Abstract
Background:
Spinal cord injury (SCI) is a devastating condition with a high burden of morbidity and mortality. National guidelines state that patients should not receive enteral feeding for 48 hours after inury, which may be detrimental if a patient experiences a hypercatabolic response to polytrauma. We conducted a systematic review of the benefits and harms of delayed enteral feeding in this population.
Methods:
We searched MEDLINE, EMBASE and Cochrane CENTRAL for studies which had a time parameter as part of their evaluation of feeding in the acute phase of spinal cord injury in a critical care setting. Required outcomes for inclusion were neurological improvement, neurological complications, time spent in an ICU, time to ICU discharge, incidence of secondary complications, other adverse effects and mortality. Risk of bias was assessed with the Downs and Black checklist.
Results:
Four studies met the inclusion criteria. There was no high-quality evidence of worsened outcomes with earlier feeding compared to delayed enteral feeding. One study demonstrated that patients fed before 24 h in conjunction with a broader bundle of care had improved neurological outcomes compared to previous non-standardised practice. There was no evidence of difference in frequency of infections or mortality in early or late feeding groups.
Conclusions:
We find no clear evidence of increased risk of harm from earlier enteral feeding strategies, nor clear evidence of benefit of earlier feeding as an isolated intervention.
Introduction
Spinal cord injury (SCI) is a cause of life changing disability, with devastating consequences to patients and relatives alike. The annual incidence of SCI in the UK is estimated to be 19 cases per million, 1 with an estimated mortality of up to 22.2%. 2
Patients frequently need nutritional support in the acute phase of spinal cord injury. However, the possibility of ileus and/ or gastroparesis in the early stages post-injury may mean that delaying nutrition in SCI patients is potentially safer than administering enteral nutrition early on in a patient’s admission.3,4 This viewpoint is affirmed by the recommendation of the Multidisciplinary Association for Spinal Cord Injury Professionals (MASCIP), who recommend that the patient receives ‘nil enterally for the first 48 h if the individual is at risk of paralytic ileus’. 5
Such an approach seems counterintuitive. This would not be the case in polytrauma cases without SCI due to the need to combat the acute hypercatabolic response, 6 to avoid well documented sequelae of malnutrition in critically ill patients. 7
The benefit of early enteral nutrition has been established in critically ill patients since the 1990s. 8 A 2009 meta-analysis of non-neurological ICU patients validated these theoretical benefits, finding that, despite mechanistic reasoning about risk of ileus, pneumonia (including that secondary to aspiration) was in fact less likely in patients receiving enteral nutrition. 9
International guidelines do not provide a consensus on when to initiate feeding either. The American Association of Neurological Surgeons identify diet as an important factor in the management of neurogenic bowel. Early and effective neurogenic bowel management is prioritised; however, no timeframe is stated to correlate with ‘early’ unlike the current European Society for Clinical Nutrition and Metabolism (ESPEN) guidelines which identify early as less than 48 h. 10
As such, there is a lack of clarity on optimal timing to introduce nutrition in this patient population, and current guidelines in the UK appear to contradict international guidelines and evidence in similar patient populations. Therefore, we conducted a systematic review of the available published literature on feeding in the acute phase of SCI in critical care units to provide clarity on the benefits and risks of early versus delayed enteral feeding in this patient population.
Methods
This review follows the guidelines set out in the PRISMA statement. 11 The protocol was registered a priori on PROSPERO (CRD42023389016).
Eligibility criteria
Participants had to have radiologically or clinically diagnosed acute spinal cord injury who were admitted to a critical care/ high dependency unit setting in the acute phase of their illness. Chronic spinal cord injury patients or patients in rehabilitation ward settings were excluded. Participants needed to receive enteral feeding with an explicit statement about time or range of times from admission or injury over which feeding was commenced. Studies where time parameters were not stated were excluded. We included studies that measured any clinical outcome including change in neurological status (e.g. motor function, sensory function), length of stay, complications secondary to spinal cord injury, other adverse effects, mortality and quality of life measures. This was for any available timepoints.
Participants/population
Patients admitted to an intensive care unit with an acute spinal cord injury. Acute spinal cord injury can be clinically or radiologically confirmed.
Intervention/exposure
Feeding which is initiated at a timepoint that is specified within studies
Comparator(s)/control
Feeding at a different timepoint or usual practice. Control is not a requirement for inclusion. Lack of a control group will not lead to exclusion.
Outcomes: Clinically relevant endpoints for the ICU clinician, rather than biochemical or metabolic parameters. We defined this as change in neurological status (e.g. motor function, sensory function) neurological complications, length of stay in ICU, length of stay in hospital, complications secondary to spinal cord injury, other adverse effects, mortality and quality of life measures.
There were no study design restrictions, however fully published manuscripts in peer-reviewed journals were required. Abstracts and poster presentations were excluded due to their brief format leading to inability to fully appraise methodology. Language was restricted to publications in English or where a translation of the original publication was available in English.
Information sources
We searched Medline via Ovid, EMBASE via Ovid and the Cochrane CENTRAL trials database on the 12th February 2023 (Appendix 1). For studies that met inclusion after full text review, we also conducted forward and backward citation searching on papers which met full text inclusion criteria using Google Scholar.
Search strategy
Our search strategy is outlined in Appendix 1.
Selection process
Two reviewers (AG and CO) screened all titles and abstracts independently through Rayyan. Disagreements between reviewers were resolved through discussion. The same process was used for full text review of eligible abstracts (See Figure 1).
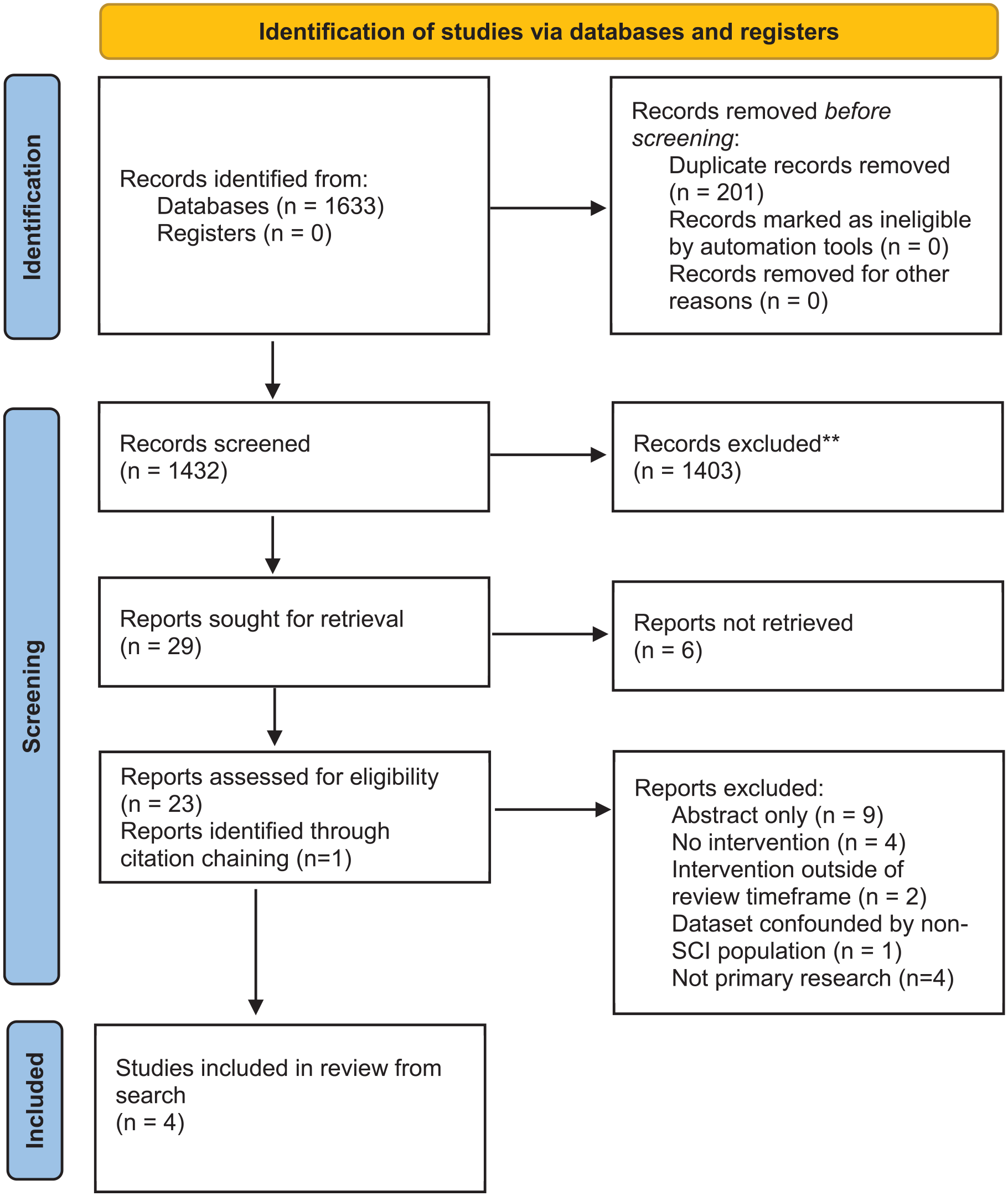
PRISMA flow diagram.
Data collection process
Data extraction was conducted independently by the same reviewers using a pre-prepared Google Form that was piloted on one of the studies to ensure it was able to extract the relevant variables. Differences in extraction were discussed and amended.
Data Items
Participant characteristic data on level of spinal cord injury and modality of ascertaining this (radiological/clinical/both) and number of patients was extracted. We extracted data on nutrition requirement calculation, rate of feed, caloric content and constitution of enteral feed. We also looked at any confounding co-intervention.
Study risk of bias assessment
Individual study risk of bias was assessed using the Downs and Black tool due to the heterogeneity of study design as this allows reasonable comparison between studies. 12
Effect measures
All effect measures are reported as they were in their original papers.
Synthesis methods
Narrative synthesis was undertaken due to the methodological heterogeneity between studies.
Results
Study selection
The study selection process is detailed in Figure 1.
Study characteristics
The four studies included in this review include data from 333 patients (Table 1). The studies were published between 1989 and 2019. One was based in France, 13 two in North America,14,15 and one in New Zealand. 16
Study characteristics.
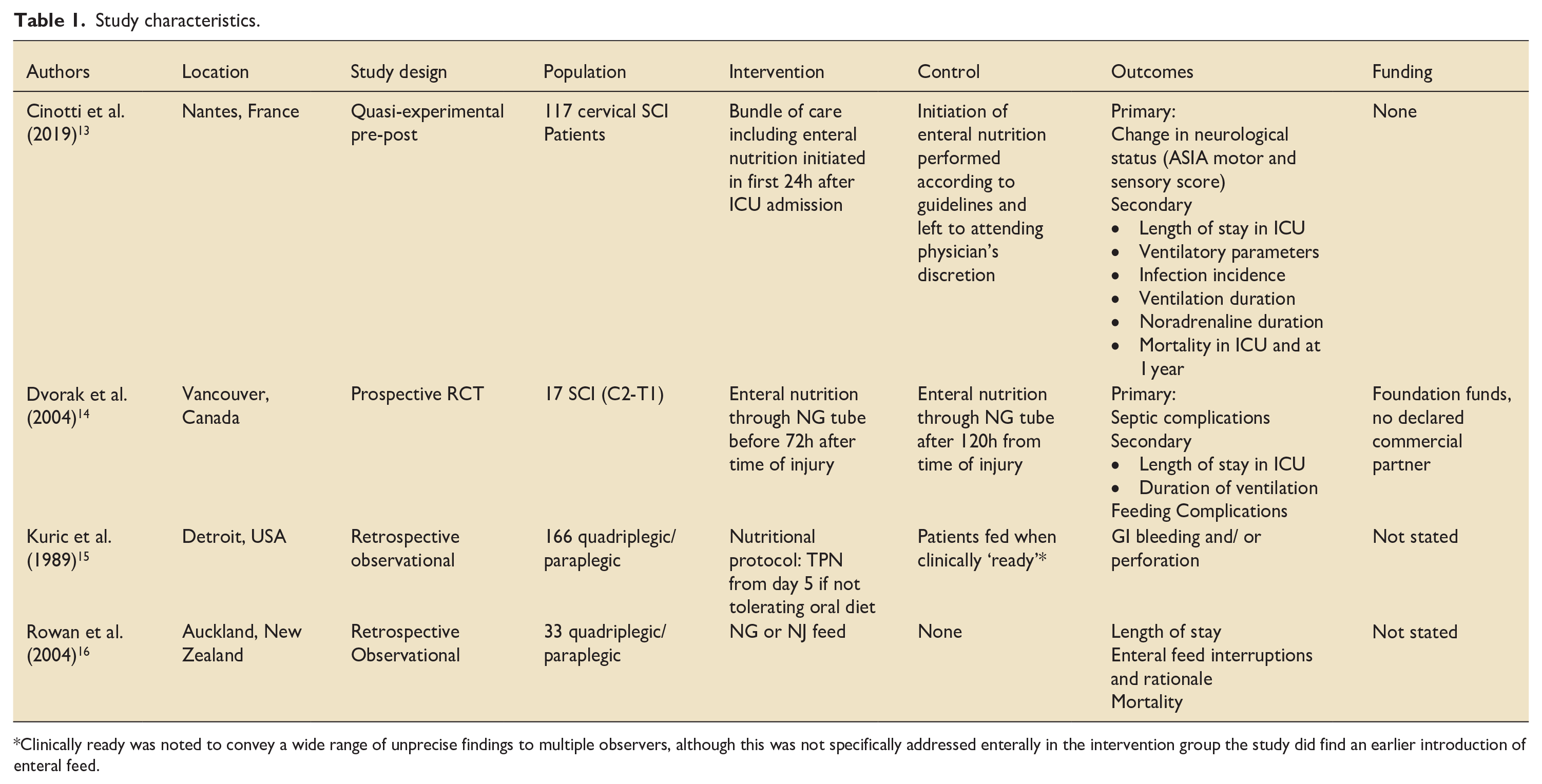
Clinically ready was noted to convey a wide range of unprecise findings to multiple observers, although this was not specifically addressed enterally in the intervention group the study did find an earlier introduction of enteral feed.
Cinotti et al. 13 conducted a pre-post quasi-experimental study evaluating multiple clinical outcomes in SCI patients given a bundle of care (including enteral feeding in the first 24 h) versus routine care. 13 The RCT conducted by Dvorak et al. 14 primarily examined difference in septic complications when patients received enteral nutrition before 72 h post-injury versus after 120 h post-injury. 14 The earliest study, conducted by Kuric et al. 15 in Detroit, USA examined gastrointestinal complications in 166 patients with a nutrition protocol that commenced feeding before 120 h if able versus feeding patients at clinician discretion. 15 The fourth study, conducted by Rowan et al. 16 in New Zealand, was a retrospective study which examined the effects of enteral feeding via nasogastric or nasojejunal feed on length of stay, mortality and enteral feed interruption. 16
Patient and intervention characteristics from individual studies are shown in Table 2.
Patient and intervention characteristics for studies.
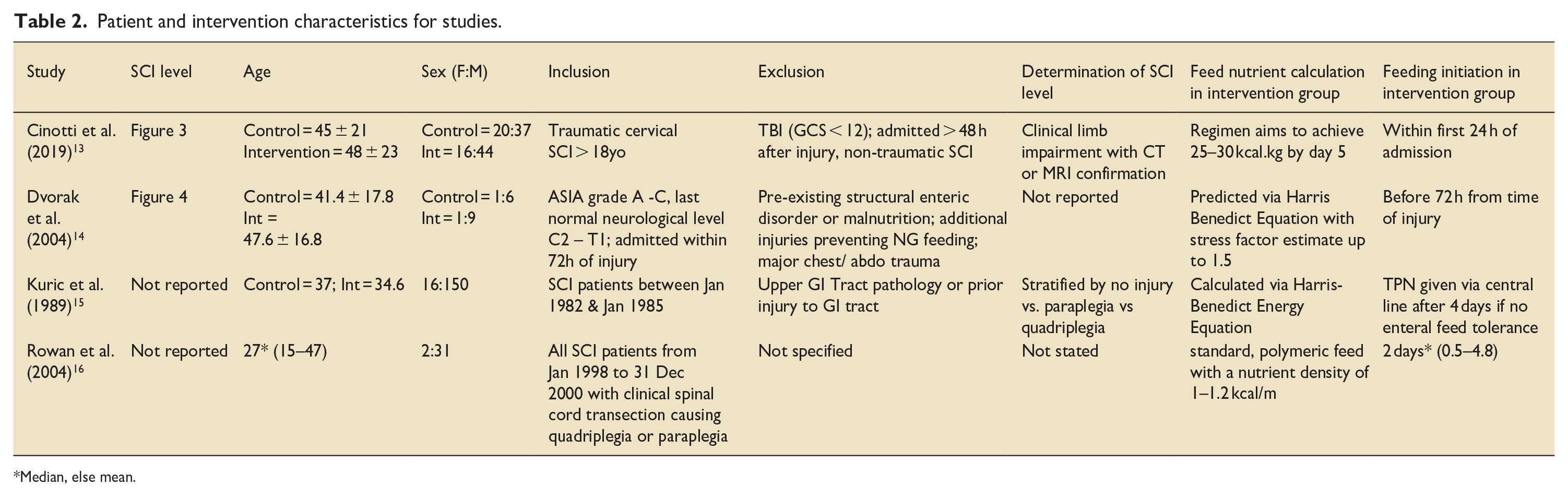
Median, else mean.
Only Cinotti et al. 13 and Dvorak et al. 14 reported the specific injury level of their patients, with all being above the level of T1 (Figures 2 and 3).13,14 The average age of the patients ranged from 27 to 48 years old. About 83% of participants in studies were men. Only the Cinotti et al. 13 reported using imaging to determine SCI level. Half of the studies report using the Haris Benedict Energy equation to calculate feeding requirement. In intervention groups, enteral feeding was started at a maximum of 24 h after admission up to 4.8 days from time of injury; Kuric et al. 15 started total parenteral nutrition at 4 days if enteral feeding was not tolerated by this point.

Bundle of interventions employed in the study by Cinotti et al. (2019). 13
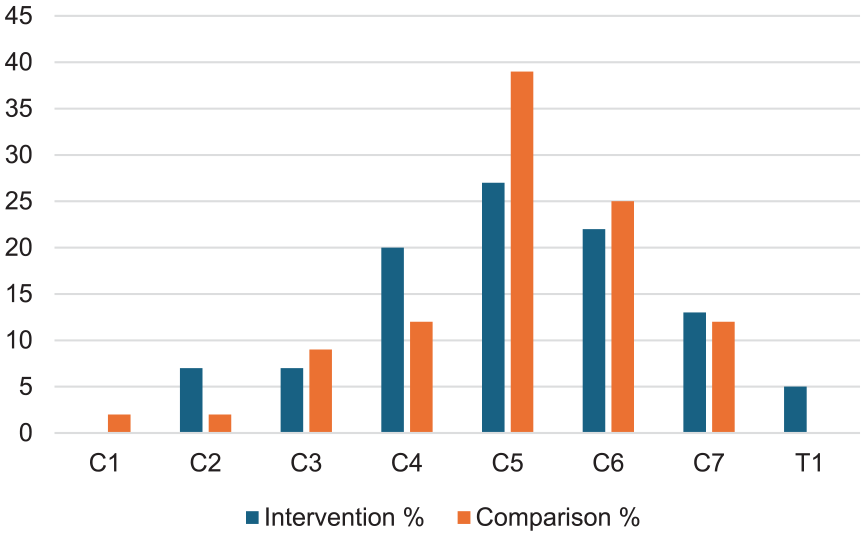
Distribution of SCI level across treatment groups in participants in the trial by Cinotti et al. (2019). 13
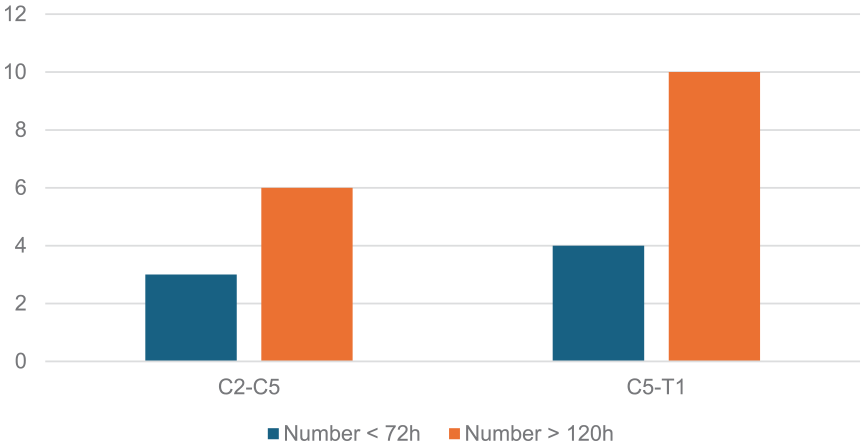
Distribution of SCI level across treatment groups in participants in the trial by Dvorak et al. (2004). 14
Risk of bias
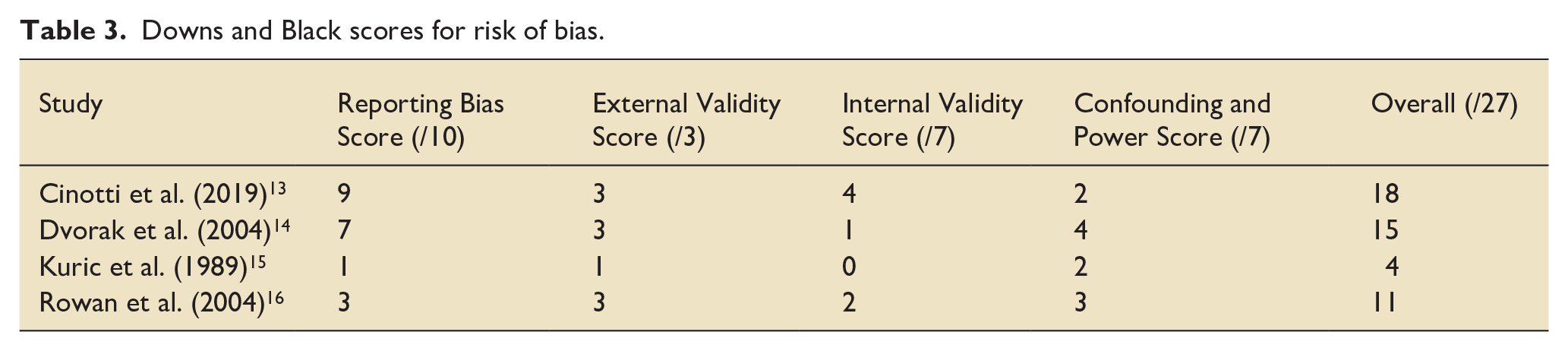
Risk of Bias scoring via Downs and Black is shown in Table 3. Due to being at particularly high risk of bias (4/27), we have excluded the study conducted by Kuric et al. 15 from the narrative synthesis of outcomes.
Downs and Black scores for risk of bias.
Narrative synthesis of results of individual studies
Change in neurological status
Initiating feed in under 24 h was associated with improvements in neurological status when used as part of a broader bundle of care. Cinotti et al.
13
were the only group to report this outcome. Earlier initiation of enteral feeding was associated with a statistically significant improvement in ASIA motor score at discharge from ICU (+16 points vs +6 points;
Length of stay in ICU
Earlier feeding regimens were not clearly associated with changes in length of ICU admission. The single RCT demonstrated that feeding before 72 h post-injury was associated with a 15.1 day longer stay than starting feeding after 120 h; however no statistical testing was conducted. 14 Cinotti et al. 13 found no statistically significant difference between their treatment and control groups. The New Zealand study found that patients who started enteral feeding at an average of 48 h remained for a median of 8.8 days (range 2–36.2 days). 16
Mechanical ventilation
There was no clear indication of differences in length of time mechanically ventilated in association with earlier enteral nutrition. Overall, Cinotti et al.
13
found no significant difference in the length of time patients spent being mechanically ventilated between the control group and those receiving a specific care bundle (median 15 days vs 17 days;
While Dvorak et al. 14 observed that patients who started feeding before 3 days had a longer average mechanical ventilation time (762 h) compared to those after more than 5 days (502 h), no statistical analysis was performed on this difference.
Infection
There is no evidence of a change in infection risk with earlier feeding. Cinotti et al. 13 reported no statistically significant septic complications between groups. Likewise, in the RCT there were equal numbers of septic complications in the group fed early versus the group fed late.
Mortality in ICU and at 1 year
There was no evidence of association between mortality and earlier feeding. There was no difference in mortality between the groups in the Cinotti et al. 13 quasi-experimental study. The mortality rate in ICU in the patient group observed by Rowan et al. 16 was 2/33 (6.1%).
Feeding complications/enteral feed interruptions and rationale
Feeding complications associated with early enteral nutrition were generally low across reporting studies. Dvorak et al. 14 reported 39 complications in the early feeding group and 59 in the late feeding group. Nausea and excessive diarrhoea were more common in the late group, while early feeding saw a higher use of prokinetics due to intolerance.
Rowan et al. 16 also noted low overall feeding complication rates, with an average of 2 days of feeding interruption per patient; this was mostly due to high gastric aspirates (>100–200 ml), vomiting, tube issues and ileus. Most patients experienced fewer than four feed interruptions overall.
Discussion
This systematic review highlights an interplay between timing of enteral feeding and clinical outcomes in SCI patients which is challenging to interpret, with many equivocal results from low quality studies that are all prone to bias.
There is possible benefit in initiating feeding within 24 h to improve neurological recovery, however the study this comes from was confounded by the fact earlier feeding was used as part of a broader bundle of care in that trial.
The impact of earlier feeding on other key outcomes, such as ICU length of stay, duration of mechanical ventilation, infection risk and mortality, is unclear. While delayed feeding in some studies showed a signal indicating shorter ICU stays and reduced ventilation times, the difference was not statistically significant. No difference in infection or mortality was observed between early and delayed feeding. This suggests that early enteral nutrition may be safe.
Feeding complications were generally low, with a possible increase in feed intolerance seen in those fed earlier. However, the 100–200 ml volume used to define feed intolerance in the reporting study is a very conservative volume when one considers that aspirates as high as 500 ml have been shown to not lead to any increased risk of adverse events in general ICU patients. It is also the volume currently recommended by ESPEN guidance to contribute to the multimodal assessment of feed intolerance. 10
Limitations
The evidence included in this review is of low quality. It is challenging to determine true effect size of earlier feeding; only one study recorded objective diagnoses of spinal cord injury level as inclusion criteria. Across the included studies, there was no overt control for comorbid injury status amongst many other confounders. There was no time-series based analysis when measuring most outcomes.
The review process itself may be limited by only using conventional databases for medical research. We did not use Scopus or Web of Science, and excluded non-English publications (although this was limited to a single text after title and abstract screening). Similarly, our decision to exclude conference posters and abstracts may have reduce the amount of data available to us. We are however confident that our review has found appropriate peer-reviewed data due to covering the same papers as have been covered in prior reviews on this topic.17,18
Future practice, policy and research
A finding that was consistent across all three studies in the narrative synthesis was uniformity in approach to feeding regimen, which may not be optimal. Substantial heterogeneity in caloric expenditure was shown by Rowan and Kazemi 19 in a 2020 study with some SCI patients having a difference of almost 1500 kcal in terms of energy expenditure. There is therefore a risk of both under and overfeeding without accurate calculation, both of which would be detrimental to patient outcomes.
There may therefore need to be a change in the titration of caloric intake for these patients, with use of indirect calorimetry as opposed to formulae such as the Harris-Benedict equation. Thus, in future we may need to look at conducting a multicentre study of titration based on indirect calorimetry with Harris-Benedict or current ESPEN guidelines as a control, in conjunction with wider implementation of early feeding regimens. Single specialist centres would not be able to admit enough patients to conduct a trial with enough statistical power, therefore any trial would need to have a multicentre basis.
Conclusion
There is very low-quality evidence that early enteral feeding (initiated less than 72 h into an ICU admission) is as safe as delayed feeding in patients with acute spinal cord injury. There is insufficient evidence to determine that earlier feeding benefits long term neurological outcome. Whilst the metabolic effects of spinal cord injury are known to be different to other traumatic injuries, in the absence of significant evidence of harm, early nutrition is likely to be safe with monitoring for complications.
Footnotes
Appendix 1
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JW is an associate editor for JICS. AG and CO have no conflicts of interest to declare. AG and JW conceptualised the work. All authors conducted the work described; AG and COH wrote the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics
No ethical approval was required for this paper
