Abstract
Background and aim:
Monitoring of gastric residual volume (GRV) to assess for enteral feeding intolerance is common practice in the intensive care unit (ICU) setting; however, evidence to support the practice is lacking. The aim of this study was: (i) to gain a perspective of current practice in adult ICUs in the UK around enteral feeding and monitoring of GRV, (ii) to characterise the threshold value used for a high GRV in clinical practice, (iii) to describe the impact of GRV monitoring on enteral feeding provision and (iv) to inform future research into the clinical value of GRV measurement in the adult ICU population.
Methods:
A web-based survey was sent to all UK adult ICUs. The survey consisted of questions pertaining to (i) nutritional assessment and enteral feeding practices, (ii) enteral feeding intolerance and GRV monitoring and (iii) management of raised GRV.
Results:
Responses were received from 101 units. Ninety-eight percent of units reported routinely measuring GRV, with 86% of ICUs using GRV to define enteral feeding intolerance. Threshold values for a high GRV varied from 200 to 1000 ml with frequency of measurement also differing greatly from 2 to 12 hourly. Initiation of pro-kinetic medication was the most common treatment for a high GRV. Fifty-two percent of respondents stated that volume of GRV would influence their decision to stop enteral feeds a lot or very much. Only 28% of units stated that they had guidelines for the technique for monitoring GRV.
Conclusions:
Measurement of GRV is the most common method of determining enteral feeding intolerance in adult ICUs in the UK. The practice continues despite evidence of poor validity and reproducibility of this measurement. Further research should be undertaken into the benefit of ongoing GRV measurements in the adult ICU population and alternative markers of enteral feeding intolerance.
Introduction
Monitoring of gastric residual volume (GRV) to assess for enteral feeding tolerance is common practice in the intensive care unit (ICU) setting. 1 This practice continues despite evidence suggesting that GRV measures do not correlate with enteral feeding tolerance, gastric emptying, incidence of regurgitation, aspiration or ventilator associated pneumonia.2 –4 However, a lack of agreement between the guidelines of scientific societies and absence of an alternative bedside measure of feeding intolerance may hamper the widespread discontinuation of this practice. The most recent guidelines from the American Society of Enteral and Parenteral Nutrition (ASPEN) advise that GRV measures should no longer be monitored in patients in the ICU. 5 However, the European Society of Clinical Nutrition and Metabolism (ESPEN) recommend that enteral nutrition should be delayed if GRV is above 500 ml/6 h and that monitoring of GRV may be necessary to safely deliver enteral nutrition in the context of feed intolerance. 6 In addition to this different guidance on the validity of GRV monitoring, there is no consensus in research or clinical practice as to what constitutes a high GRV with studies quoting values ranging from 75 to 1200 ml. 7 A recent Cochrane review described an uncertain relationship between GRV and clinical outcomes (i.e. pneumonia, vomiting and length of hospital stay). 8 Despite this there may be some critical care patient groups (i.e. receiving high dose opioid sedation, catecholamines and following GI surgery), who may warrant closer monitoring of gastric aspirates.9,10
In critically ill patients an elevated GRV is one of the most common reasons for withholding enteral nutrition,1,11 negatively impacting on provision of adequate nutritional support. 12 Systematic reviews report a high GRV is the most common criterion used in studies to define enteral feeding intolerance in critically ill adults 79%, 7 critically ill children 48% 13 and premature infants 81%. 14 However, there was considerable variation with regards to the cut off values for GRV used to define enteral feeding intolerance and importantly there was no association between GRV and enteral feeding intolerance prevalence. 7 Several studies have shown no adverse effects from discontinuing GRV measurements in critically ill adults4,15,16 or children.17,18 The systematic review of enteral feeding intolerance in the adult critical care population did not find sufficient evidence to include GRV within the proposed definition of intolerance. 7 Despite these results suggesting there is insufficient evidence to support the use of GRV measurement, the practice appears to continue to be commonplace.
Surveys of paediatric ICUs in the UK identified that routine GRV measurement is standard practice in this patient group 19 with nursing staff relying heavily on GRV measurements to guide decision making surrounding initiating and withholding enteral feeds. 20 Work has subsequently commenced in the paediatric ICU population to determine the clinical and cost-effectiveness of not performing routine GRV measurements.21 –23 There is however a paucity of data on current practice around GRV monitoring in the adult population. Failure to achieve nutritional targets during an ICU admission has been associated with worse clinical outcomes24 –26 and it is therefore imperative that practices within ICUs do not hinder provision of adequate nutrition support.
The aim of this study was: (i) to gain a perspective of current practice in adult ICUs in the UK around enteral feeding and monitoring of GRV, (ii) to characterise the threshold value used for a high GRV in clinical practice, (iii) to describe the impact of GRV monitoring on enteral feeding provision and (iv) to inform future research into the clinical value of GRV measurement in the adult population.
Methods
A web-based survey based on the paediatric ICU GRV practice survey 19 was conducted; the paediatric survey was adapted for use with permission. Modifications were made from the paediatric survey due to differences in clinical practice between paediatric and adult patients. The survey was piloted by 10 respondents consisting of dietitians, doctors and nurses at University Hospital Southampton with minor iterations made following feedback. The final version of the survey was made available via a secure web-based provider (Microsoft Forms). One hundred forty-two adult ICUs were approached to take part in the survey and the target response rate for survey completion was set a priori at 70%.
The survey was distributed in July 2022 to all adult ICUs in the UK via the British Dietetic Association Critical Care Specialist Group (BDA CCSG) and by email to non-members. Email contacts for non-BDA CCSG member ICUs were found online or by telephoning the individual dietetic department or ICU. The survey was further promoted on social media (i.e. Twitter). Where low response rates were identified regional BDA dietetic and ICU groups were contacted and asked to promote the survey within their regions. Reminders were sent via the BDA CCSG online forum and email to non-responders. Up to two further emails or telephone reminders were targeted to non-responder units. Unit names were collected in order to contact non-responder units and check for duplication. The survey suggested completion should be undertaken collaboratively between an ICU dietitian, clinical nurse of any band and senior doctor (specialist registrar or above) and requested that any written guidelines or protocols on GRV monitoring be emailed to the study team.
The survey consisted of three domains: (i) nutritional assessment and enteral feeding practices, (ii) enteral feeding intolerance and GRV monitoring and (iii) management of raised GRV. The survey contained 54 questions including 32 closed question, 19 open questions with free text boxes for comments and three ranked questions. Respondents were asked to consider their current ICU clinical practice when completing the survey. Data were collected into a spreadsheet (Microsoft Excel). Data were summarised using description statistics for quantitative data. Thematic analysis was undertaken on qualitative data provided as free text comments using the methods described by Braun and Clarke whereby the comments were transcribed, and themes explored using an inductive approach. The following process was undertaken: data familiarisation, generation of initial codes, search for themes, review of themes, defining and naming themes and sub-themes. 27
The study was considered a service improvement project by the Clinical Effectiveness Department at University Hospital Southampton NHS Foundation Trust and therefore ethical approval was not required. The survey was registered as a service improvement project (number QI/0042) via the Trust Clinical Effectiveness Department. Consent was obtained from all respondents prior to them completing the survey.
Results
One hundred forty-two adult ICUs were approached to take part in the survey. One hundred one responses were received representing 71% of adult ICUs in the UK. The majority of responses (75%[76/101]) were from units with solely a general/mixed ICU; 19% (19/101) of responses included more than one type of ICU (general/mixed ICU plus one or more specialist ICU including cardiothoracic, neurosciences, burns, trauma and liver); and 6% (6/101) of surveys were from a specialist ICU with no general/mixed ICU caseload. Despite the instructions that the survey should be completed collaboratively between a doctor, nurse and dietitian only 2% (2/101) of surveys were completed jointly by all three professionals. Three percent (3/101) of surveys were completed jointly by a nurse and dietitian, 2% (2/101) were completed by solely a nurse and the majority (93% [94/101]) were completed by a dietitian alone. Non-responder ICUs accounted for 29% (29/101) of adult ICUs in the UK. There were no common factors identified between non-responder units (i.e. number of ICU beds, presence of a dietitian, type of ICU).
Nutritional assessment and enteral feeding practices
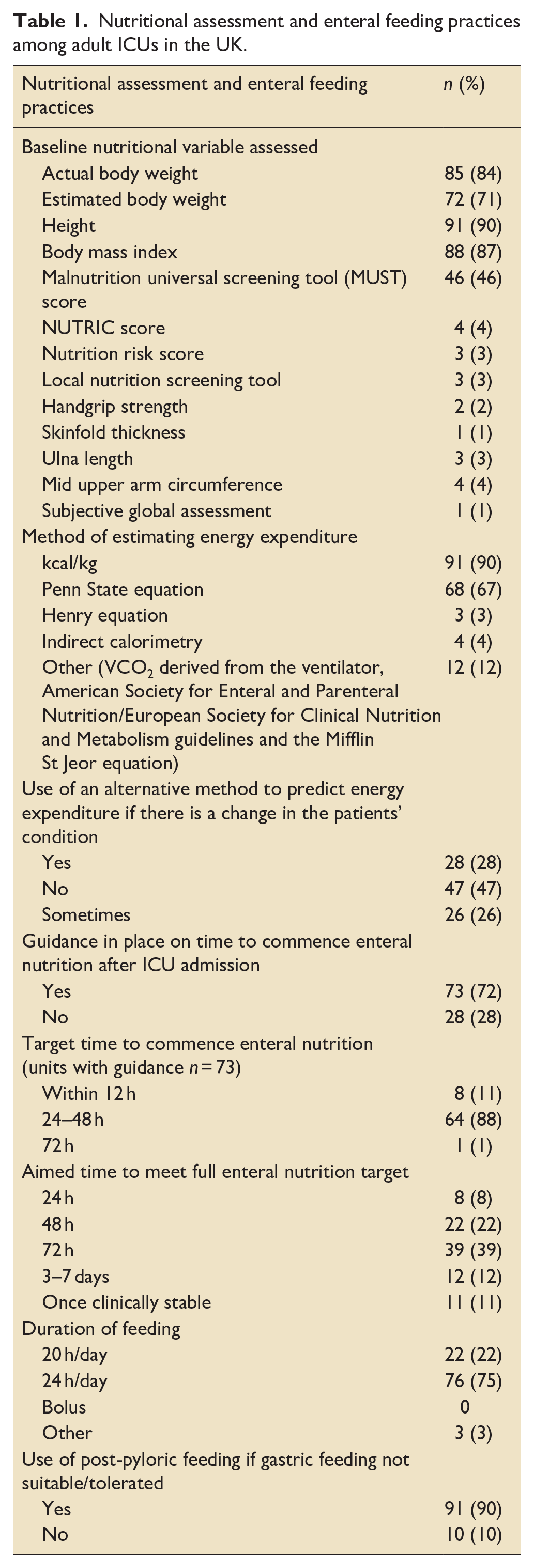
Ninety-eight percent (99/101) of units responded that they have written enteral feeding guidelines. Twenty-three percent (23/101) units provided a copy of these guidelines. All units undertook some form of nutritional assessment on ICU admission. Most units estimated energy requirements using a predictive equation (Table 1). Reasons for re-estimating nutritional requirements were primarily due to changes in mechanical ventilatory status (endo-trachael extubation, tracheostomy wean or moving to non-invasive ventilation) or use of extra corporeal membrane oxygenation. Ninety-one percent (92/101) of units reported utilising post-pyloric feeding with reasons for initiation being enteral feeding intolerance, raised gastric aspirates despite pro-kinetic medication and clinical conditions such as pancreatitis and post GI surgery.
Nutritional assessment and enteral feeding practices among adult ICUs in the UK.
Enteral feed tolerance and GRV monitoring
Seventy-eight percent (79/101) of ICUs reported having an agreed definition of enteral feeding intolerance, of which the most common was GRV measurement (Figure 1). Twenty-two units reported no agreed definition; however, eight of these provided details of the criteria used to monitor for enteral feeding intolerance.
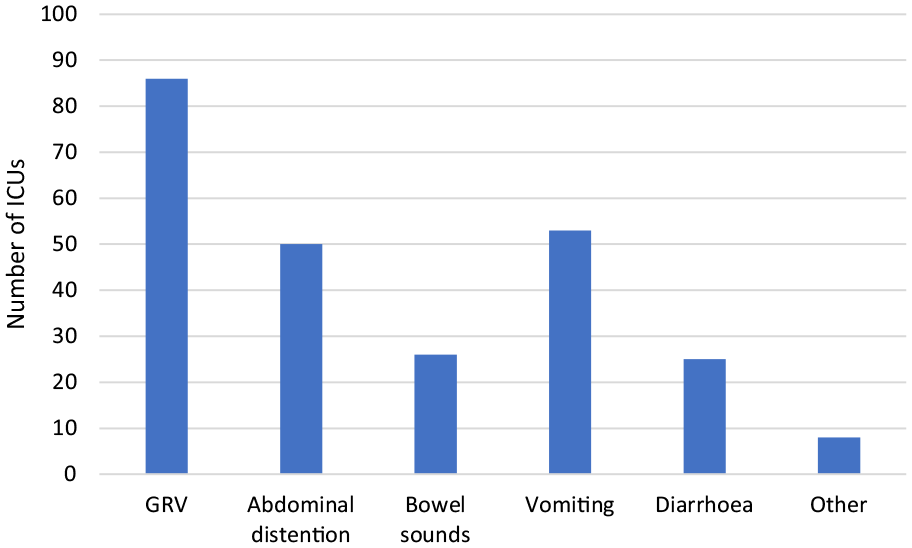
Criteria used to assess enteral feeding intolerance among adult ICUs in the UK.
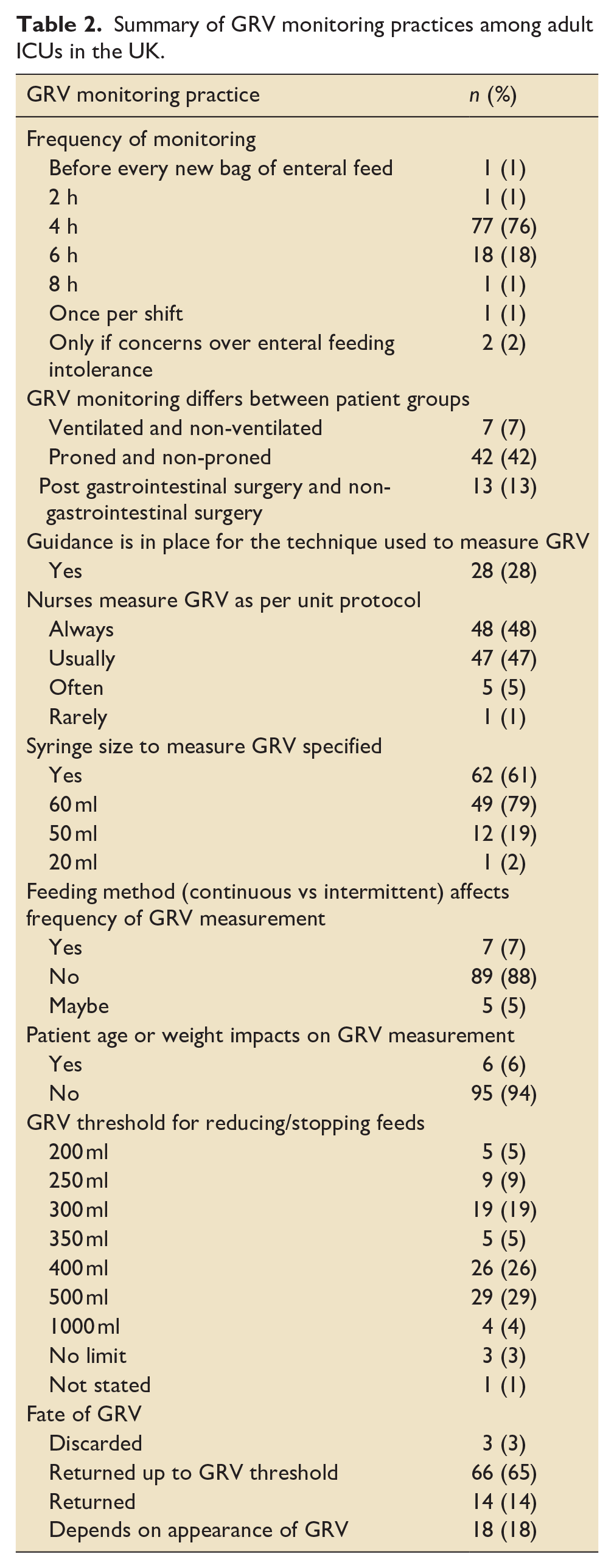
Ninety-eight percent (99/101) of units reported routinely measuring GRV with two units reporting only measuring if they had concerns over feeding intolerance (Table 2).
Summary of GRV monitoring practices among adult ICUs in the UK.
Thematic analysis
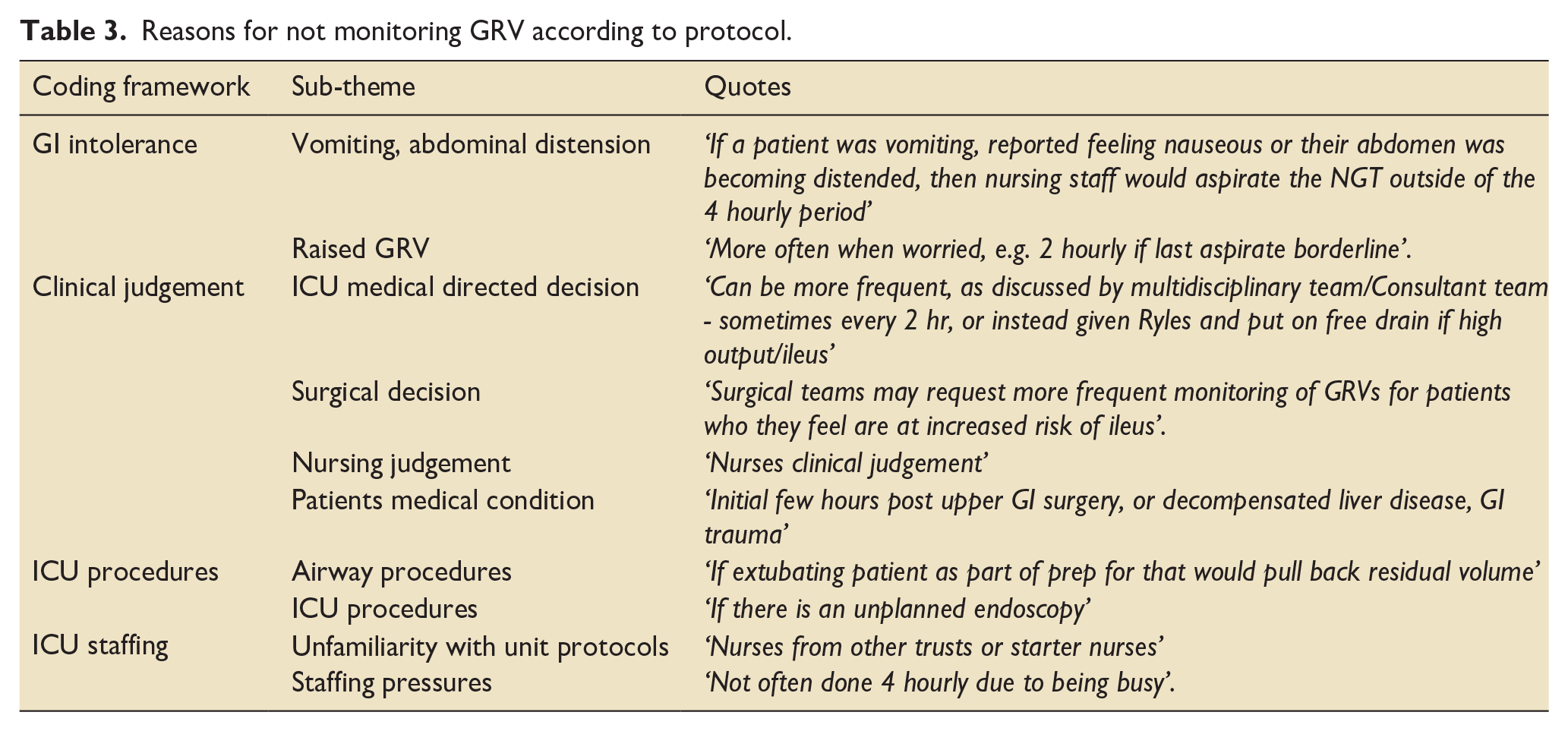
There were 52 comments relating to variations in monitoring practices grouped into four overarching themes. Forty-eight percent (48/101) of respondents reported that GRV is always monitored according to protocol, with 52% (53/101) of ICUs reporting that monitoring does not always follow the protocol. On qualitative analysis of the comments, four main themes emerged: clinical judgement, GI intolerance, ICU procedures and ICU staffing, with 10 sub-themes (Table 3). The dominant theme related to concerns over GI intolerance with 17% (17/101) of respondents reporting GRV would be measured more frequently if there is a suspicion of GI intolerance. Five percent (5/101) of units indicated they would measure GRV more frequently if the GRV measurement was raised above the unit threshold, with this threshold ranging from 250 to 500 ml. 8% (8/101) of ICUs reported that GRV may be monitored outside of the unit protocol at the request of the ICU medical team and 4% (4/101) units commented that visiting surgical teams may request increased GRV monitoring.
Reasons for not monitoring GRV according to protocol.
GRV threshold for reducing feeds
Two free text questions were included in the survey regarding the practice and threshold for reducing feed rate in response to a high GRV measurement. The threshold for a high GRV varied from 200 ml to no limit. Unit practice regarding what to do in response to a GRV measurement above the designated threshold also varied. Fifty-eight percent (59/101) of units responded that feed may be reduced following one high GRV, 15% (15/101) of units responded that two consecutive high GRV measurements were required to necessitate a change in feed rate. Six percent (6/101) of units reported awaiting three consecutive high values, one unit stated four consecutive high GRV measurements and 8% (8/101) of units responded that feed rate was only reduced after persistently high GRV but did not quantify this. Two percent (2/101) of units reported basing the decision to reduce feeds on trends rather than single values and 7% (7/101) of ICUs stated that feed rate is only reduced after initiation of pro-kinetic medication.
Management of raised GRV
Respondents were asked to rate how they would treat a high GRV in their unit from most likely to least likely. Most units (66% [67/101]) reported that they would prescribe pro-kinetic medication as the first line response to a raised GRV. The second most common treatment for raised GRV was to change the enteral nutrition formula, followed by instigation of post-pyloric feeding (Figure 2). Other strategies to manage a raised GRV included ensuring that the patient is regularly opening their bowels and continuing with enteral nutrition but at a reduced rate.
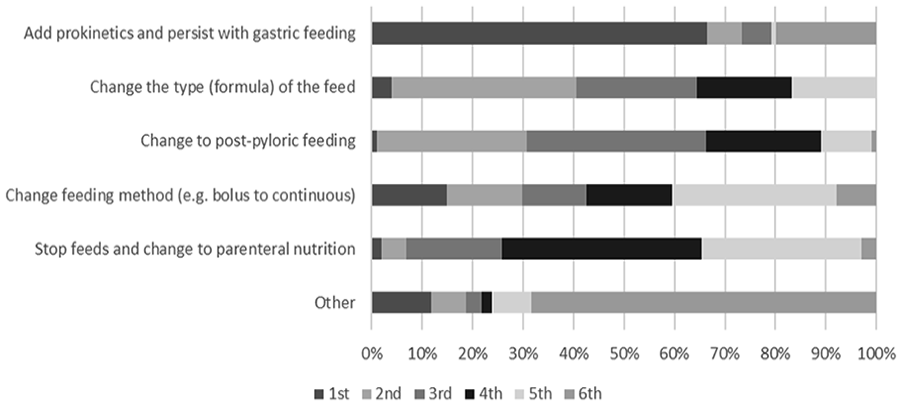
Strategies for management of raised GRV measurements.
Withholding enteral feeds
Sixty-three percent (64/101) of units reported that if EN is stopped then this decision is made jointly between the nurses and a doctor or dietitian. Six percent (6/101) of ICUs reported that the decision to withhold enteral feeds is primarily a medical decision and 30% (30/101) of units reported only stopping feeds based on the ICU feeding protocol. The volume, colour and consistency of GRV aspirated affects the majority of ICU staff decisions on stopping feed (Figure 3).
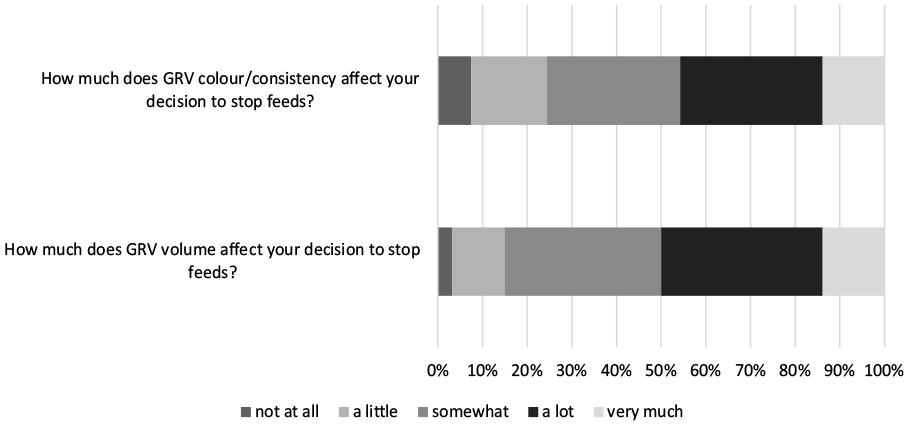
Effect of high GRV measurements on decision to stop EN.
The majority of respondents commented that the decision to stop feeds based on GRV was usually due to trends in volume rather than a one-off measurement and the GRV needed to be taken into consideration alongside other signs of intolerance such as abdominal distention and vomiting or risk of aspiration. However, other units noted that feeds are often stopped or reduced based solely on protocolised GRV measurements without further clinical interpretation. Forty percent (40/101) of respondents commented that they would stop enteral feeds if GRV appeared to contain blood or faecal matter. Twelve percent (12/101) of respondents reported enteral nutrition would be stopped if GRV contained feed, whilst 4% (4/101) noted that GRV containing bile would cause feed to be discontinued. If enteral feeds are stopped due to raised GRV 36% (36/101) of units reported it would be restarted upon a medical review, 18% (18/101) of respondents stated that feed would be restarted once GRV is reduced, 16% (16/101) stated they would wait 4–6 h before reviewing and 15% (15/101) stated feed would be restarted on dietetic review.
Discussion
This survey provides the first description of UK practice regarding measurement of GRV in adult critical care. The vast majority of adult ICUs in the UK that responded to this survey reported routinely measuring GRV to guide enteral nutrition delivery, with a small minority reporting measuring GRV only when there are clinical concerns. Most UK ICUs currently use GRV as an indicator of enteral feeding intolerance with GRV providing the sole measure in a number of units. Threshold values for a high GRV in the UK vary greatly with frequency of measurement also differing between units.
The ongoing reliance on GRV as a measure of GI dysfunction and enteral feeding intolerance may be as a result of the absence of other reliable functional markers that can be used to assess feed tolerance at the bedside.7,10 A scoping review considering potential biomarkers and functional measures of GI dysfunction and feeding intolerance in ICU patients published in 2022 concluded that there are currently no markers suitable for daily clinical use. 28 This review investigated the use of 11 potential biomarkers, two physiological markers (faecal weight and intra-abdominal pressure) and ultrasound as a functional marker, and concluded that none of the above markers are currently available for clinical use without further research and validation studies. Potential markers of interest include heparin binding protein, intra-abdominal pressure, cholecystokinin and acetylcholine however all require further research. GRV provides a convenient, albeit unreliable, assessment of GI dysfunction for busy ICU clinicians in the absence of an alternative suitable bedside measurement. 9 ICU clinicians may be concerned that not measuring GRV will result in increased incidence of aspiration and pneumonia, although several studies have shown that increasing the GRV threshold,29,30 or not measuring GRV at all4,15,16 does not result in increased incidence of complications in either adults or children.17,18 However, in light of the reported high incidence of GI dysfunction and associated adverse outcomes in some patient groups (i.e. surgical patients, those receiving high dose opioid sedation and catecholamines) ongoing GRV monitoring may be warranted in some critical care populations. 9 The majority of respondents to our survey reported that the colour and consistency of GRV aspirated affects decisions on stopping feed. Presence of blood or faecal matter in the aspirate signals GI dysfunction and cessation of routine measurements of GRV could lead to these symptoms being missed.
Our survey showed that 64% of units have a GRV threshold of less than the 500 ml cut off advocated in the ESPEN critical care nutrition guidelines; the paediatric survey also reported varying GRV thresholds. 19 Several units in our survey reported planning to review the evidence on GRV measurements and thresholds and that their feeding protocols may be amended following this. Recent studies have highlighted the lack of robust evidence for ongoing GRV monitoring4,15,16 and a systematic review of enteral feeding intolerance in the adult ICU population found no correlation between incidence of feed intolerance and reported GRV threshold and concluded that routine use of GRV to define enteral feeding intolerance could not be recommended. 7 This review concluded that a diagnosis of enteral feeding intolerance should be made after an assessment of symptoms throughout the GI tract and ability to achieve adequate enteral intake, not based solely on delayed gastric emptying as determined by GRV. 7
In a busy ICU, the practice of measuring GRV adds to the burden of nursing workload and has considerable cost implications. Parrish and McClave 31 made a conservative estimate of the costs involved in regular GRV measurement in the United States and concluded the practice had an estimated health care cost of $22,680,000 per year excluding the cost of syringes and other equipment needed. At a time where healthcare costs as well as the sustainability of healthcare practices 32 are being increasingly scrutinised, ICU clinicians must ensure that all practice is based on the best available evidence and that the nursing time required for any procedures can be clearly justified. GRV measurements have not been shown to accurately reflect gastric emptying and risk of aspiration2,3 and the volume obtained on aspiration will vary depending on the position of the nasogastric tube in the stomach, the position of the patient when the aspirate is taken, the size of the nasogastric tube, frequency of aspiration and the technique used for aspiration.9,31 Of note, this survey reports that only 28% of UK adult ICUs have a guideline in place regarding the technique that should be used for aspiration of GRV. This lack of standardised practice is likely to add to the inaccuracies of GRV volumes obtained. However, even if techniques for GRV measurements were standardised, this would not account for individual patient variation in what could be considered a ‘normal’ gastric aspirate. Gastric aspirates are made up from a combination of endogenous GI secretions and exogenous fluids (feed, medications, water flushes), both of which vary between patients. In health endogenous secretions are estimated to be around 4000–5000 ml per day. However, in critical illness the use of proton pump inhibitors, absence of chewing, smell and taste in the digestive process and post-pyloric feeding can dramatically alter endogenous production of GI secretions. 31
The results of our study are similar to a previous survey conducted in 2018 investigating nursing practices around GRV measurement in Turkey and Belgium. 33 That study reported measuring GRV to be commonplace with 98% of respondents reporting doing so. Over 50% of nurses reported measuring GRV to monitor for feed tolerance with threshold values varying; 45% of respondents reported a GRV of 50–200 ml to be considered high. Interestingly, more experienced ICU nurses appeared to measure GRV more commonly than their less experienced colleagues. 33 If GRV measurements in the ICU are to be abandoned, ICU nurses must be reassured as to the safety of this course of action.
Work carried out in the paediatric population investigating practitioner and parent views on not measuring GRV has shown that a trial of not measuring GRV in this population is acceptable; however potential concerns regarding increased risk of aspiration, pain and delayed diagnosis of GI problems were identified and this needs careful consideration in future work looking at ongoing GRV measurement.21,22,34 A large-scale prospective randomised controlled trial investigating not measuring GRV is due to commence shortly in the paediatric population 23 and a similar study would be beneficial in the adult population.
Measurement of GRV has been shown to be the most significant factor in failure to meet nutritional targets in the adult ICU population, with a 38% increase in risk of having a low ratio of delivered versus prescribed calories. 11 This combined with other factors that hinder enteral feed provision in the ICU setting such as fasting for procedures contributes to a frequent failure to meet EN targets in critical care patients12,35 and is associated with impaired clinical outcomes.24 –26 Taking this into consideration, in addition to the evidence on poor validity of GRV measurements2,3 and lack of detrimental consequences to not measuring GRV,4,15,16 raises a question around the ongoing use of GRV to guide enteral nutrition delivery. Further research on the usefulness of ongoing GRV measurements in adults should build on the work being undertaken in the paediatric population.21 –23 In the meantime, strategies for catching up on lost nutrition due to feeding interruptions such as volume-based feeding should be promoted in critical care units to ensure adequate nutritional delivery if feeding is temporarily paused. 36
There are some limitations to this study. Firstly, the survey does not represent all adult ICUs in the UK and practice may differ in non-responder units. Secondly, the survey was primarily completed by ICU dietitians with only 7% of surveys completed by nursing staff. In the paediatric survey of GRV monitoring practices in the UK, a more collaborative approach to survey completion was seen with nurses and dietitians completing 96% of surveys and doctors completing 92%. The findings may therefore represent perceived practice from dietitians rather than actual nursing or medical practice. Repeating this survey and ensuring responses are completed by nursing and medical staff in addition to dietitians may produce different results. However, in the UK the role of the critical care dietitian is well developed, 37 and dietitians are well placed to comment on nutritional practices within the ICU.
Conclusions
Measurement of GRV is the most common method of determining enteral feeding intolerance in adult ICUs in the UK. The practice continues despite evidence of poor validity and reproducibility of this measurement. This is likely to be due to lack of alternative bedside measures of feed tolerance. However, the ongoing measurement of GRV is likely to be contributing to inefficient use of nursing resource and failure to meet nutritional targets. Further research should be undertaken into the benefit of ongoing GRV measurements in the adult ICU population and alternative markers of enteral feeding intolerance such as monitoring of symptoms, functional measures of GI dysfunction and biomarkers of feeding intolerance.
Footnotes
Authorship statement
Authors made the following contribution to the manuscript: BJ, LVM and PCC formulated the question; BJ adapted the survey from previously published work; BJ piloted and set up the web-based survey, contacted non-responder units and collated the data; BJ conducted the analysis of results and drafted the manuscript; BJ, LVM and PCC critically reviewed and revised the manuscript; BJ, LVM and PCC provided final approval of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: None of the authors has any conflict of interest to declare. Dr Marino reports an unrestricted grant from Abbott Laboratories for the development of Pedi-R-MAPP, in addition to honoraria from Abbott Laboratories and Danone-Nutricia outside the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Bethan Jenkins, Pre-Doctoral Clinical Academic Fellow (NIHR301168) is funded by Health Education England (HEE)/National Institute for Health Research (NIHR) for this research project. The views expressed in this publication are those of the author(s) and not necessarily those of the NIHR, NHS or the UK Department of Health and Social Care. Luise Marino is supported by the Research Leaders Programme, Southampton Academy of Research, University Hospital Southampton NHS Foundation Trust.
