Abstract
Background:
Beta-blockers have been studied for potential benefits in traumatic brain injury (TBI). This study aimed to investigate the association between early beta-blocker exposure and brain injury biomarkers following moderate-severe TBI.
Methods:
We conducted a retrospective cohort study using data from the Transforming Clinical Research and Knowledge in TBI (TRACK-TBI) study. Patients ⩾ 17 years with moderate-severe TBI (Glasgow Coma Scale 3–12) admitted to an intensive care unit (ICU) were included. Early beta-blocker exposure was defined as administration within the first 72 h of admission. The primary outcome was blood-based brain injury biomarker levels on day 3 post-injury. Biomarkers included glial fibrillary acidic protein (GFAP), ubiquitin C-terminal hydrolase-L1 (UCH-L1), neuron-specific enolase (NSE), S100 calcium-binding protein B (S100B), and the inflammatory biomarker C-reactive protein (CRP). Propensity-weighted models analyzed the association between beta-blocker exposure and biomarker levels.
Results:
Among 450 patients, 31 (7%) received beta-blockers (BB+). The mean (SD) age of BB+ patients was 51.4 (16.2) years, compared to 39.5 (17.0) years for unexposed patients (BB−). BB+ group was associated with a decreased NSE level on day 3 (ratio = 0.71, 95% CI 0.52–0.96, p = 0.026), although this was not significant after adjusting for multiple comparisons (p = 0.13). For secondary outcomes, UCH-L1 levels increased on day 5 in the BB+ group (ratio = 1.62, 95% CI 1.12- 2.36, p = 0.011), but this was not significant after adjustment (p = 0.55). The NSE level on day 14 decreased in the BB+ group (ratio 0.45, 95% CI 0.30–0.66, p < 0.001) and remained significant after adjustment (p = 0.005).
Conclusions:
There was no association between early beta-blocker exposure and the primary outcome which was blood-based brain injury biomarker levels on day 3. In exploratory analysis, we found that early beta-blocker may associated with decreased NSE level on day 14. Due to the retrospective nature of the study and the use of propensity-weighted analysis to identify associations, direct clinical practice changes cannot be recommended. However, the significant association with NSE level warrants further investigation through prospective studies or randomized controlled trials to confirm the potential neuroprotective effect of early beta-blocker exposure on neuronal cellular injury.
Introduction
Traumatic brain injury (TBI) remains a significant global health problem. The Global Burden of Disease study in 2019 reported approximately 27.16 million new cases of TBI globally. 1 TBI is a complex brain disease with considerable heterogeneity in patient pathology, leading to challenges in creating standardized therapeutic protocols. After the primary injury to the brain, cerebral blood flow autoregulation enables the brain to maintain relatively constant blood flow despite the variation of blood pressures, thus protecting the brain from both hyperemia and ischemia. However, autoregulation commonly becomes dysfunctional after secondary brain injury following TBI. 2 Additionally, the profound hemodynamic instability and catecholamine surge observed in many patients after moderate to severe TBI can exacerbate the cascade of metabolic insults that further contribute to secondary brain injury. 3 Despite this challenge and the complex mechanism of secondary brain injury, no therapeutic interventions have been approved for the prevention of secondary brain injury.
Several lines of research have examined the association of beta-blockers after TBI with reduced mortality4 –6 and improved functional outcomes. 7 Additionally, there has been a growing interest in biomarkers for their potential in diagnosing 8 and prognosticating after TBI, 9 as well as gaining improved insights into the cascade of neuronal secondary brain injury that ensues following TBI. An improved understanding of the impact of early beta-blockers on neuronal cellular injury is necessary to guide therapeutic protocols for beta-blockade following TBI. To address this gap, our study examined the association of early exposure to beta-blockers with brain injury biomarkers following moderate to severe TBI.
Methods
Study design and database
We conducted a retrospective cohort study using prospectively collected data from patients enrolled in the Transforming Clinical Research and Knowledge in TBI (TRACK-TBI; ClinicalTrails.gov#NCT02119182) study, which comprises data from non-penetrating TBI patients at 18 Level 1 trauma centers in the United States between 2014-2018. 10 Detailed inclusion and exclusion criteria for the TRACK-TBI dataset have been described previously. 11 Briefly, TRACK-TBI subjects presented to the emergency department (ED) of participating institutions within 24 h of a TBI. 12 Data collected included demographic and clinical information, and blood samples (aliquoted and frozen at −80°C within 1 h per TBI-CDE protocol 10 ). Data were collected by trained research personnel, using structured data collection tools conforming to the National Institute of Neurological Disorders and Stroke (NINDS) TBI Common Data Elements. 13 Procedures were followed by the ethical standards of the responsible committee on human experimentation, and with the Helsinki Declaration of 1975. The study was approved by the Institutional Review Board at Duke University (Date of IRB approval, August 16th, 2022).
Study population
We included adults (age ⩾ 17 years) in the TRACK-TBI cohort with moderate to severe TBI, defined as the Glasgow coma Scale (GCS) < 13 after resuscitation, who were admitted directly to an intensive care unit (ICU).
Exposure, outcomes, and covariates
The primary exposure was early beta-blocker administration, defined as beta-blocker exposure within 72 h of admission. We have included both cardio-selective (acebutolol, atenolol, bisoprolol, metoprolol, and nebivolol) and non-selective (nadolol, propranolol) beta-blockers. The primary outcome was brain injury biomarker levels on day 3 following injury, glial fibrillary acidic protein (GFAP; pg/mL),14,15 ubiquitin carboxy-terminal hydrolase L1 (UCH-L1; pg/mL)16,17, S100 calcium-binding protein B ( S100B; μg/L),17,18 Neuron Specific Enolase (NSE; ng/mL) released form neuronal cell body injury,19,20 and high-sensitivity C-reactive protein (hs-CRP; mg/L).17,21 The details of each biomarker are in Supplemental Table 1. Secondary outcomes included the aforementioned brain injury biomarker on days 5 and 14. Covariates included: hospital site, age, sex, race, ethnicity, educational levels, injury causes, Glasgow Coma Scale (GCS) at Emergency Department (ED) arrival, systolic blood pressure and mean arterial pressure at ED, injury cause, injury mechanism, non-head/neck Injury Severity Score (ISS), Abbreviated Injury Scale (AIS) head score, initial head CT finding, Rotterdam Score, blood transfusion in the ED, hyperosmolar therapy, the history of preinjury beta-blocker used, the history of TIA, and baseline (day 1) brain injury biomarkers.
Statistical analysis
Differences in patient and injury characteristics between those who did and did not receive beta-blockers within the first 72 h of admission were assessed for statistical imbalance using Mann-Whitney tests for continuous and ordinal variables, as well as Fisher’s exact tests for categorical variables (Table 1). Biomarker levels were log-transformed to achieve sufficient Gaussian distributions. Differences in biomarker levels were evaluated using linear mixed-effects regression on the log-transformed values. For each biomarker type, we incorporated all biomarker values at Days 1, 3, 5, and 14 into a single mixed model, fitting a random intercept for each subject. All analyses used inverse probability weighting to account for differences in the baseline characteristics as a result of the non-random group allocation. The weights were derived by using a boosted regression algorithm to generate predicted probabilities of being in the beta-blockers cohort based on all baseline characteristics found in Table 1. Changes in biomarker values over time were assessed by adding a group-by-time interaction term to the model. Effect sizes are expressed as ratios of the untransformed biomarker values, which are the unstandardized B coefficients from the log-transformed regression. All analyses were run as complete case without accounting for survivorship bias due to a lack of outcome assessment. Statistical significance was determined using a two-sided threshold of p < 0.05. Both primary and secondary results were adjusted for multiple comparisons using a 5% false-discovery rate per Benjamini-Hochberg. The boosted regression was implemented using the Toolkit for Weighting and Analysis of Nonequivalent Groups (TWANG) application by Rand Corporation (downloaded September 2023). All other analysis was carried out using SPSS version 26 (Armonk, NY) and SAS version 9.4 (Cary, NC) statistical software.
Patients Demographic Data.
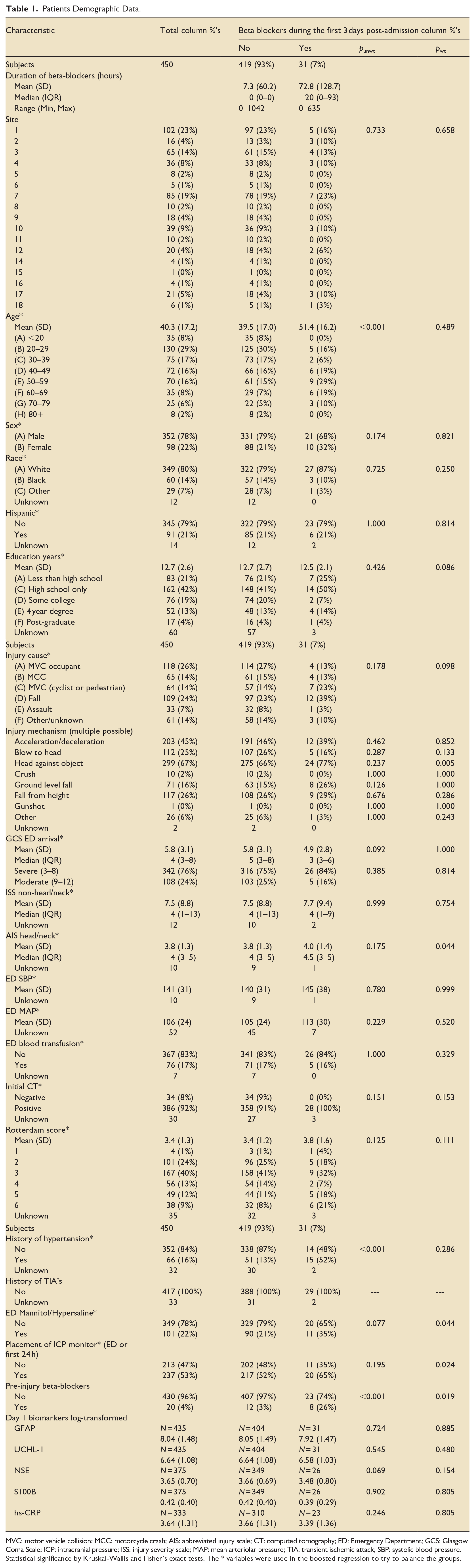
MVC: motor vehicle collision; MCC: motorcycle crash; AIS: abbreviated injury scale; CT: computed tomography; ED: Emergency Department; GCS: Glasgow Coma Scale; ICP: intracranial pressure; ISS: injury severity scale; MAP: mean arteriolar pressure; TIA: transient ischemic attack; SBP: systolic blood pressure.
Statistical significance by Kruskal-Wallis and Fisher’s exact tests. The * variables were used in the boosted regression to try to balance the groups.
Results
Demographic and clinical characteristics of the Cohort
After applying the eligibility criteria, the total study population comprised 450 subjects (Figure 1). The number of males was 352 (78%) and the mean (SD) age was 40.3 (17.2) years. The most common causes of TBI were motor vehicle collision (MVC) occupants (n = 118, 26%) and falls (n = 109, 24%). The median (IQR) GCS at ED arrival was 4 (3–8). The median (IQR) Injury Severity Score (ISS) non-head/neck was 4 (1–13), while the median (IQR) Abbreviated Injury Scale (AIS) Head/Neck was 4 (3–5). The mean (SD) of the Rotterdam score was 3.4 (1.3). The majority of patients initially presented with a positive CT scan (92%). In our study population, 101 (22%) of patients received hyperosmolar therapy, 76 (17%) received a blood transfusion in the ED, and 237 (53%) had an ICP monitor placed during the first 24 h of ICU admission.
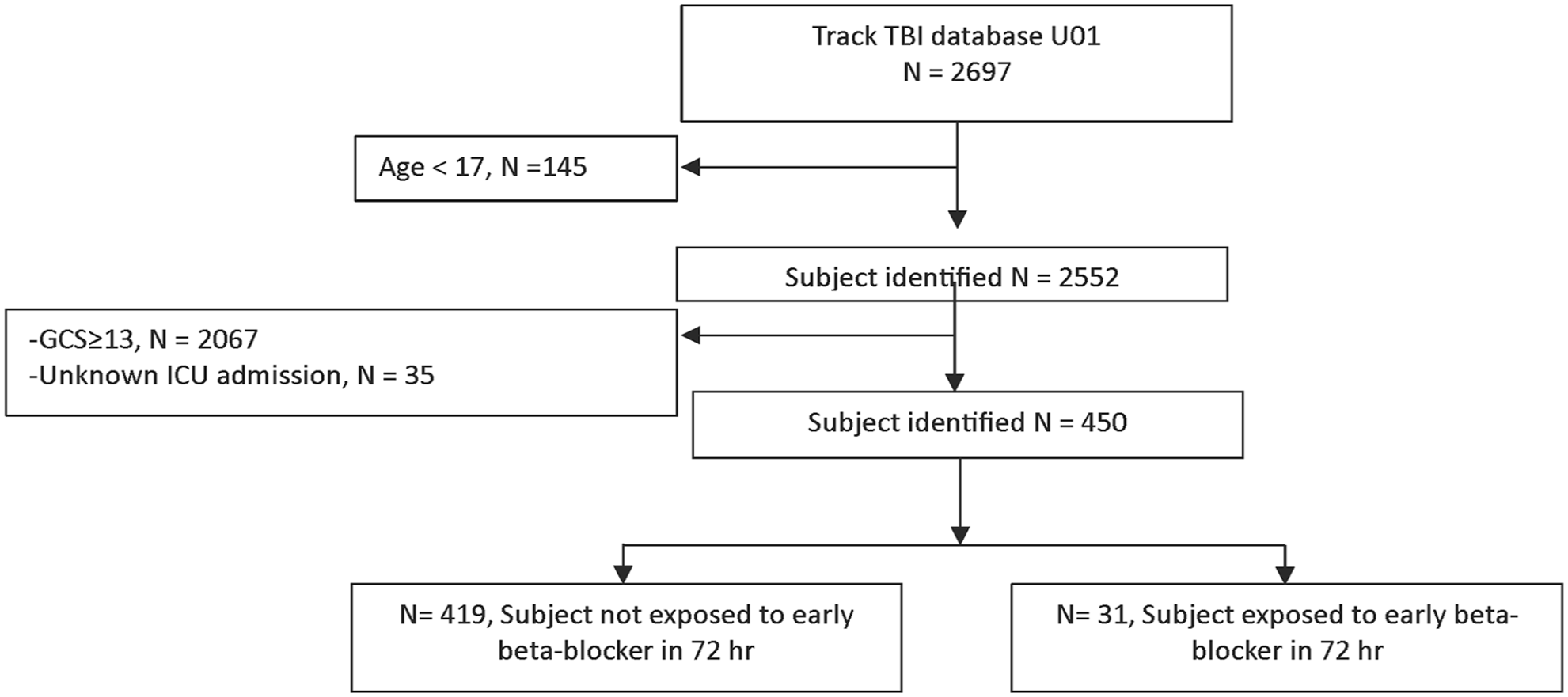
Study flow diagram.
Among the study population, 31 patients (7%) received beta-blockers (BB+) in the first 72 h. The mean age (SD) of the BB+ group was significantly higher than the non-exposed group (BB−), 51.4 (16.2) years versus 39.5 (17) years, (p < 0.001). The rate of history of hypertension was also significantly higher in the BB+ group at 52% versus 13% (p < 0.001). Treatment with mannitol was more common in the BB+ group, with 11/31 (35%) versus 90/419 (21%), as well as the placement of an ICP monitor, which was found more in the BB+ group, with 20/31 (65%) versus 217/419 (52%). Patients in the BB+ group also had significantly greater exposure to preinjury beta-blockers compared to the BB-, with 8/31 (26%) versus 12/419 (3%), (p < 0.001). Supplemental Figure 1 illustrates the daily average mean arterial pressure (MAP) and heart rate (HR) in patients with and without beta-blockers over the first 5 days, showing that MAP and HR tend to be higher in the BB+ group.
The log-transformed level of day 1 biomarkers, including GFAP, UCH-L1, NSE, S100B, and hs-CRP had no statistically significant associations between the two groups. Differences in pre- and post-propensity weighting are described in Table 1.
Association of early beta-blocker exposure and day 3 biomarkers
Regarding the primary outcome, early beta-blocker exposure was associated with a decreased day 3 biomarker level of NSE (ratio = 0.71, 95% CI 0.52–0.96, p = 0.026). However, this significance was lost after adjusting for multiple comparisons, (p* = 0.13). Other biomarker levels were not significantly different compared to the BB- group: GFAP (ratio = 0.91, 95% CI 0.57–1.45, p = 0.68), UCH-L1 (ratio = 1.40, 95% CI 0.98–2.01, p = 0.063), S100B (ratio = 1.03, 95% CI 0.94–1.12, p = 0.55), hs-CRP (ratio = 0.92, 95% CI 0.51–1.64, p = 0.77), as described in Table 2.
Outcomes (modeling change from day 1 biomarker value).
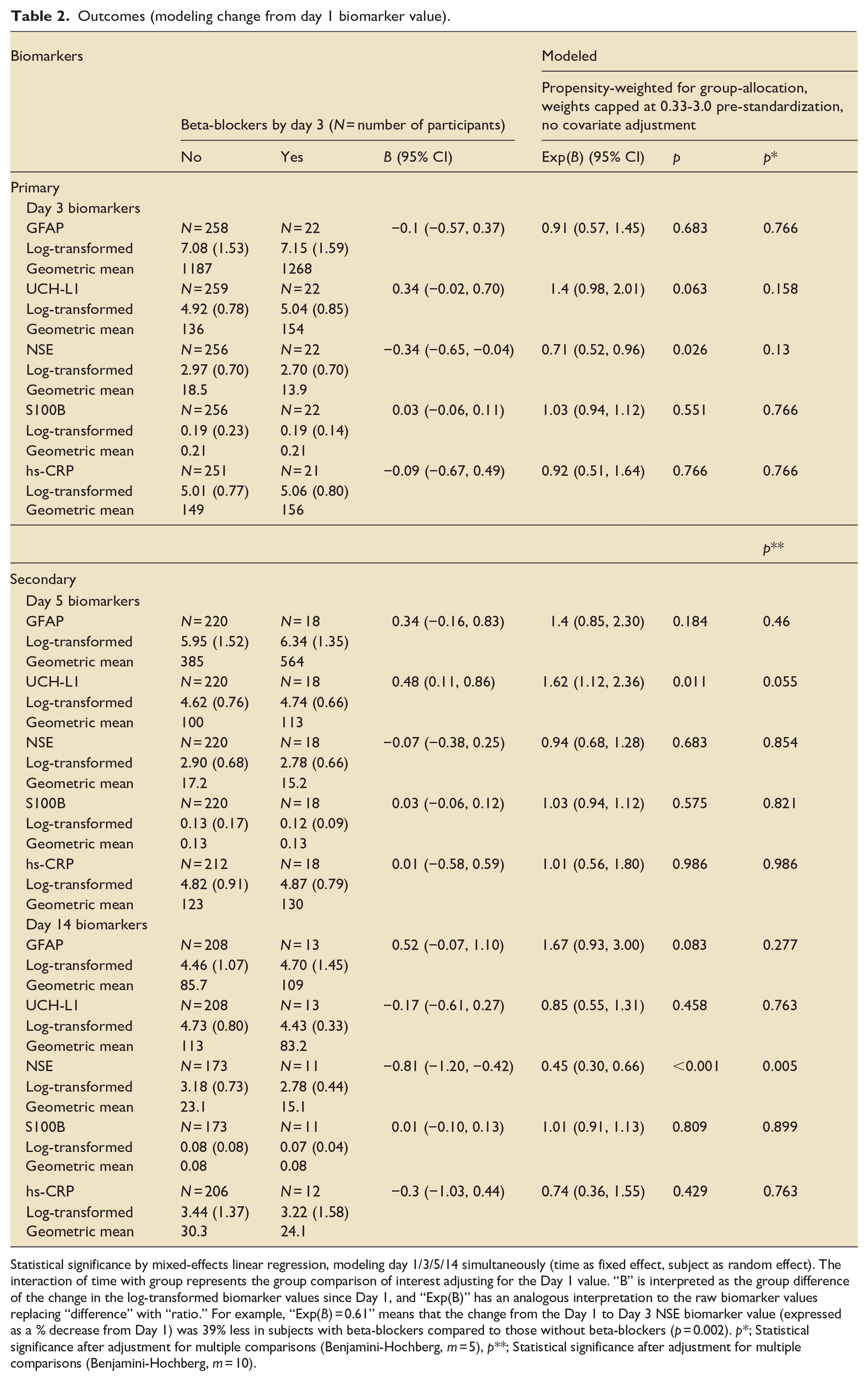
Statistical significance by mixed-effects linear regression, modeling day 1/3/5/14 simultaneously (time as fixed effect, subject as random effect). The interaction of time with group represents the group comparison of interest adjusting for the Day 1 value. “B” is interpreted as the group difference of the change in the log-transformed biomarker values since Day 1, and “Exp(B)” has an analogous interpretation to the raw biomarker values replacing “difference” with “ratio.” For example, “Exp(B) = 0.61” means that the change from the Day 1 to Day 3 NSE biomarker value (expressed as a % decrease from Day 1) was 39% less in subjects with beta-blockers compared to those without beta-blockers (p = 0.002). p*; Statistical significance after adjustment for multiple comparisons (Benjamini-Hochberg, m = 5), p**; Statistical significance after adjustment for multiple comparisons (Benjamini-Hochberg, m = 10).
Association of early beta-blocker exposure and day 5 and 14 biomarkers
For the secondary outcomes, the day 5 biomarker level of UCHL-1 was increased from day 1 in the BB+ group (ratio = 1.62, 95% CI 1.12–2.36, p = 0.011), but this was not significant after adjusting for multiple comparisons, (p** = 0.055). Other biomarkers were not statistically different compared to the BB- group: GFAP (ratio = 1.40, 95% CI 0.85–2.30, p = 0.18), NSE; (ratio = 0.94, 95% CI 0.68–1.28, p = 0.68), S100B (ratio = 1.03, 95% CI 0.94–1.12, p = 0.58), hs-CRP (ratio = 1.01, 95% CI 0.56–1.80, p = 0.99), as shown in Table 2.
On day 14, the level of NSE decreased from day 1 in the BB+ group (ratio = 0.45, 95% CI 0.30–0.66, p < 0.001) and remained significant after multiple comparisons. (p** = 0.005). No statistically significant associations were observed in other biomarkers: GFAP (ratio = 1.67, 95% CI 0.93–3.00, p = 0.083), UCH-L1 (ratio = 0.85, 95% CI 0.55–1.31, p = 0.46), S100B (ratio = 1.01, 95% CI 0.91–1.13, p = 0.81), hs-CRP (ratio = 0.74, 95% CI 0.36–1.55, p = 0.43), as shown in Table 2. The trajectories of log-transformed biomarkers are described in Figure 2.
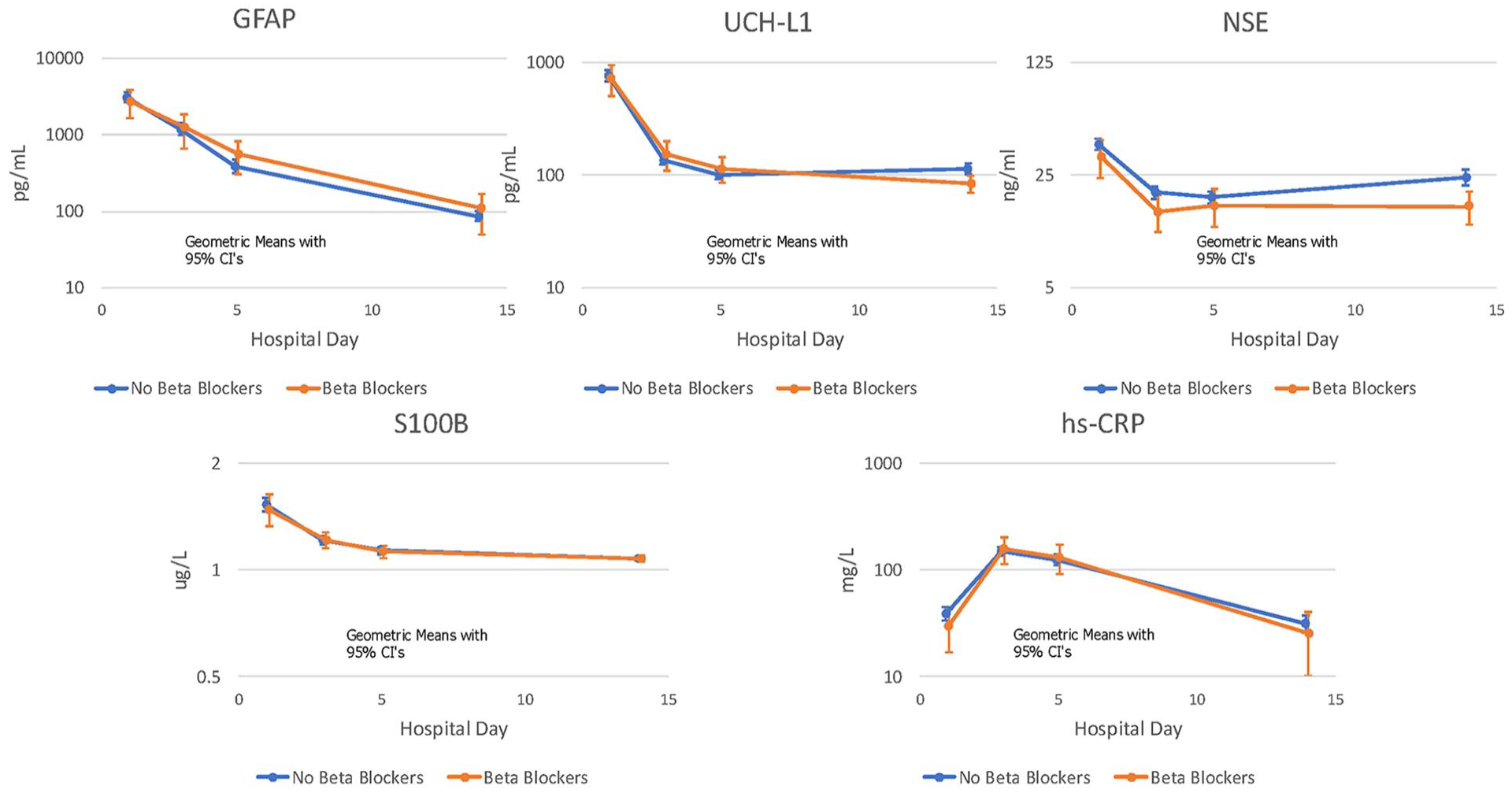
The trajectories of each biomarker and means.
After conducting a sensitivity analysis excluding subjects with pre-injury beta-blocker exposure, we found an association with decreased day 3 NSE biomarker levels (ratio = 0.51, 95% CI: 0.31–0.83, p = 0.007). Additionally, the sensitivity analysis also showed a significant association for NSE on day 14 (ratio = 0.36, 95% CI: 0.20–0.36, p = 0.001; Supplemental Table 2 and Supplemental Figure 2).
Discussion
Our report is the first study to analyze the association between beta-blockers and brain injury biomarkers in humans. This retrospective cohort study investigated the association of beta-blockers with brain injury biomarkers. Only 7% of eligible patients received beta-blockers within the first 72 h following ICU admission. In adjusted analyses, we observed that early beta-blocker exposure was associated with a decreased NSE level on day 3 prior to, but not after, adjustment for multiple comparisons. A decreased NSE level was observed on day 14 and remained significant after adjustment for multiple comparisons. No significant differences were observed in other brain injury biomarkers on days 3, 5, and 14.
Beta-blockers exert their benefits by blocking the activation of the sympathetic nervous system and subsequent release of catecholamines after TBI. 22 These catecholamines impact not only the stress response but also the inflammatory response, hypermetabolism, and host protective immunity. The surge of sympathetic response is linked to poorer patient outcomes following TBI.22,23Additionally, beta-blockers may also work by preventing the impairment of cerebral autoregulation, 24 improving cerebral perfusion, 25 and decreasing cerebral hypoxia.26,27 While beta-blockers have been proposed protocols like the Lund concept to modulate sympathetic hyperactivity and control systemic hypertension, maintaining adequate cerebral perfusion pressure (CPP) remains a priority in TBI management. Many studies have accepted higher systolic blood pressure targets, especially in the early phase, to ensure sufficient brain perfusion.28,29 However, excessive elevations particularly in the context of autonomic dysfunction such as paroxysmal sympathetic hyperactivity can disrupt cerebral autoregulation, increase cerebral blood volume, and raise intracranial pressure (ICP), potentially worsening secondary brain injury. 29 Therefore, while maintaining blood pressure high enough to optimize CPP is crucial, careful modulation of blood pressure is needed to avoid the harmful effects of excessive sympathetic activity.
Several lines of evidence support this potential benefit of beta-blockers. Observational studies 25 and retrospective reviews7,30 suggest an association with lower mortality and improved functional outcomes. Arbabi et al. 31 found a reduced risk of death in trauma patients receiving beta-blockers, particularly those with a severe head injury. Finally, a randomized controlled trial (RCT) by Khalili et al. 3 demonstrated a reduction in mortality and improved functional outcomes with beta-blockers.
However, other clinical studies evaluating the use of beta-blockers in patients with TBI have yielded conflicting results. A study using the TRACK-TBI data found no difference in the Glasgow Outcome Scale-Extended (GOS-E) score at 6 months, possibly due to the variation in beta-blocker doses and timing. 32 Additionally, one underpowered RCT did not observe a significant difference in catecholamine level after TBI with beta-blocker use. 33 Safety data on beta-blockers for TBI patients is mixed. While some studies suggest the beta-blockers are safe with minimal impact on heart rate variability and blood pressure,30,34 others report increased infection rates, longer ventilation support, extended ICU stay, and longer hospital stays overall.35,36 Despite potential benefits, these side effects require careful consideration, given the potential for hypotension to compromise cerebral perfusion and exacerbate secondary brain injury.
Despite promising evidence for improved mortality and functional outcomes with beta-blockers in TBI, our study highlights the need for further investigation regarding the impact on biomarkers. We observed that early administration of beta-blockers was associated with reduced day 14 NSE levels, a marker of direct neuronal injury. Elevated NSE is linked to worse outcomes in TBI, including increased intracranial pressure (ICP), decreased cerebral perfusion (CPP), and greater severity.19,37,38 As one of the most common TBI biomarkers used clinically, 39 NSE may help monitor treatment effectiveness. A previous study found a correlation between higher serum NSE levels and unfavorable outcomes. 40 This observed reduction of NSE with beta-blockers aligns with the prior studies suggesting their benefit, potentially supporting their use in moderate to severe TBI populations. However, we did not observe an association with other biomarkers.
In our study of moderate to severe TBI patients treated with beta-blockers, we observed 41 an interesting pattern in NSE levels. NSE levels typically rise within the first 12 h after a TBI due to passive release from cellular destruction. A second peak, observed within 7–94 h in cerebrospinal fluid (CSF), is believed to represent a secondary wave of neuronal death.41 –43 However, our exploratory blood-based NSE data showed a rise in NSE levels at 14 days post-injury. The biphasic peak in NSE levels suggests the possibility of secondary brain injuries like edema or additional tissue damage, or an increased risk of secondary organ injury or hemolysis.
Thus, it may be possible that NSE in early BB use is lower than no early BB on day 14. Further research is needed to better understand the effect of early BB use on secondary brain injuries.
Additionally, we observed a contrasting trend in the BB+ group concerning UCHL-1 level on day 5 and NSE patterns. However, the significance of these trends was not maintained after adjusting for multiple comparisons. Both NSE and UCH-L1 are acute biomarkers associated with neuronal cell body injury.
Our study has several limitations. First, the observed data were based on a small sample size of patients exposed to beta-blockers, which may impact our ability to stratify the preinjury beta-blockers exposure for sensitivity analysis. Additionally, due to the heterogeneous types of beta-blockers, combined with the lack of universal guidelines for their use and the variability in protocols among institutions, such as indication, timing of and the duration of administration, may influence our findings. Furthermore, the definition of our population (where the exposed group is defined as those exposed within 72 h) could also impact the results. These factors might affect the exploratory secondary outcomes, which are the biomarker on day 5 and 14.
The observational nature of the study may not establish a true causal relationship between beta-blocker exposure and brain injury biomarker levels. Consequently, this approach may introduce bias, making it challenging to determine whether the observed effects stem from the beta-blockers themselves or are confounded by other factors. The heterogeneity in the types of beta-blockers used reflects variations in physician preference, institutional protocols and the availability of medications, further complicate the analysis. Additionally, given the small number of patients receiving beta-blockers, we recognize the limitations in our propensity score modeling and the potential for residual confounding. Although we attempted to use propensity weights and other statistical methods to minimize confounding factors, the residual confounder of the observational cohort persisted.
Second, the limitations regarding the interpretation of NSE as a biomarker for TBI arise from the fact that the brain is not the sole source of NSE. It is also found in erythrocytes and can be influenced by hemolysis, 44 which may significantly impact the utility of serum NSE as a biomarker. The interpretation of NSE levels in relation to beta-blocker exposure should be with caution, given that NSE is not specific to the brain and due to the exploratory nature of this outcome. The observed association may not indicate a causal benefit of beta-blocker use. Third, because we are selecting patients who received beta-blockers within the first 72 h, and considering that nadolol, the beta-blocker with the longest half-life in the study (24 h), the results of biomarkers after day 5 might not accurately reflect the true benefit of early beta-blocker administration. Considering the poor biomarker specificity, the current consensus favors the use of a combination of markers in a panel, which may provide a more robust measure of concussion than a single biomarker. 45 Lastly, despite observed differences in brain injury biomarker levels, and given the above limitations, our results should be considered exploratory and hypothesis-generating for future studies to find the biological plausibility of how beta-blockers could influence on biomarkers.
Conclusion
Early beta-blocker exposure was not associated with changes in biomarker levels on day 3 and 5. However, in exploratory analysis, we found that early beta-blocker use may be associated with decreased NSE levels on day 14. Early beta-blocker exposure may affect neuronal cellular injury, but this will require confirmation in future larger and well-powered studies. If the implementation of early beta-blockers into TBI management is to be considered, high-quality RCTs to elucidate the optimal type, dosage, and timing of treatment to mitigate secondary brain injuries while controlling for systemic comorbidities and concomitant extracranial injuries are needed.
Supplemental Material
sj-docx-1-inc-10.1177_17511437251349680 – Supplemental material for Early beta-blocker exposure and association with brain injury biomarkers following moderate to severe traumatic brain injury: A TRACK-TBI study
Supplemental material, sj-docx-1-inc-10.1177_17511437251349680 for Early beta-blocker exposure and association with brain injury biomarkers following moderate to severe traumatic brain injury: A TRACK-TBI study by Pattrapun Wongsripuemtet, Tetsu Ohnuma, Nancy Temkin, Jason Barber, Jordan Komisarow, Geoffrey T Manley, Jordan Hatfield, Miriam Treggiari, Katharine Colton, Cina Sasannejad, Nophanan Chaikittisilpa, Ramesh Grandhi, Daniel T Laskowitz, Joseph P Mathew, Adrian Hernandez, Michael L James, Karthik Raghunathan, Joseph B Miller, Monica S Vavilala, Ben Goldstein, Vijay Krishnamoorthy and the TRACK-TBI Investigators in Journal of the Intensive Care Society
Supplemental Material
sj-docx-2-inc-10.1177_17511437251349680 – Supplemental material for Early beta-blocker exposure and association with brain injury biomarkers following moderate to severe traumatic brain injury: A TRACK-TBI study
Supplemental material, sj-docx-2-inc-10.1177_17511437251349680 for Early beta-blocker exposure and association with brain injury biomarkers following moderate to severe traumatic brain injury: A TRACK-TBI study by Pattrapun Wongsripuemtet, Tetsu Ohnuma, Nancy Temkin, Jason Barber, Jordan Komisarow, Geoffrey T Manley, Jordan Hatfield, Miriam Treggiari, Katharine Colton, Cina Sasannejad, Nophanan Chaikittisilpa, Ramesh Grandhi, Daniel T Laskowitz, Joseph P Mathew, Adrian Hernandez, Michael L James, Karthik Raghunathan, Joseph B Miller, Monica S Vavilala, Ben Goldstein, Vijay Krishnamoorthy and the TRACK-TBI Investigators in Journal of the Intensive Care Society
Supplemental Material
sj-tiff-3-inc-10.1177_17511437251349680 – Supplemental material for Early beta-blocker exposure and association with brain injury biomarkers following moderate to severe traumatic brain injury: A TRACK-TBI study
Supplemental material, sj-tiff-3-inc-10.1177_17511437251349680 for Early beta-blocker exposure and association with brain injury biomarkers following moderate to severe traumatic brain injury: A TRACK-TBI study by Pattrapun Wongsripuemtet, Tetsu Ohnuma, Nancy Temkin, Jason Barber, Jordan Komisarow, Geoffrey T Manley, Jordan Hatfield, Miriam Treggiari, Katharine Colton, Cina Sasannejad, Nophanan Chaikittisilpa, Ramesh Grandhi, Daniel T Laskowitz, Joseph P Mathew, Adrian Hernandez, Michael L James, Karthik Raghunathan, Joseph B Miller, Monica S Vavilala, Ben Goldstein, Vijay Krishnamoorthy and the TRACK-TBI Investigators in Journal of the Intensive Care Society
Supplemental Material
sj-tiff-4-inc-10.1177_17511437251349680 – Supplemental material for Early beta-blocker exposure and association with brain injury biomarkers following moderate to severe traumatic brain injury: A TRACK-TBI study
Supplemental material, sj-tiff-4-inc-10.1177_17511437251349680 for Early beta-blocker exposure and association with brain injury biomarkers following moderate to severe traumatic brain injury: A TRACK-TBI study by Pattrapun Wongsripuemtet, Tetsu Ohnuma, Nancy Temkin, Jason Barber, Jordan Komisarow, Geoffrey T Manley, Jordan Hatfield, Miriam Treggiari, Katharine Colton, Cina Sasannejad, Nophanan Chaikittisilpa, Ramesh Grandhi, Daniel T Laskowitz, Joseph P Mathew, Adrian Hernandez, Michael L James, Karthik Raghunathan, Joseph B Miller, Monica S Vavilala, Ben Goldstein, Vijay Krishnamoorthy and the TRACK-TBI Investigators in Journal of the Intensive Care Society
Footnotes
Author contribution
Pattrapun Wongsripuemtet: Conceptualization, Investigation, Writing - Original Draft, Writing - Review & Editing Tetsu. Ohnuma: Conceptualization, Methodology, Investigation, Writing - Original Draft, Writing - Review & Editing, Supervision. Nancy Temkin: Conceptualization, Methodology, Formal analysis, Validation, Writing - Original Draft, Writing - Review & Editing. Jason Barber: Conceptualization, Methodology, Formal analysis, Validation, Writing - Original Draft, Writing - Review & Editing Jordan. Komisarow: Conceptualization, Investigation, Writing - Original Draft, Writing - Review & Editing. Geoffrey T. Manley: Resources, Writing - Review & Editing Jordan Hatfield: Conceptualization, Investigation, Writing - Original Draft, Writing - Review & Editing. Miriam Treggiari: Writing - Review & Editing. Katharine Colton: Resources, Writing - Review & Editing. Cina Sasannejad: Conceptualization, Investigation, Writing - Original Draft. Nophanan Chaikittisilpa: Writing - Review & Editing. Ramesh Grandhi: Resources, Writing - Review & Editing. Daniel T. Laskowitz: Resources, Writing - Review & Editing. Joseph P. Mathew: Resources, Writing – Review & Editing. Adrian Hernandez: Resources, Writing - Review & Editing. Michael L. James: Resources, Writing - Review & Editing. Karthik Raghunathan: Resources, Writing - Review & Editing. Joseph B. Miller: Resources, Writing - Review & Editing. Monica S.Vavilala: Resources, Writing - Review & Editing. Ben Goldstein: Writing - Review & Editing. Vijay Krishnamoorthy: Conceptualization, Methodology, Investigation, Writing - Original Draft, Writing - Review & Editing, Supervision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Institutes of Health: R01NS10832 (Krishnamoorthy).
Ethical consideration
The study adhered to ethical guidelines with IRB. The study was approved by the Institutional Review Board at Duke University (Date of IRB approval, August 16th, 2022).
Consent to participate
Inform consent was obtained from participants enrolled in the study. This study was conducted by using data from the Transforming Clinical Research and Knowledge in Traumatic Brain Injury Study (TRACK-TBI; ClinicalTrials.gov #NCT02119182). Data were collected by trained research personnel, using structured data collection tools conforming to the National Institute of Neurological Disorders and Stroke (NINDS) TBI Common Data Elements (Hicks R, Giacino J, Harrison-Felix C, Manley G, Valadka A, Wilde EA. Progress in developing common data elements for traumatic brain injury research: version two–the end of the beginning. Journal of Neurotrauma 2013;30(22):1852-1861). The replication and ongoing study are publicly available at ![]() . This study was approved by the Institutional Review Board at Duke University. Detailed inclusion and exclusion criteria for the TRACK-TBI dataset have been described previously (Yuh EL, Cooper SR, Mukherjee P, et al. Diffusion tensor imaging for outcome prediction in mild traumatic brain injury: a TRACK-TBI study. J Neurotrauma 2014;31(17):1457-77. DOI: 10.1089/neu.2013.3171.).
. This study was approved by the Institutional Review Board at Duke University. Detailed inclusion and exclusion criteria for the TRACK-TBI dataset have been described previously (Yuh EL, Cooper SR, Mukherjee P, et al. Diffusion tensor imaging for outcome prediction in mild traumatic brain injury: a TRACK-TBI study. J Neurotrauma 2014;31(17):1457-77. DOI: 10.1089/neu.2013.3171.).
Consent for publication
Not applicable.
Copyright permission statement
The authors hereby grant written permission to the Journal of Intensive Care Society to reproduce all figures and materials included in this manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
