Abstract
Introduction:
The neutrophil elastase (NE) inhibitor is a potential treatment strategy for acute lung injury (ALI) or acute respiratory distress syndrome (ARDS). However, the clinical effectiveness of sivelestat sodium, a selective NE inhibitor, remains controversial. We performed a systematic review and meta-analysis to evaluate the effects of sivelestat in patients with ALI/ARDS.
Method:
The literature search, selection, and data extraction were conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analysis Statement (PRISMA) guidelines. The randomized controlled trials (RCTs) with reference lists were retrieved from Scopus, PubMed, and Cochrane Library, using the Cochrane risk-of-bias tool for the quality assessment. Logarithm relative risk (logRR), risk difference (RD), and standardized mean difference (SMD) were calculated using the fixed effects model or random effects model, depending on heterogeneity.
Result:
Ten RCTs involving 1170 patients (583 receiving sivelestat and 587 receiving standard care or placebo) were included. Sivelestat was associated with a significant reduction in 28–30-day mortality, shorter duration of mechanical ventilation, and improvement in the ratio of arterial oxygen partial pressure to fractional inspired oxygen (PaO2/FiO2 ratio) in a time-dependent manner. These beneficial effects were more pronounced in patients with sepsis-related ALI/ARDS. The incidence of adverse events did not differ between groups.
Conclusion:
Sivelestat might be a promising adjunctive treatment for ALI/ARDS, especially in the sepsis etiology, as evidenced by reduced mortality, shortened mechanical ventilation duration, and improved oxygenation. The large-scale, well-designed RCTs are warranted.
Keywords
Introduction
Acute lung injury (ALI) and acute respiratory distress syndrome (ARDS), especially in patients with systemic inflammatory response syndrome (SIRS), are common conditions with high morbidity and mortality in most intensive care units (ICUs). 1 According to the American College of Chest Physicians and the Society of Critical Care Medicine,1,2 SIRS is triggered by direct pulmonary damage (pneumonia, gastric aspiration, and inhalation injury) and indirect lung injury (sepsis, trauma, pancreatitis, burns, drug overdose, multiple blood transfusions, and shock), which induces excessive inflammation in the lung epithelium and vascular endothelium.2,3 While the respiratory supportive care for ALI/ARDS is continuously improving, the novel pharmacologic therapies are also mentioned. 3 Although various pharmacological treatments, including corticosteroids, alprostadil, acetylcysteine, surfactant, and pentoxifylline, have been investigated, data regarding their effectiveness are limited and remain controversial. 1 Neutrophils are the first line of defense against infections,4,5 but might induce excessive inflammatory responses and organ injuries in ALI/ARDS,6,7 as observed in patients with sepsis and severe COVID-19.8,9
Neutrophil elastase (NE) is one of the physiological and pathological proteolytic enzymes (serine proteases) released from the activated neutrophils to digest the extracellular matrix and promote neutrophil chemokines that exacerbate lung damage. 10 Indeed, elevated NE in alveolar lavage fluid is demonstrated in animals with endotoxin- and bacteria-induced ALI /ARDS, while elevated serum NE levels in patients with ARDS are closely related to the disease severity.10,11 Hence, NE inhibition might be beneficial in ALI/ARDS through the reduction of lung damage.11,12 Although the potential therapeutic role of NE inhibitors (endogenous or synthetic) is mentioned in different models of inflammatory tissue damage, the results are still inconclusive, and the translation of these findings to clinical practice remains a challenge. Sivelestat sodium, discovered by the Ono Pharmaceutical Company (Osaka, Japan), 13 is a selective small molecular weight (434 Da) NE inhibitor with a reversible competitive inhibitory property, which is stable in vitro in the presence of the strong oxidants (such as hypochlorous acid). 13 Although sivelestat was approved in Japan for the treatment of “ALI-associated SIRS” in 2002, the Pharmaceuticals and Medical Devices Agency (PMDA), Japan’s regulatory authority, mandated a post-market clinical study to reassess the efficacy and safety of sivelestat in real-world clinical settings. 14 However, the efficacy of sivelestat in patients with ALI/ARDS remains controversial. Some medical institutions were hesitant to use sivelestat due to a desire for further clinical evidence, particularly after the STRIVE study was published with negative results in 2004. 15 Over the past two decades, numerous RCTs have been further conducted, and the efficacy of sivelestat on ALI/ARDS should be reassessed. Here, a systematic review and meta-analysis of the available randomized controlled trials (RCTs) was conducted to evaluate the effectiveness of sivelestat in patients with ALI/ARDS. The larger sample size from the pooled analysis may allow a better quantitative assessment of sivelestat efficacy than the individual study.
Method
Study protocol and search strategy
This study was conducted and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-analysis Statement (PRISMA) guidelines (Supplemental Material) 16 and registered at the international PROSPERO database for prospective systematic reviews (CRD420250645302). The literature search was independently performed by two authors (K.S-k. and A.C.) using a standardised approach to enroll the trials of selective NE inhibitor (sivelestat) for patients with ALI/ARDS published from January 2000 to September 2024. Online electronic databases, including PubMed, Scopus, the Cochrane Library, and CliniclTrials.gov were searched using the following keywords: (“sivelestat” OR “elaspol” OR “neutrophil elastase inhibitor”) AND (“ARDS” OR “acute respiratory distress syndrome” OR “ALI” OR “acute lung injury” OR “lung injury” OR “respiratory insufficiency” OR “respiratory failure” OR “systemic inflammatory response syndrome” OR “shock” OR “sepsis”) AND (“humans” OR “clinical trials” OR “RCT” OR “randomized controlled trials” OR “observational studies” OR “cohort”). Manual searches of the reference lists were also conducted from all relevant original and review articles to identify additional eligible studies. Preprint articles were also enrolled. The included studies met the following criteria: (I) RCT or observational studies evaluating the use of sivelestat in humans, (II) the intervention group receiving sivelestat compared against a control group (did not receive sivelestat) with standard care or placebo, (III) patients confirmed with ALI/ARDS and needing to be supported with a mechanical ventilator, and (IV) research data represented interested outcomes such as 28–30-day mortality, the ratio of arterial oxygen partial pressure to fractional inspired oxygen (PaO2/FiO2 ratio), or ventilation-free day. Several included studies used the term “ALI,” which was commonly applied before the 2012 Berlin definition of ARDS. For consistency with the original study terminology, we report outcomes as “ALI/ARDS,” but acknowledge that in current clinical practice ALI has been replaced by mild, moderate, and severe ARDS under the Berlin definition. Studies were excluded if they were not written in the English language or if the full text was not available. When the outcomes of studies were reported in multiple publications, the publication with the largest patient cohort with relevant outcome data was included in this review. Results from case reports, reviews, abstracts, and replies/commentaries were also excluded.
Data collection and quality assessment
Study selection was carried out by two independent reviewers who applied the eligibility criteria to identify relevant studies for inclusion (K.S-k. and A.C.). The studies were first screened at the abstract, followed by full-text screening to determine the final inclusion, and the duplicates were removed. Any inconsistencies were settled by a group discussion (with the reviewers who are pulmonologists, M.P., N.V., and P.M.) until a consensus was reached. The medical subject heading, methods, patient disease status, study design, intervention, and outcome variables were used to identify relevant studies. The eligibility decisions and the reasons for excluding or including participants were recorded using the Excel spreadsheets. For the data extraction, two reviewers autonomously extracted data from the included studies. The data were extracted from study design, study protocol (e.g., doses of sivelestat and duration of administration), sample size, country, patient demographics, baseline characteristics, and clinical outcomes (mortality rate, PaO2/FiO2 ratio, and ventilator use). Ventilator days (VD) and ventilator-free days (VFD) were treated as separate outcomes and were not combined, as they represent different clinical endpoints. As such, VD reflects the total duration of mechanical ventilation, whereas VFD incorporates both survival and freedom from ventilation within a defined time frame.
The data that were primarily presented with median and range or interquartile range (IQR) were estimated as mean and standard deviation (SD) by a previously published statistical formula. 17 Two investigators independently evaluated the bias risk of the included studies and cross-checked the evaluation results. Disagreements were discussed or adjudicated by a third investigator. The risk of bias tools were assessed by the Cochrane risk-of-bias tool for randomized trials version 2 (RoB2) tool for included RCTs. 18 These assessments were done for each individual study to determine the overall risk of bias in the included studies.
Statistical analysis
Log relative risks (logRR), risk difference (RD), and standard mean differences (SMD) with 95% confidence intervals (CI) were calculated using outcomes extracted from each study before data pooling and presented using the forest plots. Pooled relative risk (RR) with 95% CI was represented to estimate the effectiveness of sivelestat treatment on dichotomous outcomes, such as the risk of 28–30-day mortality. Pooled SMD with 95% CI was used to estimate the sivelestat efficacy on continuous outcomes, such as the 28-day ventilation-free days and PaO2/FiO2 levels, because outcomes were reported using different scales. Effect sizes were interpreted as small (~0.2), moderate (~0.5), and large (>0.8), in accordance with current methodological guidance. 19 The fixed-effects model or random-effects model was used to pool the data and to calculate the results due to the presence of relevant statistical heterogeneity. Statistical heterogeneity between the trials was evaluated using Cochran’s Q test, with a p < 0.05 and the I2 value >50%. 4 Asymmetry in the funnel plot and Egger’s test was considered as the presence of potential publication bias. 20 The subgroup analysis was also performed based on study design, patient characteristics (e.g., causes of ALI/ARDS, baseline of PaO2/FiO2 ratio, and the Acute Physiology and Chronic Health Evaluation II (APACHE-II)). All reported p values were two-tailed, and p < 0.05 were considered as statistically significant. Statistical analyses were performed using the IBM SPSS package software version 22 (IBM Corp., New York, USA).
Results
Study inclusion and study characteristics
A total of 361 records were retrieved from the four databases according to the keyword search strategies. After the elimination of duplicate articles, 161 of 361 records were initially reviewed and excluded according to the title and abstracts. The remaining 21 full-text articles were examined. A manual search of the reference lists of these trials added three eligible studies. Finally, a total of 10 articles15,21–29 containing 1170 patients (583 in the sivelestat intervention group and 587 in the control group) were assessed to be included in this systematic review according to the inclusion and exclusion criteria (Figure 1). Details of included studies were represented (Table 1). The majority of trials were single-center studies conducted in Japan and China, while three trials were multi-center studies.15,22,27 In details of etiology, four of the included trials focused on sepsis-related ALI/ARDS,21,27–29 two on postoperative ALI/ARDS,24,25 and four on multiple causes.15,22,23,26
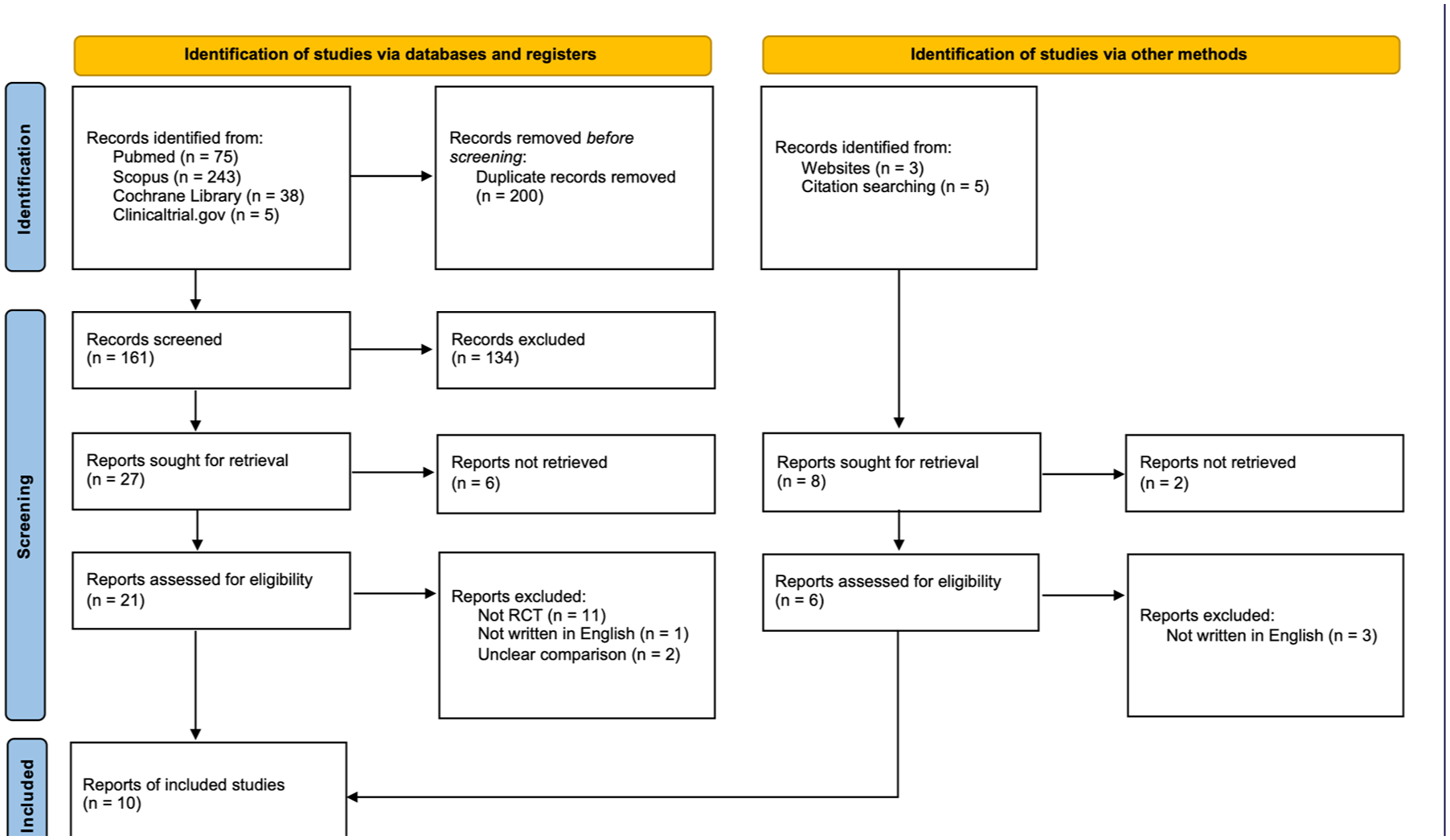
PRISMA diagram of selected included studies.
Characteristics of included studies.
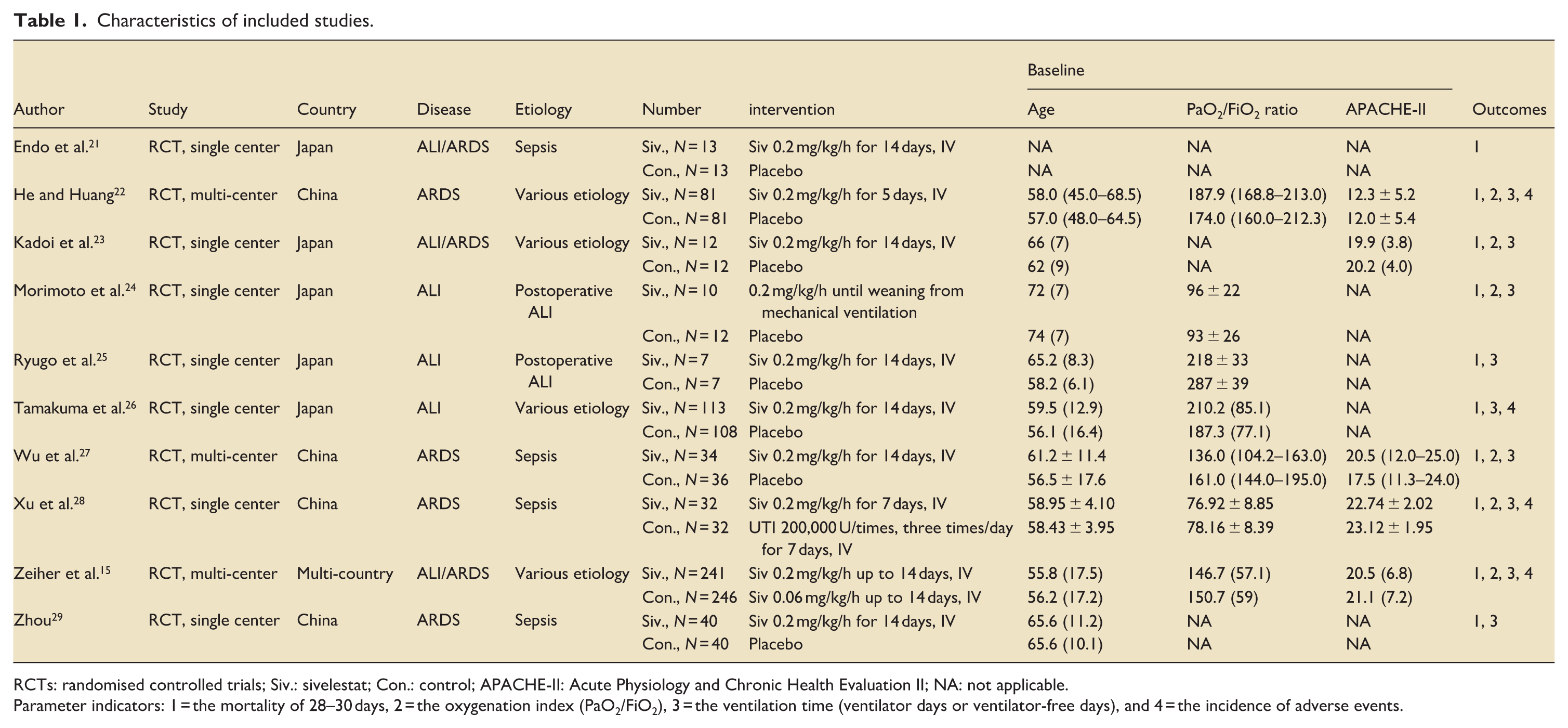
RCTs: randomised controlled trials; Siv.: sivelestat; Con.: control; APACHE-II: Acute Physiology and Chronic Health Evaluation II; NA: not applicable.
Parameter indicators: 1 = the mortality of 28–30 days, 2 = the oxygenation index (PaO2/FiO2), 3 = the ventilation time (ventilator days or ventilator-free days), and 4 = the incidence of adverse events.
Risk of bias assessment
The risk of bias among included RCT studies was assessed by the RoB2 tool (Figure S1). All of the studies were considered to have RoB risks. Five studies may have had a problem with the experimental design, which resulted in a RoB score of “high risk” due to the possible contamination of substances. Three of five were rated regarding allocation concealment due to significant differences between treatment and control arms.21,27,29 All studies were defined as unclear biases because they failed to elaborate in the article.
Efficacy of sivelestat treatment on mortality outcome
A total of 10 included trials reported the effect of sivelestat on the risk of 28–30-day mortality. The overall results of 10 studies,15,21–29 containing a total of 1170 participants (583 in the sivelestat group and 587 in the control group), indicated significant improvement of survival in sivelestat treatment compared to controls for the risk of 28–30-day mortality (logRR: −0.37; 95% CI: −0.56 to −0.18; p < 0.01; Figure 2(a) and RD: −0.10; 95% CI: −0.15 to −0.05; p < 0.01; Figure 2(b)), without evidence of heterogeneity (Figure 2(a) and (b)) and publication bias with Egger’s test (RR: p = 0.14 and RD: p = 0.20) and funnel plot (Figure S2(a) and (b)).
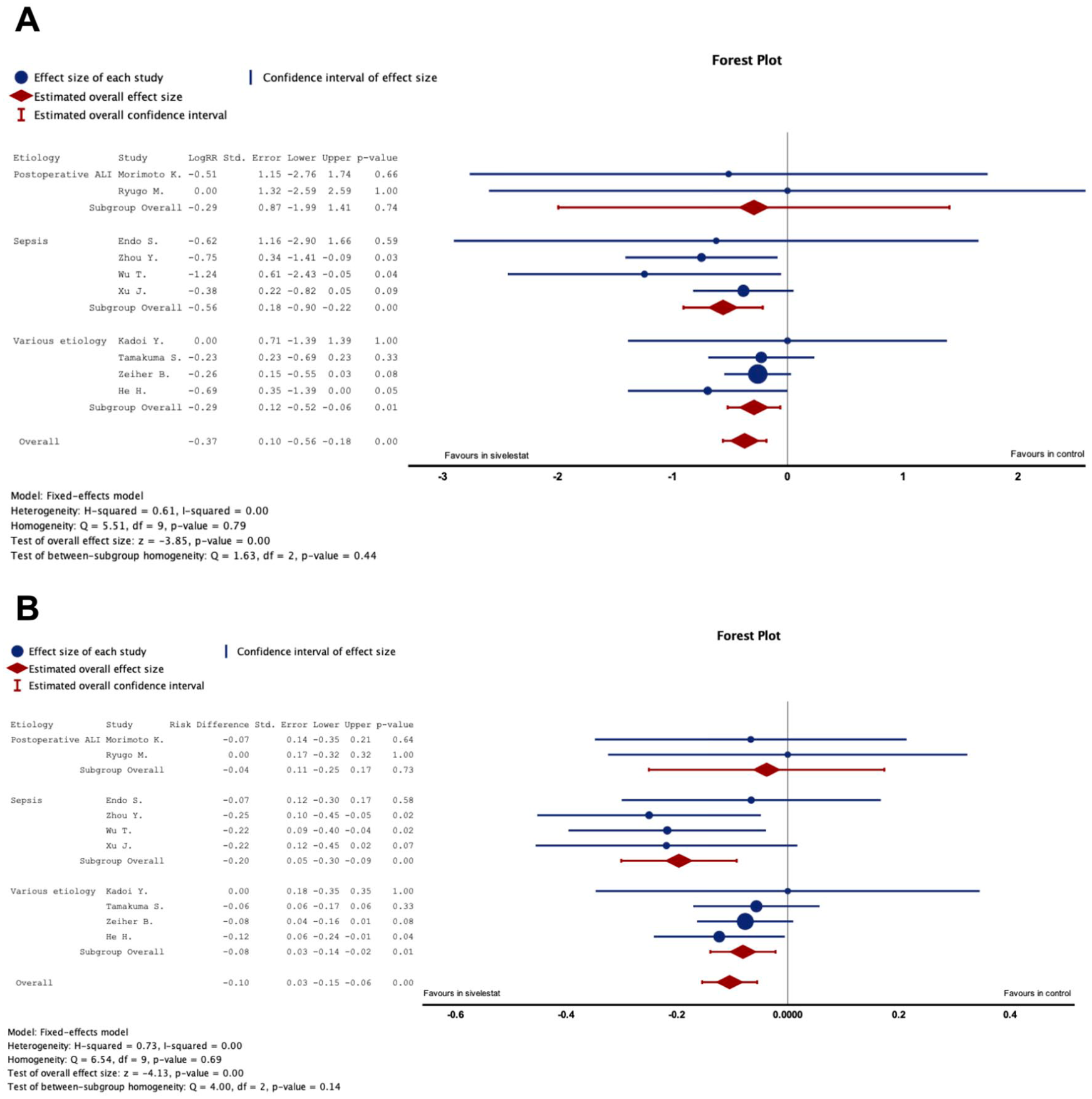
Representative forest plot to demonstrate the efficiency of 28–30-day mortality measured by logRR (a) and RD (b).
Subgroup analysis revealed that the mortality benefit of sivelestat was most pronounced in studies focusing exclusively on sepsis-related ALI/ARDS21,27–29 (logRR: −0.55; 95% CI: −0.56 to −0.18; p < 0.01; I2: 0%; Figure 2(a) and RD: −0.17; 95% CI: −0.07 to −0.27; p < 0.01; I2: 0%; Figure 2(b)). In contrast, no statistically significant reduction in mortality was observed in studies limited to postoperative ALI24,25 (logRR: −0.29; 95% CI: −1.99 to 1.41; p = 0.74; I2: 0%; Figure 2(a) and RD: −0.17; 95% CI: −0.07 to −0.27; p < 0.01; I2: 0%; Figure 2(b)). Other parameters, including age, PaO2/FiO2, and APACHE-II score at enrollment, were also evaluated (Table 2).
Subgroup analysis of 28–30-day mortality.
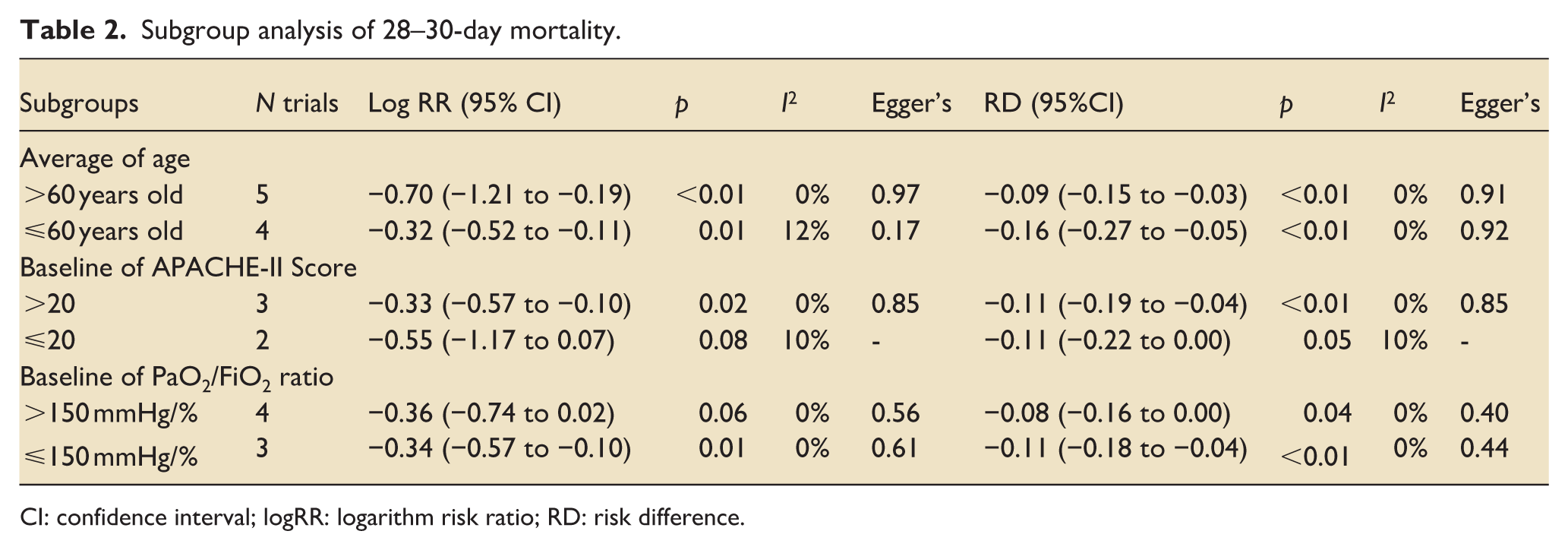
CI: confidence interval; logRR: logarithm risk ratio; RD: risk difference.
Efficacy of sivelestat treatment on clinical respiratory outcomes
In the clinical respiratory outcomes, this study provided the PaO2/FiO2 ratio and ventilator times. There were six included studies15,22–24,27,28 involving 829 patients that provided the PaO2/FiO2 ratio for 7 days after sivelestat treatment. Overall analysis of meta regression in the included studies showed that sivelestat significantly increased the PaO2/FiO2 ratio in patients with ALI/ARDS compared with the control group in a time-dependent manner (coefficient test = 28.86; p < 0.01; Figure 3(a)). After removal of one study 27 with a high risk of bias and substantial clinical heterogeneity during sensitivity analysis, the pooled effect was altered, but the association remained statistically significant (coefficient test = 46.82; p < 0.01; Figure 3(b)).
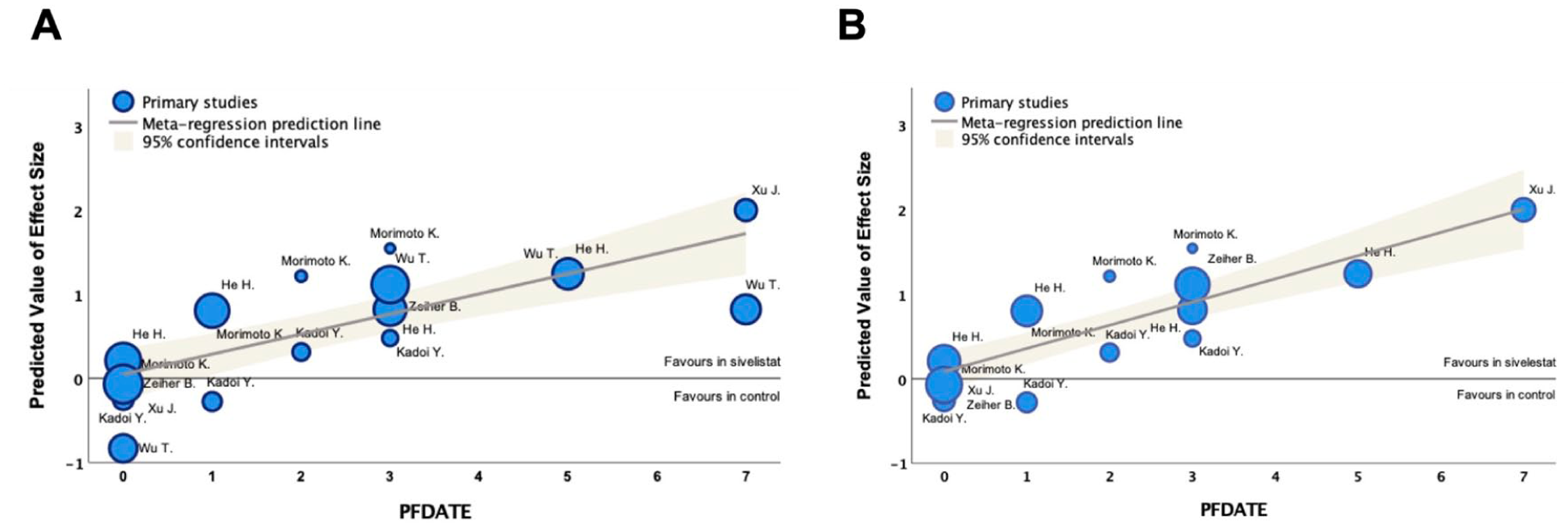
Representative bubble plot to demonstrate the efficiency of PaO2/FiO2 levels for 7 days measured by metaregression analysis of all included studies (A) and removal study with a high risk of bias (B).
Regarding ventilator use-related outcomes, five studies involving 204 patients reported ventilator days (VD),23–25,28,29 while four studies involving 940 patients reported 28-day-ventilator-free days (VFD).15,22,26,27 Overall analysis showed that sivelestat was associated with an increase in ventilator-free days (VFD; SMD = 0.18; 95% CI: 0.01–0.35; p = 0.03; Figure 4) in patients with ALI/ARDS, corresponding to a reduced duration of ventilator days (SMD = −0.95; 95% CI: −1.17 to −0.16; p = 0.02; Figure 4). As such, an SMD of 0.18 for VFD represents a small effect size and may not necessarily translate into clinically meaningful benefit, while an SMD of −0.95 for ventilator days represents a large statistical effect but should be interpreted cautiously given heterogeneity and study design limitations. Other parameters, including age, PaO2/FiO2 ratio, and APACHE-II score at enrollment, were also evaluated (Table 3).
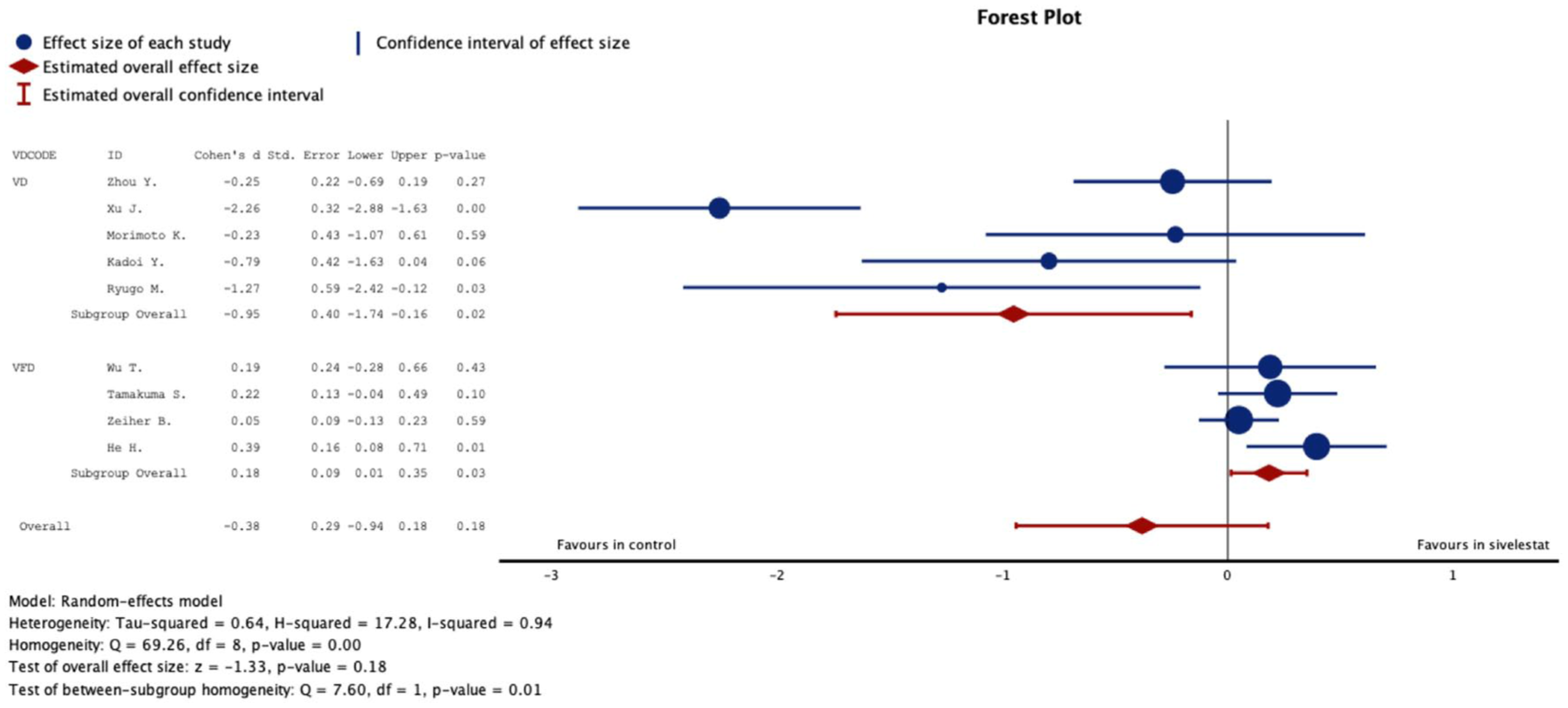
Representative forest plot to demonstrate the efficiency of VD and 28-day VFD measured by the SMD.
Subgroup analysis of VD and VFD.
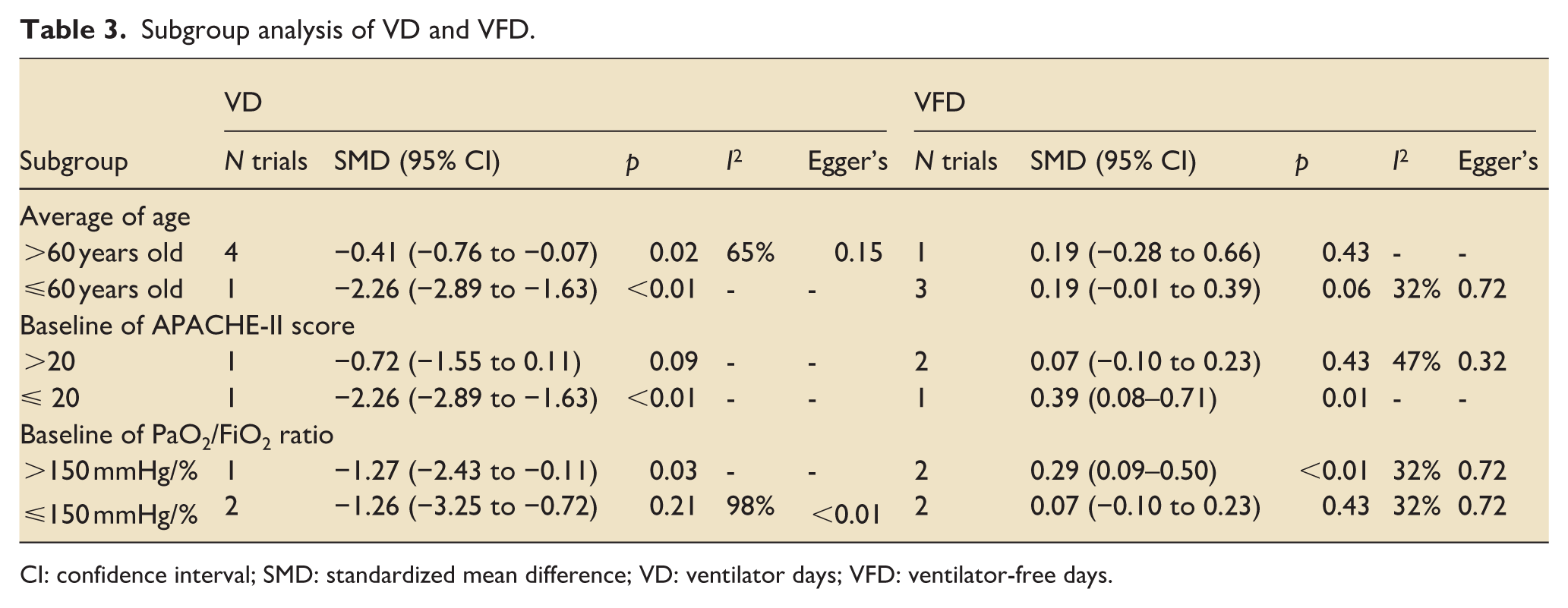
CI: confidence interval; SMD: standardized mean difference; VD: ventilator days; VFD: ventilator-free days.
Adverse events of sivelestat treatment
Regarding outcomes of adverse effects, there were four studies15,22,26,28 containing 934 patients that reported comparative adverse effects (AEs). Overall analysis showed no significant differences of AEs in ALI/ARDS patients with SIRS compared with the control group (logRR = 0.00; 95% CI: −0.07 to 0.07; p = 0.98; Figure 5), without evidence of heterogeneity (I2 = 0%, p = 0.63).
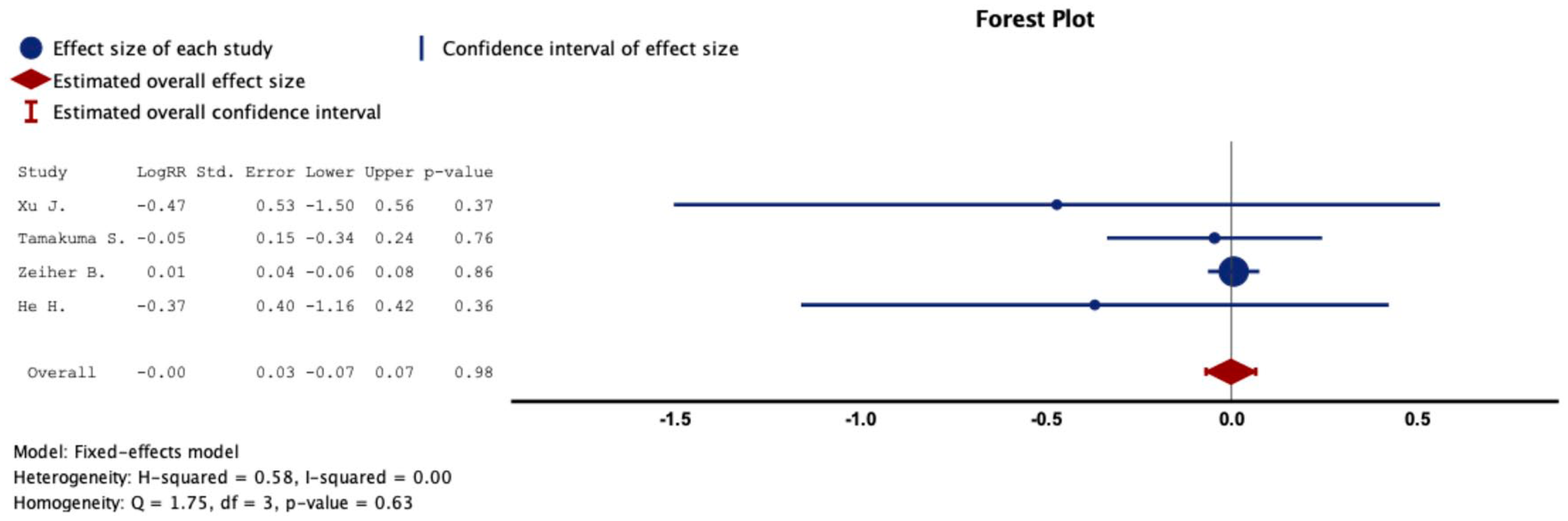
Representative forest plot to demonstrate the efficiency of AEs measured by logRR.
Discussion
In this systematic review and meta-analysis of RCTs, a total of 10 studies were included to evaluate the efficacy of sivelestat in patients with ALI/ARDS. Overall, sivelestat treatment was associated with improvements in several key clinical outcomes, including ventilator requirements and oxygenation (PaO2/FiO2 ratio). In addition, a reduction in short-term mortality was observed in pooled analyses with low statistical heterogeneity, suggesting a potential survival benefit under specific clinical conditions. These findings differ from previous Japanese meta-analyses,14,30 which demonstrated improvements primarily in oxygenation indices without a significant reduction in mortality. This discrepancy may be partially explained by the larger pooled sample size, inclusion of more recent trials, and broader patient populations in the current analysis, which increased statistical power to detect differences in clinically relevant outcomes. Earlier analyses were based on early post-marketing studies and data derived predominantly from Japanese patient populations using the ICHUSHI database. In contrast, the present systematic review incorporated larger and more contemporary trials from multiple regions and applied a predefined protocol with dual independent screening and data extraction that strictly adhered to the PRISMA guidelines. Nevertheless, when considering the overall ALI/ARDS population, the beneficial effects of sivelestat were not uniformly observed across all included studies, indicating that the benefit may not be generalizable to unselected patients. By further performing subgroup analyses, our study extends previous work by identifying sepsis-related ALI/ARDS that derived more pronounced benefits from sivelestat treatment. These findings suggest that the clinical efficacy of sivelestat is context-dependent and influenced by underlying disease etiology and severity, rather than representing a universal treatment effect across all forms of ALI/ARDS.
While the potential benefits of sivelestat are promising, optimizing its administration is crucial to improve its effectiveness. The discordance in the effect of sivelestat in two large RCTs, including a phase III study by Tamakuma et al. with improved respiratory outcomes and shortened ICU stay 26 versus a phase II study (the STRIVE study) with a negative trend in 180-day mortality without an improvement of 28-day mortality and VFDs, 15 may be explained by the different patient’s characteristics (race, age, disease severity, dose of treatment, or sepsis status). While the phase III study from Japan enrolled patients with lung injury scores lower than 2.5, 26 the STRIVE study recruited patients with lung injury scores >2.5. 15 Indeed, the post-hoc analysis of the STRIVE study demonstrates the favorable trends in mortality and VFDs after receiving sivelestat in patients with injury scores equal to or lower than 2.5. 15 Moreover, the different proportions of sepsis-related ALI/ARDS may have contributed to these discordant results. There are 59% and 69% in the STRIVE study and Tamakuma et al.’s study, respectively, with a statistical difference (p < 0.01). 31 Here, this current study demonstrated a favorable treatment effect of sivelestat in patients with ALI/ARDS from sepsis, especially severe ALI/ARDS, including PaO2/FiO2 ratio <150 and APACHE-II Score >20, with the mean age of >60. Thus, the large RCTs focusing on the comparative efficacy of sivelestat on ALI/ARDS with and without septic etiology will be needed to reach a solid clinical conclusion for the use of sivelestat in real clinical practice. Furthermore, the possibility of using sivelestat to prevent ARDS development in patients with sepsis 32 or to conduct it in children with ALI/ARDS 33 is warranted. This information may help to minimise the medical costs of these patients.34,35 Of note, a retrospective study conducted in China demonstrated that sivelestat therapy significantly reduces medical costs for patients with sepsis-ARDS. 36
Although several complications can cause ALI/ARDS, the most common cause is sepsis. The excessive neutrophil activation in sepsis facilitates neutrophil migration into the alveoli to release several substances, including proteases, cytokines, and reactive oxygen species (ROS), to destroy the endothelial and epithelial cells, causing lung injury.4,7,37–42 Compared to non-sepsis-related ALI/ARDS, neutrophil elastase (NE) is generally significantly more elevated in sepsis-related ALI/ARDS. As such, NE is released into the extracellular milieu through degranulation and the formation of neutrophil extracellular traps (NETs), playing a more prominent role in the inflammatory cascade and lung tissue damage in response to systemic infection.39,43,44 Therefore, targeting NE with selective NE inhibitors, such as sivelestat, may be a promising treatment strategy for sepsis-related ALI/ARDS. While NE directly digests extracellular matrix in the lung, NE stimulates protease-activated receptors and induces proinflammatory cytokines, IL-8, tumor necrosis factor-alpha (TNF-α), and high-mobility group box chromosomal protein 1 (HMGB1), which amplify sequestration and exacerbate further inflammation.45–47 Additionally, NE induces vascular endothelial injury, increases vascular permeability, and damages epithelial cells 48 via several signaling pathways, including cyclooxygenase-2 (COX-2) and mitogen-activating protein kinases (MAPK) pathways.49,50 These pro-inflammatory factors of ARDS in the lungs are attenuated by sivelestat12,46 along with inflammation in other organs (liver, pancreas, and kidney).51,52 Although NE is a significant contributor, other inflammatory mediators and mechanisms are involved in both sepsis- and non-sepsis-related ARDS. 53 Additionally, there are other NE inhibitors in different respiratory disorders in current clinical trials, including alvelestat for COPD, 54 lonodelestat for cystic fibrosis, 55 or BAY 85-8501 for non-cystic fibrosis bronchiectasis, 56 that might also be beneficial in sepsis ALI/ARDS. More studies are interesting.
There are several limitations. First, a limited number of databases were searched, and studies published in languages other than English were excluded. Given that sivelestat has been primarily studied in Japan, relevant non-English studies may have been missed, potentially introducing selection bias. Although these studies may provide important insights, our methodology required dual independent screening and data extraction, which was not feasible without access to validated translations. Future research may benefit from systematic translation and inclusion of Japanese studies to enhance the robustness and global applicability of meta-analytic evidence. Second, the number of included studies was smaller than expected and mostly conducted in Japan and China, which might introduce ethnic and racial biases. Third, this meta-analysis included preprint articles to reduce publication bias. The information about ALI/ARDS classification is available in most of the included studies, but not all studies. Several included studies used the term “acute lung injury” (ALI), which was commonly applied before the 2012 Berlin definition of ARDS. For consistency with the original study terminology, we report outcomes as “ALI/ARDS,” but acknowledge that in current clinical practice, ALI has been replaced by mild, moderate, and severe ARDS under the Berlin definition. Other treatment methods, such as mode of ventilation, use of other medications, or fluid administration, might alter the outcomes. Fourth, due to data inefficiency, the effect of sivelestat on ICU stays with different doses cannot be performed. Finally, the analysis used pooled data (individual data were not available), which prevented a detailed analysis to obtain more comprehensive results.
Conclusion
In summary, this systematic review and meta-analysis suggest that sivelestat use is associated with a reduction in 28–30-day mortality, shorter duration of mechanical ventilation, and improved oxygenation (PaO2/FiO2 ratio) in patients with ALI/ARDS. The beneficial effects appear more pronounced in patients with sepsis-related ALI/ARDS than in those with non-sepsis etiologies. However, given the heterogeneity of available evidence and methodological limitations, further large-scale, high-quality RCTs focusing on well-defined patient subgroups with sepsis and non-sepsis-related ALI/ARDS are warranted before the recommendation to use sivelestat in routine clinical use.
Supplemental Material
sj-docx-3-inc-10.1177_17511437261425058 – Supplemental material for The effectiveness of selective neutrophil elastase inhibitors (sivelestat) in acute lung injury or acute respiratory distress syndrome: A systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-docx-3-inc-10.1177_17511437261425058 for The effectiveness of selective neutrophil elastase inhibitors (sivelestat) in acute lung injury or acute respiratory distress syndrome: A systematic review and meta-analysis of randomized controlled trials by Awirut Charoensappakit, Kritsanawan Sae-khow, Nuntanuj Vutthikraivit, Patinya Maneesow, Kent Doi, Monvasi Pachinburavan and Asada Leelahavanichkul in Journal of the Intensive Care Society
Supplemental Material
sj-jpg-2-inc-10.1177_17511437261425058 – Supplemental material for The effectiveness of selective neutrophil elastase inhibitors (sivelestat) in acute lung injury or acute respiratory distress syndrome: A systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-jpg-2-inc-10.1177_17511437261425058 for The effectiveness of selective neutrophil elastase inhibitors (sivelestat) in acute lung injury or acute respiratory distress syndrome: A systematic review and meta-analysis of randomized controlled trials by Awirut Charoensappakit, Kritsanawan Sae-khow, Nuntanuj Vutthikraivit, Patinya Maneesow, Kent Doi, Monvasi Pachinburavan and Asada Leelahavanichkul in Journal of the Intensive Care Society
Supplemental Material
sj-tiff-1-inc-10.1177_17511437261425058 – Supplemental material for The effectiveness of selective neutrophil elastase inhibitors (sivelestat) in acute lung injury or acute respiratory distress syndrome: A systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-tiff-1-inc-10.1177_17511437261425058 for The effectiveness of selective neutrophil elastase inhibitors (sivelestat) in acute lung injury or acute respiratory distress syndrome: A systematic review and meta-analysis of randomized controlled trials by Awirut Charoensappakit, Kritsanawan Sae-khow, Nuntanuj Vutthikraivit, Patinya Maneesow, Kent Doi, Monvasi Pachinburavan and Asada Leelahavanichkul in Journal of the Intensive Care Society
Footnotes
Acknowledgements
The authors gratefully thank Chulalongkorn University for data accessibility and/or materials made available for the present study.
Author contributions
Awirut Charoensappakit: literature search, data extraction, data validation, risk of bias, analysis, and wrote the manuscript. Kritsanawan Sae-khow: literature search, data extraction, risk of bias, analysis, and wrote the manuscript. Nuntanuj Vutthikraivit: data extraction, risk of bias, and assessment of discussion. Patinya Maneesow: data extraction, risk of bias, and assessment of discussion. Kent Doi: data validation and assessment of discussion. Monvasi Pachinburavan: data validation and assessment of discussion. Asada Leelahavanichkul: data validation, risk of bias, analysis, assessment of discussion, and wrote the manuscript. All authors conceived the study, corrected the manuscript, and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Research Council of Thailand (NRCT; N84A680764, N32A680647, and N42A680063), Ratchadapiseksompotch Fund, Faculty of Medicine, Chulalongkorn University (RA-MF-08/69, RA-MF-36/68). A.C. was funded by Grants for Development of New Faculty Staff, Ratchadaphiseksomphot Fund, Chulalongkorn University. The funder of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
