Abstract
Background:
Personal protective equipment has important environmental impacts, assessing these impacts is therefore an important element of personal protective equipment design. We applied carbon footprinting methodology to Bubble-PAPR™, a novel, part-reusable and part-recyclable powered air-purifying respirator, designed at our institution. Current guidance states that disposable respirator masks can be worn for 1-h in the United Kingdom, whilst the Bubble-PAPR™ allows prolonged use.
Methods:
Following a detailed use-case analysis, the carbon footprint of each component was estimated using a bottom-up (attributional) cradle-to-grave process-based analysis. Modelling considered the use of virgin or closed loop recycled polyvinyl chloride for the disposable hood element, and disposal via infectious or recycling waste streams to estimate a per-use carbon footprint.
Results:
The per-use carbon footprint with manufacture from virgin polyvinyl chloride and disposal via incineration is 0.805 kgCO2e. With nine cycles of closed loop recycling and manufacture of the polyvinyl chloride hood (10 uses), the carbon footprint falls to an average of 0.570 kgCO2e per use.
Conclusion:
Carbon footprinting may contribute to the value proposition of this novel technology. We estimate that a single Bubble-PAPR™ use has a higher carbon footprint than disposable respirator mask-based PPE. However, this is mitigated in circumstances when multiple disposable mask changes are required (e.g. prolonged use) and use may be justifiable when user comfort, visualisation and communication with patients and colleagues are essential.
Introduction
Personal protective equipment (PPE) has been a constant feature of the COVID-19 pandemic. The worldwide environmental impacts of the unprecedented amount of PPE used in health and care settings are unknown. However, the carbon footprint of PPE is estimated to have totalled 106,478 tonnes of carbon dioxide equivalents (CO2e) in England alone during the first 6 months of the pandemic. 1
Healthcare workers caring for patients with high-consequence infections such as SARS-CoV-2 are required to wear PPE during their clinical work. A system of PPE usually includes an impermeable long-sleeved gown, suit or apron, gloves, and respiratory protective equipment (RPE). If there is a risk of exposure to aerosolised infectious material, eye protection and a respirator are also required. Unlike standard surgical masks, respirators filter small, aerosolised particles. The most commonly used respirators in the UK are disposable filtering facepiece (FFP) masks made from layers of synthetic non-woven material with interleaved filtration layers. They can be classified by their filtration efficiency from FFP1 (lowest) to FFP3 (highest). 2 Though reusable respirator masks are available, single-use items remain more common, and are disposed into the carbon intensive infectious clinical waste streams. Respirators are difficult to recycle due to their layered multi-material construction, contributing to an unprecedented rise in PPE-related waste. 1
During the initial stages of the COVID-19 pandemic some clinical settings, such as the intensive care unit (ICU), required near constant use of respirators for patients with high-consequence respiratory infections due to the perceived risk of aerosol exposure associated with invasive ventilation. 2 Although respirators are not universally required in the ICU currently they continue to be worn for some procedures such as airway suctioning and tracheostomy insertion, and by individuals who may be at higher risk from infection. Much of the RPE worn in ICUs during the COVID-19 pandemic was designed for non-healthcare (predominantly industrial) settings. To work effectively, a respirator mask must have a tight-fitting seal against the skin and fit the contours of the wearer’s face precisely. This renders respirator masks uncomfortable for many healthcare workers, particularly when worn repeatedly or continuously. 3 Multiple brands and versions of face masks are required to ensure appropriately fitted masks are available for the different face shapes of a diverse workforce, and some staff members are unable to find an appropriately fitted respirator from existing stocks. 4 Furthermore, respirator masks inhibit communication by obscuring the mouth; compounded when visors or head coverings are also required.
Whilst the COVID-19 pandemic highlighted many research achievements, there remains a lack of comfortable RPE which provides sufficient protection for high-risk environments, and the environmental impacts of PPE remain a pressing concern. In recognition of this challenge, the Greener Net Zero strategy incorporates research aimed at reducing the carbon footprint of PPE, particularly face masks. 5 In an attempt to address some of the clinical limitations, supply-line concerns and environmental impacts of existing respirators, a novel item of RPE was designed and created during the pandemic by front-line healthcare staff at Manchester University NHS Foundation Trust (MFT) in collaboration with product designers and engineers at Designing Science Ltd (Middlesex, UK) and The University of Manchester. Bubble-PAPR™ is a powered air-purifying respirator (PAPR) designed to: protect staff from environmental aerosol exposure; facilitate communication between staff and patients; be more comfortable to wear than FFP3 face masks; and, importantly, be part-reusable and part-recyclable. The reusable element has a wipeable outer surface and a simple shape, so can be cleaned and reused by different staff members. This offers a significant advantage over other PAPR systems adopted for healthcare which are typically single use, or have a complex shape that is difficult to clean. The design, development and testing of Bubble-PAPR™ are described elsewhere.6,7
A carbon footprint is the sum of all direct and indirect greenhouse gas (GHG) emissions which are produced throughout the lifecycle of a product and are expressed in carbon dioxide equivalents (CO2e). 8 These data should inform the value proposition of a product when considering the ‘triple bottom line’ approach to social, financial and environmental impacts. Similar work has been undertaken to assess anaesthetic drugs, laryngoscopes and surgical techniques, as summarised by McGain et al.9,10 In this study, we aimed to calculate the carbon footprint per use of Bubble-PAPR™. Our objectives were to estimate the carbon footprint over its lifecycle, including associated consumables, based on anticipated usage and alternative disposal scenarios.
Methods
The carbon footprint of each component of Bubble-PAPR™ was estimated using a bottom-up (attributional) carbon footprint analysis. This type of analysis takes account of the extraction of raw materials, manufacturing, transport and disposal of the components. 11 Once introduced to the hospital environment, a detailed use-case analysis was undertaken (IRAS ID:288493; REC Ref:21/WA/0018; ClinicalTrials.gov NCT04681365). The results of this part of the project have been reported elsewhere; all available information from the implementation study was used to inform the carbon footprint. 7
The Bubble-PAPR™ consists of several reusable elements and a single-use polyvinyl chloride (PVC) hood. The re-usable elements include a neck collar made from integral skin polyurethane foam (a material with a foam core and a smooth outer skin, also used to create operating theatre table mattresses). The collar draws air in through a filter via an impellor powered by an external battery. The semi-rigid hood is pulled over the collar before donning and is secured by integrated straps. When worn, the hood fully encircles the wearer’s head. See www.bubble-papr.com and Figure 1. 6 The reusable elements are cleaned with a disinfectant wipe before and after use. The implementation study used Clinell® Universal Wipes (GAMA Healthcare, Hemel Hempstead, UK), supplied in packs of 200. 7 The single-use hood is destroyed during doffing.
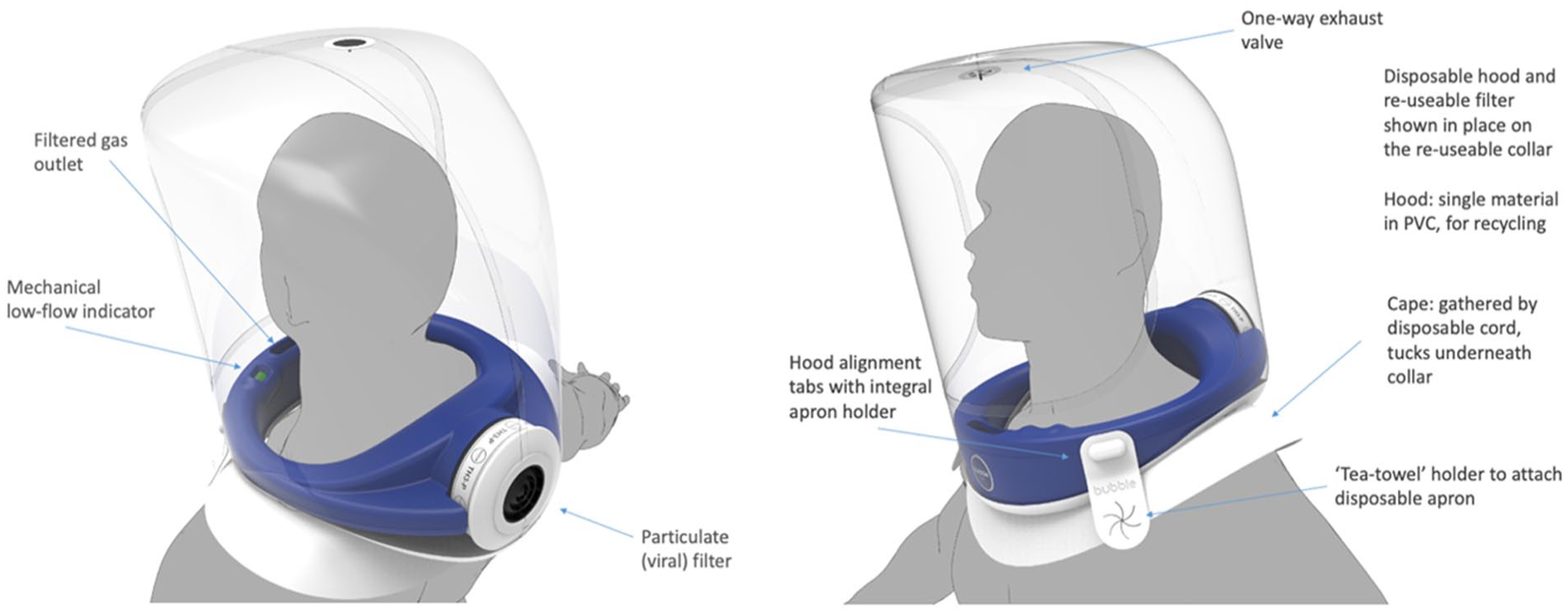
Annotated schematic of the Bubble-PAPR™.
A spreadsheet package (Google sheets, Google LLC, Mountain View, California, USA) was used to perform the calculations. Specialised lifecycle assessment software (e.g. SimaPro, PRé Sustainability BV, Amersfoort, Netherlands) was not used.
Materials
The material compositions of the Bubble-PAPR™ collar were obtained from the prototype manufacturer and/or by examination. The hood is made of high-grade PVC which can be recycled following doffing directly into a specific container. An external lithium polymer battery is used to power the collar. The Inventory of Carbon and Energy (ICE) Summary, V3.0 and UK Government Greenhouse Gas conversion factors for company reporting 2021 databases were used to select the closest material match to calculate the weight-based carbon footprint of each item over its lifecycle.12,13 These values, together with information on the expected usage of each component, were used to calculate the estimated carbon footprints.
Manufacture modelling assumptions
We assumed that the Bubble-PAPR™ (collar, including all re-usable elements and hood) would be manufactured from virgin materials in UK factories. Electricity use for Bubble-PAPR™ manufacture has not been measured. However, based on similar work, we assessed that the contribution of electricity would be small. For example, Rizan et al. calculated 0.016 kWh required for electricity for manufacturing a single-use FFP3 facemask and visor, which, based on UK electricity supply, equates to emissions of 0.00467 kgCO2e, around 1.5% of total emissions for the products. 1 Therefore, we added an additional 1.5% to the carbon footprint of each component of the Bubble-PAPR™ in our modelling, to account for electricity use in manufacture.
The Bubble-PAPR™ prototype used in this study was manufactured in the UK, and transportation distances have been calculated from the respective manufacturing locations (S40 and WS10) to Wythenshawe Hospital, Manchester, UK. We have assumed transportation by an averagely laden, average-sized, rigid lorry and obtained emissions factors from the UK Government Greenhouse Gas conversion factors for company reporting database. 13 Delivery frequency was estimated at three-monthly for the single-use hoods (which require replenishment as they are used) and 12-monthly for the reusable components (the minimum expected life-span of the reusable elements).
For packaging used for the PVC hood, we assumed that the carbon emissions and weights are the same as that estimated for apron packaging, comprising of a low density polyethylene packaging film and a corrugated board box (due to their similar sizes and packaging style) in the analysis by Rizan et al. 1
Per-use modelling assumptions
The collar uses a rechargeable 5V lithium polymer battery. A typical 2-h use of Bubble-PAPR™ is estimated to use 10% of the capacity of a fully-charged battery, with the battery then recharged via UK mains electricity. 7
End-of-life disposal modelling assumptions
Our modelling considered that the reusable collar of the Bubble-PAPR™ would be used for a 12-month lifespan, then disposed-of. At the end of its lifecycle, the collar can be disposed of in domestic waste or recycling where available. Information regarding the carbon footprint of hospital waste streams has been gathered from a recently published study based in three hospitals in one UK NHS organisation, which likely provides the most accurate emissions data for hospital waste streams in the UK. 14
Scenario modelling assumptions
The hood is designed for single use, prior to disposal via either via the infectious waste stream or recycling (Figure 2). It could be made from virgin PVC or recycled PVC, providing that sufficient high-grade recycled material is available. Infectious waste streams may undergo high temperature incineration or an alternative treatment (e.g. chemical disinfection) process prior to disposal alongside other waste streams which may enter landfill or undergo low temperature incineration. 14 For the purpose of this study it is assumed that the infectious waste stream undergoes autoclave decontamination followed by low temperature incineration with energy from waste recovery. We have modelled two scenarios: Scenario 1 models manufacture from virgin PVC and disposal via the infectious waste stream; scenario 2 models closed-loop recycling of single use hoods initially manufactured from virgin PVC. Polyvinyl chloride obtained from open loop recycling has not been considered as this may not be of sufficient quality.
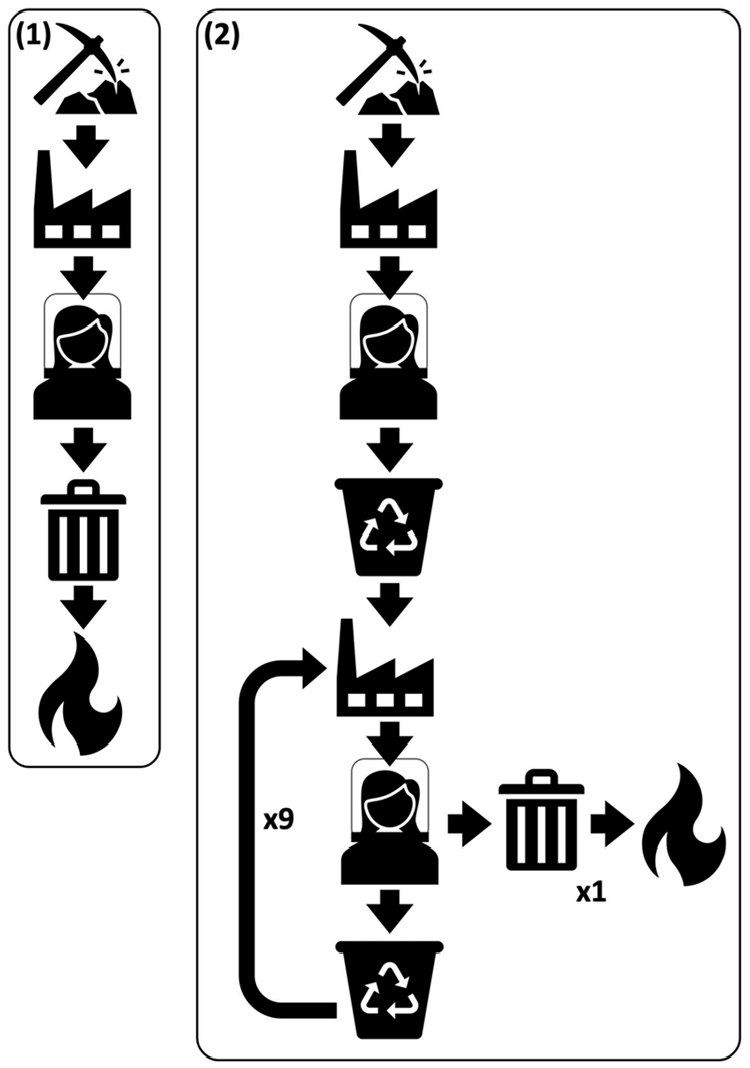
Manufacture/disposal options modelled. (1) hoods manufactured from virgin PVC and disposed of via the infectious waste stream; (2) closed-loop recycling whereby hoods, initially made from virgin PVC, are recycled to produce new hoods an average of nine times, with eventual disposed by incineration.
Our modelling for usage is based on observations in the implementation study set in a tertiary NHS hospital with most users working in the ICU. 7 We estimated each collar would be used 10 times per 24 h, with each hood estimated to be used for 2-h stints. Based on this model, in a 1-year period, each collar would be used with 3650 hoods. The collar would require cleaning with a disposable disinfectant wipe before and after each of these uses.
Exclusions
Packaging for the reusable components has not been included as packaging is yet to be determined, nor has the container in which the recyclable hood could be collected following doffing. These emissions are judged to be small compared to the recyclable elements themselves.
Results
The carbon footprint associated with a single use of Bubble-PAPR™ for each of the proposed scenarios was calculated (Table 1). The elements which do not vary across both the scenarios have a carbon footprint of 0.062 kgCO2e per use (see Supplemental Appendix for full calculations) and includes: a proportional amount of the manufacture and disposal of the re-useable elements, hood packaging, two disinfectant wipes, battery charging, and transport of main Bubble-PAPR™ components to the hospital.
Materials and associated emissions per use of the Bubble-PAPR™.

Representative calculation based on an assumed average of nine cycles of closed-loop recycling (10 uses). See Supplemental Appendix for full calculations.
For scenario 1, where there is no recycling involved, the total carbon footprint per use is 0.805 kgCO2e. The majority (92%) of emissions are attributed to the manufacture and disposal of the single-use hood.
Figure 3 demonstrates the cumulative average of carbon emissions with each use of the Bubble-PAPR™ with closed-loop recycling of the hood, following an initial use with a hood manufactured from virgin PVC, and including one disposal via the infectious waste stream (see Supplemental Appendix for calculations). Over 30 uses, the carbon emissions fall from 0.805 kgCO2e (virgin PVC hood disposed of by incineration) to 0.553 kgCO2e (after 30 uses, involving 29 closed-loop recycling processes and one disposal via the infectious waste stream). The most significant carbon emissions reduction occurs with the first cycle, with a fall in the cumulative average carbon emissions of 0.130 kgCO2e, subsequent reductions become smaller with each cycle. We used the figures for nine cycles of recycling (10 uses total) for scenario 2, with a cumulative average carbon footprint of 0.570 kgCO2e per use, however more or fewer closed-loop cycles may be achievable in real-world use.
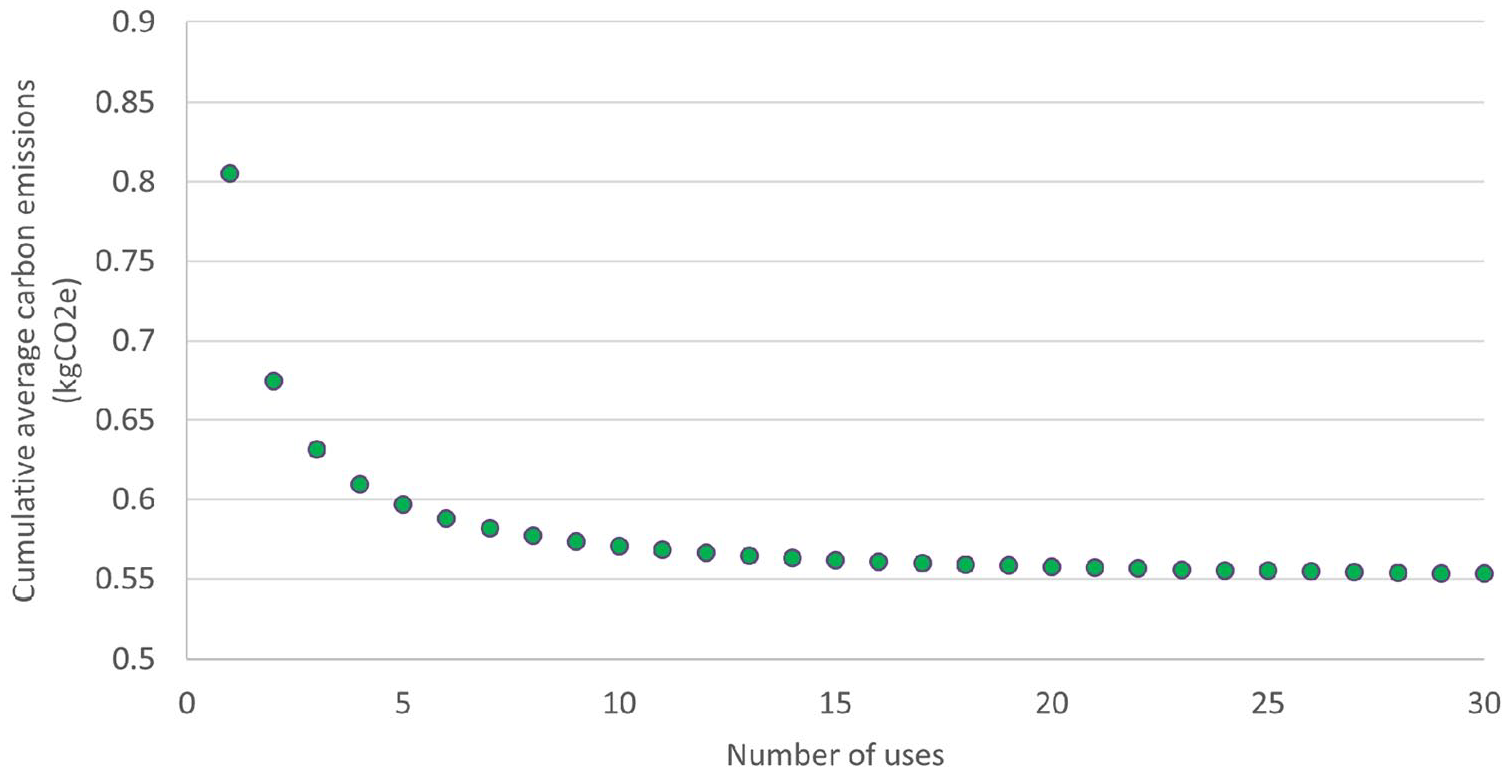
Cumulative average carbon footprint per use of the Bubble-PAPR™ in the closed-loop recycling scenario (scenario 2).
Discussion
Bubble-PAPR™ fulfils the function of both a respirator and full-face protection. A single 2-h use of the Bubble-PAPR™ is associated with emissions of between 0.570 and 0.805 kgCO2e, depending on waste handling and production material. Of this, the majority of emissions are due to the single-use PVC hood. Existing studies have calculated the carbon footprint per use of a disposable FFP respirator to be 0.125 kgCO2e and a disposable face shield to be 0.231 kgCO2e (total 0.356 kgCO2e). 1
The UK Health and Safety Executive (HSE) recommends that respirator masks be used for no more than 1 h, 15 whilst loose-fitting PAPRs can be used for extended periods. Therefore, Bubble-PAPR™ may have a lower carbon footprint than alternatives if HSE guidance is followed. For example, a 2-h stint would require the wearer to doff and don alternative RPE at least twice – a total carbon footprint of 0.712 kgCO2e. This would mean that the Bubble-PAPR™ would have a lower carbon footprint if manufactured and recycled via closed loop recycling and a marginally higher carbon footprint if manufactured from virgin PVC and disposed of in the infectious waste stream (Figure 4).
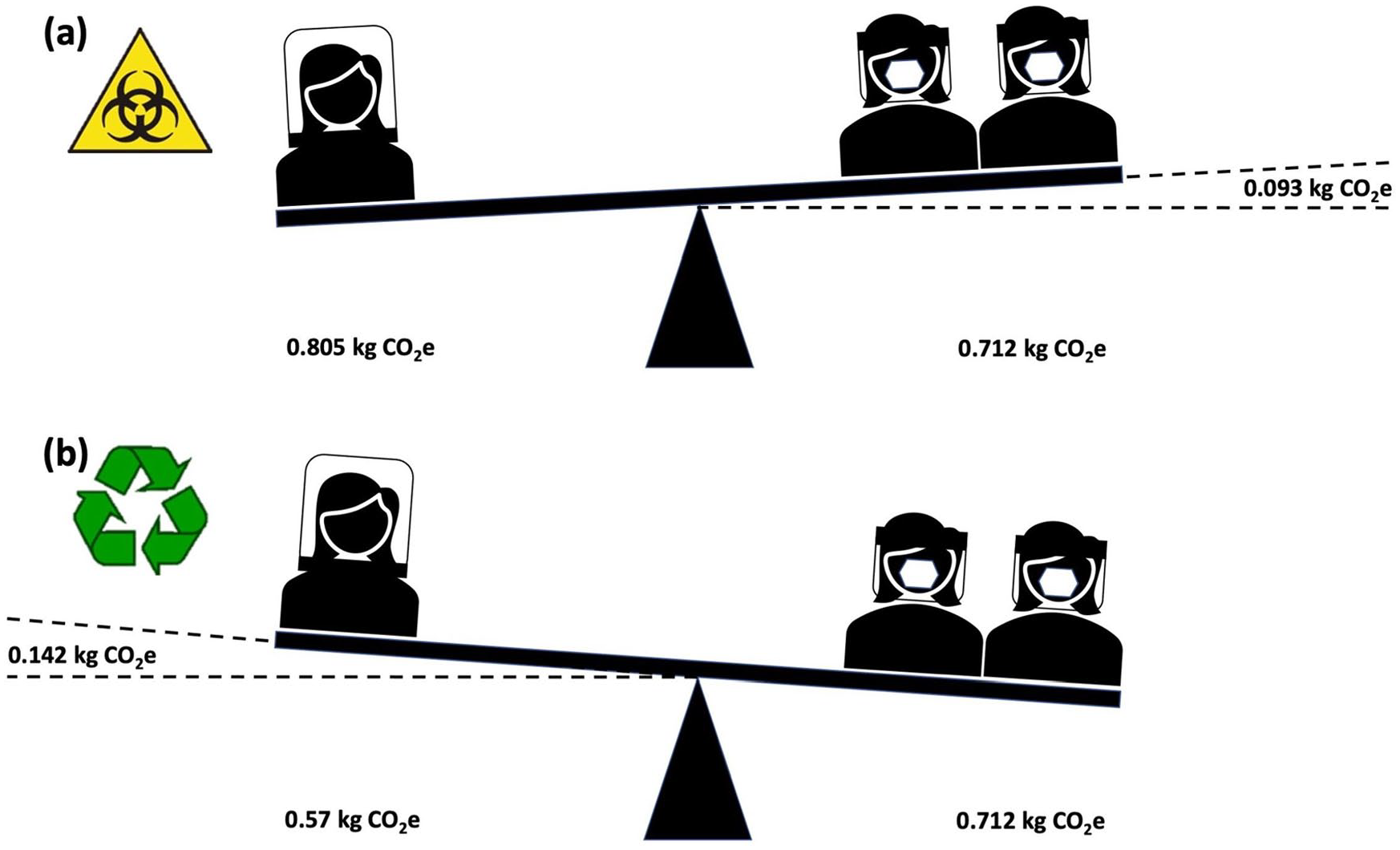
The carbon footprint of 2 h of use, based on HSE wear limits of less than 1 h for respirator masks and prolonged use for PAPRs: (a) comparison with scenario 1 and (b) comparison with scenario 2.
Despite the HSE guidelines for the duration of use, clinical practice does not always reflect regulatory ideals, and some studies have found that single-use respirator masks are often worn for longer than 1 h.3,15 ‘Real world’ usage may therefore result in a Bubble-PAPR™ carbon footprint higher than that of disposable respirator mask-based PPE (Figure 5).
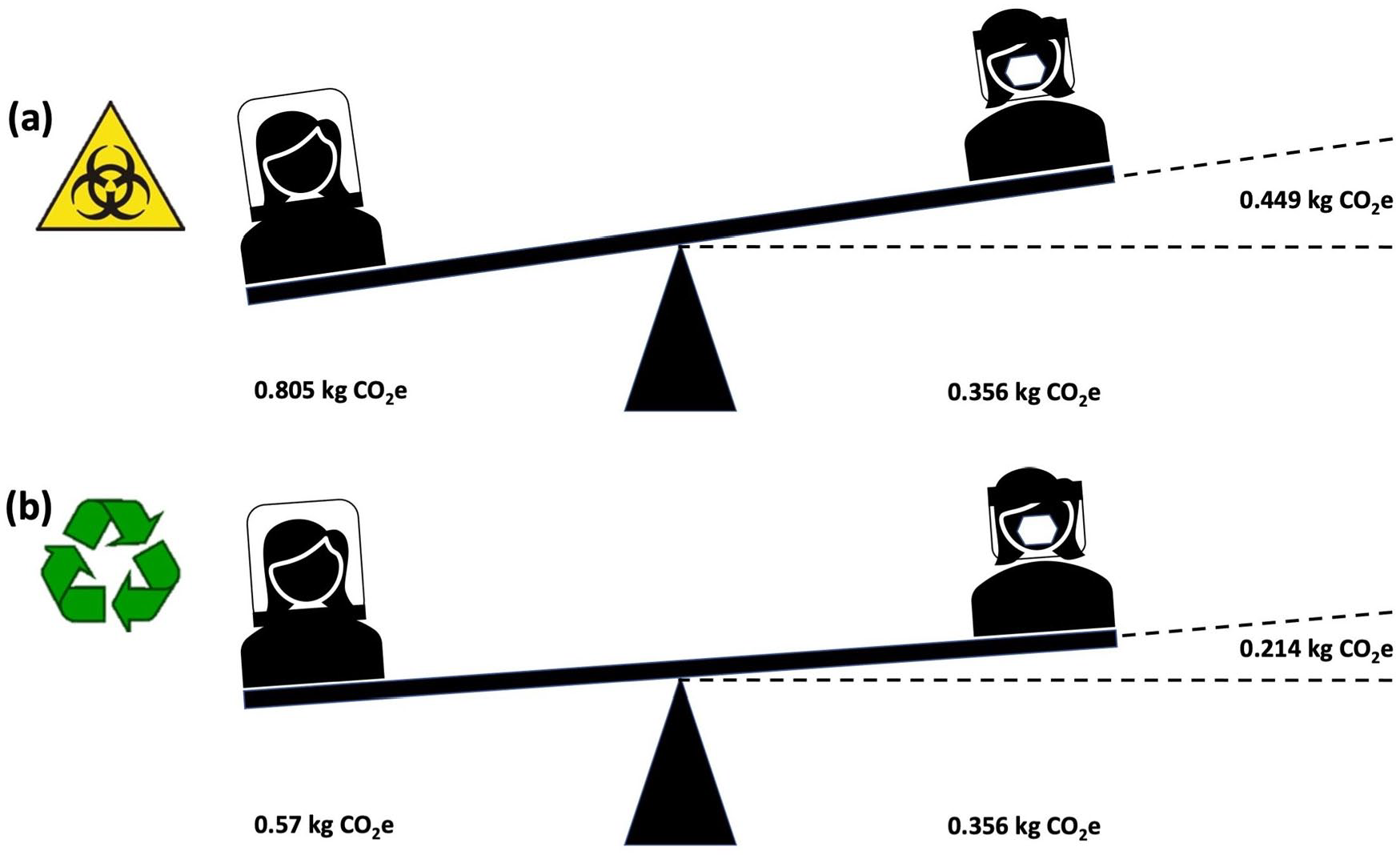
The carbon footprint of a 2-h period of use, assuming a single FFP mask and face shield or Bubble-PAPR™ worn for the whole period: (a) comparison with scenario 1 and (b) comparison with scenario 2.
Closed-loop recycling could significantly decrease the emissions associated with single-use PVC. Recycling of medical PVC (e.g. oxygen masks) has already been implemented in UK healthcare, providing a viable potential route to recycling for Bubble-PAPR™. 16 It is likely, however, that each time the hood undergoes closed loop recycling, some additional virgin PVC will be required due to degradation of the original material, increasing the overall carbon emission.
Where PVC recycling is available and HSE time limits are observed, the adoption of Bubble-PAPR™ would be preferable from a climate perspective for scenarios involving more than 1 h of continuous use. 17
The comparison of carbon footprint does not consider the other potential benefits of the Bubble-PAPR™ over disposable PPE alternatives which may justify the higher carbon footprint in some circumstances. For example, where tight-fitting face masks are unsuitable, such as when a staff member has failed fit testing for available respirator mask designs, sustained facial injuries relating to face mask use or has facial hair, meaning that a tight mask seal cannot be obtained. 4 Powered air-purifying respirators may have advantages over face masks with comfort and communication, which are amplified in critical scenarios. In cases where either system could be used, considering the other aspects of the ‘triple bottom line’ – involving the social and economic impacts, in addition to environmental aspects, may provide a useful theoretical framework for use 8,18:
Planet – environmental impacts
From a carbon footprint perspective, the Bubble-PAPR™ currently does not offer more environmentally sustainable PPE than alternative disposable devices in most scenarios. However, it can be the lower-carbon option where staff members must remain in PPE for prolonged periods of time, as is common in the ICU. The Bubble-PAPR™ would remain comfortable, preventing the need to repeatedly don and doff single-use PPE. 7 This may be of particular benefit in very long-duration use scenarios (e.g. airway surgery): if the Bubble-PAPR™ were to be worn in place of three single-use FFP3 facemasks and visors, the carbon footprint for the Bubble-PAPR™ would be lower, even in the absence of recycling.
Public Purse – economic impacts
The Bubble-PAPR™ has been designed to be no more expensive per use than currently available FFP masks, which are reported to cost between £3 and £5 per item in the NHS setting.15,19 This is achievable because the Bubble-PAPR™ hood is made of a single material and can be mass-produced with minimal assembly. Factoring in the cost of disposable face shields would likely mean that the Bubble-PAPR™ has a small financial advantage over disposable PPE systems. Furthermore, because fit testing is not required, there is no loss of devices to this process.
People – social impacts
It has been repeatedly noted throughout the COVID-19 pandemic that the use of PPE has impacted the ability to communicate effectively.20 –22 Communication is vital in many situations, particularly critical scenarios encountered in the ICU. Some staff and patients rely to some extent on lip reading in addition to hearing. 22 Furthermore, being able to visualise staff members’ faces may be therapeutically important in some circumstances – such as during speech and language therapy where facial expressions are used to convey techniques to patients. Using PAPRs could therefore allow more effective and efficient communication and delivery of care. Scarcity of FFP-3 facemasks during the COVID-19 pandemic led to huge anxiety amongst healthcare staff. 23 Lack of supply chain resilience contributed to this lack of PPE. The introduction of Bubble-PAPR™, particularly in the case of closed loop recycling, could ease pressure on supply chains during future demand surges.
It is our assessment that the overall value proposition of Bubble-PAPR™ (vs disposable FFP-based PPE) depends on the reconciliation of the environmental and social impacts under most usage conditions. It may, for example, be justifiable to use the Bubble-PAPR™ when being able to visualise the healthcare worker’s face is important for therapeutic reasons or when communication is otherwise challenging. Furthermore, staff comfort may be a priority when involved in a complex case or when recovering from facial injuries associated with respirator mask use.3,4 In the short term, there may be an acceptable environmental trade-off favouring staff comfort and effective delivery of healthcare. All healthcare comes at an environmental cost and calculating and characterising such costs are important for new devices and systems in the future.
Study limitations
The limitations of our study include that it is solely based on carbon footprinting, and there are other environmental considerations which would require further investigation to assess the full environmental impact of the Bubble-PAPR™. For example, the manufacture and incineration of PVC involves the production of potentially harmful by-products, which may contribute to air and groundwater contamination if released into the environment. 24 Location of recycling centres are currently not confirmed, but candidate sites have been identified, with similar transport implications to non-recycled disposal. The carbon footprint of manufacturing was derived from other work,1,12,13 and although uncontaminated PVC (e.g. surplus material from manufacturing processes) is suitable for closed-loop recycling without loss of quality, additional virgin PVC may need to be added during recycling of used Bubble-PAPR™ hoods due to loss, contamination or degradation of original material.25,26 The impact of these factors would need to be quantified in future work based on the production version. We did not have access to specialised life cycle analysis software for this study, so instead used values derived from emissions factor databases.12,13 This means that our modelling lacks some of the advanced analytical features offered by these packages (e.g. Monte Carlo analysis, which offers an indication of the certainty of carbon footprint estimates). However, this was accounted for in-part through the simulation of different use scenarios. Finally, emissions related to the packaging of reusable items and collection of recyclable materials, which has yet to be defined at the time of the study, have not been included so the carbon footprint would increase once these are included, however the implications of these are estimated to be is minimal given the prediction of 3650 uses per re-usable device.
Our closed-loop recycling model (scenario 2) assumes that each disposable hood will be recycled an average of nine times. However, we acknowledge that there are human factors-related issues whenever clinical staff are relied upon to segregate waste. Various behavioural ‘nudges’ have been found to improve waste segregation at source, 27 and the introduction of this equipment should be accompanied by a recycling system designed to maximise yield. Existing healthcare-related PVC recycling schemes that use re-usable collection bins demonstrate the viability of this approach, however data on the proportion of potentially-suitable PVC recycled are not available, to our knowledge. 16 We suspect that consistent recycling may be more likely in closely-controlled environments, such as intensive care units, where staff are familiar with specialised equipment handling. Nevertheless, some circumstances may make disposal by incineration unavoidable – for example, if the Bubble-PAPR™ is used by patient transfer teams to locations where recycling facilities are not available institutions.
The future
Our modelling has identified the carbon hotspots within the lifecycle of the Bubble-PAPR™, enabling further design modifications to target these areas. The current design does not allow for reuse of the hood as it is destroyed during removal. Future modifications may allow the introduction of a reusable hood, but this will rely on appropriate washable materials and decontamination procedures. As with PVC recycling, there are examples of reusable fabrics being introduced into UK healthcare instead of disposable options. 28 However, there remain logistical challenges with the large-scale cleaning, drying and storage. Further developments could see the reusable collar being refurbished at the end of its lifespan, prolonging use, and further reducing its carbon footprint. We anticipate that if these measures were implemented, the associated emissions would reduce in line with reusable PPE.
We believe this study is one of the first to report the carbon footprint of a novel PPE device. This is an essential consideration within the innovation process as environmental concerns become more prominent, and is of particular relevance in the UK, where environmentally focused procurement regulations are already in place. These will become stricter in future: from 2028, carbon footprinting must be available for products supplied to the NHS, with a commitment to only procure goods and services from suppliers who can demonstrate progress towards ‘net zero’ by 2030. 29
In conclusion, Bubble-PAPR™ has a carbon footprint comparable to that of disposable FFP-based PPE when PVC recycling is available and HSE usage conditions are observed. 17 When users extend the use of FFP3-based PPE and/or the Bubble-PAPR™ hood is disposed of by incineration, Bubble-PAPR™ has a larger carbon footprint. Trade-offs may be justified, but justification depends largely on the benefits for patients and healthcare staff. We hope that this study opens debate about the acceptable environmental costs of PPE with a better user experience, whilst providing a challenge to manufacturers and users to collaborate to minimise the impact of new equipment.
Supplemental Material
sj-docx-1-inc-10.1177_17511437231173349 – Supplemental material for The carbon footprint of Bubble-PAPR™: A novel item of personal protective equipment
Supplemental material, sj-docx-1-inc-10.1177_17511437231173349 for The carbon footprint of Bubble-PAPR™: A novel item of personal protective equipment by Annie Pinder, Li Fang, Rosie Hillson, Ingeborg Steinbach, Brendan McGrath and Cliff Shelton in Journal of the Intensive Care Society
Footnotes
Authors’ note
Presented in part at the 14th Annual Infection Prevention Conference in Bournemouth, October 2022. Presented in part at the BJA Spring scientific meeting in Manchester, June 2023.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BM and CS were involved in the design and testing of Bubble-PAPR™, and their institution (Manchester University NHS Foundation Trust) may receive financial benefits relating to its sale.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by unrestricted grants and funding from the Engineering and Physical Sciences Research Council (Impact Acceleration Account 302), the Oxford Road Corridor, the Health Innovation Manchester ’Momentum’ special projects fund 2021, the Acute ICU Charitable Research Fund, Manchester University NHS Foundation Trust, and Manchester University NHS Foundation Trust.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
