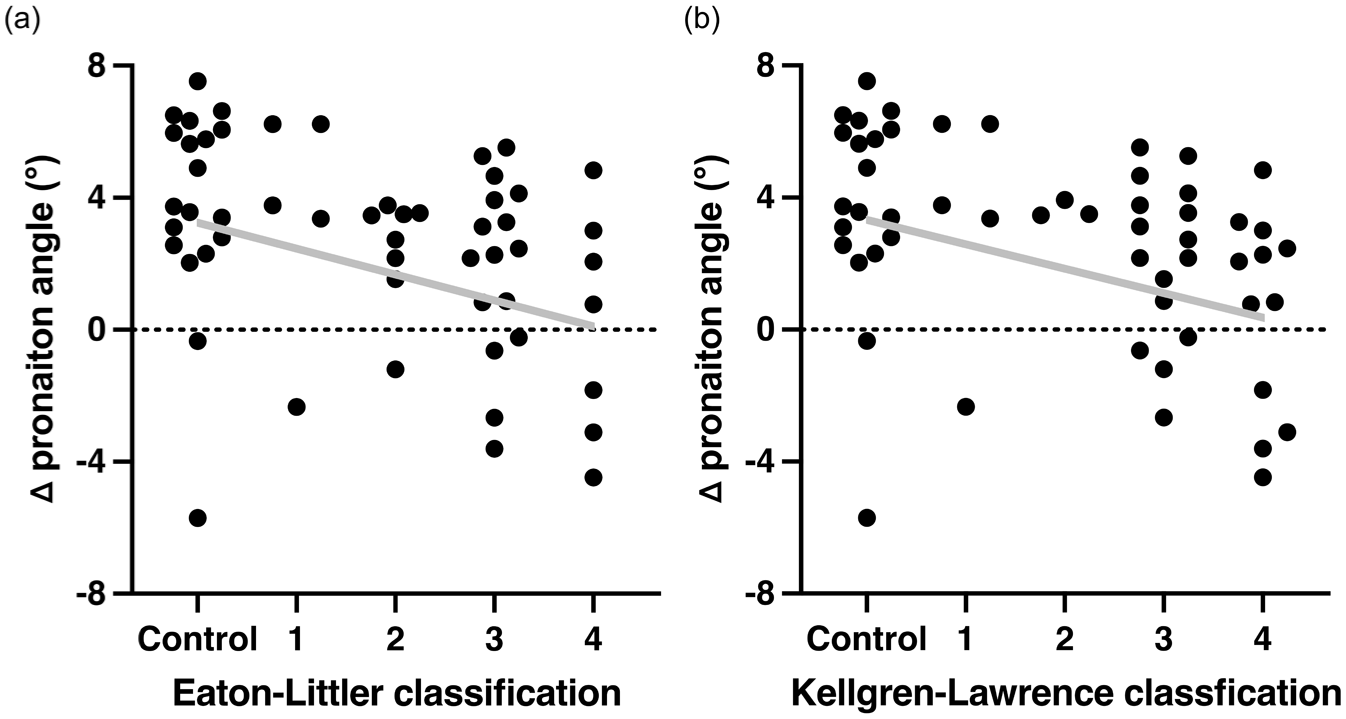
Scatterplots of Δ pronation angles in the control and trapeziometacarpal joint osteoarthritis groups for each (a) Eaton–Littler and (b) Kellgren–Lawrence classifications. Each dot denotes the mean of the Δ pronation angle of each participant. The grey line indicates the regression line.
During pinch, both thumb pronation and supination patterns were observed in both groups (Figure 2). The proportion of the thumb supination pattern during pinch was higher in the TMC OA group (25% (27/108 pinch times in 36 patients)) than in the control group (8.8% (5/57 pinch times in 19 controls); p = 0.013) (Figure S1). In the control and TMC OA groups, two and four hands, respectively, changed pronation and supination patterns during performing three pinches.
Discussion
We employed a three-axis gyroscope to assess thumb rotation patterns during pinch in patients with TMC OA and healthy individuals. The Δ pronation angle was significantly lower in the TMC OA group than in the control group. Additionally, the pronation angle in patients with TMC OA exhibited a moderate negative correlation with the Eaton–Littler and Kellgren–Lawrence classifications. Furthermore, while both groups demonstrated cases of thumb supination during pinch, the TMC OA group demonstrated a significantly higher frequency of supination.
Reports on thumb rotation during pinch are controversial. Cooney et al. (1981) and Brand and Hollister (1993) described TMC joint pronation during pinch, whereas Halilaj et al. (2014b) and Inaba et al. (2024) reported cases of TMC joint supination. This inconsistency elucidates the uncertainty regarding thumb rotation during pinch movement. Biomechanical studies of TMC OA specimens have primarily focused on the position of the first metacarpal and joint space (D’Agostino et al., 2017; Morton et al., 2023), with limited focus on thumb rotation. Although both healthy controls and patients with TMC OA demonstrated a pronation pattern while pinching, the mean pronation angle was lower in patients with TMC OA than in healthy controls. Additionally, the frequency of the supination pattern while pinching was significantly higher in the TMC OA group than in the control group. Therefore, thumb rotation patterns vary between healthy and pathological conditions.
Changes in thumb rotational movements during pinch may contribute to the early pathology of TMC OA. Regarding thumb rotation, 1DI supinates the thumb (Boutan, 2000; Brand and Hollister, 1993), whereas the OP pronates it (O’Brien et al., 2022). These muscles maintain balance through a competitive interplay (Boutan, 2000; Adams et al., 2018; Boutan, 2000). Thumb pronation and supination are synchronized with abduction and adduction, respectively (Crisco et al., 2015; Halilaj et al., 2014b). Supination during pinch results in radial and dorsal deviation of the first metacarpal (Kuo et al., 2014). Sahrmann et al. (2002) proposed that directional susceptibility to movement is associated with musculoskeletal pain in various joints, such as the shoulder, knee, and lumbopelvic region (Caldwell et al., 2007; Kajbafvala et al., 2016; Kim et al., 2013). TMC OA is characterized by a dorsoradial subluxation and adduction deformity (Bettinger et al., 1999). The TMC joint is specifically susceptible to the supination direction along with disease progression. Healthy individuals maintain thumb pronation during pinch. However, if the OP muscle output decreases because of muscle fatigue (Hu et al., 2018; Todd et al., 2010), compensatory activation of the 1DI may trigger thumb supination. The decreased Δ pronation angle and increased frequency of supination pattern in the TMC OA group support our hypothesis. Furthermore, the correlation between the pronation angle and arthritic progression suggests a decline in the pronation angle even in the early stages of TMC OA. While our findings suggest that muscle imbalance contributes to reduced pronation in TMC OA, we cannot exclude the possibility that structural changes and stiffness of TMC joint also play a role in limiting thumb pronation (D’Agostino et al., 2018). We assumed that these relationships could be seen as a chicken-and-egg effect and influence each other to contribute to the TMC OA progression.
Regarding therapeutic interventions, previous studies have emphasized the importance of strengthening the 1DI muscle for the treatment of TMC OA as a stabilizer of TMC joint during pinch (McGee et al., 2015; McVeigh et al., 2022; O’Brien et al., 2013). This study did not establish a causal association between thumb supination during pinch and TMC OA development. However, based on the findings, it is suggested that improving thumb pronation by facilitating OP muscle during pinch might serve as a valuable therapeutic intervention, maintaining proper thumb movement patterns. The reduced pronation angle observed in the TMC OA group, along with the increased frequency of the thumb supination pattern, indicates that OP dysfunction may contribute to altered kinematics and compensatory 1DI and adductor muscle activation. Therefore, while 1DI strengthening has been previously recommended, rehabilitation strategies should also focus on improving OP function to restore a muscular balance between 1DI and OP muscles. Neuromuscular re-education, including OP strengthening, enhances the dynamic stabilization of TMC joint (DeMott, 2017; O’Brien et al., 2022), supporting our therapeutic suggestion.
This study has some limitations. First, skin displacement between the sensor and the first metacarpal may have influenced angle measurements during pinch. However, most skin movement occurs along the longitudinal axis, minimizing its effect on pronation angle measurements (Gehrmann et al., 2010). Second, OP muscle contraction was not directly evaluated. Future studies incorporating surface electromyography or ultrasound imaging could assess OP activity and contraction during motion. Third, this study adopted a cross-sectional design, necessitating prospective research to clarify the association between TMC OA pathology and impaired OP function.
In conclusion, this study demonstrated thumb rotation patterns during pinch in both healthy controls and patients with TMC OA. In the TMC OA group, pronation angles were lower, showing a moderate negative correlation with the Eaton–Littler and Kellgren–Lawrence classifications. The TMC OA group also exhibited a higher frequency of thumb supination during pinch. Therefore, altered thumb rotation patterns during pinch may contribute to joint misalignment and the development of TMC OA. Our findings may influence targeted rehabilitation interventions.
Supplemental Material
sj-jpg-1-jhs-10.1177_17531934251383073 – Supplemental material for Thumb rotation patterns during pinch in patients with trapeziometacarpal osteoarthritis
Supplemental material, sj-jpg-1-jhs-10.1177_17531934251383073 for Thumb rotation patterns during pinch in patients with trapeziometacarpal osteoarthritis by Kazuya Tsukamoto, Koji Fujita, Tomoyuki Kuroiwa, Mio Norose, Toshitaka Yoshii and Akimoto Nimura in Journal of Hand Surgery (European Volume)
Footnotes
Acknowledgements
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by a grant from JA Kyosai Research Institute (Agricultural Cooperative Insurance Research Institute); JSPS KAKENHI (grant number JP21H03485); and the Japanese Society for Surgery of the Hand.
Ethical approval
Ethical approval for this study was obtained from the ethics committee of Institute of Science Tokyo.
Informed consent
Written informed consent was obtained from all subjects before the study.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
