Abstract
Background:
Increasing numbers of patients with cancer are being admitted to intensive care units (ICU) with sepsis. The mortality from sepsis and septic shock in these patients is unclear. This study aimed to establish mortality from sepsis and septic shock in patients with cancer admitted to ICU and assess mortality trends over time.
Methods:
We conducted a literature search using MEDLINE and EMBASE. Included studies enrolled adult patients with cancer admitted to ICU with sepsis or septic shock and reported outcomes of interest. Studies were assessed using the Newcastle-Ottawa Scale for risk of bias and the quality assessment tool for observational cohort and cross-sectional studies. We performed a meta-analysis to estimate pooled ICU, hospital and 30-day mortality from sepsis and septic shock and a multivariate meta-regression to assess mortality trends over time. The study was registered on PROSPERO (CRD42022341277).
Results:
Twenty-five articles were included. The pooled ICU, hospital and 30-day mortality for sepsis was 44% (95% CI 38%–50%), 54% (95% CI 49%–60%) and 49% (95% CI 44%–55%) respectively. The pooled ICU, hospital and 30-day mortality for septic shock was 51% (95% CI 45%–57%), 62.6% (95% CI 56%–69%) and 54% (95% CI 46%–61%) respectively. There was significant heterogeneity between studies. The meta-regression identified decreasing ICU and hospital mortality from sepsis, and decreasing ICU mortality from septic shock.
Conclusion:
Patients with cancer admitted to ICU with sepsis face a significant mortality risk greater than that of the general population, despite decreasing mortality over time. Further research is required to improve outcomes.
Introduction
Due to the increasing incidence of cancer and improvements in diagnosis and management, there is a growing number of people living with cancer.1,2 The immunosuppressive effects of cancer and cancer treatments render this patient population highly susceptible to sepsis, a life-threatening organ dysfunction caused by a dysregulated host response to infection.3,4 The number of patients with cancer admitted to intensive care units (ICU) has been increasing over recent years, with sepsis amongst the leading causes of ICU admission.5,6 It has been estimated that up to 20% of patients admitted to the ICU have a history of cancer.5,6 This proportion is set to continue to increase given the increasing incidence of cancer, the development of new immunosuppressive therapies and further improvements in survival after diagnosis.1,2
Despite the increasing burden of sepsis in patients with cancer, there is a paucity of data describing outcomes in this cohort. Most published studies investigating clinical factors influencing sepsis-related mortality in patients with cancer have not been based in the ICU setting.4,7–9 Furthermore, there is uncertainty regarding the absolute case fatality rates (CFR) of sepsis in ICU among patients with cancer, and how these might have changed over time with improvements in the early detection and treatment pathways for sepsis. The paucity of data on the subject is further complicated by the variability in the definitions of sepsis across studies, and this is highlighted by the discrepancies in measured trends of sepsis when the case definition is based upon clinical criteria in comparison to administrative codes. 10
Our primary hypothesis is that sepsis-related ICU mortality in patients with cancer has reduced over time, in line with the decrease in sepsis-related mortality in the general population. The aim of this systematic review and meta-analysis is to establish ICU mortality among patients with cancer admitted to ICU with sepsis or septic shock, and determine changes in mortality over time.
Methods
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement has been used to structure this report. 11 The protocol was registered on the International Prospective Register of Systematic Reviews (PROSPERO; reference number CRD42022341277). 12
Search strategy and eligibility criteria
The electronic databases MEDLINE and Embase were searched using the Ovid search engine in February 2024. The full search strategy is presented in the Supplemental Material. Studies that enrolled adult patients (age ⩾ 18 years) with cancer admitted to ICU with sepsis or septic shock, and provided ICU, hospital or 30-day mortality data were included.
Data collection
The data collected included: the study characteristics (author, publication date, study design), the characteristics of the study participants (number of patients with cancer, mean or median age, sex distribution, type of cancer, top three sites of infection, type of organism causing sepsis), physiological scoring systems for organ dysfunction and disease severity (Sequential Organ Failure Assessment score (SOFA), Simplified Acute Physiological Score II (SAPS II), Acute Physiology and Chronic Health Evaluation II score (APACHE II)), use of organ support (vasopressors, mechanical ventilation, non-invasive ventilation and renal replacement therapy) and outcomes (ICU, hospital, 28- or 30-day mortality and ICU length of stay). 28- and 30-day mortality were combined and referred to herein as 30-day mortality. The primary outcome was ICU mortality and secondary endpoints were hospital and 30-day mortality. Data were collected manually by a single reviewer, and there were no additional processes for obtaining or confirming data from study investigators.
Risk of bias assessment
The Newcastle Ottawa Scale (NOS) tool and the quality assessment tool for observation cohort and cross-sectional studies were used to examine each study for risk of bias.13,14
Synthesis of results
We performed a meta-analysis of ICU, hospital and 30-day mortality from both patients with sepsis and patients with septic shock across all the included studies. Additionally, we examined mortality data from studies conducted from 2010 to obtain an updated understanding of current mortality. The proportion of patients who died within the prespecified timeframes and settings in each study was recorded. Logit transformation of the proportions was carried out prior to meta-analysis. A random effects model was used due to the observed variability in design of included studies. The τ2 statistic was utilised to assess between-study variance, and the proportion of outcome variability, explained by between-study heterogeneity, was assessed using the
To assess the trends in sepsis-related mortality over time measured in years, we performed a multivariate meta-regression for three outcomes: ICU, hospital and 30-day mortality among patients with sepsis. The study year assigned to each study was defined as the year at the midpoint of the study period, for studies that were longer than 1 year. The
Results
Study selection
The literature search generated 2764 articles. Following exclusions, 25 met the inclusion criteria.18–42 Figure 1 describes the process of study selection and the numbers of articles excluded on the basis of each criterion in accordance with PRISMA recommendations. 11
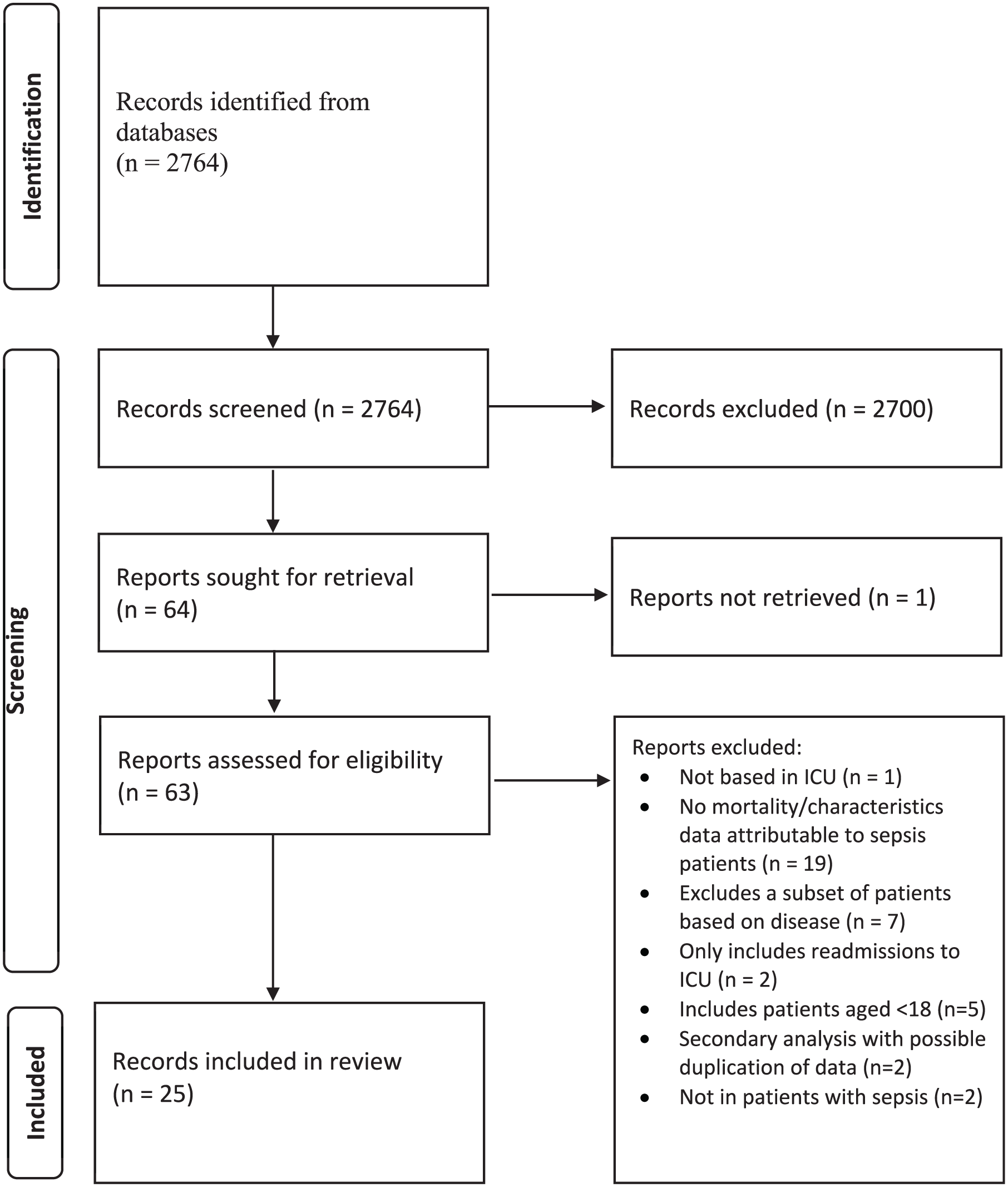
PRISMA flowchart for inclusion and exclusion of reviewed studies.
Study characteristics
Of the 25 articles included in the review, there were 17 retrospective observational studies, five prospective observational studies, one retrospective cross-sectional study, one secondary analysis of a prospective observational study and one subgroup analysis of a prospective observational study. The studies collected data from patients treated between 1994 and 2020. Ten studies were based in Europe, eight in Asia and seven in South America. 11 studies involved patients in specialised oncological ICUs. 14 publications reported outcomes in patients with sepsis (including septic shock) whereas 11 reported on septic shock alone (Supplemental Table 1). Most studies used the most recent international definition of sepsis at the time of the study with 11 studies using the Sepsis-3 definition. 3 In three publications, the authors did not reference the use of an internationally recognised definition of sepsis.
The median number of patients with cancer per study was 238 [96, 353]. The mean age of patients was 66 (13) years. 64% of the patients were male. The proportions of haematological and solid cancers across studies, when reported, were 9067/54,722 (17%) and 45,456/54,722 (83%) respectively. When stated, the most common solid cancer present was gastrointestinal and the most common haematological malignancy was leukaemia. When stated, the most common site of infection was the respiratory tract, and the most common causative organism type was gram negative bacteria (Supplemental Table 1).
Physiological scoring and organ support
Across all studies, 16 recorded a SOFA score, with 12 reporting that the score was measured within the first 24 h of admission. The mean SOFA score was 7.3 (4.1). Seven studies recorded a SAPS II score and nine recorded an APACHE II score. The mean SAPS II score was 61.3 (24.6) and the mean APACHE II score was 22.7 (7.9). In the ‘septic shock cohort’ the mean SOFA score was 9.6 (3.9), the mean SAPS II score was 61 (5.3) and the mean APACHE II score was 22.8 (7.9).
Records of the requirement for organ support varied between the studies. 76% of studies recorded the use of mechanical ventilation, 28% recorded the use of non-invasive ventilation, 72% recorded the use of renal replacement therapy and 92% recorded the use of vasopressors. Of these, the proportion of patients requiring mechanical ventilation, non-invasive ventilation, renal replacement therapy and vasopressors was 58%, 14%, 22% and 96% respectively (Supplemental Table 3).
Pooled mortality: All patients
The pooled ICU, hospital and 30-day mortality across the studies included in the meta-analysis was 44% (95% CI 38%–50%), 54% (95% CI 49%–60%) and 49% (95% CI 44%–55%) respectively. The
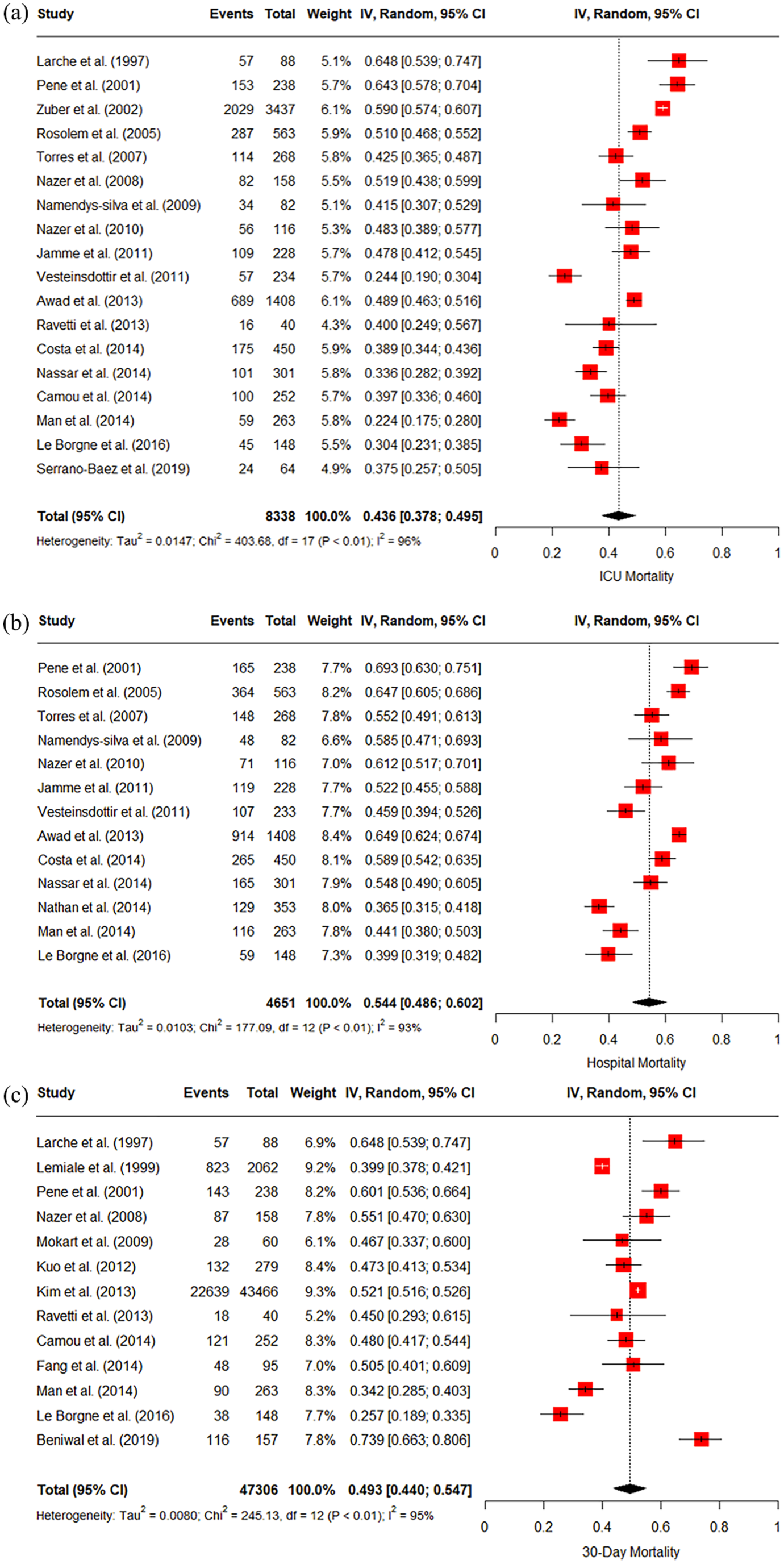
(a) Forest plot of all studies reporting ICU mortality (
The pooled ICU, hospital and 30-day mortality across all studies from 2010 was 37% (95% CI 31%–44%), 51% (95% CI 43%–59%) and 47% (95% CI 39%–55%) respectively. The
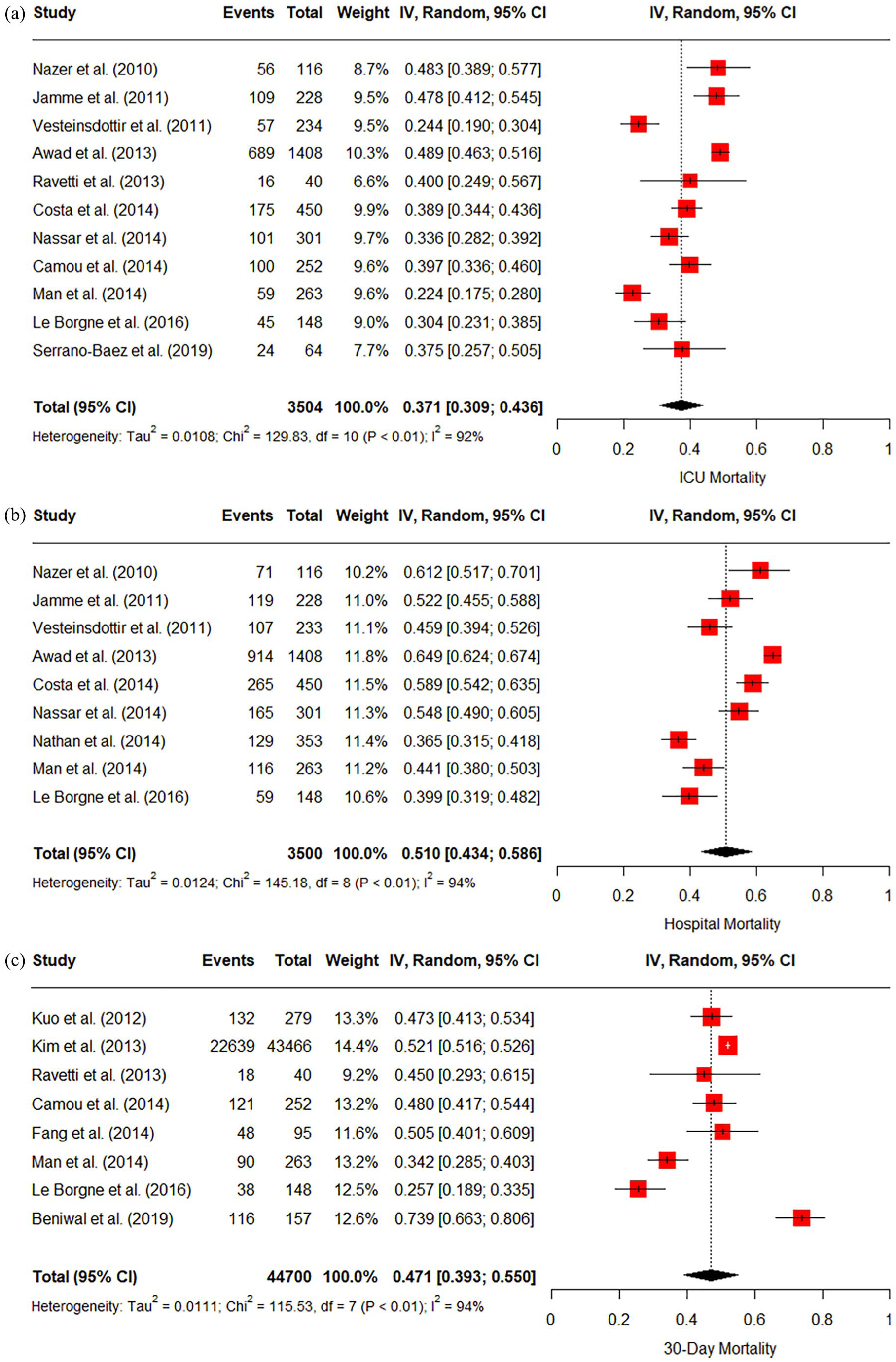
(a) Forest plot of all studies reporting ICU mortality from 2010 (
Pooled mortality: Septic shock cohort
The pooled ICU, Hospital and 30-day mortality across studies reporting on patients with cancer with septic shock was 51% (95% CI 45%–57%), 63% (95% CI 56%–69%) and 54% (95% CI 46%–61%) respectively. The
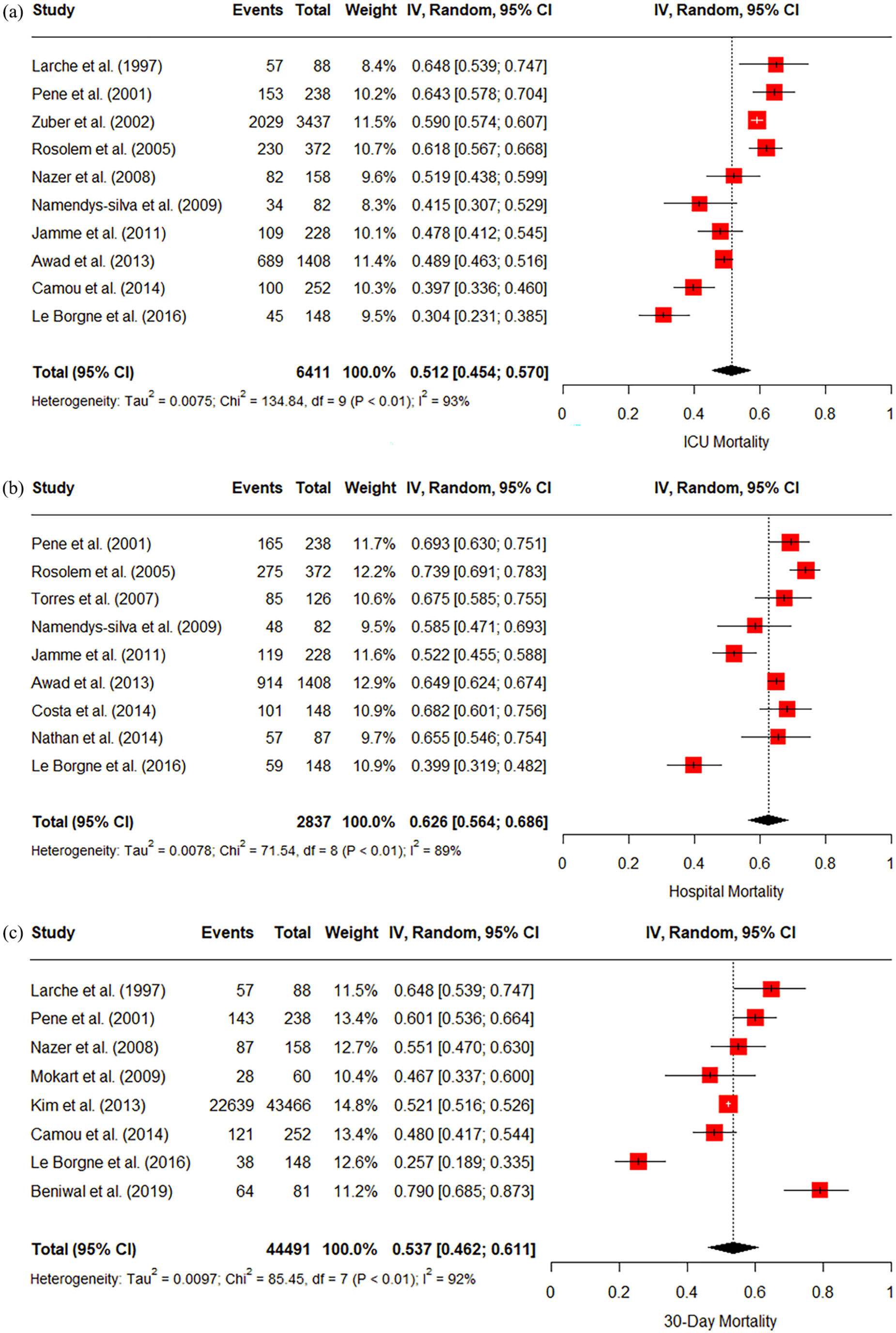
(a) Forest plot of all septic shock studies reporting ICU mortality (
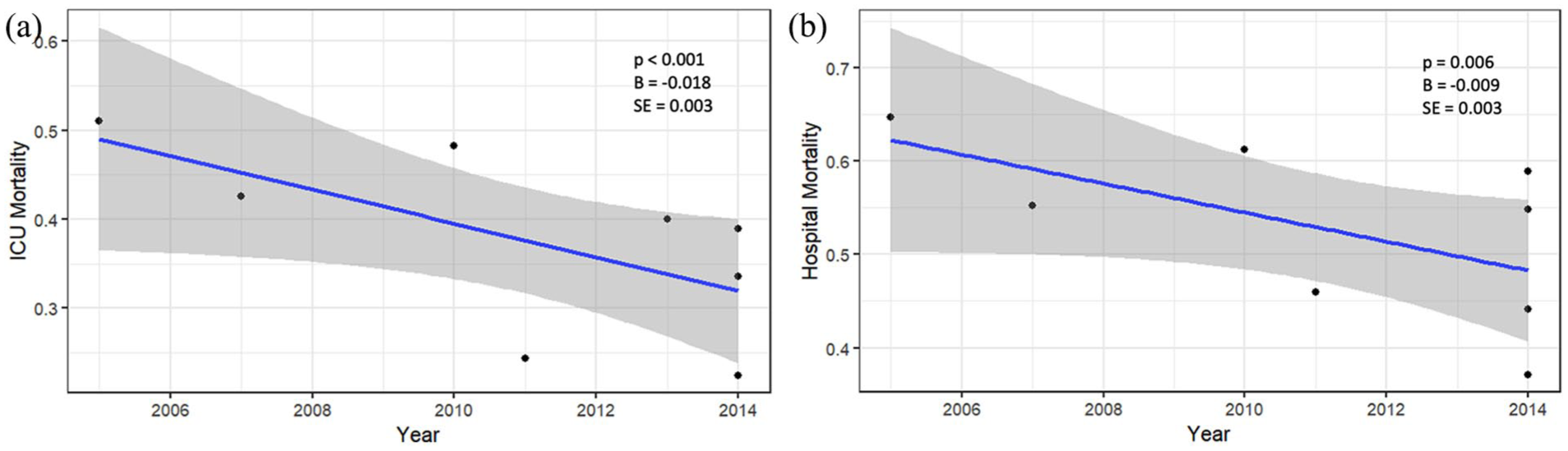
(a) Scatter plot of ICU mortality over time (
The pooled ICU, hospital and 30-day mortality across studies reporting patients with cancer with septic shock since 2010 was 42% (95% CI 35%–50%), 58% (95% CI 49%–68%) and 51% (37%–64%) respectively. The
Trend in mortality over time
After adjustment for potential confounders of mean age, proportion of haematological malignancy and definition of sepsis used in the study, a decrease in ICU mortality over time was observed on meta-regression (Coefficient: −0.018, Standard Error (SE): 0.003,
In the septic shock cohort, a decrease over time was also observed in ICU mortality (Coefficient: −0.019, SE: 0.006,
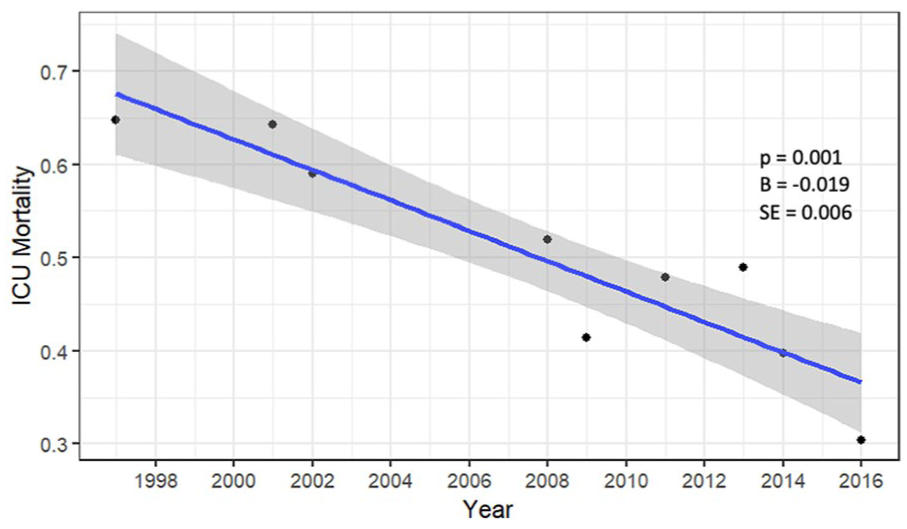
Scatter plot of ICU mortality in the septic shock studies.
Discussion
In this systematic review and meta-analysis, we estimated the mortality for patients with cancer admitted to intensive care with sepsis and septic shock across all studies, and across studies carried out after 2010, and evaluated trends in various measures of sepsis-related mortality over time for studies between 1994 and 2020. The studies were based at centres across Europe, Asia and South America. We found that pooled estimates of ICU, hospital and 30-day mortality for patients with cancer with sepsis were 44%, 54% and 50%, respectively across all studies, and 37%, 51%, 47% respectively for studies completed from 2010. We found that pooled estimates of ICU, hospital and 30-day mortality for patients with cancer with septic shock were 51%, 63% and 54% respectively across all studies, and 42%, 58% and 51%, respectively for studies completed from 2010. In addition, after accounting for differences in age, proportion of solid versus haematological malignancy and the definition of sepsis used in the studies, ICU and hospital mortality decreased over time while ICU mortality also decreased in the septic shock cohort.
Our findings suggest that in the ICU setting the expected proportion of mortality, and therefore the case fatality rate from sepsis, is high. As expected, mortality in the septic shock cohort was higher than that of all studies. Additionally, the pooled mortality estimates for both cohorts were lower for studies completed from 2010 suggesting that mortality in this cohort improved over time. As a comparison, a systematic review including studies from a similar time period assessing patients from the general population treated in ICU for sepsis found a hospital mortality of 42%, which is lower than our finding for patients with cancer. 43 Nazer et al. performed a meta-analysis of mortality from sepsis in patients with cancer in papers published from 2010 and found similar mortality estimates to our findings: 48% (95% CI 43%–53%), 62% (95% CI 58%–67%) and 50% (95% CI 38%–62%) for ICU, hospital and 30-day mortality respectively. 44 Other studies enrolling non-ICU patients have reported an increased risk of mortality from sepsis in patients with cancer when compared to patients without cancer.7,9,45 Of note, our estimate of 30-day mortality in this cohort was lower than hospital mortality. This may be related to longer hospital stays exceeding 30 days in critically unwell patients with cancer. In these patients, a fatal outcome would contribute to ICU and hospital mortality, but not 30-day mortality.
Our findings build on existing evidence that mortality from sepsis has been improving in recent years. Other studies have also reported improving survival in patients with cancer admitted to hospital with sepsis, and our results extend this pattern to patients who require ICU care.7,26,45,46 This is despite more patients, globally, with cancer being admitted to ICU for a therapeutic trial, who historically may have been limited to ward-based care.47,48 However, there are limited studies that examine this trend .
Improved survival from sepsis has also been observed in the general population. This has been attributed to greater awareness, earlier recognition and earlier administration of antibiotics driven by the education efforts as part of the Surviving Sepsis Campaign.49,50 This may also explain the improved survival from sepsis observed in patients with cancer. However, an observational study found that while mortality from sepsis in patients with cancer has improved, mortality from sepsis in patients without cancer remained static. 7 This suggests that factors specific to patients with cancer may have contributed to improved survival. One such factor may be the use of less cytotoxic and more targeted anti-cancer treatment which can reduce side effects and facilitate faster recovery. Another factor may be the earlier admission to intensive care due to more inclusive attitudes towards ICU admission for patients with cancer.
We recognise that patients with cancer and sepsis have significant heterogeneity. For example, patients with cancer who have undergone recent surgery appear to have improved survival if they develop sepsis. A large retrospective study in the US demonstrated that severe sepsis was more common in medical versus surgical hospitalisations in patients with cancer. 8 A recent study from our centre found that patients with solid organ cancer, admitted to ICU with sepsis, who had had recent surgery, had better survival compared to non-surgical admissions. 51 The surgical patient subgroup may have better baseline physiological reserves that allow them to be considered for surgical intervention in the first place, and sources of infection that are amenable to source control. These patients also benefit from structured perioperative standards of care that reduce the incidence and severity of postoperative complications such as sepsis. For example, the National Emergency Laparotomy Audit (NELA) in the UK has promoted earlier senior surgical and anaesthetic input, prompt imaging and surgery, and routine postoperative ICU admission for high-risk patients, which has brought general improvements in 30-day mortality (7th NELA). 52
Furthermore, those with haematological malignancies are susceptible to sepsis secondary to profound immunosuppression. 53 Similarly, patients receiving cytotoxic agents face broad myelosuppression and an increased risk of neutropenic sepsis while those receiving immunotherapies can be at risk of atypical infections.54,55 Finally, patients admitted to oncological ICUs with sepsis may benefit from the specialised expertise and protocols in managing cancer or treatment-related sepsis, and from the close collaboration between intensivists and oncologists or haematologists. 56
Limitations
There was a high degree of heterogeneity amongst the included studies and a wide range of reported mortality. This study included papers with differing definitions of sepsis including the Sepsis-1, Sepsis-2 and Sepsis-3 definitions, and alternative methods of defining sepsis, which may affect the accuracy of the pooled mortality estimates. 3 Several studies enrolled patients in specialised oncological ICUs which reduces the generalisability of their data to general ICUs. Given evident improvements in mortality over time, the estimated mortality derived from this review, which encompasses data from 1994 to 2020, may overstate the present-day mortality from sepsis in patients with cancer. Finally, as demonstrated in this paper, mortality trends are liable to change over time due to changes in cancer and sepsis treatments. Therefore, the literature search and analyses should be repeated on a regular basis to provide up-to-date information for clinical practice. Specifically, future studies should aim to distinguish between important subgroups such as surgical versus non-surgical ICU admissions, elective versus emergency ICU admissions, solid versus haematological malignancies, and administration of recent anti-cancer therapies.
Conclusion
Patients with cancer admitted to ICU for sepsis have a high risk of mortality. However, mortality from sepsis may be decreasing over time. This may be attributable to improvements brought about by the Surviving Sepsis Campaign and improvements in the management of cancer. Since sepsis is a preventable and often reversible cause of mortality, the presence of cancer should not be a barrier to ICU admission and provision of the highest standard of care for patients. Furthermore, regular reassessment of these trends over time is of paramount importance to inform clinical prognosis and decision-making.
Supplemental Material
sj-docx-1-inc-10.1177_17511437251363762 – Supplemental material for The evolution of mortality from sepsis in patients with cancer: A systematic review and meta-analysis
Supplemental material, sj-docx-1-inc-10.1177_17511437251363762 for The evolution of mortality from sepsis in patients with cancer: A systematic review and meta-analysis by Luke Edwards, Elizabeth Nelmes, Maddalena Ardissino, Helen Lin Jia Qi, Shaman Jhanji, David B. Antcliffe and Kate C. Tatham in Journal of the Intensive Care Society
Footnotes
Author contributions
KT designed the study and was a co-senior author with DA. LE and EN performed the literature search. MA performed the statistical analysis. LE wrote the first draft of the manuscript. All authors contributed to the study design and manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded by the Royal Marsden Cancer Charity with Luke Edwards being supported by a Royal Marsden Cancer Charity fellowship. This study represents independent research, which, along with authors Kate Tatham and Shaman Jhanji, is supported by the National Institute for Health and Care Research (NIHR) Biomedical Research Centre at The Royal Marsden NHS Foundation Trust and the Institute of Cancer Research, London. Kate Tatham has received previous funding from NIHR and Academy of Medical Sciences, outside the remit of this work. David Antcliffe is supported by the NIHR Imperial Biomedical Research Centre, the UK Intensive Care Foundation and an NIHR DSE award (NIHR303768). The views expressed in this article are those of the authors and not necessarily those of the National Health Service, NIHR or Department of Health and Social Care.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
