Abstract
Viscoelastic tests (VETs) have transformed assessment of haemostasis and transfusion practices in trauma, cardiac and liver transplantation centres. Impaired haemostasis is a common problem on the general intensive care unit (ICU), but routine use of VETs is rare. We have accordingly reviewed the evidence to determine whether there is evidence to support the use of VETs as a standard point of care test on all ICUs in assessing and managing patients. The benefits of using VETs in the management of major haemorrhage, namely faster identification of a coagulopathy, in particular early detection of fibrinolysis, and reduced transfusion requirements for blood products have been seen in the general ICU. Validation of treatments algorithms is now required to standardise practice. There is also emerging evidence to support the use of VETs to guide urgent treatment decisions in patients with a coagulopathy or in patients taking anti-coagulants or anti-platelet therapies. We recommend that departments independently review the feasibility of setting up a viscoelastic point of care service which considers the applicability to their patient cohort, the financial cost and the personnel required.
Introduction
Viscoelastic tests (VETs) represent a global assessment of haemostasis grounded on the cell-based model of coagulation. This dynamic, in vivo process involves both cellular and non-cellular components of whole blood. Accordingly, the initiation of coagulation occurs at the site of trauma, following exposure of whole blood to cells expressing tissue factor. Signal amplification follows through platelet activation and thrombin generation. Propagation through large scale thrombin generation ensues, which promotes the stabilisation of a fibrin-enmeshed, platelet plug – or clot – at the site of tissue injury. This model better reflects the physiology of haemostasis and supersedes the protein-based, cascade model of coagulation. 1
VETs first appeared at the end of the 1940s and several different analysers have since been developed. All devices measure how the viscoelastic properties of blood changes with clot formation but differ subtly in how these measurements are obtained. 2 Thromboelastography (TEG), thrombelastometry (ROTEM) and Sonoclot are the analysers most used in clinical practice. Technologies continue to be refined to offer more sophisticated and user-friendly analysis.2,3 Uptake in use of VETs has been relatively slow. Use is largely restricted to trauma, cardiac and liver transplantation centres, where an established evidence base exists to supports its use in both assessing haemostasis and guiding transfusion in these cohorts.4 –8
By comparison, VET devices are not frequently seen on general intensive care units (ICUs). Impaired haemostasis is, however, a significant problem on ICUs, with the incidence of coagulopathy and thrombocytopaenia reportedly as high as 66% and 38%, respectively. 9 This corresponds with high rates of transfusion; just under 40% of patients receiving red cells, platelets, or fresh frozen plasma during their ICU stay. 10 Common causes of impaired haemostasis on ICU include physiological disturbances (e.g. hypothermia, acidosis, hypocalcaemia), consumptive processes (e.g. sepsis, extra-corporeal circuits), decreased synthesis of blood components, which can be acquired (e.g. liver disease, renal failure, drugs) or hereditary (e.g. haemophilia), and blood loss. Multiple causes often coexist with significant heterogeneity between patients. 11 Understanding and appropriately managing these complex haematological disturbances presents a daily challenge for intensivists, however, the Guideline for Provision of Intensive Care Services (GPICS) makes no recommendation regarding the availability of VETs to aid clinical decision making. 12
The main tools guiding diagnosis and management are clinical assessments, conventional coagulation tests (CCTs) and advice from haematologists. In this review we have considered whether there is an evidence base to support the use of VETs, alongside current CCTs, on all ICUs in assessing and managing patients.
Making the diagnosis
Identifying significant haematological disturbances in patients is essential for making a correct diagnosis. Unfortunately, no test can make a complete assessment of haemostasis. CCTs make a very limited assessment of haemostasis as they measure only the acellular components in plasma. They exclude the contribution of platelets, von Willebrand Factor (vWF), natural anti-coagulants and are also unable to measure fibrinolytic activity. 3 A quantitative assessment, rather than a functional assessment, of pro-coagulant factors is made. Equally, VETs are limited by their insensitivities to vWF, protein C and mild-to-moderate fibrinolysis (Figure 3). 3 Neither VETs or CCTs, confers a 100% sensitivity and specificity in identifying or excluding the presence of abnormalities in haemostasis. In addition, there are no universally agreed parameters with which to define hypo-, hyper- or normo-coagulability (Figure 3). Accurate interpretation of results from both CCTs and VETs is therefore difficult.
Significant discrepancies between CCTs and VETs in diagnosing impaired haemostasis in ICU patients has been reported.
In a retrospective audit of bleeding surgical and non-surgical patients on the ICU, most patients had a normal ROTEM. 13 In approximately 50% of patients with a normal ROTEM, thrombocytopaenia, prolonged APTT and elevated INR was identified on CCTs. This was interpreted as an impairment in haemostasis and used to guide transfusion. This may be deemed as unnecessary given the existence of a normal ROTEM in these patients (Figure 1). 13
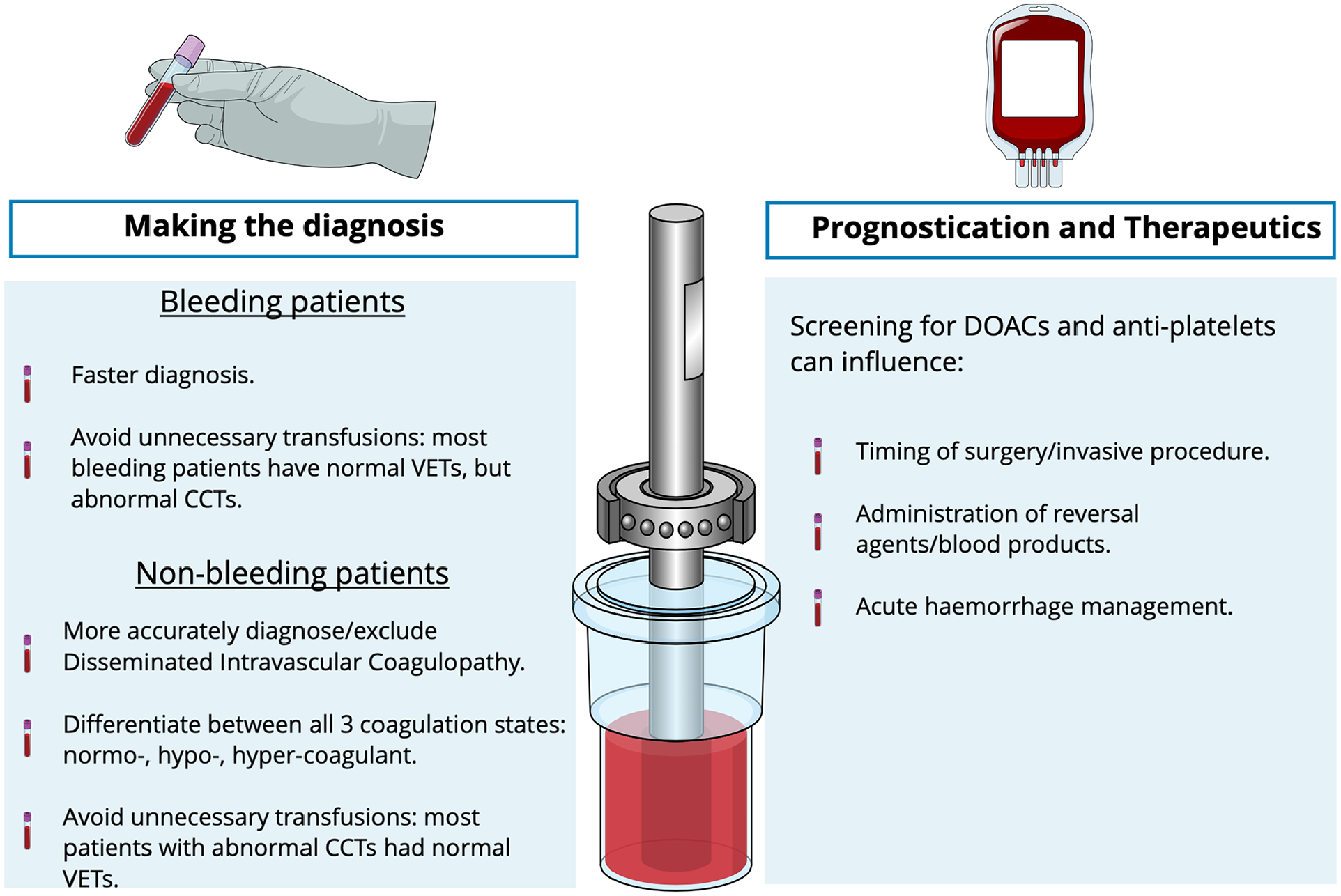
Potential benefits of using viscoelastic tests on intensive care units.
In non-bleeding ICU patients, a systematic review found evidence to suggest VETs may be able to more accurately diagnose or exclude disseminated intravascular coagulopathy (DIC) (Figure 1). 14 Although, in the absence of trials specifically comparing CCTs with VETs it was unable to definitively conclude whether VETs offer additional value in assessing haemostasis. It has since been shown that VETs are better able to differentiate between all three coagulation states (hypo, hyper or normo-coagulant), unlike CCTs which are not designed to assess hypercoagulability (Figure 1). 15 Discrepancies in test interpretation have also been demonstrated in this cohort. A normal TEG was identified in 60% and 81% of non-bleeding ICU patients with a deranged INR and thrombocytopaenia, respectively (Figure 1). 15
This is not a case of either/or, but rather whether VETs should be used in addition to CCTs. On balance, evidence suggests that there are benefits of using VETs to aid diagnosis in critically ill patients who are either actively bleeding or in whom impaired haemostasis is suspected on CCTs. In major haemorrhage, VETs confer an obvious time advantage which can influence outcomes, both in terms of morbidity and mortality.13,16 Another secondary benefit may include more conservative use of blood products on the ICU. 13
Prognostication and therapeutics
The ability of VETs to predict bleeding and/or thrombo-embolic events and in turn influence decisions regarding therapeutics is a theory that is yet to be proven. Current evidence, albeit limited, suggests a low predictive capacity.17 –19 The high incidence of thrombo-embolic complications seen in critically ill patients with coronavirus infection during the pandemic drove an interest to look at VET patterns in these patients. Findings suggested that VETs lack discriminatory value. Hypercoagulability did not confer a higher rate of thrombo-embolic complications, and in fact a higher rate of thrombo-embolic complications was observed in patients who were not hypercoagulable.18,19
The use of VET as a monitoring tool for the adequacy of anti-platelet and anti-coagulation therapies remains unvalidated. There is however growing evidence to support their use as a screening tool to facilitate urgent clinical decision-making (Figure 1).
Specific assays for direct oral anticoagulants (DOACs) are now available for most devices. A recent systematic review concluded that DOACs have a clear influence on VET parameters and DOAC specific assays have a suitably high sensitivity for detecting clinically relevant blood concentrations. 20 Again, the speed with which these tests can be performed makes it an ideal choice for emergencies. Testing for DOACs may influence several decisions on the ICU including timing of emergency surgery or other invasive procedures, administration of reversal agents and management of acute haemorrhage (Figure 1).
Modern analysers also incorporate separate platelet-function tests, or platelet-mapping assays which can assess the degree of platelet inhibition following anti-platelet therapy. 3 Results from small prospective trials have shown that these assays can accurately detect significant platelet inhibition after anti-platelet therapy, comparable to the gold standard laboratory-based test, optical light transmission aggregometry.21,22 These findings have been incorporated into practice in some centres which now utilise platelet mapping to support peri-operative decision making in both elective and non-elective patients. 22 Similarly, as with DOAC testing, in patients taking anti-platelets that are admitted to ICU, platelet function testing might guide decisions related to timing of emergency invasive procedures and transfusions.
Specific patient groups
Sepsis
A lot of the evidence, previously discussed, assessing the use of VETs as a diagnostic tool in the non-bleeding ICU patient has come from trials involving patients with sepsis.14,15 This is hardly surprising given the size of the sepsis caseload on general ICU.
Sepsis induced coagulopathy is a well described phenomenon, prevalent on the ICU. Key observations from trials looking at VETs in this cohort include a higher incidence of hypocoagulability in patients with septic shock and a higher incidence of hypercoagulability in patients with sepsis, without clinical evidence of shock.15,23 The development of hypocoagulability was independently associated with a higher mortality. This reflects that a correlation between the extent of coagulation derangement and mortality exists. 15
Another potential benefit of performing VETs in patients with sepsis is to diagnose DIC (Figure 2). In theory, repeated testing may allow for earlier detection, timely treatment and an improvement in outcomes. At this time, a DIC scoring system based on VET parameters awaits validation in critical illness and current evidence is limited to results from small studies.14,23
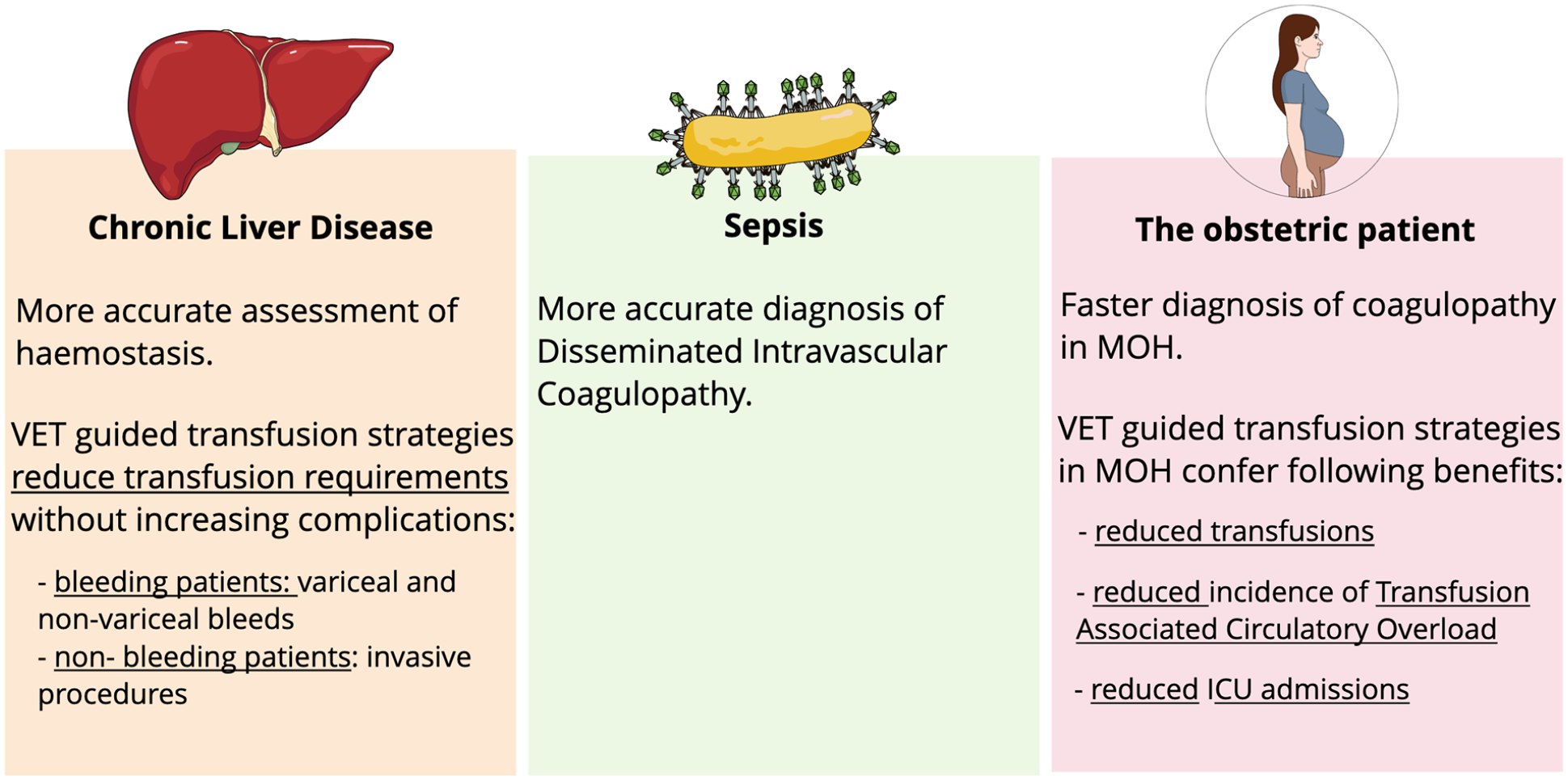
Special patient groups: possible applications of viscoelastic tests.
Chronic liver disease
Patients with chronic liver disease (CLD) account for approximately 3% of all admissions to ICU, a figure which is increasing annually. 24
In patients with stable cirrhosis haemostasis is re-balanced.25,26 During primary haemostasis, thrombocytopaenia and abnormal platelet function is balanced by both an increase in von Willebrand factor and reduction in ADAMTS-13. Thrombin generation remains unchanged during secondary haemostasis because of equal reductions in both pro- and anti-coagulant factors. At the time of fibrinolysis, the reduction in plasminogen is counteracted by elevated tissue plasminogen activator and reduced factor VIII, alpha-2 anti-plasmin and thrombin activatable fibrinolysis inhibitor. 26
This rebalanced state is easily disrupted and may necessitate or complicate an admission to ICU. During critical illness patients with CLD can exhibit both hypo- and hyper-coagulable features. It has been further elucidated that hypocoagulability and hypofibrinolysis are the most frequent disturbances seen in the sub-set of patients with CLD who develop acute-on-chronic liver failure (ACLF). Overall critical illness can predispose patients with CLD to bleeding or thrombosis. In some patients, bleeding and thrombosis risks may coexist simultaneously. Unfortunately, best practice is yet to be established, thus VETs may have a role to play.25 –27
CCTs are routinely abnormal in patients with CLD but are poor predictors of bleeding or thrombotic events. 27 There is considerable evidence to support the idea that VETs provide a better assessment of haemostasis in cirrhotics (Figure 2). 28 This has held true when comparing patients with stable cirrhosis and those with ACLF. 29 Some evidence, albeit limited, has emerged suggesting certain VET parameters may also help predict bleeding events in ACLF. 29
Empirical correction of abnormal CCTs with blood products can cause a paradoxical increased bleeding risk by increasing portal pressure and is not recommended in patients with CLD, including those with ACLF.25,27 Indeed, it has been consistently shown that worsening portal hypertension, as a result of volume overload, and not coagulopathy, is the most likely precipitant of variceal bleeding in CLD. 27 International best practice guidelines for the periprocedural management of abnormal CCTs and thrombocytopaenia in patients with cirrhosis recommend against empirical correction of INR/PT prior to procedural intervention with a high bleeding risk in the absence of concomitant vitamin k antagonist administration. 30 Likewise, they recommend caution in ascribing platelet counts below 30 × 109 to liver disease alone and against the periprocedural correction of thrombocytopaenia, unless in the context of very high-risk surgery. Finally, they recommend against prophylactic treatment of fibrinogen concentrations less than 1 g/L with cryoprecipitate or fibrinogen concentrate, even in the context of critical illness. 30 These recommendations continue to be ignored and it remains common practice to routinely correct abnormal haemostatic parameters with fresh frozen plasma, cryoprecipitate and platelet transfusions prior to undertaking invasive procedures. Empirical correction even occurs in patients with a PT/INR < 1.5 and platelet count >50 × 109/L despite these best practice recommendations. 27
VET-guided transfusion strategies in patients with stable cirrhosis undergoing invasive procedures have performed well and has demonstrated a significant reduction in the number of blood products transfused, without a significant difference in bleeding complications observed (Figure 2).29,31,32 A similar reduction in transfusion requirements has been observed in utilising a VET-guided transfusion strategy in patients with both variceal and non-variceal bleeds (Figure 2).33,34 Furthermore, re-bleeding risk was no greater in those who received VET-guided blood management following non-variceal haemorrhage and a somewhat lower following variceal bleeding. Critics of these trials will refer to their small size, heterogenous VET parameters and perhaps outdated standard-of-care protocols in the control arms. Despite these criticisms, subsequent meta-analyses have confirmed that VET-guided transfusion strategies do result in a significant reduction in blood product administration with no difference, or even a significant reduction, in bleeding complications.35 –37 Furthermore, if some standard-of-care protocols lack biological plausibility, they remain consistent with the widespread clinical practice of empirical transfusion. 27 Arguably VETs may give clinicians the confidence to move away from entirely empirical transfusion strategies in the context of haemorrhage and whilst organisations representing international expert opinion do not yet recommend routine evaluation with VETs prior to invasive procedures,38 –40 they do acknowledge that VETs may provide reassurance of normal haemostasis in such instances and may also be useful in the management of haemorrhage.27,38,39
Most of the current data have been obtained from patients with stable cirrhosis rather than critically ill patients with CLD. Further validation is therefore required in ICU patients with liver disease. Given the complexity of haemostatic disturbances in critical illness, it is reasonable to suggest that the more global assessment of haemostasis offered by VETs may yield benefits over CCTs alone. The recent guidelines from the Society of Critical Care Medicine (SCCM) have lent credence to this and supports the use of VETs in the management of patients with ACLF and acute liver failure (ALF). 41 In the initial stages of ALF a conservative approach to correcting haemostatic disturbances is favoured, as this may facilitate transplant listing, and ongoing management is usually within specialist liver centres where VET is available.
The obstetric patient
Haemorrhage is one of the leading causes of admission to ICU in the peri-partum period. 42 Whilst the absolute rate of peri-partum admissions is low (0.2%), this is anticipated to rise as the rate of post-partum haemorrhage increases.42,43 The normal physiological changes of pregnancy results in a pro-thrombotic state. This is a protective mechanism that buffers against blood loss at delivery and thereby avoids the need for transfusion in many cases. 44 The coagulopathy that ensues in major obstetric haemorrhage (MOH) is often related to fibrinogen levels, which are supra-maximal at term.44,45 There is evidence that fibrinogen falls early in MOH and hypofibrinogenemia predicts progression to severe MOH and a requirement for interventions. 45 As such the focus of managing haemostatic disturbances in obstetrics is invariably in promptly diagnosing and correcting hypofibrinogenemia.
There is consensus agreement that VETs enable more timely diagnosis of coagulopathy in the setting of MOH (Figure 2). 43 The Obstetric Association of Anaesthetists (OAA), British Society of Haematologists and Royal College of Obstetricians and Gynaecologists support its use.2,46,47 Validated VET-based treatment algorithms are awaited but there is early evidence that these could be beneficial.43,45 A large single centre observational study identified ROTEM guided transfusion as associated with a significant decrease in both the total number of units and the total volume of blood products transfused (Figure 2). 48 This was explained by the finding that less than 25% of patients with an MOH progressed to develop a coagulopathy. Improvements in morbidity were also seen, with a significant decrease in the incidence of transfusion associated circulatory overload and a non-significant decrease in ICU admissions (Figure 2). The Obs-UK trial (NIHR152057) is a national, multi-centred randomised controlled trial, which recently began, that aims to reduce excess bleeding and the need for transfusion after childbirth through the delivery of an appropriate care bundle. A key aspect of this care bundle is the use of VETs and the study hopes to comprehensively assess the value of VET based treatment algorithms, in terms of both patient outcomes and cost-effectiveness. 49
Most of the potential benefits of using VET in peri-partum women is confined to labour ward and maternity services. The appropriate use of VETs as part of a validated treatment algorithm may lead to a significant decrease in the number of patients with an MOH requiring ICU admission. Non-significant reductions in ICU admissions following the use of VETs have already been observed. 48
Practical considerations
There are several practicalities once must consider prior recommending that VETs become standard of care on ICU.
The high cost of setting up viscoelastic POCT is arguably the main factor that precludes its use. 1 The total expenditure includes the cost of the machine, the consumables, and the maintenance. North Manchester General Hospital conducted a pilot study to determine cost-effectiveness. 50 In a short initial trial period they quantified the reduction in blood components achieved when VETs were used to guide peri-operative transfusion decisions. Subsequently over a 3-month audit period they identified 150 surgical patients who required four units of packed red cells, with or without, other blood components. When this was scaled for the year, they estimated that potentially £48,000 could be saved in blood components alone if using a VET-guided approach to peri-operative transfusion. Over a 5-year period the net savings, once removing the outlay costs of the machine, the consumables, and the maintenance, were approximately £125,000. This estimate obviously has potential to be much lower depending on an individual hospital’s transfusion practices. Additional deductions may also be required for the cost of training staff, which was not accounted for in these estimates. Equally this estimate may be much higher if the other possible savings, in addition to the cost of blood products, are accounted for. This can include savings from reduced ICU admissions or length of ICU stay and decreased theatre or interventional radiology cancellations or delays.
For the full benefits of POCT to be realised a 24/7 service is required . This service must include individuals who can both accurately perform and interpret the results (Figure 3). Quality assurance has been made significantly easier with advances in the devices, but accurate interpretation requires a good understanding of the technology. 3 The discrepancies in skill-sets, out of hours is a reality both in ICU and theatre environments. The development and use of treatment algorithms may be of significant help and alleviate this burden of test interpretation (Figure 3).
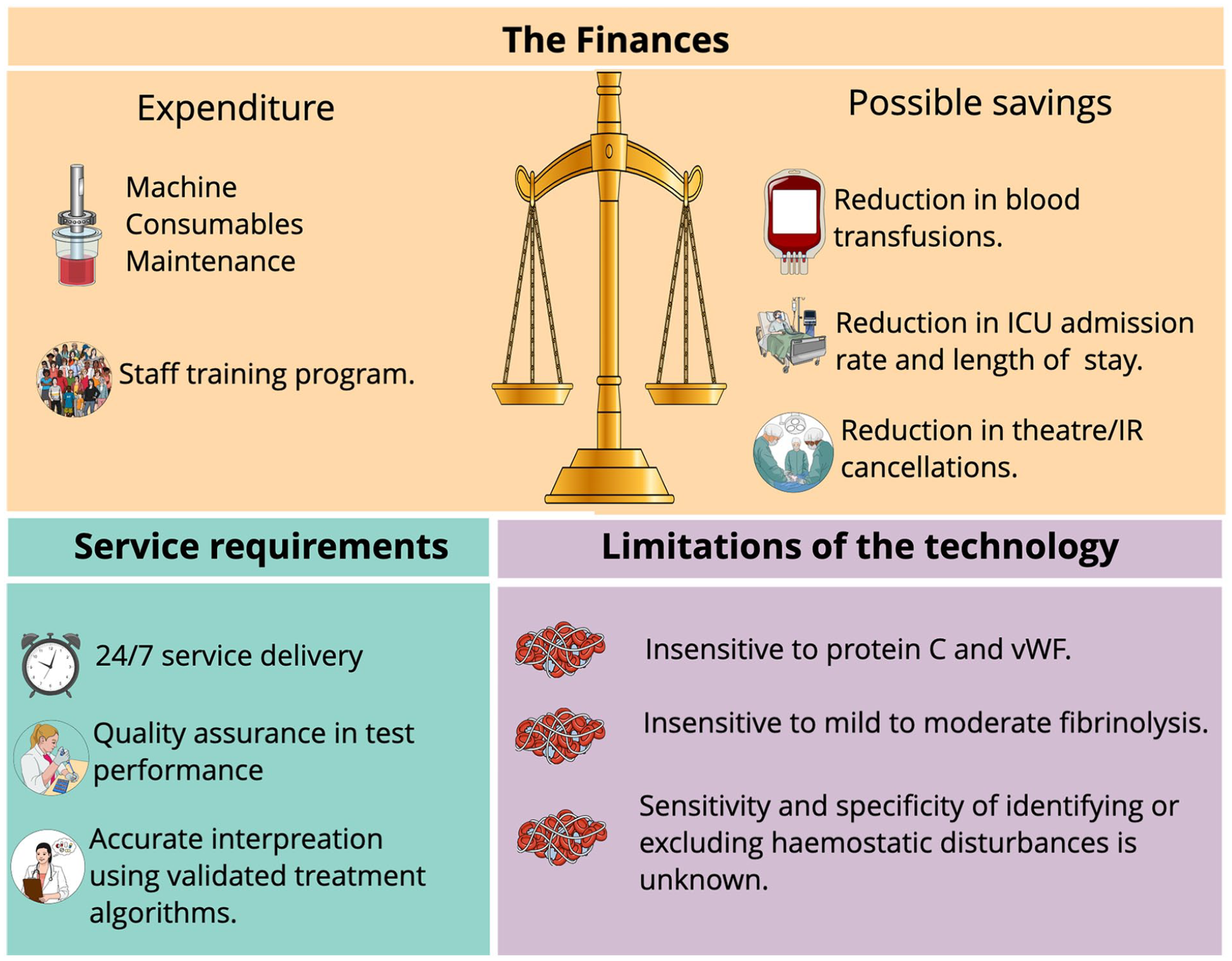
Practical considerations of setting up a viscoelastic point of care service on intensive care units.
Lastly, whilst it might seem obvious, the case mix of individual ICU must reflect the patient demographics in whom there is an evidence base to support the use of VETs.
Discussion
We have considered both the evidence from relevant trials and the practical considerations to support the use of VETs as a standard POCT on all ICUs.
There is compelling evidence to suggest that VETs are better at diagnosing haemostatic disturbances in the bleeding patient on ICU when compared with CCTs. This mirrors evidence from trauma centres where VET use is already well established. This has now been seen in surgical and non-surgical ICU patients, including cirrhotic and obstetric patients.2,12,31,32 The main advantages cited are faster time to detection of an evolving coagulopathy and in particular a better reflection of fibrinolysis. When VETs have subsequently been used to guide transfusion decisions in these patients, there has been a significant reduction in transfusion requirements, without an increase in adverse outcomes13,33,34,48 Transfusion is associated with a dose-dependent increase in morbidity, mortality and average hospital and ICU length of stay. 51 A more judicious approach in ICU patients may therefore help to reduce adverse outcomes, reduce costs and spare a finite resource. The World Health Organisation reiterated this in a recent report calling for implementation of appropriate patient blood management strategies, which it claimed is necessary to avoid another world health crisis. 51 Finally, there is an evolving role for VETs to support urgent clinical decision making in individuals taking anti-coagulants and anti-platelets which is increasingly more common amongst patients admitted to ICU.20 –22
As yet, VET-based treatment algorithms have neither been validated nor shown to improve patient outcomes, beyond reducing unnecessary exposure to blood products. Most of the available data comes from relatively small trials, with heterogenous study protocols and which are not specific to ICU patients. Having said that, current ‘best practice’ for the management of bleeding, coagulopathy and thrombosis in ICU is often outmoded and based on limited, if any, high quality evidence. 27
Given the paucity of high-quality evidence of improved patient focussed outcomes, coupled with the large capital outlay required to set up a VET POCT service, it is impossible to definitively recommend that all ICUs implement VETs as standard of care. We do however suggest that all ICUs should consider on an individual basis, as part of routine service evaluation, whether they have an appropriate patient mix and adequate patient numbers to justify the implementation of VETs whilst the results of future trials are awaited to vindicate their decision.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
