Abstract
Objective
Current obstetric guidelines for postpartum hemorrhage (PPH) vary in fluid resuscitation management. This study aimed to evaluate the effect of fluid management on coagulation parameters in early PPH.
Methods
We performed a multicenter, randomized trial. Women who had 500 mL of blood loss in the third stage of labor were randomized to receive a restrictive fluid administration strategy or a liberal fluid administration strategy. A rotational thromboelastometry panel was performed in 72 patients. We evaluated within-group and between-group differences in the EXTEM clotting time (CT), EXTEM amplitude at 10 minutes (A10), INTEM CT, and FIBTEM A10. We also evaluated the mean fibrinogen concentration, activated partial thromboplastin time, and partial thromboplastin time in the total study population (n = 249).
Results
There were no significant differences in hemostatic parameters between the groups after correction for baseline values.
Conclusions
In women with PPH <1500 mL, there is no clinically relevant effect of a restrictive or liberal fluid administration strategy on thromboelastometric hemostatic and regular coagulation parameters.
Introduction
Postpartum hemorrhage (PPH) is one of the main causes of maternal death worldwide and one of the major causes of maternal morbidity in higher income countries.1–3 PPH is defined as blood loss of more than 500 to 1000 mL within the first 24 hours after delivery.2,4–6 The overall incidence of PPH is estimated to be 4% to 8%.2,7–9 Several causes of PPH have been reported, with uterine atony accounting for over 70% of the cases. 10 An important aspect in the management of PPH is fluid resuscitation.
There is no uniformity in the fluid resuscitation strategy in the available obstetric guidelines. The Dutch Society of Obstetricians and Gynecologists has stated that there is currently no evidence to administer more fluids than lost when reaching 500 mL of blood loss. 11 In 2017, The Royal College of Obstetricians and Gynaecologists Green Top guideline recommends commencing fluid resuscitation at 500 mL of blood loss. If blood loss reaches 1000 mL or if a patient shows signs of clinical shock, the Royal College of Obstetricians and Gynaecologists recommends to infuse up to 3.5 L of clear fluids. 5 In 2019, The American College of Obstetricians and Gynecologists did not mention clear fluid transfusion. 12 Their practice guideline recommends preparing for blood (product) transfusion in women with more than 1500 mL of blood loss and abnormal vital signs. Additionally, in 2019, the Network for the Advancement of Patient Blood Management, Haemostasis and Thrombosis consensus statement mentioned a recommendation of “restrictive crystalloid resuscitation (1–2 mL of crystalloid for every 1 mL of blood loss) as initial fluid resuscitation according to the clinical condition and the estimated blood loss”. 13
Volume resuscitation can consist of crystalloids, colloids, transfusion of blood products, or a combination of either, but they all have (dis)advantages. Crystalloids and colloids are readily available and can be rapidly transfused. However, large amounts of these products may induce coagulopathy, hypothermia, and acidosis causing the classic lethal triad.14–16 Blood transfusion might cause a variety of blood transfusion-related risks, such as antibody formation, acute hemolytic reaction, anaphylactic shock, acute lung injury related to transfusions, and transfusion-related circulatory overload. 17
To evaluate hemostasis, standard plasma assays, such as fibrinogen concentrations, the activated partial thromboplastin time (APTT), the partial thromboplastin time (PT), and thromboelastometry can be used. Thromboelastometry is a dynamic, whole-blood, viscoelastic coagulation test, which evaluates the coagulation from clot formation throughout fibrinolysis. 18 Thromboelastometry provides real-time information on the intrinsic and extrinsic coagulation pathways, as well as on the platelet and fibrinogen contribution to clot formation. 18 Thromboelastometry can be used within the obstetric field with slightly adjusted reference ranges compensating for hemostatic changes in pregnancy and childbirth.18,19
In a randomized, controlled trial, we previously showed no disadvantages to a restrictive fluid administration (RFA) strategy in the early stages of PPH on the progression of blood loss. 20 We performed a randomized, controlled trial in a pre-planned subgroup of patients in whom we evaluated the effect of an RFA and liberal fluid administration (LFA) strategy on coagulation in women with PPH <1500 mL. We hypothesized that the clotting time (CT) is prolonged and the thromboelastometry amplitude at 10 minutes (A10) is decreased with an LFA policy based upon the coagulopathy induced by a large amount of crystalloid and colloid infusion.
Methods
We used data from the REFILL study, which was a randomized, controlled, multicenter trial that was conducted from August 2014 until September 2019. The REFILL study was performed in four hospitals in The Netherlands. The study was approved by the Medical Ethics Committee Maastricht University Medical Centre (approval number: NL4294206813). This trial is registered in the Netherlands Trial Register NTR3789 or NL3623 (https://www.trialregister.nl/trial/3623).
Women were considered eligible if they were 18 years and older, were pregnant and labor started after 24 + 0 weeks of pregnancy, understood the Dutch language, and were mentally competent. Vaginal and cesarean deliveries were included. Women were excluded for the following reasons: they used prophylactic or therapeutic anticoagulant therapy (carbasalate calcium within the previous 10 days or low molecular weight heparin within the previous 48 hours), or they had known coagulation disorders, pre-eclampsia, contraindication for liberal fluid therapy (e.g., cardiac causes, systemic causes such as Marfan syndrome, renal causes, or pulmonary failure), or an antenatal diagnosis of placenta accreta spectrum. We obtained written informed consent prior to active labor and oral confirmation during early labor from each patient.
Randomization was performed using a sealed opaque envelope by the treating team of health care professionals. Randomization was stratified in each center in blocks of four in a 1:1 allocation. The sealed opaque envelopes were created by an independent research nurse or medical student who was not involved in the randomization of the patient. Envelopes were distributed in each center by Maastricht University Medical Centre.
The women who had 500 mL of blood loss and ongoing blood loss in the third stage of labor and who provided written and oral consent were randomized to an RFA or LFA strategy. In the RFA group, patients received clear fluids at 0.75 to 1.0 times the blood lost. In the LFA group, patients received clear fluids at 1.5 to 2.0 times the blood lost. The first 2000 mL of volume replacement consisted of NaCl 0.9%, Ringers lactate, or a combination of both. The blood loss was measured by weighing absorption towels with exclusion of the first absorption towel, which was disposed directly after birth of the child.
Citrated (3.2%) blood samples and those with EDTA (Vacutainer; BD, Mississauga, Ontario, Canada) were drawn at three times. The first time of sampling was T1, which was when there was 500 mL of blood loss and ongoing blood loss. The patients were randomized and the first blood sample was drawn. If there was no intravenous access yet, an intravenous access was established. Hemoglobin concentrations, the hematocrit, the platelet count (Sysmex XN-9100, Sysmex, Norderstedt Germany; Cell Dyn Sapphire, Abbot, Abbot Park, IL, USA; or Advia 21202i, Siemens Healthineers, Den Haag, The Netherlands), the APTT (Siemens CS2100 with Actin FSL, Siemens Healthineers; STA-R Evolution with STA APTT, Diamond Diagnostics, Holliston, MA USA; or STA-R Max with C.K. Prest, Diamond Diagnostics), the PT (Siemens CS2100 with Innovin, STA-R Evolution with Neoplastin plus, or STA-R Max with Neoplastin plus), and Clauss fibrinogen (Siemens CS2100 with Innovance or STA-R Evolution with STA Fibrinogen) were measured. A rotational thromboelastometry (ROTEM) panel (ROTEM® Delta and Werfen; Munich, Germany) was performed in a subset of 72 women who delivered at Maastricht University Medical Centre or Zuyderland Medical Centre. ROTEM® values were procured in subsequent randomizations at T1 and T2 (45–60 minutes after T1) as the regular samples were drawn, regardless of the amount of blood loss or randomization. The other participating centers had no ROTEM® analysis available.
Fluid resuscitation was initiated at T1. At T2, a second set of blood samples were drawn. The parameters that were measured were identical to those at T1. At T3, 12 to 18 hours postpartum, a third blood sample was drawn if the patient was still in the hospital with measurement of hemoglobin concentrations and the hematocrit. T3 was not used in the analyses.
Hemodynamic parameters, such as blood pressure, heart rate, and oxygen saturation, were measured according to a local protocol. In case of a systolic blood pressure <90 mmHg, diastolic blood pressure <50 mmHg, a decrease of >20 mmHg in blood pressure, or a maternal heartrate of ≥125 beats/minute, an additional 500 mL of clear fluid was administered in 15 minutes in either group.
The underlying cause of the PPH was treated according to a national and local protocol (Dutch Society of Gynecology and Obstetrics). If 1500 mL of blood loss was reached, the randomized study arm was abandoned, and the patients were treated according to the local massive hemorrhage protocol. Blood samples were still drawn, and the patients’ data were analyzed according to intention-to-treat.
Data were collected from the patients’ charts and study files, and stored anonymously in Maastricht University Medical Centre in a restricted access file. A trial number was assigned to each patient at randomization. Severe adverse outcomes were defined as the requirement of intensive care admittance, four or more units of packed cells, embolization, or hysterectomy. Severe adverse outcomes were registered and analyzed by a data safety monitoring board. Throughout the study, there was no need to abandon the trial prematurely.
Results regarding the primary outcome (progression to more than 1000 mL blood loss) of the REFILL study have been published previously. 20 The sample size was not calculated because this study reports a subgroup of the original study. The limited number of samples may have affected the statistical significance of our results.
The secondary outcome was the differences in coagulation parameters. ROTEM® values of the EXTEM CT, EXTEM A10, INTEM CT, and FIBTEM A10 at T2 were recorded in the subset of 72 patients. We also recorded APTT, PT, and fibrinogen values in the total study population at T2. A correction for baseline and the change over time between T1 and T2 were calculated.
A comparative analysis was performed using Student’s t test for continuous data or the chi-square (Fisher’s exact) test for dichotomous outcomes. To compare between-group differences in the change over time, a linear regression was performed with adjustment for the baseline value of the outcome under analysis. The data were analyzed according to intention-to-treat. All analyses were performed using IBM SPSS 24.0 software (IBM Corp., Armonk, NY, USA).
The reporting of this study conforms to the CONSORT statements. 21
Results
In the subset of 72 patients, 36 were randomized to the RFA group and 36 to the LFA group. Maastricht University Medical Centre provided 63 datasets, and Zuyderland Medical Centre provided 9 datasets. The baseline characteristics are shown in Table 1.
Baseline patients’ characteristics.
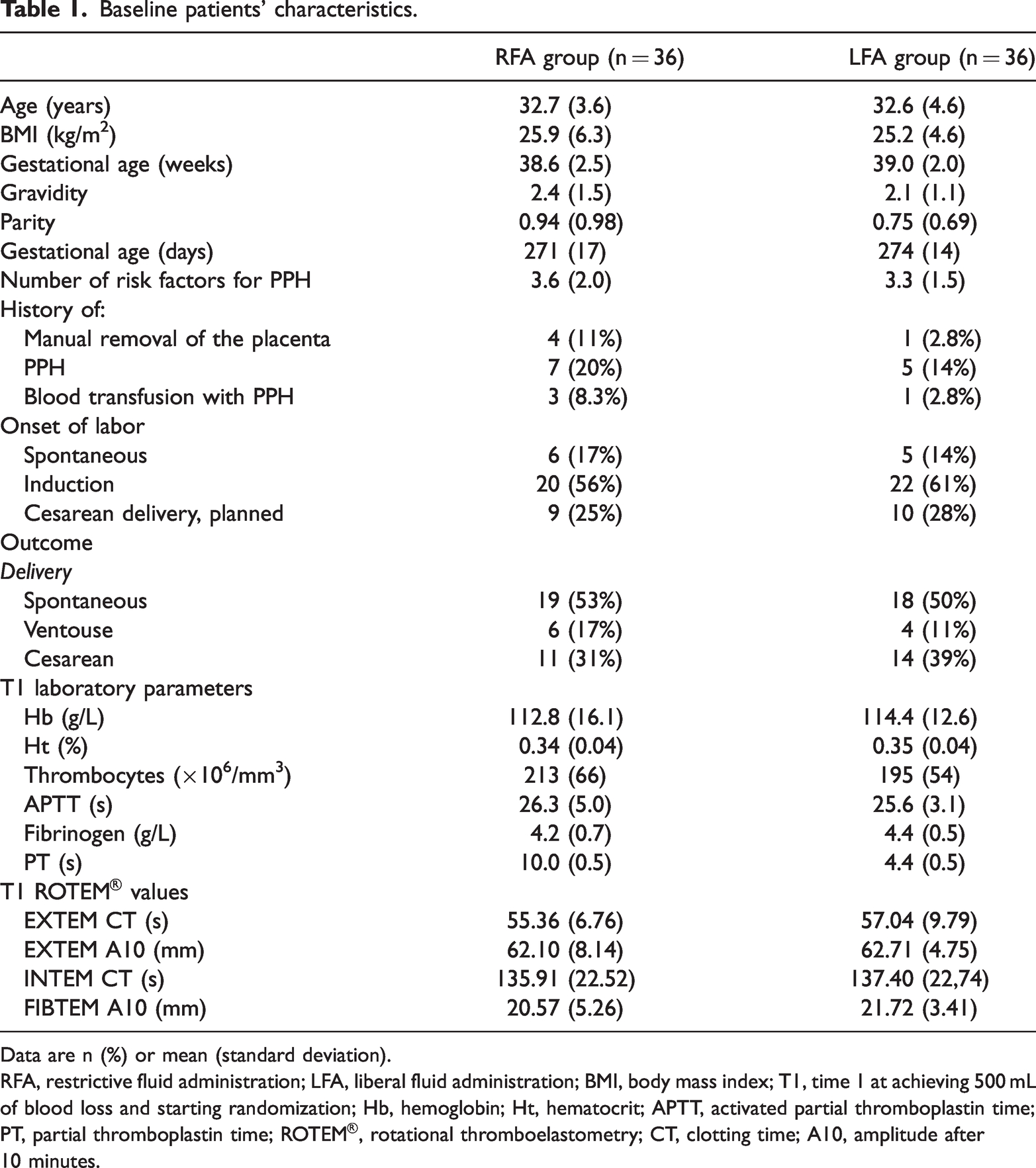
Data are n (%) or mean (standard deviation).
RFA, restrictive fluid administration; LFA, liberal fluid administration; BMI, body mass index; T1, time 1 at achieving 500 mL of blood loss and starting randomization; Hb, hemoglobin; Ht, hematocrit; APTT, activated partial thromboplastin time; PT, partial thromboplastin time; ROTEM®, rotational thromboelastometry; CT, clotting time; A10, amplitude after 10 minutes.
The mean (±standard deviation) fluid administration in the 72 patients was 1214 ± 1250 and 1588 ± 982 mL in the RFA and LFA groups, respectively. The mean blood loss in the RFA group with available ROTEM® values was 1307 ± 728 mL, and the mean blood loss in the LFA group with available ROTEM® values was 1055 ± 376 mL. Nine patients reached more than 1500 mL of blood loss, with three patients in the LFA group and six in the RFA group. These patients were treated according to a local massive hemorrhage protocol.
Unfortunately, data on thromboelastometric parameters were not available for all 72 patients. Data were unavailable because of a shortage of blood drawn to perform the complete ROTEM® panel. Table 2 shows ROTEM® values.
Mean ROTEM® values and the change over time at T1 and T2.

Reference range coagulation values at peripartum: fibrinogen (g/L): 3.6 to 6.8; APTT (s): 24 to 34 (MUMC, Zuyderland Medical Centre, and UMCN) and 26 to 36 (JBZ); PT (s): 9.6 to 10.4 (MUMC and Zuyderland Medical Centre), 12.0 to 17.8 (UMCN), and 12.0 to 15.0 (JBZ).
*p < 0.05.
ROTEM, rotational thromboelastometry; T1, time 1 at a blood loss of 500 mL and randomization; T2, time 2 at 45 to 60 minutes after T1; CT, clotting time; A10, amplitude after 10 minutes; RFA, restrictive fluid administration; LFA, liberal fluid administration; CI, confidence interval; APTT, activated partial thromboplastin time; MUMC, Maastricht University Medical Centre; UMCN, University Medical Centre Nijmegen; JBZ, Jeroen Bosch Hospital; PT, partial thromboplastin time.
At T2, there were no differences in the mean ROTEM® values for EXTEM CT, EXTEM A10, INTEM CT, and FIBTEM A10 between the RFA and LFA groups. In the LFA group, we found a significant difference in the mean FIBTEM A10 value of 1.43 mm between T1 and T2 (p = 0.041). None of the other ROTEM® values showed any significant change between T1 and T2 in either group. After correction for baseline values, there were no significant differences in changes over time between the randomized groups (Table 2).
We compared fibrinogen, APTT, and PT values in the complete study population (n = 249) at T2 between the randomized groups because the original subset analysis was underpowered. The mean fibrinogen concentration, APTT, PT, and thrombocyte count at T2 were not significantly different between the RFA and LFA groups at T2. Linear regression with correction for baseline values at T1 showed no significant effect of the fluid resuscitation strategy on fibrinogen concentrations, the APTT, the PT, and the thrombocyte count (Table 3).
Mean coagulation values and the change over time at T1 and T2.

Reference range coagulation values at peripartum: fibrinogen (g/L): 3.6–6.8; APTT (s): 24–34 (MUMC, Zuyderland Medical Centre, and UMCN) and 26–36 (JBZ); PT (s): 9.6–10.4 (MUMC and Zuyderland Medical Centre), 12.0–17.8 (UMCN), and 12.0–15.0 (JBZ).
APTT, activated partial thromboplastin time; PT, partial thromboplastin time; RFA, restrictive fluid administration; LFA, liberal fluid administration; T1, time 1 at a blood loss of 500 mL and randomization; T2, time 2 at 45–60 minutes after T1; CI, confidence interval; MUMC, Maastricht University Medical Centre; UMCN, University Medical Centre Nijmegen; JBZ, Jeroen Bosch Hospital.
Discussion
This study showed no significant differences in plasma or whole-blood coagulation parameters between the RFA and LFA strategies in women with PPH <1500 mL. Mean ROTEM® values at T1 and T2 were well within refence ranges in pregnancy. 19
Standard laboratory coagulation tests show a correlation with ROTEM® results. EXTEM CT is correlated with PT results, INTEM CT with APTT results, and FIBTEM A5, A10, and maximum clot firmness with Clauss fibrinogen results. The platelet count affects clot stability, as measured by INTEM and EXTEM.19,22–24 Our study showed no significant differences in fibrinogen, APTT, or PT values between the RFA and LFA groups. Linear regression corrected for the baseline value showed no significant effect of the fluid administration strategy in early PPH. The confidence interval ranges of all parameters analyzed were small. Additionally, if the extremes of these intervals were met, these values would not indicate a clinically relevant difference. Therefore, the ROTEM® parameters did not appear to be affected by the RFA or a LFA strategy in early PPH in this study.
We did not find any clinically relevant differences in coagulation parameters in women with PPH up to 1500 mL between the RFA and LFA groups. Our data are limited by the amount of blood loss. Although the relative difference in fluid administered between groups was 0.93 versus 1.5 times the volume of blood loss, the absolute difference was only 374 mL and was unlikely to cause a clinically relevant change in hemostasis. Fluid resuscitation protocols and their effect on coagulation might become more relevant when larger amounts of clear fluids are transfused at a greater amount of blood loss. This speculation is supported by a large retrospective cohort study by Gillissen et al who examined the amount of infused clear fluids and the differences in hemostatic parameters in 1038 women with severe PPH. 25 They showed a more severe deterioration of coagulation parameters with administration of larger volumes of clear fluids. More prospective research is required to evaluate the effect of resuscitation strategies on coagulation parameters in the population with >1500 mL of blood loss.
This study has some strengths and limitations. The strengths of our study were the randomized, controlled design of the study and the adherence to the study protocol. In the RFA group, 0.93 times the blood loss was replenished compared with 1.5 times in the LFA group. An important limitation of our study is the limited number of patients evaluated. Even though this was a multicenter study, not every hospital adopted ROTEM® analysis in the management of PPH. The subset analysis was underpowered. Therefore, we analyzed the available data on fibrinogen, APTT, PT, and thrombocyte values in the complete study group at T2 as representative data of the fibrinogen status and intrinsic and extrinsic pathway status. Another limitation of this study is the inter-center differences in the PT analysis. The reference range of the PT analysis in Maastricht University Medical Centre and Zuyderland Medical Centre was 9.6 to 10.4 s, whereas the reference ranges of the PTs analyzed in University Medical Centre Nijmegen and Jeroen Bosch Hospital were different, with 12.0 to 17.8 s and 12.0 to 15.0 s, respectively. However, the mean PT time did not exceed 11 s and stayed well within 1.5 times the upper limit of the smallest reference range.
In conclusion, this study shows no clinically meaningful effect of the fluid resuscitation strategy on hemostatic parameters by thromboelastometry and traditional coagulation parameters in women with PPH <1500 mL. Future studies are required to evaluate the effect of fluid resuscitation on coagulopathies in severe PPH.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231171007 - Supplemental material for Restrictive versus liberal fluid administration strategy (REFILL study) in postpartum hemorrhage and its effects on thromboelastometry (ROTEM®) values: a randomized, controlled trial
Supplemental material, sj-pdf-1-imr-10.1177_03000605231171007 for Restrictive versus liberal fluid administration strategy (REFILL study) in postpartum hemorrhage and its effects on thromboelastometry (ROTEM®) values: a randomized, controlled trial by Pim B.B. Schol, Natascha de Lange, Yvonne Henskens, Luc J.M. Smits, Nicol A.C. Smeets and Hubertina C.J. Scheepers in Journal of International Medical Research
Footnotes
Acknowledgements
We would like to thank every health care professional and laboratory worker who made this study possible. In particular we would like to thank Rick Wetzels for his preparations regarding the ROTEM® analyses.
Author contributions
NL, YH, LS, and HS were involved in the conception and design of the trial. PS drafted the primary manuscript, was the overall investigator of the trial, and is the corresponding author. PS, HS, and LS analyzed the data. NS was the local executor at Zuyderland Medical Centre for arranging the ROTEM® analysis. All authors edited the manuscript, and read and approved the final draft.
Data availability statement
Data are available upon request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
