Abstract
Heatstroke represents the most severe end of the heat illness spectrum, and is increasingly seen in those undergoing exercise or exertion (‘exertional heatstroke’) and those exposed to high ambient temperatures, for example in heatwaves (‘classical heatstroke’). Both forms may be associated with significant thermal injury, leading to organ dysfunction and the need for admission to an intensive care unit. The process may be exacerbated by translocation of bacteria or endotoxin through an intestinal wall rendered more permeable by the hyperthermia. This narrative review highlights the importance of early diagnosis, rapid cooling and effective management of complications. It discusses the incidence, clinical features and treatment of heatstroke, and discusses the possible role of intestinal permeability and advances in follow-up and recovery of this condition. Optimum treatment involves an integrated input from prehospital, emergency department and critical care teams, along with follow-up by rehabilitation teams and, if appropriate, sports or clinical physiologists.
Introduction
Heatstroke represents the extreme and life-threatening end of the spectrum of heat-related illness. Heat illness is common; it is responsible for tens of thousands of deaths in heatwaves, 1 is predicted to rise by over 2.5-fold in the next 30 years, 2 and is in the top three causes of deaths in athletes. 3
Heatstroke is often a multi-system disease, commonly causing coagulopathy, respiratory failure, renal and liver injury, and gastrointestinal dysfunction. If severe, organ support in an intensive care unit (ICU) may be required. Mortality is high, up to 80% if untreated, 4 and is related to the duration and extent of the hyperthermia and the degree of organ dysfunction. 5 In patients requiring critical care, mortality may still be above 60%. 5
Optimal outcomes are likely to require prompt recognition and immediate treatment by prehospital, emergency medical and critical care teams, followed by effective rehabilitation and sports exercise medicine input as required.
With the UK recently experiencing its highest recorded temperature, this narrative review describes its presentation, the need for immediate cooling and supportive care, and the associated morbidity and risks. It also describes some of the new findings around the mechanism, risk factors predisposing to its development, and the need for long-term follow-up and guidance to survivors.
Definition
The most commonly used and widely known definition of heatstroke was originally proposed by Bouchama and Knochel, 6 defined as a core body temperature greater than 40°C associated with central nervous system (CNS) dysfunction, such as delirium, convulsions or coma. An alternative definition has been proposed by the Japanese Association for Acute Medicine (JAAM), which defines heatstroke as patients exposed to high environmental temperature, with one or more of the following:
disseminated intravascular coagulopathy,
CNS manifestation (impaired consciousness, cerebellar symptoms, convulsive seizures), or
hepatic or renal impairment. 7
The JAAM defines heatstroke as the most severe (Stage 3) of three heat illness categories, with heat cramp (e.g. dizziness and muscle cramps) representing Stage 1 and heat exhaustion (e.g. headache and vomiting) Stage 2. Core body temperature is not included in the diagnostic criteria; several fatal cases have been reported where the core body temperature was less than 40°C, 7 and good tolerance of core temperatures above 40°C can be observed in trained athletes.
This article will concentrate on heatstroke; other heat illnesses lie outside the scope of this review.
Subtypes of heatstroke
There are two subtypes of heatstroke, both relating to the inability to dissipate excessive body heat, unlike a pyrexia accompanying an infection, where the hypothalamic thermoregulatory setpoint is increased.
Classical heatstroke (CHS)
This is due to passive heat exposure with poor dissipation mechanisms and is typically seen during heat waves in those considered more vulnerable, such as the elderly, those with underlying health conditions associated with a reduced thermoregulatory capacity, or pre-pubertal children.
Exertional heatstroke (EHS)
This typically occurs in otherwise healthy individuals undertaking vigorous exertion, such as endurance sporting events and military activity, and in certain occupational workers, such as riot police and fire personnel. Unlike in CHS, in EHS, large quantities of heat are produced from skeletal muscle activity; it can therefore occur at low ambient temperatures. 8
Risk factors for heatstroke
A number of factors predispose to the development of heatstroke (Table 1), which has been adapted from a review article discussing drugs that may increase the risk of hyperthermia and the purported mechanisms 9 . In one series, over 57% of patients with CHS had been taking at least one drug known to increase the risk of heat illness, 10 such as diuretics and antihypertensive treatment affecting blood flow and heat loss, and antipsychotic and antidepressant medications that can further increase heat gain.
Conditions and medications that may increase the risk of heatstroke.

Presentation and progression of disease
Clinical features
Patients typically present with the following features:
Core body temperature > 40.0°C
Malaise, fatigue, dizziness, nausea, vomiting, headache, fainting, weakness and heavy sweating
Limbs paradoxically cool and clammy (peripherally shut down)
Tachycardia, hypotension
Neurological impairment (irritability, ataxia, confusion, coma).
Heatstroke is a systemic disorder that can also lead to multi-organ dysfunction, within around 48 h of presentation.
Neurological system
Neurological dysfunction is a cardinal feature of heatstroke. Patients may present with various forms of neurological impairment, which may manifest as behavioural changes, confusion, lethargy, delirium or weakness; more severe features include seizures and alteration in levels of consciousness. 11
Cardiovascular system
Tachycardia is commonly seen, and is often associated with an increased cardiac output, peripheral vasodilatation and hypovolaemia acutely. Shock may be present, in up to 43% of patients. 12 The pathogenesis of the shock is not yet fully elucidated, but may be due to redistribution of blood from the central intravascular circulation to the periphery in an attempt to lose heat, direct myocardial injury from the thermal stress, or from nitric oxide-induced vasodilatation. Hypodynamic shock with decreased cardiac output and increased peripheral and pulmonary vascular resistance has also been observed, particularly in CHS. Electrocardiographic changes during heatstroke are common, and include malignant tachyarrhythmias, conduction defects, prolongation of the QT interval, ST changes and T wave abnormalities. 13
Respiratory system
Tachypnoea is a consistent feature in patients with heatstroke, due partly to heat-induced hyperventilation, of unclear aetiology. This may progress to respiratory failure, reported in 47% of patients, 12 requiring endotracheal intubation and mechanical ventilation in around 20% of patients. 14
Renal system
Renal dysfunction varies from mild proteinuria to acute kidney injury (AKI); of heatstroke patients hospitalised with CHS during the 1995 Chicago heatwave, 53% had at least moderate renal impairment. 15 AKI is more common in EHS than CHS, affecting 91% of patients in one series, 16 and may require renal replacement therapy (RRT). Rhabdomyolysis, with its associated complications, is observed in exercise and in heat illness, where it may be related to an underlying genetic susceptibility.
Hepatic system
Abnormal liver function may be seen, and can in rare cases progress to acute liver failure, particularly in patients with EHS. Early mild elevations in aspartate aminotransferase and lactate dehydrogenase are seen, followed by a rise in bilirubin on the second or third day after the heat insult.
Haematological system
Coagulation disturbances range from petechial haemorrhage and ecchymosis to a severe disseminated intravascular coagulation picture.
Prognostic factors
Short-term outcomes
Several studies have identified factors associated with increased mortality and morbidity in both non-exertional and exertional heatstroke patients. Unsurprisingly, frailer patients and those with more significant organ dysfunction, such as coagulopathy,5,17 hypotension,5,17,18 renal failure 17 and hypoxaemia 18 have a worse outcome. The extent and duration of the hyperthermia also predicts outcome,5,19,20 necessitating emergent and rapid cooling (see below).
Long-term outcomes
A number of factors appear to affect prognosis (see Table 2). In one single observational study of 1456 patients admitted with heat illness in the Paris 2003 heatwave, the presence of particular risk factors in combination reduced the 1-year survival rate from 85% to 18%. 21
Prognostic factors affecting long-term outcomes following heatstroke.
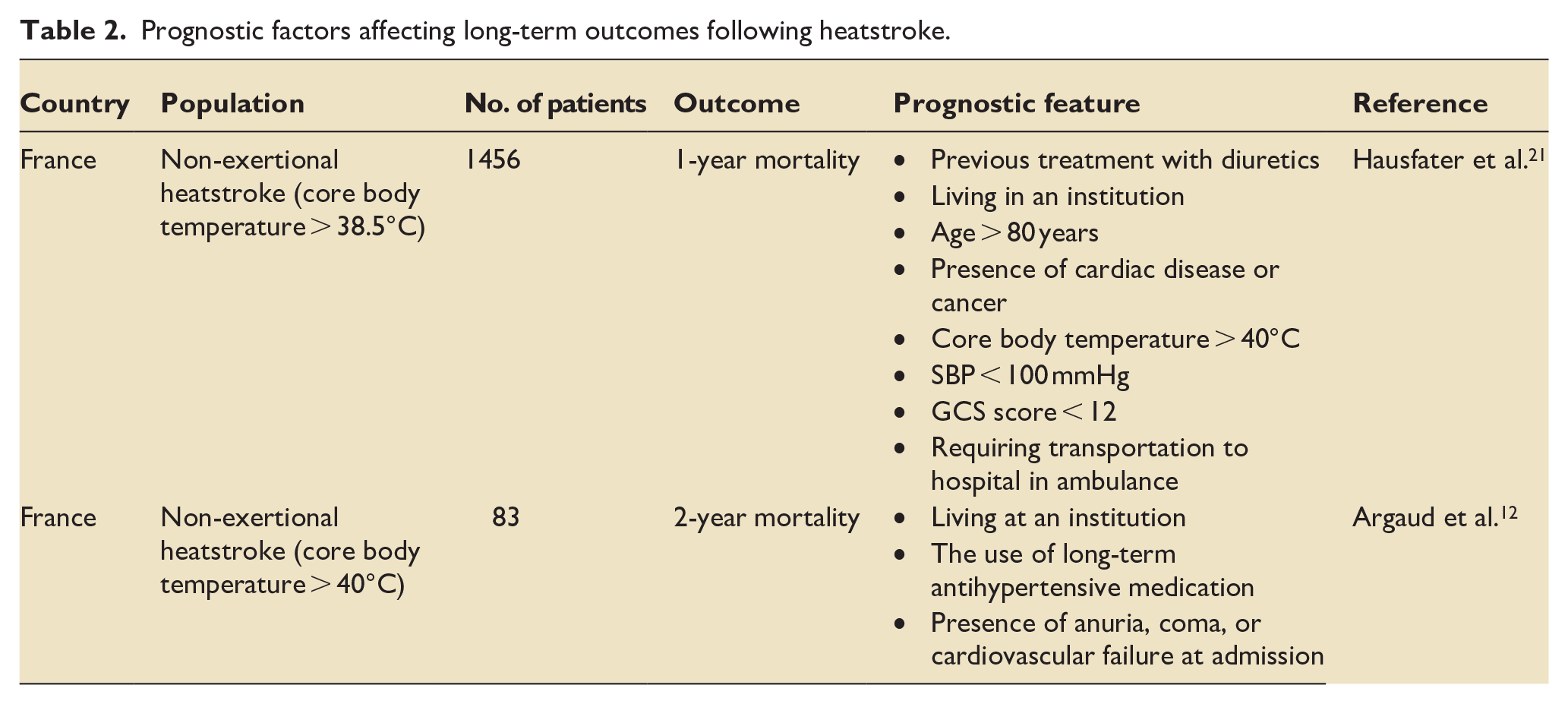
Survivors may be at risk of long-term complications. In one study of CHS patients, the 28-day mortality was 58%, but had risen to 71% by 2 years. 12 At 1-year follow-up, the proportion of survivors with severe limitation in activity, defined as being unable to work or institutionalised, had risen from 3.7% to 40.7%. 12
The risk of developing ischaemic heart disease after heatstroke increases by 3.5-fold at 12 years 22 and acute myocardial infarction 2.7-fold compared to matched controls in a group of Taiwanese patients. 22 The risk of heart failure is increased by 26-fold at 14 years. 23
Persistent neurological dysfunction has been observed in up to 66% of survivors in one study. 15 The cerebellum is frequently affected in cases of persistent neurological dysfunction, given the extreme sensitivity of Purkinje fibres to hyperthermia. 24 However, persistent damage to many other parts of the brain has also been reported. 11
Pathophysiology
A number of mechanisms for organ damage in heatstroke are proposed (see Figure 1).
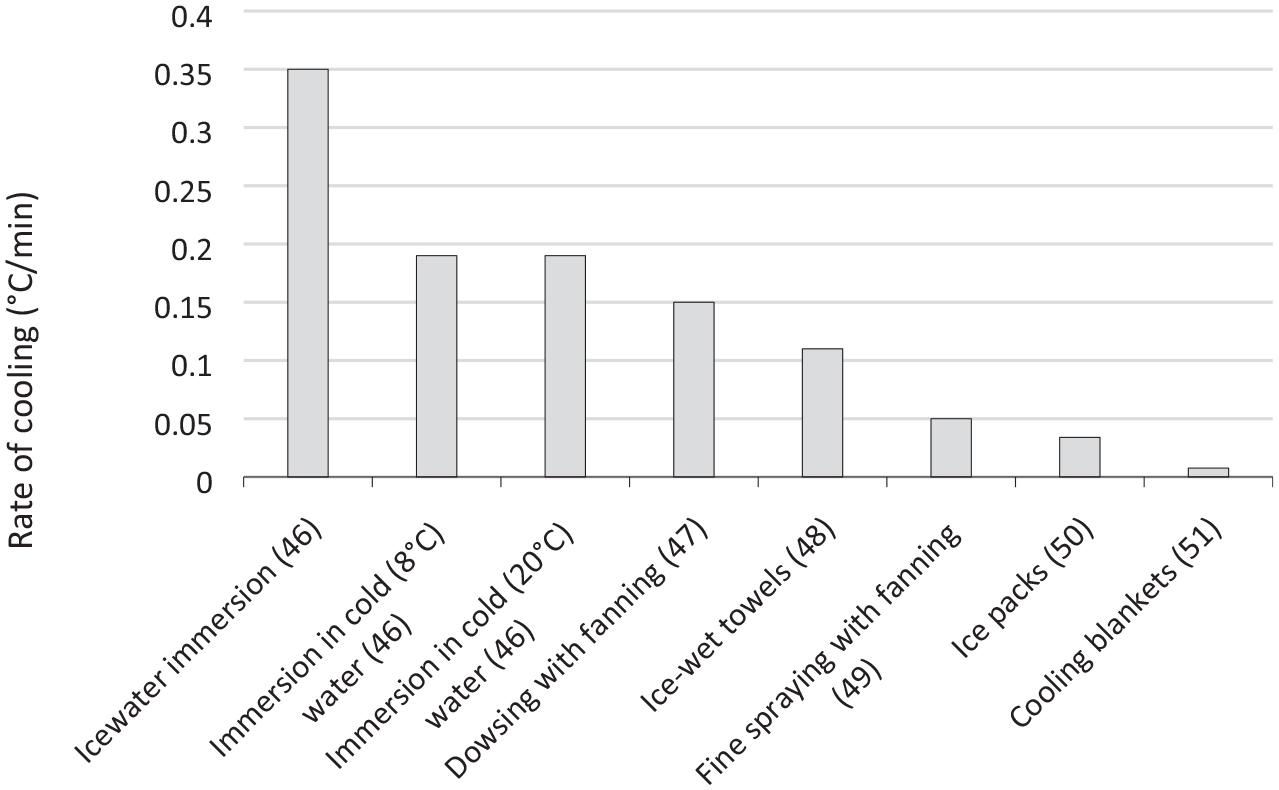
Proposed mechanisms of systemic dysfunction in hyperthermia. Adapted from Walter et al. 25
Hyperthermia is directly cytotoxic, affecting membrane stability, transmembrane transport protein function, leading to intracellular electrolyte derangement. Protein, RNA and DNA synthesis is impaired, with the latter disrupted for longer. 26 Direct cell death in humans occurs at temperatures of around 41°C. 26 The thermal energy required for cell death is similar to that required for protein denaturation, suggesting that hyperthermic cell death occurs primarily through its effect on protein structure. 26 The cellular and surrounding microenvironment is susceptible to direct thermal damage after only a short period; interstitial changes occur after only 30 min at 40.5°C. 27
In addition to direct thermal damage, organ dysfunction may be due to hyperthermic damage rendering the intestinal barrier more permeable, allowing intestinal toxins such as bacteria, 28 endotoxin and lipopolysaccharides (LPS) 29 to enter the systemic circulation. Changes to the intestinal permeability can occur at modest temperatures, around 39°C, 30 and after a short period of hyperthermia, 1–2 h. 30 Intestinal permeability increases have been observed in humans during exertional hyperthermia.31,32 Endotoxaemia and raised LPS levels are thought to be responsible for the inflammatory response and stimulation of inflammatory mediators in both CHS and EHS. 33
Changes in interleukin levels show some correlation with outcome; the rise in IL-6 and the duration of the increased expression is related to mortality in one study, independent of the maximum core temperature obtained. 34 In an animal study, pre-treatment with IL-6 before exposure to heat and antagonism of IL-1, a pro-inflammatory cytokine, improves survival. 35
Mechanisms of cerebral damage
Additional mechanisms for hyperthermic cerebral dysfunction are proposed. The permeability of the blood-brain barrier increases at temperatures above 38–39°C, 36 exposing the brain to systemic and intestinal toxins, such as LPS 37 (‘gut-brain axis’). Cerebral oxygen and glucose consumption is reduced, either due to impaired cellular uptake or reduction in cerebral metabolic activity. 38 Cerebral blood flow, while it initially increases, 39 falls to baseline or below at temperatures above 40–41°C.38,40
Synergistic effects of heat and exercise
Combining hyperthermia and exertion, such as in EHS, has synergistic effects. Intestinal permeability in exertional heat stress is higher than in exertion at normothermia 31 or passive hyperthermia alone. 32
Treatment of hyperpyrexia
Optimal treatment of heatstroke is currently based around immediate and rapid cooling and supportive care, which may include organ support in an ICU setting. No drugs are currently recommended routinely.
Physical cooling
Rapid cooling in patients with heatstroke, regardless of the underlying cause, improves mortality. In CHS, the mortality of those cooled to a core temperature of below 38.9°C within an hour of presentation more than halves, compared to those who remain above 38.9°C. 19 Cooling rates of greater than 0.15°C/min reduce mortality in EHS. 20 Rapid cooling should therefore begin on-site as soon as possible and prior to transfer. A target core temperature of 38.6°C has been suggested, to avoid hypothermia through over-cooling. 41 Core temperature (e.g. rectal) measurement is more accurate than peripheral measurement, and is recommended. 42 Additional passive cooling measures include moving the patient out of direct sunlight and removing constrictive clothing.
A variety of active cooling measures can be deployed (Figure 2), depending on the setting and resources available. In EHS, cooling by immersion in cold water is the most effective and rapid, 43 and is recommended. 44 Public events with an increased risk of exertional heat illness often have facilities available for cold-water immersion (CWI) therapy on site. Cooling with water via dousing or tarp-assisted approaches are effective in the absence of CWI. Spraying the patient with a mist of water whilst fans blow air over the moistened skin is also effective; ice packs are less effective in isolation and pose the risk of local tissue damage but may be used in combination with other techniques. 45 These techniques may be easier to facilitate than CWI, especially when faced with multiple casualties.
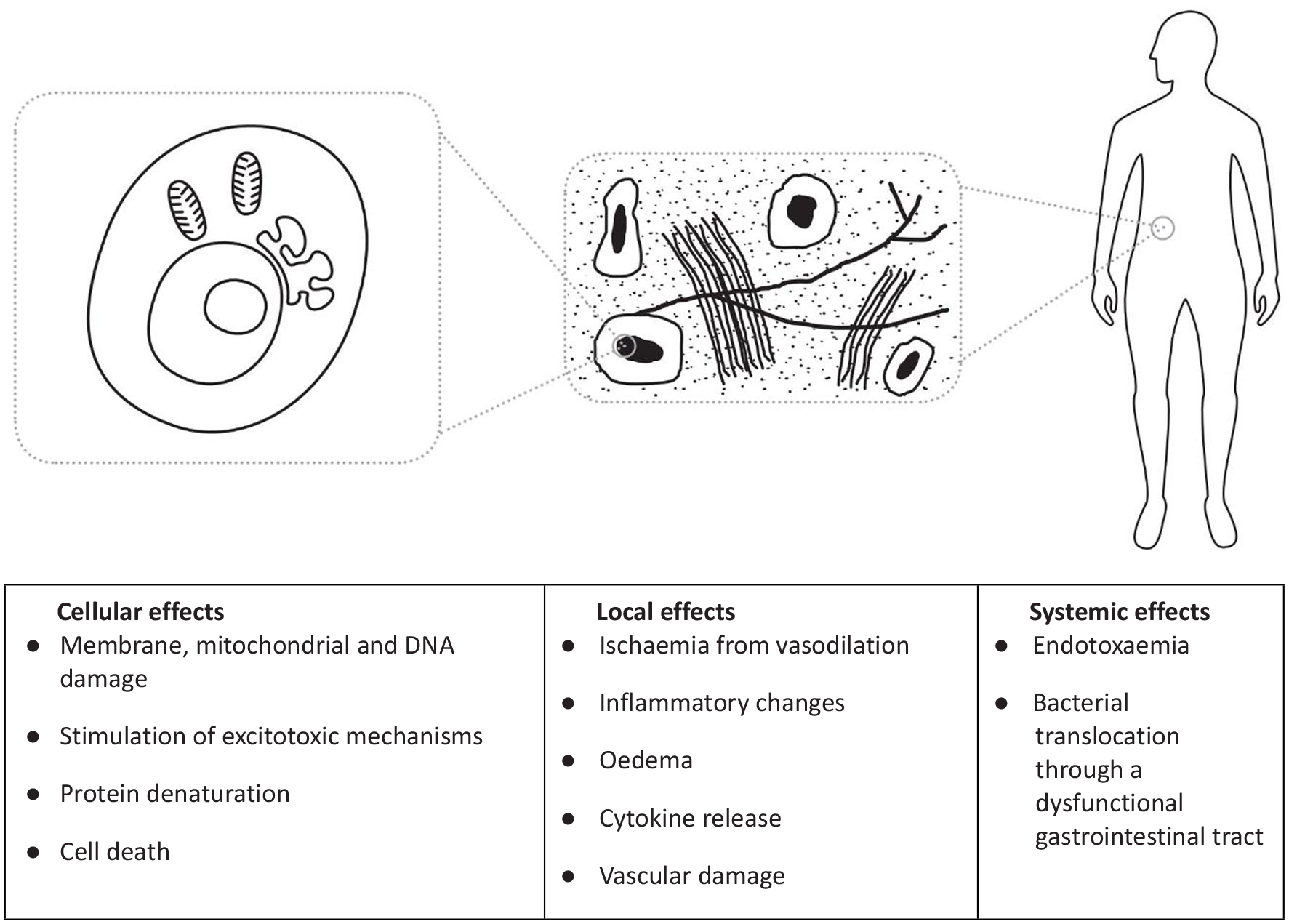
Rates of cooling for specific methods. Data from McDermott et al. 43
Cold (4°C) intravenous fluids may be used as an adjunct to the primary cooling techniques but should not be relied on as a sole method of cooling. There is no strong evidence for a particular resuscitation fluid; however, 0.9% saline may be preferred in EHS if there is an associated exercise-induced hyponatraemia. Although newer devices to aid active cooling have been developed, such as intranasal cooling, endovascular cooling catheters, and blankets made from high heat-conductive biomaterials, their use and effectiveness remain to be validated.
Supportive therapy
Despite rapid cooling, some patients with heatstroke develop multi-organ failure and require admission to an ICU. Patients may require critical care support immediately on presentation; alternatively, organ dysfunction may worsen in the first few days, 52 necessitating delayed admission.
Organ dysfunction in heatstroke is generally managed conventionally. Specific aspects of management are detailed below.
Optimum temperature target after the initial cooling is unclear. A temperature of 37.5°C or above has a trend towards reduced survival in critically ill non-infective patients compared with 36.5–37.4°C, becoming significant above 38.5°C, suggesting a core temperature below 37.5°C may be prudent after the initial cooling. 53
Patients may require intubation for airway protection, respiratory failure or neurological dysfunction. Agitation may warrant the use of sedation, with benzodiazepines suggested to have a theoretical benefit in blunting the shivering reflex and reducing oxygen consumption. 54 Arterial blood gases may be difficult to interpret in hyperthermia, due to changes in solubility and dissociation with haemoglobin. 55
Cardiovascular dysfunction may be multi-faceted, and require management of dehydration, cardiogenic and vasoplegic shock. In addition, both hypernatraemia and hyponatraemia may be seen in EHS, warranting close electrolyte monitoring. There is no evidence to advise alternative management to conventional treatment.
Liver dysfunction may be severe enough to warrant assessment for transplantation. Standard referral criteria are advised for liver transplantation, although one series reports that as three out of four transplant recipients did not survive and that there was spontaneous recovery in two-thirds, a more conservative approach might be warranted. 56 A prothrombin time of more than 10 times normal, persisting after 3 days, has been suggested as a further criterion for transplantation. 57 The AKI may be severe enough to require RRT. RRT in heatstroke with multi-organ failure may improve survival and a faster return to normal of serum markers and organ function, independent of the renal failure. 58 Measurement of creatine kinase levels is advised, to detect rhabdomyolysis.
Patients may be at higher risk of infection, which could be related to gastrointestinal permeability. In one study, over 50% of CHS patients showed evidence of bacterial infections. 15 In a further study, procalcitonin, which has a high sensitivity and specificity for detecting bacteraemia, was elevated in 58% of patients with CHS and was associated with increased mortality. 59
During recovery, regular assessment of arterial blood gases, full blood count, biochemistry and coagulation is important; blood tests typically return to normal within 16 days, despite different recovery kinetics. 52
On discharge from ICU, survivors may benefit from follow-up with general medical and rehabilitation teams; if they recover well, advice from a geneticist, and sports or clinical physiologist may be beneficial (see below).
Pharmacological management
Dantrolene is used in the management of MH to reduce excessive muscle contraction and heat production. While there are similarities between EHS and MH, dantrolene is not currently routinely recommended in heatstroke 60 ; however, the authors of this study suggest its use may be rational in severe cases or in cases where no improvement is observed after cooling. 60
Although antipyretics such as ibuprofen, aspirin and paracetamol may be useful in sepsis-induced pyrexias, the mechanism of the pyrexia in heatstroke is different. Antipyretics are therefore not advised and may worsen the associated coagulopathy, and hepatic and renal injury. 61
Recent systematic reviews have suggested that both systemic steroids and antibiotics against intestinal bacteria may be effective in reducing intestinal bacterial translocation, inflammatory response, organ dysfunction and mortality in heatstroke.62,63 Trials are ongoing, but neither are currently recommended.
Future work, follow-up and recovery
Individuals who recover may be at risk of a future episode of heat illness. After an episode of EHS, the risk of a further episode has been reported as over three times higher within the first 2 years. 64 Whether this is related to pre-existing genetic, phenotypic or behavioural risks, or post-exposure biochemical or genetic changes is not clear.
While follow-up of patients is currently very limited, the following areas may need to be considered.
Return to play guidelines after EHS
Some authorities recommend that in the first 14–21 days following exertional heat illness, an individual is confined to rest and essential activities of daily living before progressing to low-intensity activity in cool conditions to ensure exercise can be tolerated.42,65 Medical assessment may be an occupational requirement prior to the commencement of any exercise. 65
Once exercise in cooler conditions can be tolerated, intensity can increase and thermal stress added. To maximise resistance to heat, a full heat acclimation/acclimatisation protocol may be warranted 66 ; advice from a sports physiologist may be beneficial.
Heat tolerance testing
The use and optimal approach of exercise-heat stress tests to further guide return to fitness remains uncertain.42,65 A typical heat tolerance test is one created by the Israeli Defence force, which currently requires participants to walk at 5 km/h at a 2% gradient for 120 min in conditions of 40°C and 40% relative humidity, 6–8 weeks following heat illness. 67 Testing is discontinued and the participant deemed heat intolerant if core temperature surpasses 38.5°C or the heart rate exceeds 145 beats per minute. 67 Reassessment follows 2–3 months later.
Genetic abnormalities in heatstroke
Exertional heatstroke has clinical and biochemical similarities to malignant hyperthermia (MH), with case reports of patients with both conditions. Mutations in the ryanodine receptor (RYR), which mediates cellular calcium release, account for the majority of MH cases. 68 Some patients with EHS also display mutations in the RYR1 gene 69 ; some authorities therefore advise consideration for testing for susceptibility for MH, 70 especially if the EHS is recurrent or if no other obvious precipitant is identified. 71
The genetic basis of EHS may be more heterogeneous than MH; mutations in the sarcoplasmic skeletal muscle protein calsequestrin (CASQ1), which appears to modulate RYR1 function, and in the TrpV1 gene, involved with detecting and sensing thermoregulation and nociception, have been identified.72,73
Role of diet
A number of nutritional components, such as vitamins A and D, probiotics, arginine and glutamine appear to improve intestinal barrier function, raising the possibility of manipulating the risk of developing heat illness in high-risk individuals. Further research on the clinical significance is warranted.
Conclusion
Heatstroke, either exertional or environmental, is a common syndrome and becoming commoner. In a world where heat waves may become increasingly frequent, heatstroke is a condition with which critical care clinicians should be familiar. The priorities are urgent diagnosis, immediate cooling and supportive care, which may include multi-organ support in ICU. It is associated with a significant risk of multi-organ failure and death, even with intensive care. There is a role for further research in this area given the current paucity of high-level evidence, which would help identify individuals at risk, improve follow-up and offer advice for recovery, return to fitness and reducing future risk. There is interest in the effect of intestinal dysfunction and translocation of intestinal toxins, and whether these may be affected to reduce the effects of heat illness.
Footnotes
Acknowledgements
Figure 1, taken from Walter EJ, Hanna-Jumma S, Carraretto M, Forni LG. The pathophysiological basis and consequences of fever. Crit Care. 2016; 20: 200. https://doi.org/10.1186/s13054-016-1375-5 and published by Springer Nature, is reproduced under the terms of the Creative Commons Attribution 4.0 International Licence (![]() ).
).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Daniel Martin is Editor-in-Chief of the Journal of the Intensive Care Society. Edward Walter is on the editorial board of the Journal of the Intensive Care Society. No other authors declare a conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
