Abstract
The COVID-19 pandemic presented clinical and logistical challenges in the delivery of adequate nutrition in the critical care setting. The use of neuromuscular-blocking drugs, presence of maxilla-facial oedema, strict infection control procedures, and patients placed in a prone position complicated feeding tube placement. We audited the outcomes of dietitian-led naso-jejunal tube (NJT) insertions using the IRIS® (Kangaroo, USA) device, before and during the COVID-19 pandemic. NJT placement was successful in 78% of all cases (n = 50), and 87% of COVID-19 cases. Anaesthetic support was only required in COVID-19 patients (53%). NJT placement using IRIS was more difficult but achievable in patients with COVID-19.
Introduction
Patients with COVID-19 requiring respiratory support presented logistical and clinical challenges, and, whilst enteral nutrition was tolerated, placing patients in a prone position, and high-dose neuromuscular-blocking drugs resulted in high gastric residual volumes. 1
Bedside placement of naso-jejunal tubes (NJT) has evolved over the last 20 years with the development of electromagnetic devices, 2 and more recently the IRIS device (Kangaroo, US) which allows direct vision of the internal anatomy. 3
We conducted a retrospective audit to analyse outcomes of a dietitian-led service using the IRIS device.
Methods
All patients who had an IRIS NJT placed between January 2019 and April 2021 were included. Data were collected from dietetic, medical and intensive care records and subdivided to compare patients with, and without COVID-19 (Local audit approval 1098). Data were analysed in SPSS Version 26 (IBM, USA)
The IRIS NJT placement device (Kangaroo, USA) is a 10 F gauge, 140 cm polyurethane feeding tube with a camera on the tip of the tube. The feeding tube connects to a bedside console, which provides real time visualisation of anatomical markers to aid progression of the tube. 10 mg of metoclopramide was administered intravenously 30 min before tube placement as a pro-kinetic and all placements were confirmed by abdominal X-ray, assessed by two senior physicians (Figure 1). All NJT placements were carried out by a single operator (MEP). Maxillo-facial oedema was identified by the managing clinical team, and included oedema of both the lips and oral mucosa.
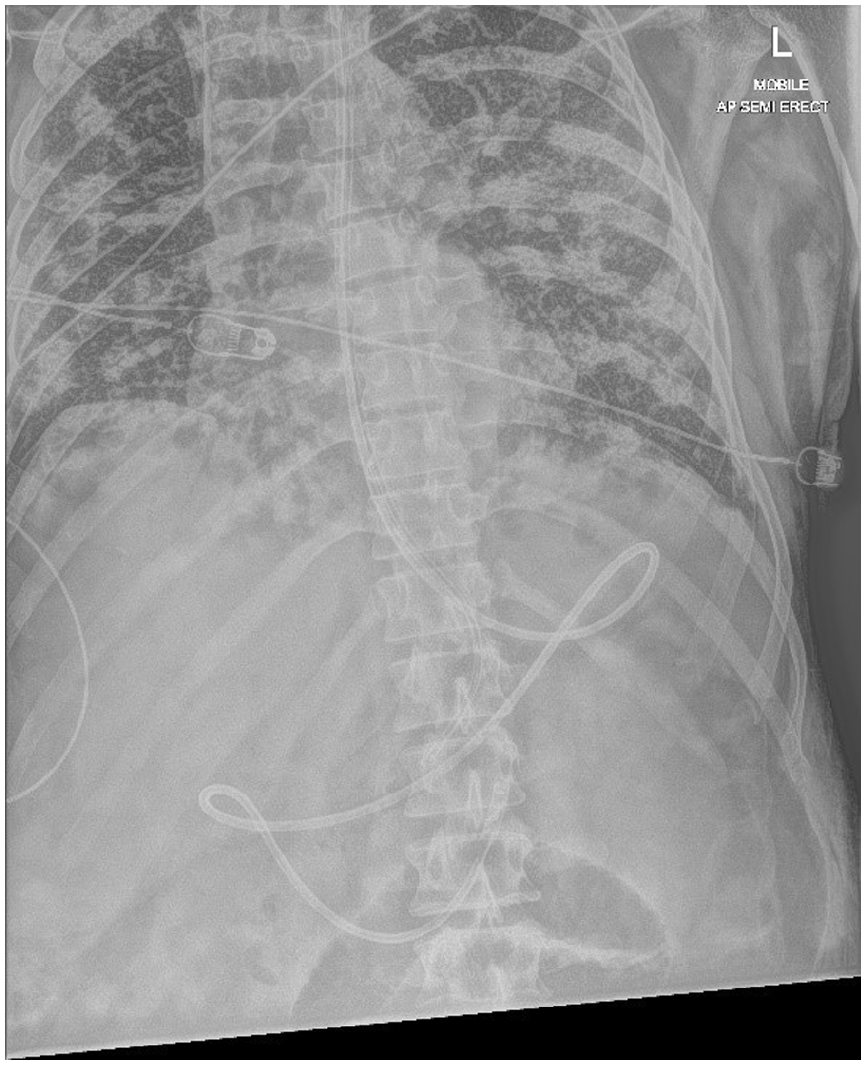
Naso-jejunal feeding tube placed in a patient with COVID-19.
Results
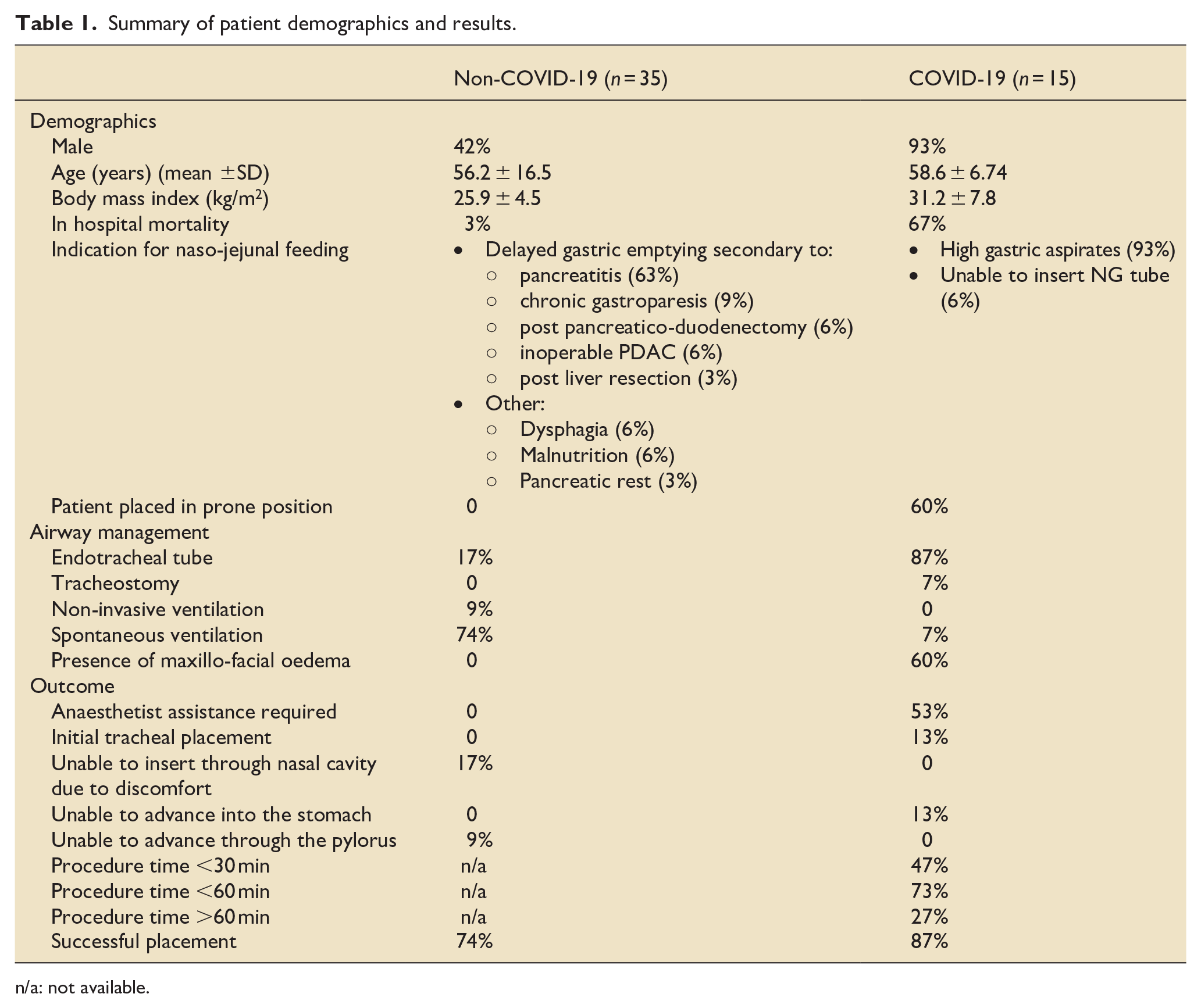
Fifty-three cases were identified, with 50 complete data sets. In three cases medical notes were not available, and these cases were excluded. Patients with COVID-19 were predominantly male, had a higher body mass index (BMI) and higher mortality (Table 1).
Summary of patient demographics and results.
n/a: not available.
Delayed gastric emptying was the primary reason for NJT insertion in both groups. Patients with COVID-19 were more likely to be intubated, and anaesthetic support using forceps and laryngoscope was required to facilitate placement of the NJT into the oesophagus in 53% of cases. In all of these cases this was due to extensive maxillo-facial oedema and secretions, which prevented identification of anatomical markers and progression of the tube.
Patient discomfort prevented placement in 17% of the non-COVID-19 cohort, where only 26% of patients were sedated. Tracheal placement only occurred in the COVID-19 cohort (13%), but was identified in real time with visualisation of the carina and corrected immediately.
Placement was successful in 87% of COVID-19 patients and 74% of non-COVID-19 patients (78% total cohort).
In 13% of COVID-19 patients, tubes could not be advanced through the oesophagus, and 11% of the non-COVID-19 cohort tubes failed to pass through the pylorus (Table 1).
Discussion
Bedside NJT placement using IRIS presents a viable alternative to endoscopic and fluoroscopic placement in both normal clinical practice and in a complex medical environment, without the risks of moving an isolated patient through the hospital to radiology, or bringing in a large endoscopy stack into an overcrowded medical environment.
Dietitians can be trained to carry out bedside placement and this is documented in other specialities. 4 Technical success in this cohort was 78% of all cases, which is consistent with other studies where success rates of 75% with IRIS 3 and 83%–84% with other devices are reported.5,6
Procedure time was longer in patients with COVID-19 when compared to published data. Median procedure time is documented at 13.5 min with IRIS 3 compared with 22.07 ± 5.78 min for ultrasound, 7 14.8 ± 4.9 min for endoscopic 8 and 14.9 ± 2.9 fluoroscopic placement, 8 whereas in our cohort only 47% of COVID-19 cases took less than 30 min, and 27% took more than 60 min. Procedure time was not collected in the non-COVID population, which is a limitation of this data set.
In 53% of COVID-19 patients, a laryngoscope and forceps were required to place the NJT into the oesophagus. This, in addition, to the restrictions of additional PPE, and the high prevalence of sedated patients (and therefore less patient discomfort), resulted in longer procedure times.
The use of vasopressors, mechanical ventilation, and an APACHE II scores >20 are associated with lower rates of technical success, 9 thus it could be anticipated that success rate would be lower with the COVID-19 population, but this was not the case within our cohort. We used a prokinetic (Metaclopramide) which has been associated with more successful NJT placement. 9
The limitations of this study include the small sample size, and lack of complete outcome data in the non-COVID-19 population and this should be included in further prospective studies.
Conclusion
Bedside placement using the IRIS device was more complicated, but is an effective technique to facilitate NJT insertion in patient with or without COVID-19.
Footnotes
Author contributions
MEP conceptualisation, methodology, investigation, writing – original draft, RK, RL, and AEF writing – review and editing, supervision. JK-B, JZ and AP writing – review and editing
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MEP has received Honoria for teaching from Viatris, Nutricia Clinical Care, Merck and Sanofi, RK has received Honoria for teaching from Viatris.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
