Abstract
Background:
Hypermetabolism and enteral nutrition delivery challenges, result in trauma patients, becoming malnourished during their hospital, admission. This study aimed to explore enteral nutrition delivery, predictors of suboptimal delivery and the relationship with clinical outcomes in patients admitted to a critical care unit following major trauma.
Methods:
An exploration of nutrition related data collected as part of a multicentre prospective major trauma study was conducted. Nutrition related data included anthropometry, nutrition risk screening, feeding route, nutrition products, target volume, nutrition delivery and causes of enteral feeding interruptions. Multivariate logistic regression analysis was used to evaluate the strongest associations with suboptimal nutrition delivery.
Results:
Of 1036 participants, 71% (n = 732) required enteral nutrition for a mean of 15.7 (7.9) days. Suboptimal nutrition delivery was prevalent throughout the admission. Mean energy target was 23.8 (6.37) versus 15.81 (3.43) kcal/kg/day delivered (p < 0.001). Mean protein target was 1.27 (0.34) versus 0.89 (0.48) g/kg/day delivered (p < 0.001). Factors associated with suboptimal nutrition delivery included male sex (OR, 1.82, 95% CI 1.27–2.60; p < 0.001), traumatic brain injury (OR, 1.67, 95% CI 1.16–2.40; p = 0.006) or high NUTRIC score (OR, 1.17, 95% CI 1.08–1.27; p < 0.001); early enteral nutrition reduced the risk of underfeeding (OR, 0.49, 95% CI 0.30–0.81; p = 0.006). Lower energy and protein delivery were associated with increased days of mechanical ventilation (p < 0.001) and longer length of stay in both the critical care unit and overall hospital stay (p < 0.001).
Conclusion:
Trauma patients experience inadequate enteral nutrition delivery which potentially negatively impacts clinical outcomes. Additional investigation is required to further understand the barriers and facilitators to adequate nutrition provision in critically ill trauma patients.
Introduction
The metabolic changes after major trauma are distinctive, complex and make trauma patients more susceptible to developing malnutrition following hospital admission. 1 Critically ill patients lose approximately 2% of muscle mass per day during the first week of admission. 2 The rate of muscle loss in patients following major trauma is often higher due to hypermetabolism after injury where energy expenditure can increase to 200% of patients’ usual values.3,4 Trauma patients are vulnerable to becoming malnourished as increased nutrition requirements are often paired with practical challenges in delivery. The consequence of malnutrition in trauma patients is poorly understood. However, malnutrition is an independent risk factor for poor clinical outcomes in non-trauma populations; critically ill patients who are malnourished have increased duration on mechanical ventilation, increased length of stay on the critical care unit, more infection complications and higher risk of mortality when compared to patients who are not malnourished.5 –7
Although various nutrition screening tools have been devised to determine the risk of malnutrition, none have been shown to offer clinically significant prognostic value in critical care settings. Contemporary nutrition screening tools such as the Nutrition Risk in Critically Ill (NUTRIC) score and The Nutritional Risk Screening 2002 (NRS 2002) 8 have integrated the severity of illness into their tool to reflect its role in deteriorating nutrition status, 9 yet neither of these tools are validated for use in critically ill trauma patients. Studies have demonstrated that patients with high NUTRIC scores benefit from early enteral nutrition, with improved outcomes such as reduced infections, complications, and mortality.9,10 However, the predictive value of the NUTRIC score remains a topic of ongoing debate as other studies have found no significant association between feeding strategy, mortality, and NUTRIC scores. 11 Whilst waiting for a validated screening tool to be developed, guidelines recommend a pragmatic approach should be considered for patients at risk such as those staying in the Intensive Care Unit (ICU) for more than 2 days, undergoing mechanical ventilation, infected, underfed >5 days, and/or presenting with a severe chronic disease. 12
Critically ill patients are reported to receive as little as 50% of energy and 45% of protein requirements during enteral nutrition support. 13 Barriers to delivering adequate enteral nutrition often include feeding interruptions for planned procedures and issues with enteral feed tolerance and access. 14 A large observational study 15 of ICU patients showed improved survival associated with energy delivery of ⩾80% of the prescribed target. Since this study, ‘optimal’ nutrition delivery (defined as ⩾80% delivery of target) in mixed critical care populations has also been shown to reduce infectious complications, 13 days on the ventilator, 16 risk of mortality 16 and both critical care and hospital length of stay (LOS). 17 However, despite the common belief regarding the importance of nutrition following major trauma the evidence remains limited.
Therefore, the aims of this study were to: (1) describe nutrition risk and nutrition practices in patients admitted to critical care following major trauma, (2) evaluate the factors that influence enteral nutrition delivery in patients admitted to critical care following major trauma and (3) explore the relationships between enteral nutrition provision and clinical outcomes in patients admitted to critical care following major trauma.
Methodology
An exploration of nutrition related data collected as part of the Multiple Organ Dysfunction in Elderly Trauma (MODET) study 18 was undertaken. Ethical approval was granted by the Health Research Authority, London and South East Research Ethics Committee (IRAS: 209230).
MODET was a Pan London Trauma System prospective observational study which examined outcomes for trauma patients (aged 65 years or older) admitted to a critical care unit, compared to younger patient controls (aged 16 years or older). The study was carried out between 2017 and 2018 across the four major trauma centres (Level 1 equivalent hospitals) within a large regional trauma system. Consecutive patients admitted to the four critical care units were enrolled and reviewed daily until discharge or death. Critical care was defined as an ICU (level 3 beds) or a combined unit of intensive care and high dependency (level 3 and 2 beds). 18
Data were collected prospectively which included demographics, number and type of comorbidities (defined in line with the NUTRIC Score 9 ), mechanism of injury, injury type and severity, disease severity scores, organ dysfunction, days on organ support, ventilator requirements, nutrition, length of stay and mortality. Critical care length of stay was defined as day of admission to the day of discharge to ward level care and mortality was recorded on the day of death during the hospital admission (whether on the critical care unit or on the hospital ward). Traumatic Brain Injury (TBI) was defined in those with a head and neck abbreviated injury score (AIS) of greater than three. TBI without other significant injuries was defined as a head and neck AIS 4–6 and AIS < 2 in any other body region.
Nutrition related data included anthropometry (weight, height, body mass index (BMI)), nutrition risk screening on admission using the modified NUTRIC (mNUTRIC) score, 19 feeding route, enteral nutrition products prescribed, volume of enteral nutrition prescribed, volume of enteral nutrition delivered and causes of enteral feeding interruptions. Causes of enteral feeding interruptions included planned procedures, issues relating to the feeding tube and poor feeding tolerance such as vomiting and high gastric residual values (GRVs), the definition of a high GRVs was different for each site ranging from 250 to 500 ml). Height (actual or estimated) and weight at admission (estimated or actual weight) were used to calculate BMI.
Daily enteral nutrition targets (energy and protein) were calculated using the total volume of the specified enteral feed prescribed. Enteral nutrition delivery was recorded daily as the total volume received; causes of enteral feed interruptions were recorded. Enteral nutrition deficits were calculated using the difference between target volume and actual volume delivered, then calculated into energy and protein deficits based on the enteral feed prescribed. We defined ‘optimal’ enteral nutrition delivery as receiving ⩾80% of the prescribed energy target based on a large-scale observational study of ICU patients that shows improved survival associated with increased intake ⩾80% of the energy prescription. 15
Analysis of weekly enteral nutrition delivery was limited to patients who received enteral nutrition for the full duration of each respective week. Similar to previous analyses of International Nutrition Survey (INS) data, 16 no adjustment was conducted for the amount of enteral nutrition received on admission or discharge days shorter than 24 h. To enable an exploration of patient characteristics and outcomes, enteral nutrition delivery was categorised into groups (kcal/kg of energy and g/kg of protein, delivered). This method was used to allow analysis of the actual enteral nutrition received by patients, including contributions from non-nutrition sources (such as propofol and glucose infusions) rather than prescribed targets which were variable. As the majority of patients fell within the middle intake group (10–20 kcal/kg or 0.5–1.0 g/kg of protein), this group was subdivided to show the distribution of patients within this category (10–15 kcal/kg and 15–20 kcal/kg of energy and 0.5–0.75 g/kg and 0.75–1.0 g/kg of protein). This allowed for a clearer comparison of demographics and clinical outcomes between groups, revealing important differences that may have otherwise been overlooked.
Statistical analyses were conducted using IBM SPSS Statistics for Windows, version 29.0.1.0 (IBM Corp., Armonk, N.Y., USA). Categorical data are presented as counts and percentages, and continuous data are reported as mean (standard deviation) or median [interquartile range (IQR)] according to distribution. Continuous data were compared with Wilcoxon signed-rank and Kruskall Wallis tests. Categorical variables were analysed using Chi-squared tests; a p-value of <0.05 was considered significant. Univariate and multivariate logistic regression analysis were used to evaluate the strongest associations with suboptimal enteral nutrition delivery (defined as delivery of <80% of the prescribed energy target). For exploratory purposes, the multivariate model included demographic and clinical variables which are available to clinicians within the first 48 h of admission which may be predictive of the risk of underfeeding. These variables included: age, sex, co-morbidities, BMI, injury type (blunt/penetrative), ISS, presence of TBI, Acute Physiology and Chronic Health Evaluation (APACHE) II score, SOFA score, mNUTRIC score and if enteral nutrition was started early (within 48 h of admission). Variables significant at p < 0.10 level in univariate analysis were entered into the multivariate model. Co-linear variables were omitted from the model. Results are presented as odds ratios (OR) with 95% confidence interval (CI). Hosmer Lemeshow test was used to assess the model fit and area under the curve (AUC) analysis for model discrimination.
Results
Demographics
Of the 1316 patients who were enrolled into the MODET study, 276 were discharged, moved to palliative care or died within 72 h of admission, leaving 1036 for nutrition analysis. The majority of patients were male (n = 763, 73.6%) and the mean age was 53 years (21.4). The median weight and BMI were 75.0 kg [68.0–85.0] and 24.7 kg/m2 [22.8–27.8], however the majority of patients’ weights were estimated (n = 995, 96.0%). On admission 804 (77.6%) patients were considered to have a low malnutrition risk when screened using the mNUTRIC risk assessment tool. The median injury severity score (ISS) was 25 [17.0–34.0] and 564 (54.5%) patients had a TBI. The daily participant disposition is shown in Figure 1.
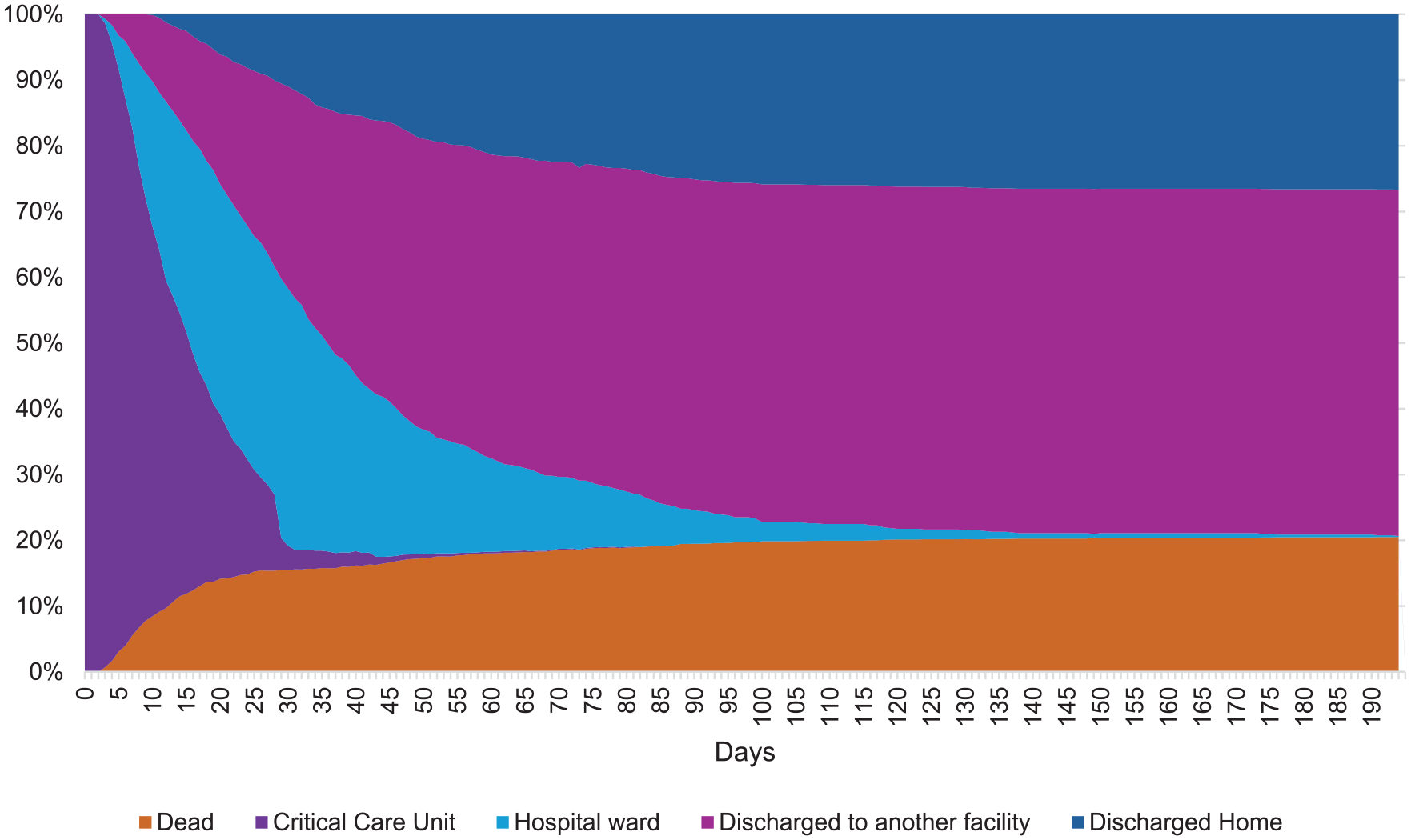
Daily participant disposition.
Nutrition support
Following major trauma, three-quarters of patients admitted to critical care (n = 768, 74.1%) required artificial nutrition support: enteral nutrition or parenteral nutrition. The majority of these (n = 732, 70.6%) required enteral nutrition via feeding tubes; 27 patients (2.6%) required a combination of enteral nutrition and parenteral nutrition and nine patients (<1%) required exclusive parenteral nutrition (Table 1). Patients who did not require artificial nutrition support were able to eat and drink (n = 238, 23.0%) or remained nil by mouth (NBM; n = 30, 2.9%). Given that the majority of those who required artificial nutrition support received enteral nutrition, the analysis then focussed on this group of patients. Early enteral nutrition (within 48 h of admission) was started in 649 (88.7%) patients and the mean days of enteral nutrition provision was 15.7 days (7.9). Polymeric enteral feed was used in 726 (99.2%) patients whilst only six patients (<1%) required a peptide enteral feed. Fibre containing enteral feed was used in 269 patients (36.7%; n = 269) whist 463 (63.3%) received a fibre-free enteral feed.
Patient characteristics and clinical outcomes by nutrition route.
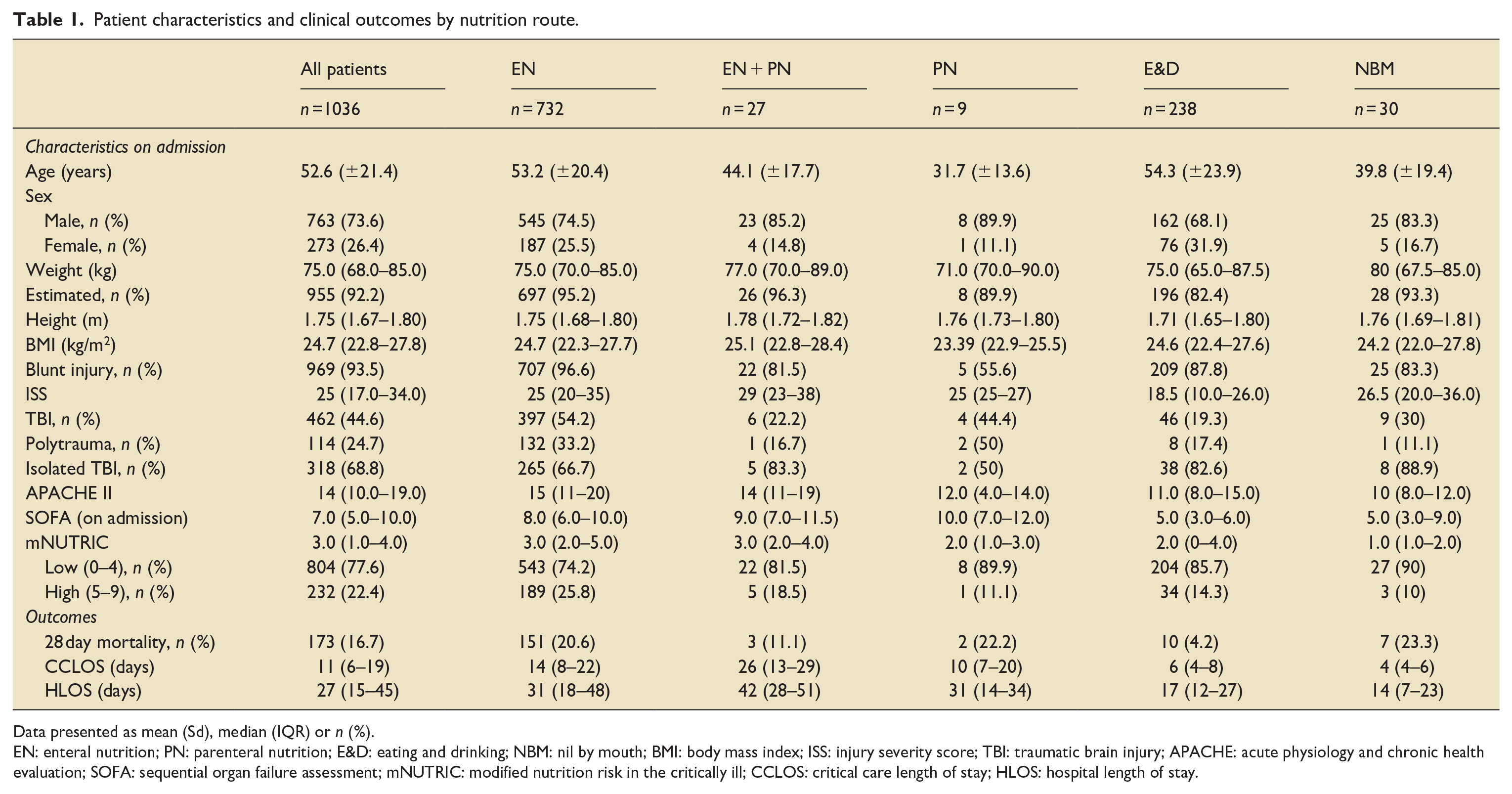
Data presented as mean (Sd), median (IQR) or n (%).
EN: enteral nutrition; PN: parenteral nutrition; E&D: eating and drinking; NBM: nil by mouth; BMI: body mass index; ISS: injury severity score; TBI: traumatic brain injury; APACHE: acute physiology and chronic health evaluation; SOFA: sequential organ failure assessment; mNUTRIC: modified nutrition risk in the critically ill; CCLOS: critical care length of stay; HLOS: hospital length of stay.
Nutrition delivery
The mean volume of enteral nutrition prescribed equated to 23.8 kcal/kg/day (6.37) and 1.27 g/kg/day (0.34) of protein per day. A significant difference was seen between the amount of energy and protein prescribed and received during each week of their critical care admission (p < 0.001; Figure 2(a) and (b)). The mean daily energy, and protein deficit throughout admission was 550 (354.3) kcal and 27 (17) g protein respectively. Of all patients on enteral nutrition, most (n = 685, 93.6%) received additional energy from propofol and/or glucose, which provided a mean of 189 (138) kcal/kg or 2.4 (1.8) kcal/kg of added energy per day. The mean delivery of energy including non-nutrition sources was 17.27 (9.3) kcal/day and 0.89 (0.5) g/kg of protein per day.
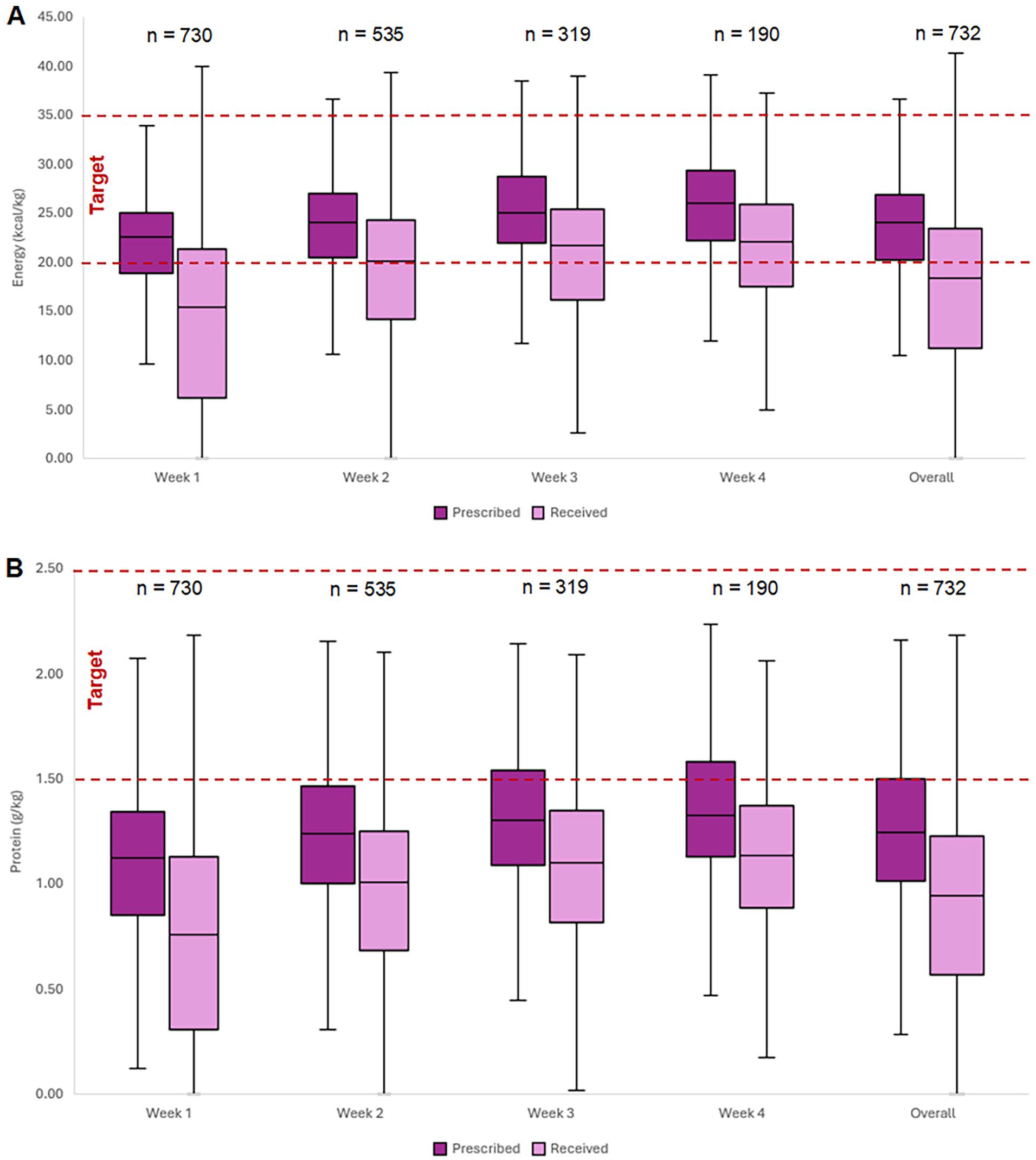
Energy and protein prescription and delivery per week and overall throughout admission. (A) Energy prescription and delivery per week and overall throughout admission and (B) Protein prescription and delivery per week and overall throughout admission.
Factors that influence nutrition delivery
Suboptimal enteral nutrition delivery was defined as the delivery of <80% of the prescribed energy target. Factors which were strongly associated with suboptimal enteral nutrition delivery included being male (OR, 1.82, 95% CI 1.27–2.60; p < 0.001), having a TBI (OR, 1.67, 95% CI 1.16–2.40; p = 0.006) or a high mNUTRIC score (OR, 1.17, 95% CI 1.08–1.27; p < 0.001; Table 2). Single system TBI was not associated with sub-optimal enteral nutrition delivery in this cohort (Supplemental Table 1). Patients who received early enteral nutrition were less likely to be underfed (OR, 0.49, 95% CI 0.30–0.81; p = 0.006).
Factors associated with risk of sub-optimal nutrition delivery.
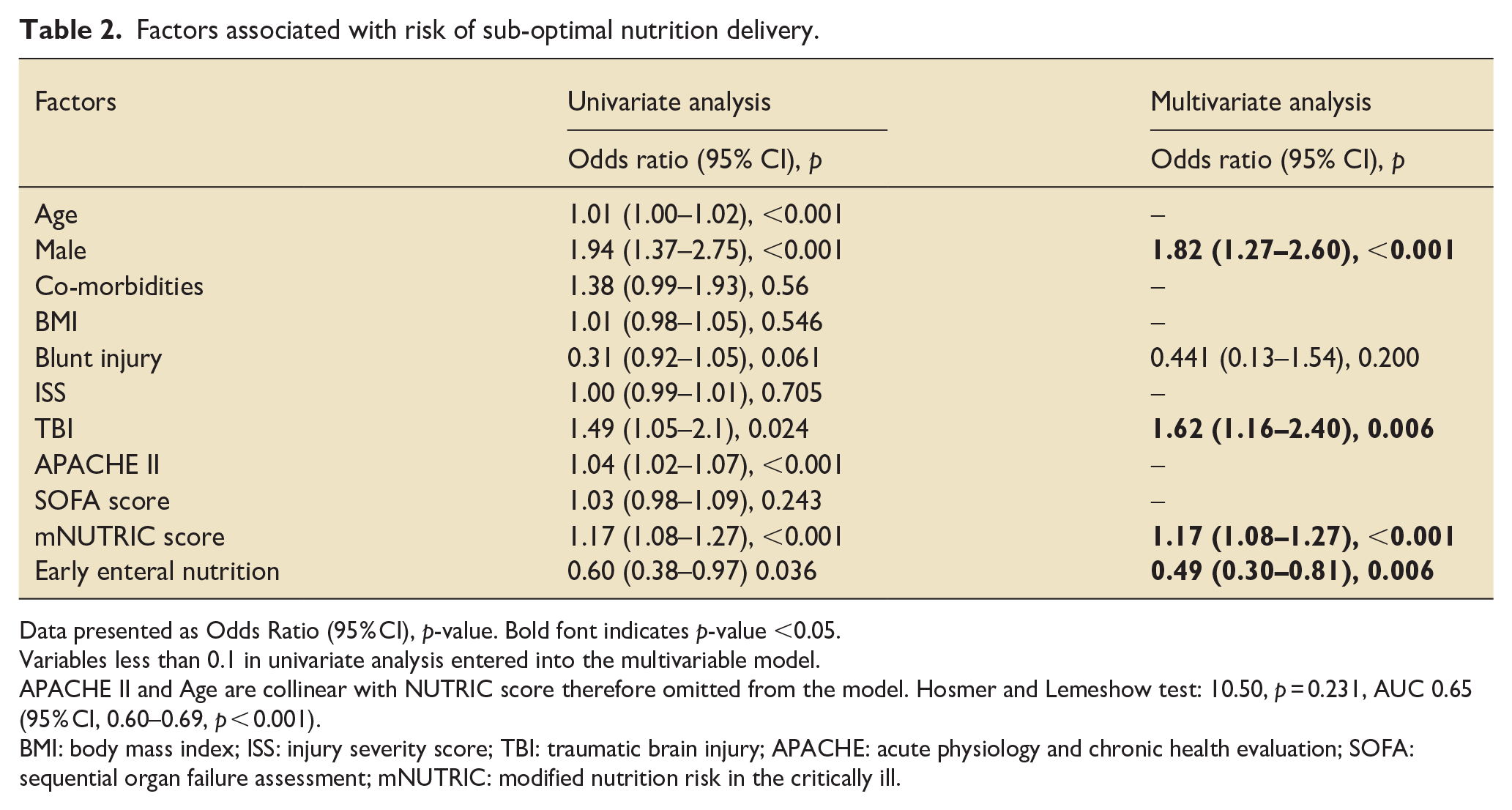
Data presented as Odds Ratio (95% CI), p-value. Bold font indicates p-value <0.05.
Variables less than 0.1 in univariate analysis entered into the multivariable model.
APACHE II and Age are collinear with NUTRIC score therefore omitted from the model. Hosmer and Lemeshow test: 10.50, p = 0.231, AUC 0.65 (95% CI, 0.60–0.69, p < 0.001).
BMI: body mass index; ISS: injury severity score; TBI: traumatic brain injury; APACHE: acute physiology and chronic health evaluation; SOFA: sequential organ failure assessment; mNUTRIC: modified nutrition risk in the critically ill.
During the study period there was 6328 episodes of enteral feeding interruption, with almost half (n = 3081, 48.6%) occurring during the first week of admission. The median length of stay on critical care was 14 days [8.0–22.0] and enteral feeding interruptions occurred on a median of 6 days [4.0–9.0]. A total of 173 patients (23.6%) had enteral feed interruptions on 10 or more days of their admission. The most common reason for enteral feeding interruptions was fasting for specific events. The most frequently observed events included periods pre and post extubation (n = 780, 41.7%), surgical intervention (n = 504, 26.9%) and scans (n = 307, 16.4%). During weeks 1 and 2 fasting related to extubation was he most common reason. During weeks 3 and 4, displacement of enteral feeding tubes was the most common reason, followed by fasting for extubation (Figure 3).
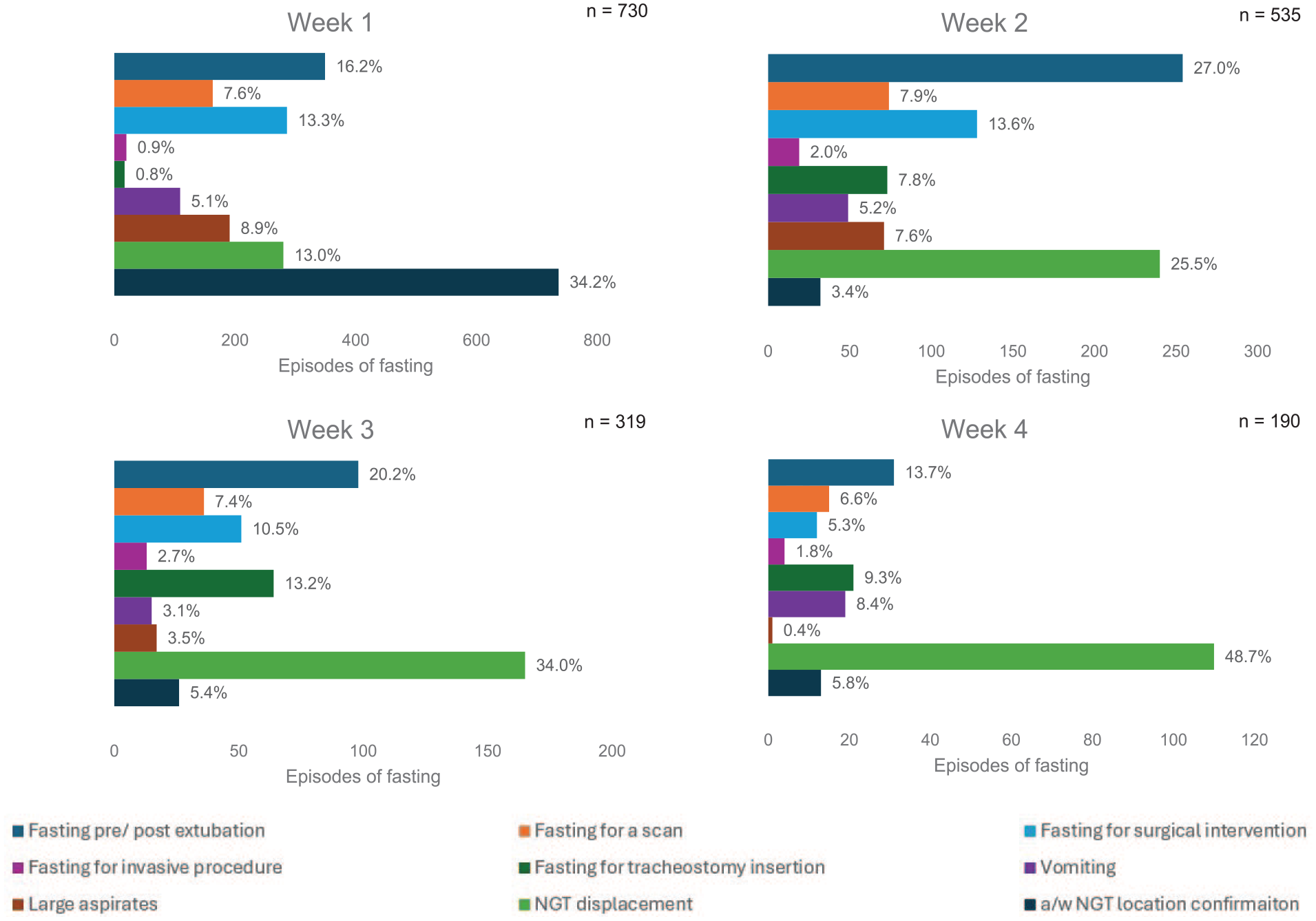
Bar charts showing frequency and cause of feeding interruptions by week of admission to the critical care unit.
A total of 264 patients (36.0%) experienced enteral feeding intolerance. There were 481 episodes of enteral feeding intolerance recorded during the study period with the majority occurring during the first week of admission. A total of 213 (29.1%) patients were prescribed prokinetics for a median duration of 6.0 days [4.0–12.0] for enteral feeding intolerance. Of those patients who received prokinetics to manage enteral feeding intolerance 140 (65.7%) had a TBI.
Relationships between enteral nutrition delivery and clinical outcomes
Lower mean delivery of both energy and protein was associated with older age (p < 0.001), higher AAPACHE II scores (p < 0.001) and higher mNUTRIC scores (p < 0.001), with few differences in injury characteristics (Table 3). Outcomes were worse for those with lower energy and protein delivery, with more ventilator days (p < 0.001), and longer lengths of critical care (p < 0.001) and hospital stays (p < 0.001; Table 3).
Characteristics and outcomes of patients in energy and protein delivery groups.
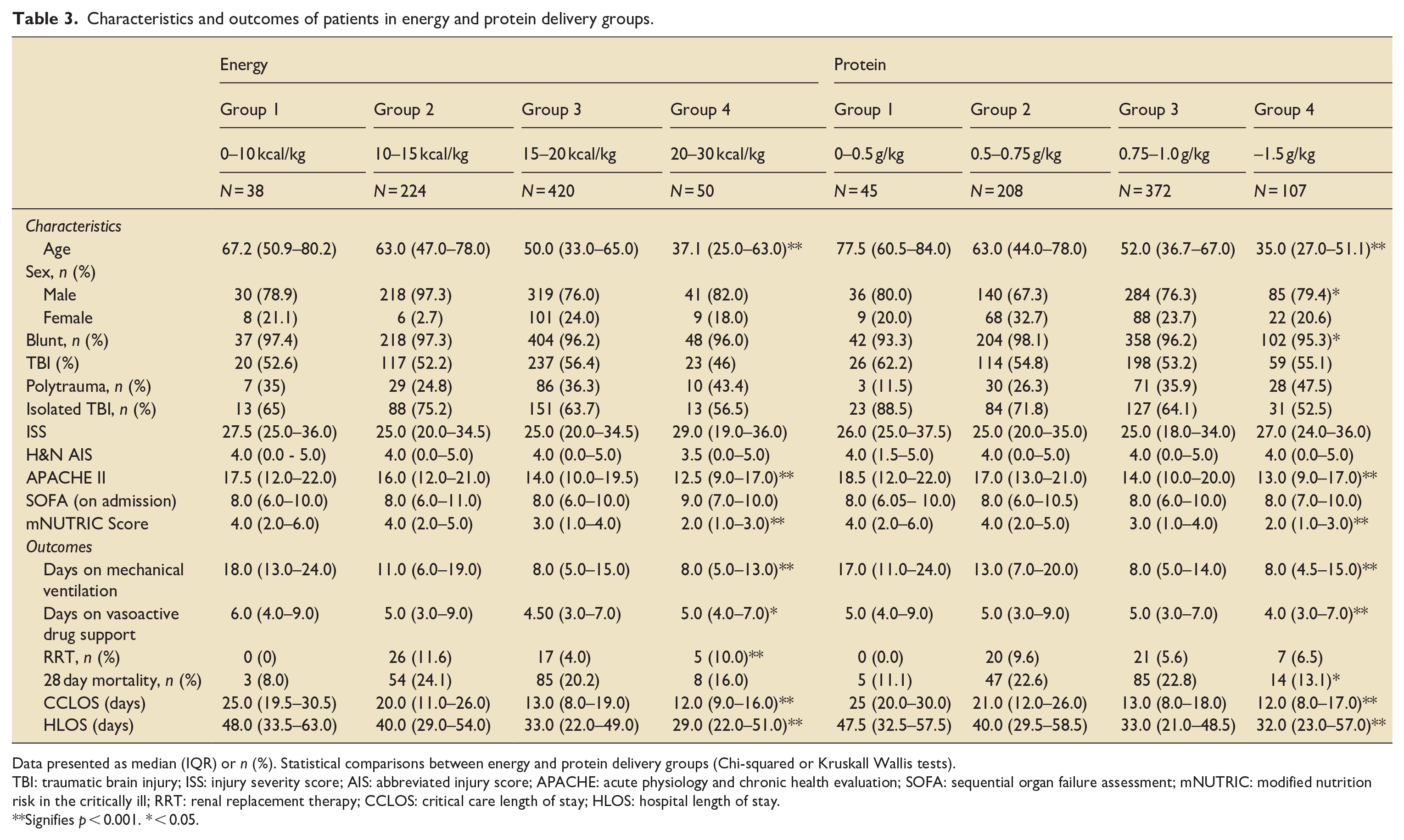
Data presented as median (IQR) or n (%). Statistical comparisons between energy and protein delivery groups (Chi-squared or Kruskall Wallis tests).
TBI: traumatic brain injury; ISS: injury severity score; AIS: abbreviated injury score; APACHE: acute physiology and chronic health evaluation; SOFA: sequential organ failure assessment; mNUTRIC: modified nutrition risk in the critically ill; RRT: renal replacement therapy; CCLOS: critical care length of stay; HLOS: hospital length of stay.
Signifies p < 0.001. * < 0.05.
Discussion
This study has investigated enteral nutrition support practices and explored factors that influenced enteral nutrition delivery and the impact on outcomes in patients admitted to critical care following major trauma. As far as we are aware is the first of its kind in the UK trauma population. Overall, the results indicate that patients admitted to critical care following major trauma do not receive ‘optimal’ enteral nutrition (⩾80% of the prescribed energy target). Male patients who present with a TBI or a high mNUTRIC score were more likely to be sub optimally fed. The main observed causes of suboptimal delivery were related to fasting and issues with the enteral feeding tubes. Patients with the lowest energy and protein delivery experienced more days on the ventilator with longer critical care and hospital length of stays, although this study design precludes us inferring a causal relationship. However, these results provide the impetus to improve enteral nutrition practices and potentially improve outcomes in critically ill trauma patients.
This study highlighted several features in nutrition support practices for critically ill trauma patients. Consistent with nutrition support guidelines,12,20 the enteral route was favoured over the parenteral route, with early initiation of enteral nutrition (<48 h) observed in most cases. Supporting this approach, a recent study 21 found that early enteral nutrition in trauma patients with moderate to severe isolated TBI significantly reduced both the number of days on the ventilator and the length of stay in critical care, reaffirming the benefits of timely enteral feeding. In terms of enteral nutrition formulation, the majority of patients received polymeric formula. Almost two-thirds of patients received fibre-free enteral feed, yet studies exploring the effects of dietary fibre on enteral feeding tolerance and clinical outcomes have demonstrated promising results with improved feeding tolerance 22 and reductions in both critical care and hospital length of stay in patients who received fibre containing enteral nutrition. 23
Interpreting the nutrition status was challenging due to the estimation of weights in 92% of cases. Although plausible, the average BMI of 24.7 kg/m² may therefore not be truly reflective of the population. A simple visual estimate of patients’ body weight has been shown to be inaccurate in critical care with an error of as much as 20% against measured values, 24 therefore it was not possible to truly assess patients anthropometry. Similar to the findings of Iwuchukwu et al., 25 a substantial proportion (n = 804, 78%) of participants had low mNUTRIC scores ranging from 0 to 4, indicating a low initial nutrition risk score. However the usefulness of mNUTRIC in trauma patients is unclear; while it identifies malnutrition risk, it has not been found effective to guide early enteral nutrition initiation or predict mortality in this patient group. 26
Determining optimal energy and protein targets for trauma patients remains a practical challenge, especially when relying on predictive equations. It is important to acknowledge that indirect calorimetry (IC) is recommended as the gold standard to measure energy expenditure, however access is often limited. 27 This method requires specialist equipment, training and adequate staffing to carry out the assessment therefore its use in clinical practice is limited by technical and financial issues. 28 In the absence of IC, weight-based or predictive equations are often used.
While guidelines provide general recommendations,12,20 the specific nutritional requirements for individual patients may vary significantly based on various factors, including injury type and severity, metabolic demands, and underlying medical conditions. Trauma specific guidelines are limited and the quality of evidence which supports these guidelines is considered low. 29 However, these guidelines suggest energy targets ranging from 20 to 35 kcal/kg and protein targets ranging from 1.5 to 2.5 g/kg for major trauma patients. 29 In examining the nutrition practices observed in this study, a notable discrepancy between the protein targets set and the recommendations outlined in prevailing trauma guidelines was observed. It is unclear why this occurred, however many studies have revealed that clinicians tend to underestimate requirements due to concerns with overfeeding.25,28 This requires further investigation.
On average, patients received 73% of their estimated energy and 70% of their protein requirements, however these targets were not aligned with the recommended guidelines for trauma patients. 29 When compared to what the guidelines recommend, 29 the mean delivery equates to 69% of estimated energy and 59% of protein requirements, with some patients potentially needing more than this due to having a TBI, open abdomen or large wounds.12,20 Similar findings are reported by Chapple et al., 30 with delivery of 58% of energy and 53% of protein in critically ill head-injured patients globally, which is lower than other studies showing delivery rates between 67% and 76%.31,32 It is important to note that changes in clinical guidelines, enteral nutrition products and dietetic staffing may have occurred since data collection began in 2017. However, trauma-specific nutrition support guidelines have remained unchanged since 2016. 29
There are ongoing challenges in achieving optimal enteral nutrition delivery in critically ill trauma patients. Whilst enteral feeding interruptions are mostly considered unavoidable, exploration and minimisation of fasting times should be considered. It is well recognised that pre-procedural fasting is a significant barrier to adequate enteral feed delivery in critically ill patients. 28 The presence of fasting guidelines has been shown to be beneficial in reducing overall fasting times, 33 however it is imperative for guidance to remain dynamic and evolve with emerging evidence. The need for guideline adaptation is highlighted by Landais et al., 34 who compared different practice of fasting before extubation in over 7000 patients in a randomised clinical trial. The study concluded that maintaining enteral nutrition until extubation in critically ill patients was non-inferior to a traditional fasting approach where enteral feed was held for 6 h beforehand, suggesting a change in practice might be considered safe. In addition to pre-procedure fasting, our study documented 264 patients (36.0%) experienced enteral feeding intolerance. Of the 213 patients who received prokinetics to manage enteral feeding intolerance, 140 (65.7%) had a TBI, this is not surprising as it is well recognised that gastric dysmotility frequently occurs following a TBI.35,36
Given the observational nature of this study, we cannot make definitive causal inferences from our findings and these results need to be cautiously interpreted as there are several likely confounding variables that were not measured or for which we were unable to adjust. The data however suggest that there may be an association between the mean delivery of both energy and protein (kcal/kg and g/kg) and days of mechanical ventilation, length of stay on critical are and total hospital length of stay. Whilst injury characteristics did not differ, those in lower enteral nutrition delivery groups had higher APACHE II scores suggesting, perhaps not surprisingly, that those who were more unwell may be more difficult to feed enterally. Further exploration is required to assess whether there is a direct link between enteral nutrition delivery and outcomes; it is possible that poorer outcomes were related to the severity of illness rather than enteral nutrition delivery.
The optimal amount of energy and protein required by critically ill patients is debated. Observational studies15,16 suggest improved survival associated with enteral nutrition delivery of ⩾80% of the prescribed energy target. However more recent randomised controlled trials indicate no benefit or even potential harm from meeting estimated energy targets. The PermiT 11 and TARGET 37 trials both demonstrated that lower energy delivery, including permissive underfeeding, resulted in similar outcomes compared to standard or high calorie feeding. Whilst trials such as EDEN 38 and NUTRIREA-3 39 found that early energy and protein restriction within the early acute phase reduced feeding intolerance and reduced critical care and ventilator dependency, without compromising functional and physical function at 6 and 12 months.
Similarly, the findings from the EFFORT protein trial 40 and the PRECISe trial 41 recently concluded that higher protein intake may not be as beneficial as once thought. It is important to note however that the trauma population within both trials was a relatively small subset of 10%. Nevertheless, we cannot currently exclude the possibility that a greater protein intake is associated with either benefit or harm. For our study, we defined ‘optimal’ enteral nutrition delivery as ⩾80% of the prescribed energy target reflecting the evidence base available at the time of data collection. However, evidence38,39 suggests that restricting energy and protein as low as 6 kcal/kg/day and 0.3–0.4 g/kg/day, respectively, during the acute phase of critical illness could enhance recovery and decrease morbidity.
This underscores the complexity of nutrition management in critically ill trauma patients and the need for further research to explore optimal energy and protein dose for each metabolic phase. Further investigation is also warranted to explore whether the relationships between enteral nutrition delivery and outcomes are true or explained by unadjusted factors such as injuries or severity of illness. These findings will be crucial for the planning of future trials. Regardless of contemporaneous best practice recommendations, the setting of lower than recommended targets, and then failing to achieve them, represent important human factors questions.
Strengths
This study stands as the sole multi-centre prospective observational investigation evaluating enteral nutrition delivery among critically ill major trauma patients in the UK. Its multicentre design may increase the generalisability of the findings, as previous studies in this field have often been limited by single-centre settings, small sample sizes or focussing on specific injuries such as TBI.
Limitations
The first limitation is that patients enteral nutrition targets were determined through the volume and feed prescribed rather than the dietitian’s primary calculation of nutritional requirements. Additionally, we were not able to adjust for the amount of enteral nutrition received on admission or discharge days shorter than 24 h, which may have impacted the estimated nutritional deficits. Our study relied on estimated weights and BMI, which could introduce inaccuracies into our findings. For information governance reasons, we were not able to collect site-specific barriers and facilitators to enteral nutrition delivery, and as the MODET study was conducted between 2017 and 2018, practice may have since changed. Lastly, this dataset did not capture longer term outcomes potentially influenced by enteral nutrition delivery, such as repeat admissions to hospital, functional status and quality of life. Future studies should incorporate such measures to provide a more comprehensive understanding of nutrition’s role in recovery following major trauma.
Conclusion
In conclusion, this study highlights that patients admitted to critical care following major trauma may not receive optimal enteral nutrition (defined in this study as ⩾80% of recommended targets). The delivery of energy and protein is considerably less than recommended, leading to potentially significant nutrition deficits. Patients with the lowest energy and protein delivery were more unwell and experienced increased number of days on the ventilator and longer critical care and hospital length of stays.
Moving forward, further investigation is warranted to explore the optimal dose of energy and protein and its direct association with clinical outcomes. Additionally, it is imperative to delve deeper into the barriers and facilitators to optimal enteral nutrition delivery in this population. This knowledge will be pivotal in the development of effective strategies to enhance enteral nutrition delivery and ultimately improve outcomes for critically ill trauma patients.
Supplemental Material
sj-docx-1-inc-10.1177_17511437251349679 – Supplemental material for Enteral nutrition delivery in patients admitted to a critical care unit following major trauma: Who’s at risk and what’s the impact?
Supplemental material, sj-docx-1-inc-10.1177_17511437251349679 for Enteral nutrition delivery in patients admitted to a critical care unit following major trauma: Who’s at risk and what’s the impact? by Carys Davies, Lina Johansson, Stephen J Brett, Elaine Cole and The MODET Investigators in Journal of the Intensive Care Society
Footnotes
Acknowledgements
The MODET investigators:
Professor Karim Brohi, Centre for Trauma Sciences, Blizard Institute, Queen Mary University of London
Mr Robert Christie, Centre for Trauma Sciences, Blizard Institute, Queen Mary University of London
Ms Bebhinn Dillane, Centre for Trauma Sciences, Blizard Institute, Queen Mary University of London
Ms Helen Farrah, St Georges University Hospital NHS Trust, London
Ms Chris Ryan, St Georges University Hospital NHS Trust, London
Mr Chris Aylwin, Imperial College Healthcare NHS Trust, London
Dr Adam Woodgate, Barts Health NHS Trust, London
Dr Philip Hopkins, King’s College Hospital NHS Foundation Trust, London
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Imperial Health Charity and the NIHR Imperial Biomedical Research Centre. The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care. The original study was funded by the Dunhill Medical Trust (R460/0216).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
