Abstract
Background:
Airway clearance is a vital aspect of respiratory care in patients with severe acute respiratory failure receiving veno-venous extracorporeal membrane oxygenation (VV-ECMO). These patients often receive high levels of sedation alongside ultra-lung protective invasive mechanical ventilation, which, while reducing ventilator-induced lung injury, significantly impairs physiological mechanisms essential for secretion clearance, such as expiratory flow bias, mucociliary transport, and effective cough.
Objectives:
This commentary explores the multifaceted challenges to airway clearance in the VV-ECMO population, evaluating current physiotherapeutic interventions, and identifying critical areas for future research.
Discussion:
Ultra-lung protective ventilation settings reduce airflow and expiratory shear forces necessary for secretion mobilisation. Concurrent sedation and neuromuscular blockade suppress cough reflexes and promote secretion stasis, while reduced airflow impairs the two-phase gas-liquid flow critical for airway hygiene. Additionally, increased secretion viscosity and altered lung mechanics further limit clearance. Current physiotherapy strategies such as suctioning, hyperinflation, cough augmentation, and manual techniques are often adapted from non-ECMO settings and lack validation in this population. Diagnostic challenges, such as reduced auscultation efficacy, highlight the growing importance of tools like lung ultrasound. Pharmacological adjuncts, including mucolytics and saline lavage, are used despite limited supporting evidence.
Conclusion:
Airway clearance in patients receiving VV-ECMO is hampered by a convergence of physiological, mechanical, and clinical factors. Existing literature remains scarce and largely extrapolated from conventional invasive mechanical ventilation populations. There is an urgent need for targeted research to define evidence-based interventions tailored to the unique pathophysiology of this complex group.
Keywords
Introduction
For patients receiving Veno-Venous Extracorporeal Membrane Oxygenation (VV-ECMO) reduced driving pressures 1 are often recommended as a means to limit baro-trauma and are associated with improved survival, however this may impact negatively on secretion clearance. The need for invasive mechanical ventilation (IMV), often combined with sedation and paralysis, also exacerbates secretion retention. These factors, along with the multitude of underlying causes of severe acute respiratory failure (SARF), contribute to compromised airway clearance mechanisms, but have been poorly investigated to date especially in the ECMO cohort of patients. 2
In critically ill patients, mucociliary transport and cough reflexes are key processes for clearing airway secretions. However, these mechanisms are impaired in the context of IMV and airway secretion retention is an underappreciated problem, especially in the sicker cohort with acute respiratory distress syndrome. 3 Secretion retention can result in lung volume loss, leading to ventilation-perfusion (V/Q) mismatch, reduced oxygenation, ventilator dependence and increased risk of bacterial colonization.3,4 This, in turn, heightens the risk of ventilator-associated pneumonia, slower recovery 5,6 and further pulmonary complications such as acute respiratory distress syndrome (ARDS). 7
To date, literature exploring airway clearance interventions has focussed on invasively and mechanically ventilated patients with normal tidal volumes (typically 6–10 ml/kg of ideal body weight). Respiratory physiotherapy often focuses on four key goals: clearing secretions, reversing atelectasis, optimising gas exchange, and assisting weaning. 8 Standard care consists of humidification, suctioning, mobilisation, patient re-positioning; with the addition of specific airway clearance techniques such as hyperinflation, manual chest wall techniques, cough augmentation and oscillatory devices. However, the evidence for these techniques is based on patients with normal tidal volumes and does not account for the profound physiological changes and altered IMV strategies seen in those requiring VV-ECMO for SARF/ARDS.
IMV strategies in this patient group aim to minimise lung injury by reducing mechanical power. 1 While international practice typically uses tidal volumes ⩽6 ml/kg, 9 current evidence supports an “ultra-lung protective” approach: tidal volumes ⩽4 ml/kg, respiratory rate < 20/min, plateau pressure <25 cmH2O, and driving pressure ⩽15 cmH2O. 10
This commentary aims to explore the physiological processes influencing secretion clearance in patients on VV-ECMO, especially where IMV settings favour an “ultra-lung protective” approach. By understanding these mechanisms, clinicians may develop more effective strategies to optimise secretion clearance and improve outcomes in this specific patient group.
Physiology of secretion clearance
Invasive mechanical ventilation
In healthy individuals, normal airway clearance relies on three key mechanisms: two-phase gas-liquid flow, a functional mucociliary escalator, and an effective cough reflex. To ensure secretion movement towards the glottis/mouth, an expiratory flow bias is required, where expiratory flow exceeds inspiratory flow.
Two bench studies have demonstrated that effective secretion movement depends primarily on the relationship between inspiratory and expiratory flow, rather than factors like tidal volume (TV), mean inspiratory flow, or inspiratory time.11,12 These studies confirmed that secretions only move towards the glottis when expiratory flow exceeds inspiratory flow. Kim et al. 12 found that inspiratory flow must be less than 90% of expiratory flow to achieve this effect. Later, Volpe et al. 11 in a bench model suggested that a difference of more than 17 L/min between expiratory and inspiratory flow is required for meaningful secretion clearance. Similarly, an animal study by Li Bassi et al. 13 demonstrated that an even higher flow difference, greater than 33 L/min (head of bed elevated), was needed to ensure secretion movement towards the glottis.
Ntoumenopoulos et al. 14 analysed inspiratory and expiratory flow patterns in mechanically ventilated patients, predominantly in spontaneous modes of ventilation, and found a consistent inspiratory flow bias across the cohort. All patients exhibited an absolute peak inspiratory to peak expiratory flow (PIF: PEF) ratio of >90%, with all but one exceeding >110%. Notably, in 40% of cases, inspiratory flow exceeded expiratory flow by more than 17 L/min—conditions known to promote caudad (toward the lungs) secretion movement and increase the risk of secretion impaction.
In patients receiving IMV, the normal physiological mechanisms of secretion clearance are significantly disrupted and are associated with significant pulmonary complications such as lobar collapse and pneumonia. 15 The mucociliary escalator, a primary mechanism of airway hygiene, is frequently impaired due to epithelial injury, cilial dysfunction, and changes in sputum properties. Mechanical ventilation itself can exacerbate epithelial damage through high airway pressures, shear stress, and inadequate humidification, all of which further compromise mucociliary transport. 16 The delivery of inadequately humidified gas contributes to airway desiccation, increasing secretion viscosity, impairing ciliary beat frequency, and predisposing to mucosal injury, bronchospasm, atelectasis, and secretion retention. 17
Additionally, IMV promotes regional heterogeneity in time constants and compliance, resulting in preferential ventilation of non-obstructed lung units and insufficient distal airflow beyond mucus obstructions, thus reducing airflow in regions with mucus plugging.
In this setting, when mucociliary clearance is ineffective, the cough reflex becomes the principal remaining defence. 18 However, this is often suppressed by sedation and neuromuscular blockade, commonly employed to facilitate ventilation, rendering patients unable to generate effective expiratory flows necessary for secretion expulsion.
Even when some degree of cough is preserved, the presence of an artificial airway, such as an endotracheal tube or tracheostomy, impairs or precludes glottic closure, which is critical for building the high intrathoracic pressures required during the compressive phase of cough. Consequently, cough efficiency is significantly reduced. Effective coughing relies on several interrelated physiological factors: the initial lung volume available, the development of intrathoracic (tracheal) pressure, the rate of rise in airflow (peak cough flow), and the ability to sustain airflow after the peak. 19
An additional key principle in secretion clearance is the necessity for airflow to reach distal to the retained secretions, either through the main airways or via collateral ventilation pathways, to facilitate mobilisation. 20 In the absence of such airflow, secretions are likely to remain static or become further impacted.
Effective secretion clearance in this population is critical to maintaining airway patency and preventing complications such as atelectasis and infection. Interventions should prioritise optimisation of lung volumes, restoration of expiratory flow bias, and augmentation of cough to support airway clearance in the setting of impaired native mechanisms.
ECMO for severe respiratory failure
In patients supported with VV-ECMO, secretion clearance is further compromised by the application of ultra-lung protective mechanical ventilation, a strategy employed to mitigate ventilator-induced lung injury in the setting of SARF. In the early phases of SARF, this typically involves the use of very low tidal volumes, reduced respiratory rates, and minimal airway pressures. While protective, these settings substantially reduce airflow, limiting expiratory flow bias, shear forces, and the frequency of breath cycles required to mobilise secretions. Additionally, the marked reduction in end-expiratory lung volume may result in functional residual capacity falling below closing volume, particularly in dependent or affected lung zones, promoting premature small airway closure during tidal breathing. This leads to gas trapping, impaired alveolar recruitment, and regional hypoventilation, further impeding secretion mobilisation from distal airways. These effects are compounded by poor compliance, elevated airway resistance, heterogeneous time constants, and impaired collateral ventilation. As lung mechanics improve during the recovery phase, and as the reliance on ultra-protective settings lessens, tidal volumes and airway pressures may be gradually increased. This can help restore more effective airflow, improve distal ventilation, and partially re-establish the physiological mechanisms necessary for airway clearance.
In the experience of the authors, patients recently commenced on VV-ECMO often have tidal volumes of less than 100 mL (dead space volumes), with inspiratory and expiratory flow rates typically ranging between 5 and 15 L/min. A bench study by Volpe et al. 11 with an airway in the horizontal position demonstrated that a minimum unidirectional flow of 10–20 L/min is required to facilitate secretion movement. Consequently, in the absence of adequate bidirectional flow, the generation of two-phase gas-liquid flow may be impaired in this patient group, potentially leading to static secretions and reduced airway clearance. To date, bench studies have not investigated secretion movement in scenarios where tidal volumes fall below 250–375 mL,11,12 which fails to reflect the physiology of early VV-ECMO support.
In addition to these mechanical challenges, secretions may also migrate from areas of lower compliance to regions of higher compliance when inspiratory flow exceeds expiratory flow. 11 This is particularly relevant in patients receiving VV-ECMO, where the “baby lung” concept applies: only a small portion of the lung remains aerated while the rest is consolidated. 21 In this setting, flow imbalances arising from heterogeneous lung mechanics can drive secretions from compromised towards healthier lung regions. As a result, this redistribution may increase the risk of infection spread, pneumonia progression, or even major airway obstruction.
Sputum viscosity further influences secretion movement. Acute lung pathologies can adversely affect airway secretions by increasing mucin production, introducing inflammatory cells and debris, and accumulating extracellular DNA and fibrin. 22 Dehydration of secretions further increases viscosity, necessitating higher flow rates to achieve movement. With greater viscosity, the PEF:PIF ratio threshold required for effective secretion clearance rises beyond the previously mentioned value. 11
When airflow and tidal volumes are insufficient for secretion mobilisation, the lungs rely more heavily on mucociliary clearance. However, acute lung pathologies can severely impair ciliary function. Inflammatory responses, as seen in ARDS, pneumonia, and viral infections can release cytokines, reactive oxygen species, and proteases, which damage the ciliated epithelium and reduce ciliary beat frequency. Direct epithelial injury from infection, aspiration, or trauma can lead to cilia loss and basal membrane exposure, further hindering secretion clearance. Goblet cell hyperplasia and excess mucus production also increase secretion viscosity, physically obstructing ciliary movement. Hypoxia and ischemia, common in severe lung injury, further disrupt energy-dependent ciliary function. 16
In addition to the impact of invasive mechanical ventilation (IMV) on cough effectiveness, patients supported with VV-ECMO face further physiological and clinical barriers to effective airway clearance. Ultra-lung protective ventilation strategies, characterised by markedly reduced tidal volumes, along with the frequent use of deep sedation and neuromuscular blockade to minimise metabolic demand, often diminish the capacity to generate a functional cough. The resulting reduction in lung volume limits the initial inspiratory capacity, which in turn impairs intrathoracic pressure generation and peak expiratory flow—both critical components of an effective cough. At higher lung volumes, expiratory muscles operate closer to their optimal length–tension relationship, and elastic recoil forces of the respiratory system are enhanced, 18 both of which contribute to greater expiratory pressures during cough. In the context of ECMO, these mechanisms are compromised, further reducing cough efficacy and the ability to clear secretions. Furthermore, the prolonged duration of ECMO support with an average duration of 16 days, 23 contributes to respiratory muscle deconditioning and global weakness, further impairing the ability to generate sufficient expiratory flow. These factors collectively reduce both the mechanical and neurological components of cough, increasing the risk of secretion retention and associated complications.
Under standard conditions, a 30-degree head-up position is recommended to prevent ventilator acquired pneumonia (VAP) and optimise lung volumes. 24 Head-down positioning has been associated with significant reductions in VAP, 25 however over prolonged periods may be associated with adverse events. 26 In ECMO patients, achieving or maintaining this positioning is often not feasible due to cardiovascular instability or the need to optimise ECMO blood flow. Therefore, the associated benefits of this position, namely the maintenance of end-expiratory lung volume and preservation of functional residual capacity, may not be realised in ECMO.
Airway clearance approaches in ECMO
Airway clearance interventions in patients supported by ECMO for SARF necessitate meticulous assessment and continuous monitoring due to the critical instability inherent in this population. Eden et al. 27 emphasise the importance of a thorough initial assessment and ongoing surveillance throughout the intervention period to ensure patient stability. They further recommend that decisions regarding modification of airway clearance strategies should be made collaboratively within a multidisciplinary team (MDT) - ensuring input from an ECMO consultant to guide clinical judgement.
Auscultation poses a significant challenge in this cohort due to markedly reduced airflow, complicating traditional respiratory assessment.27,28 Lung ultrasound has emerged as a valuable adjunct in this context, allowing for non-radiative imaging and application of validated scoring systems to monitor lung pathology, compliance and therapeutic response.29,30
The existing literature is predominantly comprised of small case series and consensus statements, with the UK ECMO Physiotherapy Network consensus agreement of 2017 27 serving as a foundational reference, albeit with some recommendations likely outdated. Current airway clearance practices frequently involve a combination of suctioning, postural drainage, and targeted techniques such as hyperinflation, manual chest physiotherapy, cough augmentation, and oscillatory devices. However, the optimal timing, dosing, and physiological justification for these interventions remain under-investigated.
Standard airway toileting commonly includes endotracheal suctioning. Saline lavage, however, remains a debated adjunct within the ICU population with caution given for its use due to possible adverse side effects (e.g. desaturation, excessive coughing, bronchospasm, tachycardia, dyspnoea). It should therefore not be used routinely and when done so, utilised sparingly. 31 At present there is no specific literature into the use of saline with patients receiving VV-ECMO. Mucolytic and mucoactive agents are frequently utilised, yet robust evidence supporting their efficacy in this context is scarce. A recent review by Roe et al. 22 highlights the limited, low-quality data underpinning mucolytic use in the ICU, underscoring the need for further investigation in both the ICU (and ECMO) population.
A critical determinant of effective secretion clearance is the facilitation of an effective cough reflex. Wu et al. 32 identify sedation reduction as a pivotal factor in enhancing cough strength within the standard ICU population. Despite inherent challenges in sedative weaning within the ECMO population, studies demonstrate that judicious sedation reduction is both feasible and safe. 33
Positive pressure techniques, including mechanical insufflation-exsufflation and ventilator hyperinflation, require careful evaluation of lung compliance prior to application. 27 Safety parameters concerning pressures and volumes should be closely monitored. 28 Notably, Cork et al. 2 documented a case wherein ventilator hyperinflation was successfully employed in a patient with sub-dead space tidal volumes (<50 mL) with airway plugging (viewed on bronchoscopy), yielding improved tidal volumes without adverse effects. 2 Given the infection control challenges in ECMO patients, particularly when interventions involve breaking the ventilator circuit or aerosol generation, strict adherence to protective measures is essential to mitigate risks to both patients and healthcare personnel. 27
Other less frequently utilised techniques include high frequency percussive ventilation and physiotherapy assisted bronchoscopy. High-frequency percussive ventilation has also been reported to improve lung compliance and oxygenation, as described in a case study by Boscolo et al. 34 , although data remain limited. Physiotherapy-assisted flexible bronchoscopy is practiced in some ECMO centres; however, current evidence supporting its routine use is minimal, primarily cited within consensus guidelines. 27
Flexible bronchoscopy is commonly employed in patients undergoing ECMO for SARF for secretion clearance. In a small study by Sharma et al. 35 they demonstrated that this technique can facilitate secretion clearance and support antibiotic selection through sampling. At present bronchoscopy is not a core skill of the physiotherapist, but in some hospitals it has been used to good effect in the ICU population. 36
In summary, there is a critical need for further research to elucidate the application of airway clearance interventions in the VV-ECMO cohort, with emphasis on their physiological mechanisms, safety and clinical outcomes. Such investigations will inform evidence-based practice and identify future priorities for optimising secretion management in this complex patient group.
Summary
As demonstrated in Figure 1, secretion retention in patients supported with VV-ECMO for SARF is a multifactorial challenge. Severely reduced tidal volumes, absent expiratory flow bias, suppressed cough reflex, limited ability for patient re-positioning contribute to secretion stagnation and distal airway embedding. These issues are compounded by underlying lung pathology, IMV strategies, and increased secretion viscosity, all of which impair airway clearance.
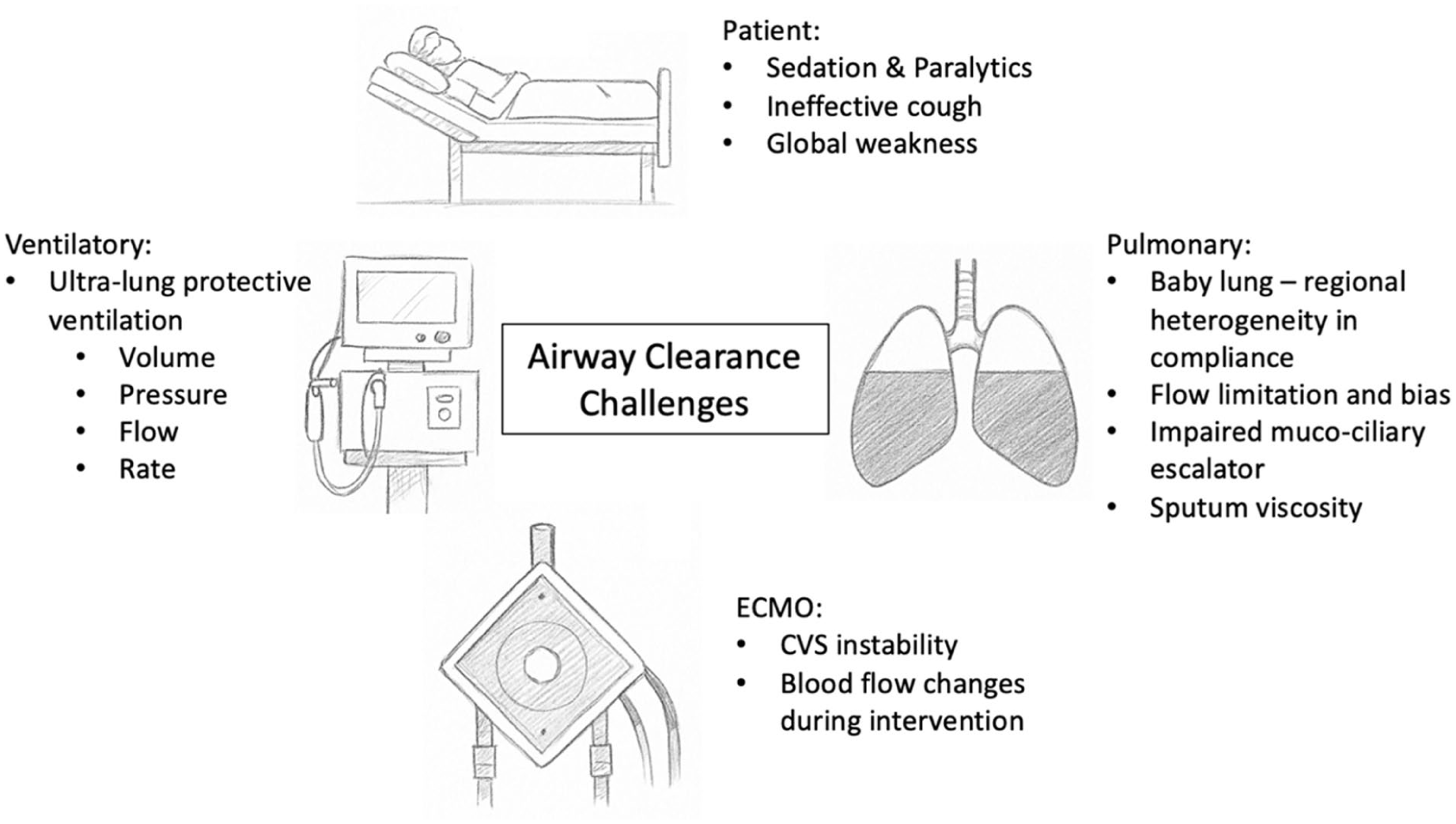
Airway clearance challenges in ECMO for SARF.
Despite the clinical importance, the respiratory physiotherapeutic management of VV-ECMO patients remains under-researched and highly variable. Intervention decisions are often influenced by the need to maintain lung protection amid extensive inflammation and minimal ventilation volumes. Existing evidence is limited, particularly regarding secretion behaviour under ultra-lung protective settings; to date, experimental studies use normal tidal volumes that do not reflect the ECMO context.
In addition, the role of mucoactive agents and saline instillation in this population is unclear. This gap in the literature is especially concerning given the unique challenges posed by VV-ECMO.
Targeted research into both physiotherapeutic techniques and pharmacological adjuncts is essential to develop evidence-based approaches that improve secretion clearance and reduce complications.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
