Abstract
We present a case of a class 3 drowning event with severe sand aspiration, successfully managed with mechanical ventilation, bronchoalveolar lavage, ECMO, and the use of a mechanical insufflation-exsufflation device (MI-E). Sand aspiration is a potentially lethal consequence of near-drowning events. It may cause respiratory failure by several mechanisms, including laryngospasm, upper or lower airway obstruction and an intrinsic acute respiratory distress syndrome (ARDS) related to the sand itself. Its standard management involves lung-protective mechanical ventilation strategies, bronchoalveolar lavage, and, more recently, extracorporeal membrane oxygenation (ECMO) in adult and paediatric cases. MI-E devices are commonly used by physiotherapists in the intensive care setting to facilitate secretion clearance in extubated patients, reducing the occurrence of ventilator-associated pneumonia. However, their use in intubated patients in the UK is not as common due to a lack of evidence and experience. The evidence supporting their use in intubated patients is of low quality, and we have not found any examples of their use for clearing aspirated particulate matter or in conjunction with ECMO in the literature. We believe this is the first time MI-E has been used this way. We argue that it could be a valuable addition to the management of sand and other particulate aspirations, even in severe cases requiring ECMO, and it warrants further exploration. Using a MI-E device may significantly decrease the duration of ECMO treatment, reducing its associated complications and expenses.
Keywords
Introduction
Sand aspiration is a significant complication of both drowning events and accidental burial. In severe cases, it may result in airway obstruction and respiratory failure, compounding the initial insult of saltwater aspiration and consequent acute respiratory distress syndrome (ARDS). It has conventionally been managed with lung-protective ventilation strategies, prone positioning, and regular bronchoalveolar lavage (BAL). 1 Following the uptake of extracorporeal membrane oxygenation (ECMO) in the management of refractory ARDS, it has also been used anecdotally in cases of respiratory failure secondary to severe sand aspiration. 2
Mechanical Insufflation-Exsufflation (MI-E) devices, also known as cough assist (CA), are commonly used by physiotherapists to facilitate secretion clearance and prevent complications in patients who are unable to produce an effective cough effort. They are used primarily as non-invasive devices that rapidly cycle positive and negative pressures, simulating the natural airflow changes during coughing, thereby increasing inspiratory and expiratory volumes and moving secretions upwards towards the upper airway. They have also been used in critical care for patients with high secretion loads to facilitate extubation and prevent re-intubation.3,4 A recent survey demonstrates clinicians are much more confident in their use among extubated patients. Barriers to their use with intubated patients include lack of experience, concerns about causing harm and a low-quality evidence base. 5 However, despite a lack of evidence and some adverse events, there is strong and growing interest in using MI-E in critical care. 6
We report a case of a class 3 drowning with sand aspiration that was treated with mechanical lung-protective ventilation, bronchoalveolar lavage, and ECMO. The removal of sand from the tracheobronchial tree was facilitated using an MI-E device, which appeared to accelerate the patient’s recovery. This case may be the first instance of an MI-E device used in an intubated patient supported by ECMO. Our manuscript was drafted in accordance with the CARE (CAse REport) guidelines. 7
Narrative
A 33-year-old male was admitted to intensive care after falling 30 feet into shallow water and suffering a class 3 drowning event. He was intubated on arrival at the hospital due to hypoxia and required a cycle of cardiopulmonary resuscitation for hypoxic cardiac arrest. Chest X-ray (Figure 1) and full body CT (Figure 2) showed extensive bilateral lung consolidation, a sacrococcygeal fracture and a degloving injury to the left thigh and buttock. His medical history included smoking, recreational drug use and a penicillin allergy. The initial treatment included tranexamic acid, tetanus immunoglobulin and antibiotic cover.
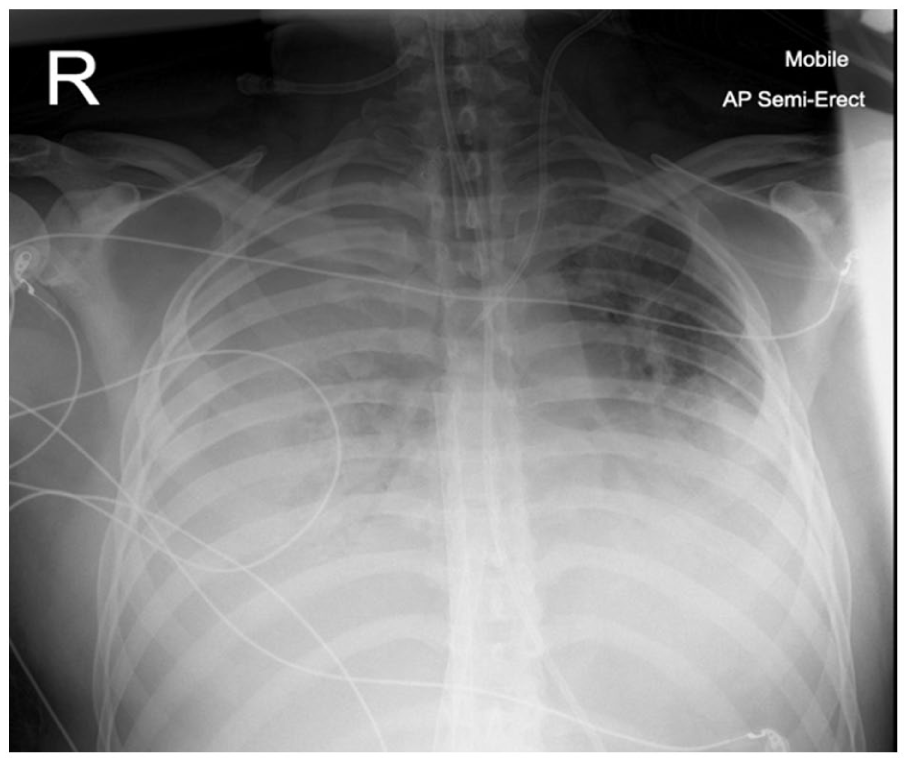
Initial chest X-ray – shows four quadrant infiltration of the lung fields.
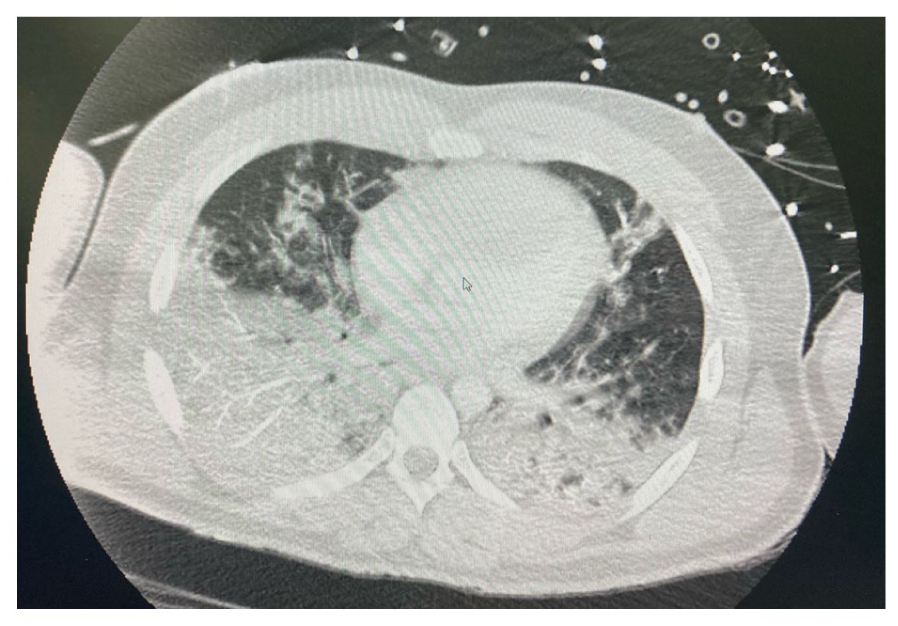
Initial CT chest – shows extensive lung infiltration.
Bronchoscopy (Figure 3) demonstrated visible watery secretions and sand particles in the trachea, and bronchoalveolar lavage (BAL) revealed a high level of mixed watery secretions and sand contamination across all lung domains. He developed a profound type 2 respiratory failure and acidosis with persistently high airway pressures despite proning.
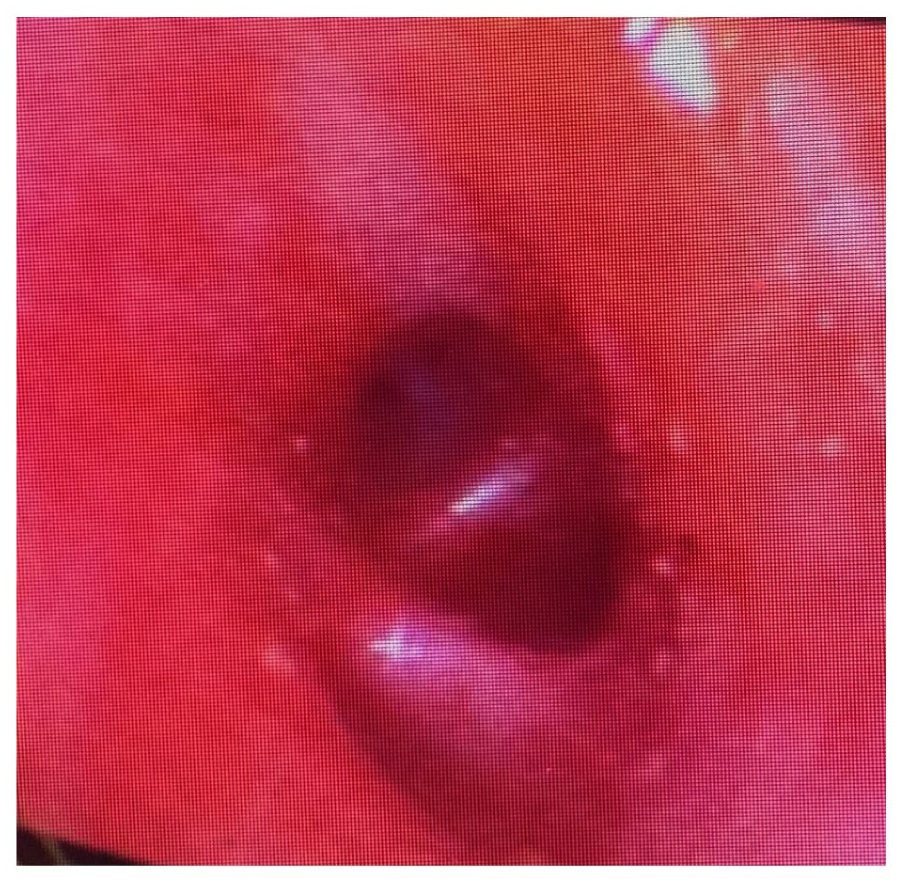
Initial bronchoscopy. Demonstrates trachea and carina with visible watery secretions and sand particles.
Due to his developing severe ARDS (see Figures 1 and 2 and Table 1) with potential reversibility, he was accepted for veno-venous extracorporeal membrane oxygenation (VV-ECMO). See Figure 4 which demonstrates ECMO cannulae in the inferior vena cava.
Pre-ECMO blood results, respiratory and vasoactive support.

Hb: haemoglobin; Plt: platelets; WCC: white cells; INR: international normalised ratio; ABG: arterial blood gas; CVS: cardiovascular; VC SIMV: volume control with synchronous intermittent mandatory ventilation; PEEP: positive end expiratory pressure; PIP: positive expiratory pressure; Pplat: plateau pressure; TV: tidal volume; MV: minute volume.
ECMO procedure and initial characteristics.
Chest X-ray post ECMO cannulation. Demonstrates ECMO cannulae in the inferior vena cava.
The patient’s vasopressor requirement improved over the following 9 days; however, he remained ECMO-dependent, with an FiO2 of 100% (ECMO) and a sweep gas of 6–8 L/min. A further three episodes of bronchoscopy and BAL removed large amounts of sand contamination with thick secretions; however, tidal volumes remained static at 100–200 mL on pressure control bilevel positive airway pressure ventilation mode (BiPAP delivered via Dräger Evita ventilator (Drägerwerk AG & Co. KGaA, Lübeck, Germany)) representative pressures on EPAP (expiratory positive airway pressure) 10 cmH2O and IPAP (inspiratory positive airway pressure) 20 cmH2O (compliance 10 mL/cmH2O). The patient was also put in the prone position on VV-ECMO.
Anticoagulation was omitted due to the high risk of bleeding from the substantial buttock wound and the fact that the ECMO circuit tubing was heparin-coated. Repeat chest X-ray demonstrated bi-basal collapse with moderate pleural effusions; however, these were felt not significant enough to drain. Microbial samples were found to have mixed growth; therefore, broad-spectrum antibiotic cover was continued.
On day 9 of admission, due to the lack of progress and the potentially harmful effects of prolonged sedation and ECMO-related side effects, a multidisciplinary team (MDT) meeting was held. This team included cardiothoracic intensivists, a respiratory physician, and the physiotherapy team. They decided to trial a mechanical insufflation-exsufflation device to help with secretion and particulate clearance, increase tidal volume, and reduce dependence on ECMO.
Four sessions of MI-E were given using a Philips E70 Cough Assist (Philips Respironics, Murrysville, PA, USA) device: two on day 10 of the admission, followed by single sessions on days 11 and 12. The patient remained profoundly sedated throughout this period.
Table 2 presents a detailed summary of the four treatment sessions involving mechanical insufflation-exsufflation (MI-E). Sessions 1, 3, and 4 were performed in manual mode, while session 2 utilised the automated mode. Across all sessions, treatment began with two initial insufflation–exsufflation cycles at +15/−20 cmH₂O, followed by three cycles of four breaths each at a 1:1 inspiratory-to-expiratory ratio, and concluded with a final insufflation. The applied pressures gradually increased across sessions, with session 1 reaching up to +23/−28 cmH₂O and session 4 maintaining similar peak pressures. Pre-treatment tidal volumes and lung compliance improved progressively from session 1 (203 mL; 10 mL/cmH₂O) to session 4 (556 mL; 27.8 mL/cmH₂O). Post-treatment measurements also demonstrated marked improvements, with tidal volumes increasing to over 500 mL and compliance exceeding 25 mL/cmH₂O by the final session. Secretions decreased substantially across treatments, from large amounts of bloodstained sandy secretions in session 1 to only small amounts in sessions 3 and 4. Concurrent ECMO parameters demonstrated a gradual reduction in RPM, flow, and sweep gas requirements over time, indicating improved pulmonary function and decreased dependence on extracorporeal support. Additional clinical interventions were noted, including temporary use of airway pressure release ventilation (APRV) after session 1, bronchoalveolar lavage (BAL) following session 2, endotracheal tube replacement in session 3, and heparinisation during session 4 to facilitate a trial reduction of ECMO support. Overall, the table highlights a stepwise improvement in lung mechanics, secretion clearance, and ventilatory efficiency throughout the course of MI-E therapy.
Procedures and results of mechanical insufflation-exsufflation interventions.
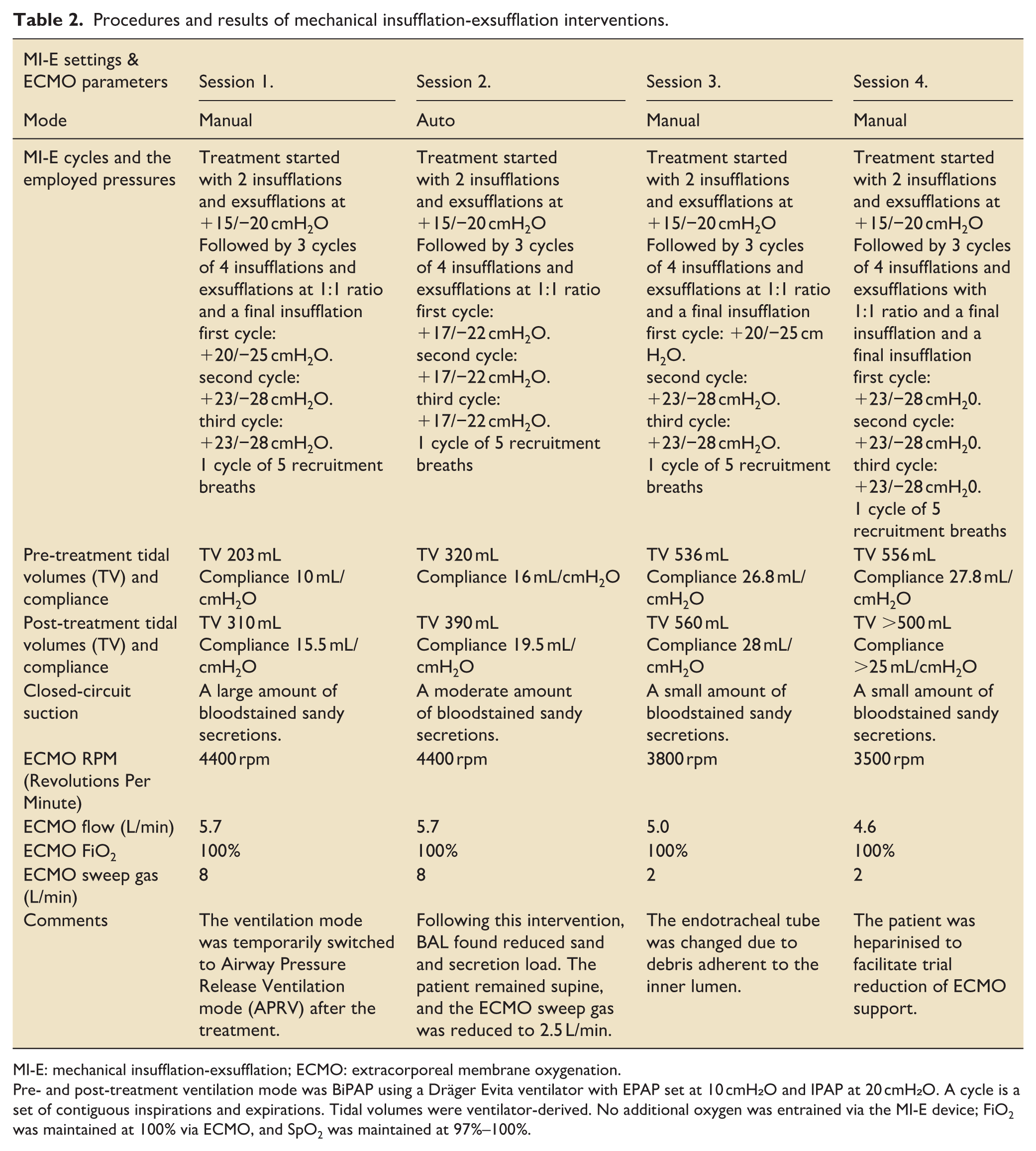
MI-E: mechanical insufflation-exsufflation; ECMO: extracorporeal membrane oxygenation.
Pre- and post-treatment ventilation mode was BiPAP using a Dräger Evita ventilator with EPAP set at 10 cmH₂O and IPAP at 20 cmH₂O. A cycle is a set of contiguous inspirations and expirations. Tidal volumes were ventilator-derived. No additional oxygen was entrained via the MI-E device; FiO2 was maintained at 100% via ECMO, and SpO2 was maintained at 97%–100%.
Figure 5 presents the tidal volumes prior to and following the MI-E interventions. No adverse events occurred during treatment; however, the patient did de-recruit several times during the first two sessions, so was given time to re-recruit between cycles.
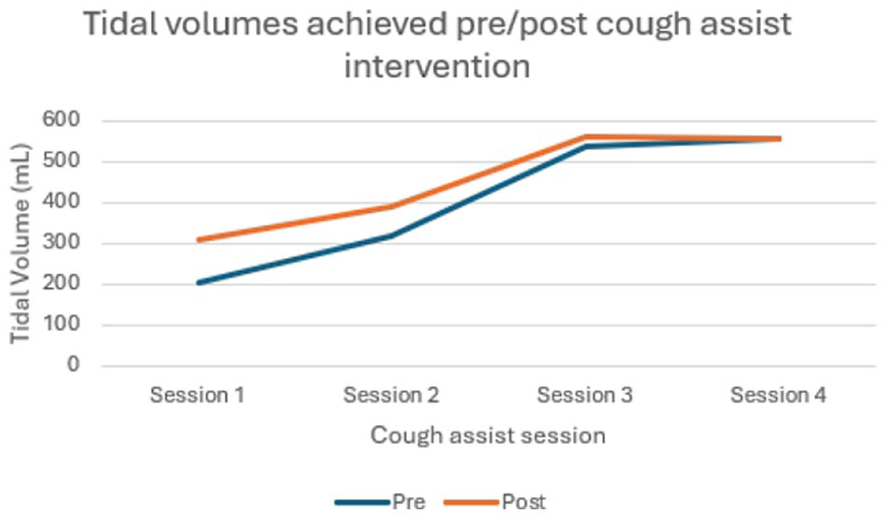
Tidal volumes before and after cough assist intervention.
On day 13 of the ICU admission, the patient was ventilated with the following parameters: BiPAP FiO2 40%, PEEP 10 cmH2O, and IPAP 18 cmH2O. Vt >400 mL. Air entry was auscultated throughout the lung fields, and the chest x-ray showed significant improvement. ECMO support was reduced to FiO2 0.7, flow 3.1 L/min, RPM 2600, and sweep 2 L/min. CVVH was commenced for fluid removal as the furosemide infusion was insufficient to reduce the strongly positive fluid balance (+13 L). Vasopressor support was weaned off, and he remained tolerant of fluid removal.
Following this success, according to standard departmental protocol, oxygen delivery to the oxygenator was gradually discontinued under close observation. Then, following several hours of monitoring, the ECMO cannulae were removed on day 16. He remained sedated and ventilated on BiPAP with an FiO2 of 0.35.
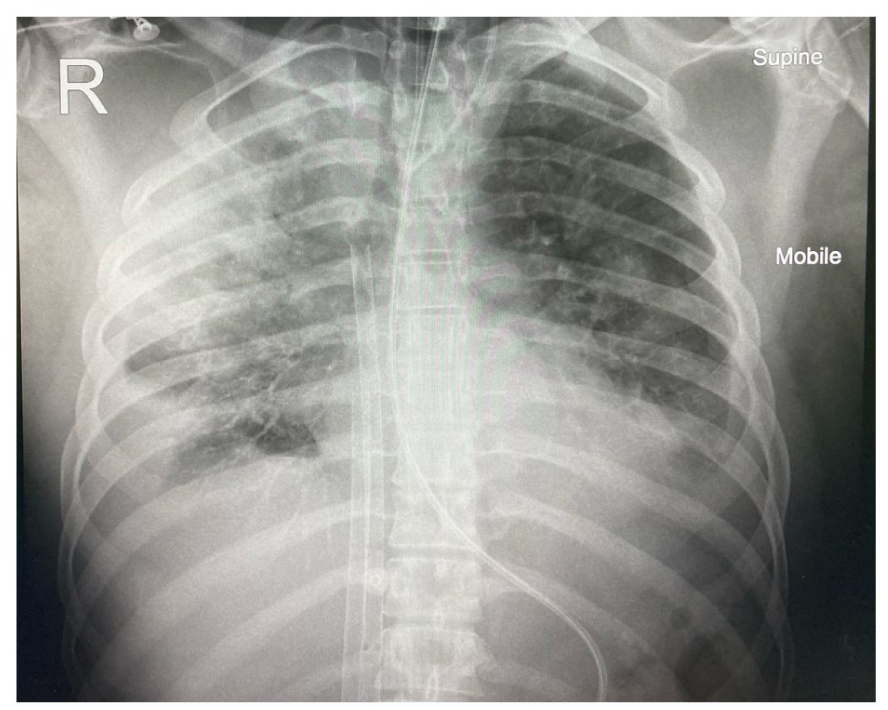
On day 21 of admission, he was transferred from the specialist cardiothoracic and ECMO unit to the general ICU, where a tracheostomy was placed and a respiratory wean commenced on day 26. Figure 6 shows the improved chest X-ray on day 22. Multiple infections complicated the recovery process; however, after 56 days of admission, he was discharged home to the care of his GP. Figure 7 presents the timeline of events.
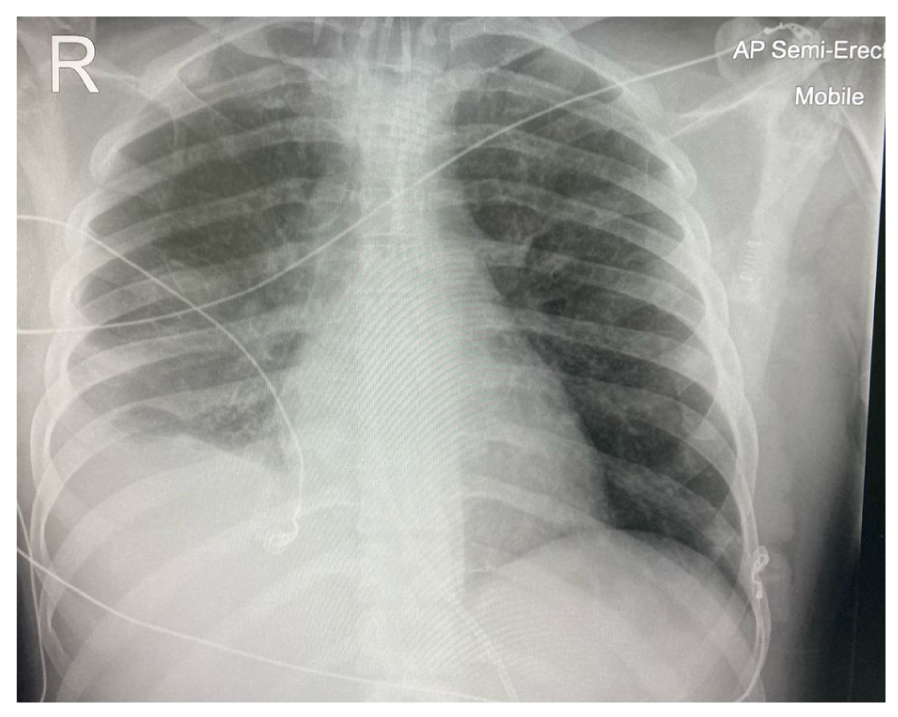
CXR taken on day 22 of admission. Demonstrates significant improvement in the previous four quadrant infiltration.
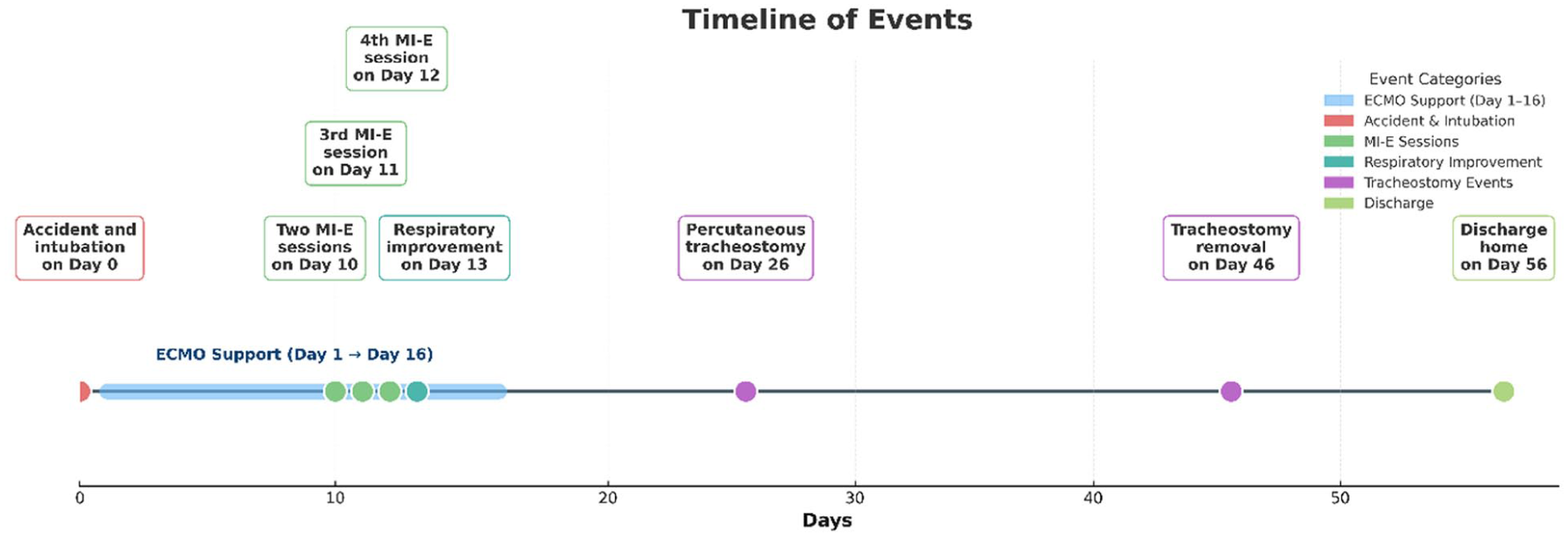
Timeline of events.
Patient experience
The patient was contacted, and verbal and written consent were gained for inclusion in this case report. He reported suffering from pain and mental health issues for 6 months after his discharge, with a significant loss of functional capacity. He returned to work as an electrician 5 months after discharge. Prior to the accident, he had been a high-level sportsman and, since discharge, was troubled by shortness of breath when trying to exercise. However, he expressed gratitude for the care he received and was optimistic about his future and prospects for physical rehabilitation.
Discussion
Sand aspiration may cause death by respiratory failure, either due to immediate physical airway obstruction or to a subsequent intrinsic ARDS response. In cases of severe ARDS, the UK Faculty of Intensive Care Medicine and the European Society of Intensive Care Medicine recommend the use of VV ECMO as an adjunct to traditional management strategies, including lung-protective ventilation, physiotherapy interventions, bronchoalveolar lavage, and medical management.8,9 The number of reported sand aspiration ARDS cases treated with ECMO seems to be very scarce. In fact, we only found one paediatric report from 2013 by Isaac et al., which describes the first successful management of sand aspiration ARDS with ECMO in a paediatric case. 10
Recently, MI-E devices have been increasingly used with intubated patients to assist with extubation and facilitate weaning from mechanical ventilation. Consensus guidelines for physiotherapy for adults on ECMO include the use of MI-E devices. They emphasise the importance of assessing overall lung compliance before use and highlight the need to prevent inadvertent harm caused by over-distension and de-recruitment. 11 In our case, profound sedation and ECMO support minimised these risks by allowing intervention times to be kept short under full airway and ventilation control.
However, the uptake of MI-E has been limited by several barriers, as highlighted in a UK-wide survey conducted by Swingwood et al. These barriers include a lack of high-quality evidence, limited experience among both physiotherapists and ICU physicians, and concerns over potential harm. 5 In a 2017 Cochrane review, Rose et al. found very low-quality evidence to support the use of MI-E technology; however, some findings suggest potential benefits without determining potential harm. 4 In a recent scoping review, Swingwood et al. concluded that there is little consistency in how MI-E is used and reported. 12
The case presented is of a patient who had suffered severe ARDS secondary to aspiration of sand and seawater. Despite repeated bronchoalveolar lavage, prone positioning and the life-sustaining safety net of VV-ECMO, his respiratory function remained static, with a persistently high sand load that limited clinical progress. Adding MI-E techniques to his management facilitated the removal of large amounts of sand, significantly improving his tidal volumes and respiratory compliance. Following a multidisciplinary approach, he was able to wean from ECMO support and ultimately progressed to a successful discharge.
The limitations of this case report include the rarity and low incidence of the reported condition, as well as the fact that very few individuals with this condition reach the hospital and survive long enough to receive ECMO care. It’s important to note that the MI-E device was used off-label in a patient who was unresponsive to standard treatment. Moreover, a recent study by Shah et al. found that MI-E-derived cough peak flow values may systematically overestimate true measurements when compared with calibrated pneumotachography, suggesting that these values should be interpreted with caution. 13 Nonetheless, ECMO served as a safety net to prevent potential complications associated with the device. This patient also had other interventions, such as bronchoscopic clearances, BALs and various ventilation modes, which all contributed to his recovery, not only the use of the MI-E device. Nevertheless, we believe that MI-E had a significantly beneficial impact on volumes and sputum clearance in this case. Until the device was used, no significant clinical improvement had been observed, which prompted the use of this technology.
Shah et al. recently reported on 359 adults with neuromuscular diseases who were established on domiciliary MI-E. 14 Despite having the largest reported cohort, they concluded that the range of prescribed settings was narrow, reflecting the limited evidence base and highlighting the need to better understand the optimal targets for titration and the impact of different MI-E settings on clinical outcomes. 14 The strength of our paper lies in its detailed description of an MI-E treatment prescription.
Given the unusual combination of the described pathology and the complex treatment modalities (ECMO and MI-E), conducting randomised controlled trials (RCTs) would likely be impossible in our presented setting. Due to these challenges, research in this area is primarily limited to case reports or case series, which can still provide valuable insights for managing similar critical situations.
Conclusion
To our knowledge, this is the first reported case demonstrating the safe and effective use of MI-E devices in managing severe ARDS due to sand aspiration, which required VV-ECMO. Our innovative, multidisciplinary approach yielded excellent outcomes in a condition with a potentially high mortality rate. The combination of ECMO and MI-E technology provided a synergistic advantage in this complex situation. We recommend that these cases be managed in specialised respiratory centres with sufficient ECMO experience and specialised ECMO and respiratory physiotherapy services.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
