Abstract
Background
Guidewire retention and sharps injury during central venous catheter insertion are errors that cause patient and healthcare professional harm. The WireSafeTM is a novel procedure safety pack engineered to prevent guidewire retention and sharps injury during central venous catheter insertion. This is a pilot study aimed to determine its acceptability, usability and safety during clinical practice.
Methods
An observational time and motion study was conducted comparing central venous catheter insertion and sharps disposal practice using standard versus WireSafeTM techniques. One-year following implementation, a structured survey was conducted to determine clinician opinion and experiences of using the WireSafeTM.
Results
15 procedures were observed using standard practice and 16 using the WireSafeTM technique. The WireSafeTM technique decreased the time taken from removal of the guidewire to disposal of sharps (standard 11.4 ± 5.6 min vs WireSafeTM 8.7 ± 1.4 min, p = 0.035), as well as total procedure time (standard 16 ± 7 min vs WireSafeTM 14.2 ± 2 min, p = 0.17), although this latter trend did not reach significance. Clinicians frequently practiced unsafe behaviour during sharps disposal in the standard group (53%), but when using the WireSafeTM technique, 100% exhibited safe practice by transferring sharps to the bin inside the sealed WireSafeTM box. One-year following implementation, 20 clinicians participated in the structured survey. Clinicians across three different departments used the WireSafeTM in varying clinical situations and reported that its use for central line insertion was either easier (10/20) or no different (10/20) compared to standard practice. All clinicians (20/20) felt that the WireSafeTM reduced the risk of guidewire retention and all stated that they approved of the WireSafeTM technique, and supported its use for convenience and safety benefits.
Conclusion
Utilising the WireSafeTM for central line insertion facilitated earlier and safer sharps disposal, and the device was well supported by clinicians for its convenience and safety benefits.
Keywords
Introduction
Central venous catheters are commonly used for administering medication and monitoring patients in the intensive care unit (ICU), emergency department and operating theatres. 1 Central venous catheter insertion via the Seldinger technique involves passing a catheter over a guidewire to facilitate venous cannulation, following which the guidewire must be removed. Although this is a commonly performed technique worldwide and has been in use for many years, there remains an inherent risk of failure to remove the guidewire. The risk of whole guidewire retention is estimated to be around 1:3000 procedures,2,3 and it is considered to be a preventable error if the correct steps have been followed.4,5 Numerous case reports describe guidewire retention,2,3,6–8 and a systematic literature review by Pokharel and colleagues determined that the incidence of reported guidewire retention has continued to increase. 9 Human error is often cited as the cause of this error 10 ; however, a number of factors are considered to contribute to such ‘mistakes’ such as distraction, interruptions, development of emergency situations, poor supervision, inexperienced operators and high workload.9,11,12 Healthcare providers should aim to reduce the potential for human error where possible, and available evidence demonstrates that guidewire retention will continue to occur without the development of novel techniques incorporating engineered solutions to nudge or force safe practice.6,9,12
In addition to guidewire retention, central venous catheter insertion is associated with the risk of sharps injury to healthcare professionals. One systematic review of reported sharps injury rates in the United Kingdom calculated an incidence of 12.74 per 100 beds per year. 13 An estimated 40,000 needle stick injuries occur annually in the British National Health Service, but due to under-reporting the incidence is likely to be significantly higher. 14 One study from the United States reported that 16% (21/131) of all needlestick injuries in residents and fellows over a period of 4 years occurred whilst they were suturing a line to the skin with a suture needle. 15 Needlestick injuries are a significant health burden and estimated to cost an average of £500,000 per year for every British National Health Service trust, 16 and Health and Safety legislation requires hospitals to take effective action to control the risk of sharps injuries. 14
The Venner WireSafeTM (Venner Medical Technologies, Singapore) is a novel safety engineered procedure pack designed to prevent guidewire retention and reduce the risk of sharps injury during central venous catheter placement. The device utilises a forcing function to ensure that the clinician removes the guidewire and uses it to open the pack and access further equipment to complete the procedure, and also acts as a safe sharps reservoir following procedure completion (Appendix 1, Figure 1). The initial steps in Seldinger central line insertion are unchanged when utilising the device and it is designed to be used after the guidewire is removed in order to suture, flush and dress the central line, as well as act as a reservoir for safe sharps disposal. The box contains a curved suture, suture holder, counter-traction forceps and scissors and whilst the operator may choose to practice their preferred method of catheter securement, having the correct equipment ‘to hand’ is likely to encourage the use of safer suturing practices. Specifically, hand suturing with straight needles is discouraged due to the immediate availability of safer non-touch suturing equipment. The steps performed when using the WireSafeTM during central venous catheter insertion: (A) venepuncture, (B) guidewire insertion, (C) catheter placed over guidewire and guidewire removed, (D and E) guidewire inserted into the light bulb shaped channel in the lid of the WireSafeTM, (F) the WireSafeTM is opened by grasping the protruding ends of the guidewire and using them as a handle to open the lid of the WireSafeTM, (G) contents of WireSafeTM are used for catheter insertion, (H) the sharps used for catheter placement are put inside the WireSafeTM and (I) the lid of the WireSafeTM is closed and sharps are sealed inside for transfer to the sharps bin. The WireSafeTM can be disposed of in the sharps bin.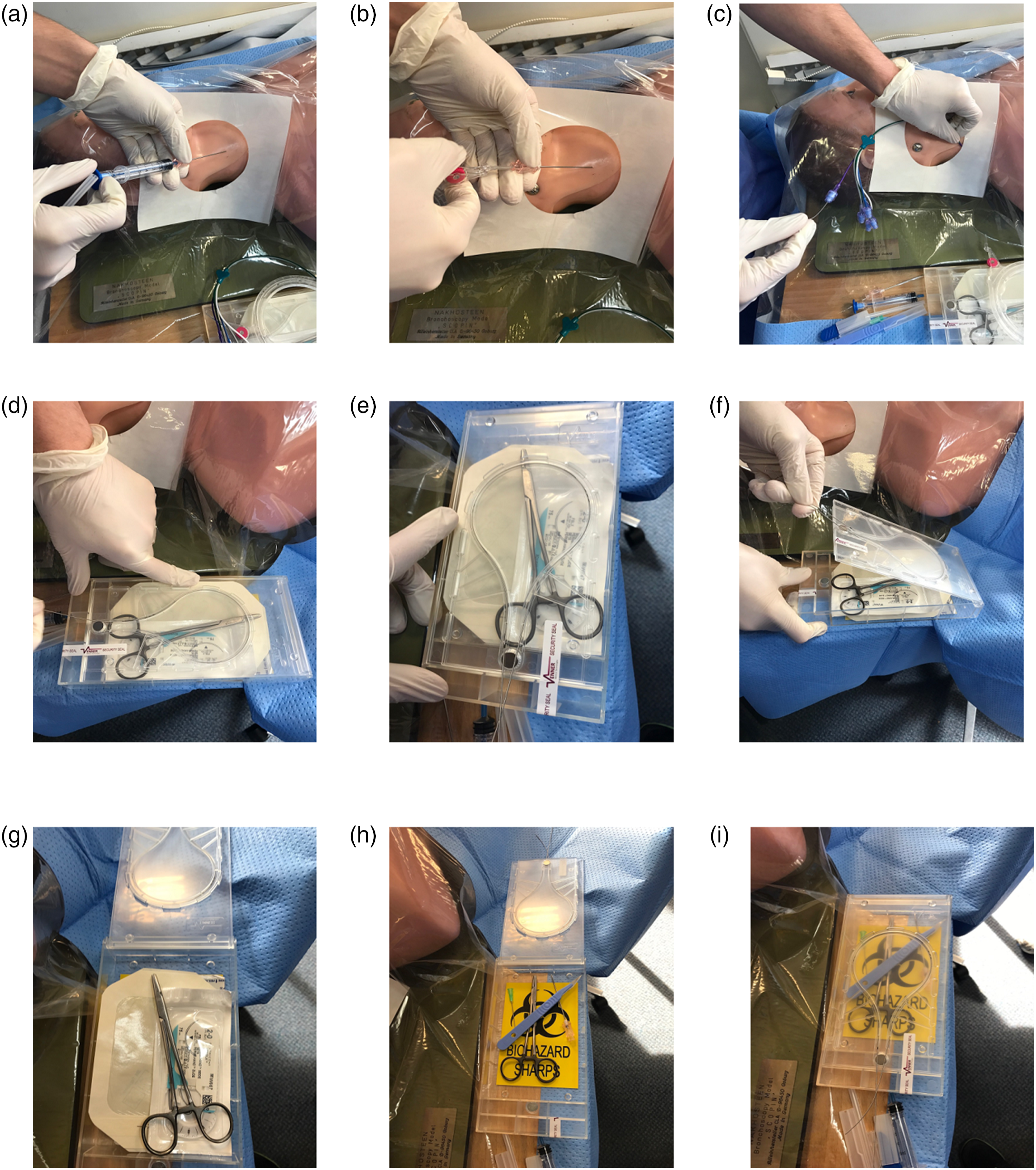
Preliminary simulation studies using the WireSafeTM have demonstrated its efficacy in preventing guidewire retention. 10 In this pilot study, the WireSafeTM was implemented in the ICU, emergency department and operating theatres of a single hospital in the United Kingdom, with the intention of improving patient and staff safety. The primary aim of this study was to assess the impact of the WireSafeTM on central line insertion in terms of procedure duration and sharps safety, and secondarily evaluate its acceptability and perceived safety benefits to staff members in terms of sharps safety and guidewire retention via a structured survey one-year following implementation of the device.
Materials and methods
The institutional review board and clinical governance committee granted approval to conduct the pilot study which was undertaken at the Queen Elizabeth Hospital, King’s Lynn, United Kingdom. The WireSafeTM has regulatory approval for clinical use in the United Kingdom and was procured by the Queen Elizabeth Hospital National Health Service Foundation Trust. A single centre clinical observational time and motion study was conducted comparing the WireSafeTM technique to standard central venous catheter insertion techniques. In both groups, the initial steps of Seldinger central venous catheter insertion up until the point of guidewire removal were exactly the same, and utilised a standard line insertion pack. In the standard group, clinicians would have the remaining equipment required to complete the procedure immediately available separately on the trolley (suture pack, needle and dressings). However in the WireSafeTM group, this equipment was all located inside the locked device, which could only be unlocked using the removed guidewire. All participants in the clinical observational study provided written and signed informed consent to take part in the study.
Appropriate departments and personnel were consulted by email and at departmental meetings and any questions or concerns were discussed. Key personnel were identified (anaesthetists, practice development nurses and operating department practitioners) and trained in the use of the WireSafeTM procedure pack for 1 minute each, and instructed to cascade this training to their team members. The WireSafeTM kit contained instructions printed on the underside of the packs pictorially demonstrating how to use the device, and during the initial implementation period, an investigator was available by telephone to ‘troubleshoot’ any issues regarding the equipment or technique. The participating clinicians were all either resident or senior anaesthetists chosen as a convenience sample over a 2-month period. They were observed by one of three (SS, TS and JG) designated investigators who observed central venous catheter insertion using both standard and WireSafeTM techniques. The group selection depended upon availability of the newly introduced WireSafeTM packs, which was not randomised. The participants were unaware that they were being timed and which specific observations were being taken, and knew only that the technique was being observed.
Data points recorded for both groups were (i) the total procedure time, (ii) the time that the WireSafeTM impacts on the procedure, that is, from guidewire removal to sharps disposal (including stitching the central venous catheter to the skin, dressing application and sharps disposal) and (iii) the method of sharps disposal used. Safe sharps behaviour was defined as either the sharps bin being brought to the procedure and utilised at the bedside, or alternatively sharps being transferred to the bin after the procedure in an open tray, container or wheeled on the procedure trolley. In the WireSafeTM group, transfer of sharps to the bin inside the closed WireSafeTM container was also defined as a safe option. Unsafe behaviour was defined as transfer of sharps to the bin in the clinician’s hands, sharps being thrown any distance into the sharps bin, or sharps being left to be cleared away by an assistant (which is counter to our institutional policy).
One year after implementation of the WireSafeTM, a structured survey of 20 clinicians who had used the WireSafeTM technique was undertaken. Clinicians were asked standardised questions on (1) the approximate number of WireSafeTM central venous catheter procedures they had completed, (2) the type of clinical scenarios in which they had utilised the WireSafeTM, (3) any problems they encountered using the WireSafeTM, (4) whether the WireSafeTM was easier, the same or harder to use compared to standard procedures, (5) whether or not they thought the WireSafeTM reduced the risk of a guidewire retention, (6) whether the WireSafeTM was more convenient, the same or less convenient for sharps disposal compared to standard procedures, (7) whether or not they approved of the WireSafeTM device for central line insertion and (8) whether or not they would support the use of the WireSafeTM going forwards for its safety and convenience benefits. Survey participants were also given white space areas to comment on any general likes or dislikes of the new technique and equipment.
Statistical analysis
All data were anonymised and collected on a secure database (Microsoft Excel, 2016). A data analysis and statistical plan was formulated, written, data-stamped and recorded in the investigator’s files before data were accessed. Continuous variables (total procedure time and the time that the WireSafeTM impacts on the procedure) were expressed as mean plus standard deviation, and compared using an unpaired t-test (Microsoft Excel 2016). Categorical variables (method of sharps disposal) were expressed in exact numbers and proportions in a 2 × 2 contingency table, and compared using a Fisher’s exact test (graphpad.com). An alpha value of <0.05 was taken as significant.
Results
Fifteen procedures were observed using standard practice and 16 using the WireSafeTM technique. In the WireSafeTM group, the mean (M) ± standard deviation (SD) total procedure duration was 14.2 ± 2 min. In the standard group, the M ± SD duration of the procedure was 16 ± 7 min (Figure 2). There was no statistically significant difference in the duration between the groups (p = 0.17). The time that the WireSafeTM actually impacts on the procedure, that is from the removal of guidewire to disposal of sharps, was significantly shorter in the WireSafeTM group compared to the standard group (8.7 ± 1.4 vs 11.4 ± 5.6 min, p = 0.035) (Figure 3). Box and Whisker graph illustrating the total time taken to complete central venous catheter insertion comparing standard practice against the WireSafeTM. Using the WireSafeTM reduced the overall time taken to perform the procedure (16 ± 7 min standard group vs 14 ± 2 min using the WireSafeTM, p = 0.17). Box and Whisker graph illustrating the time taken from guidewire removal to sharps disposal comparing standard practice against the WireSafeTM. Using the WireSafeTM reduced the time taken for sharps disposal (11.4 ± 5.6 min standard group vs 8.7 ± 1.4 min using the WireSafeTM, p = 0.035).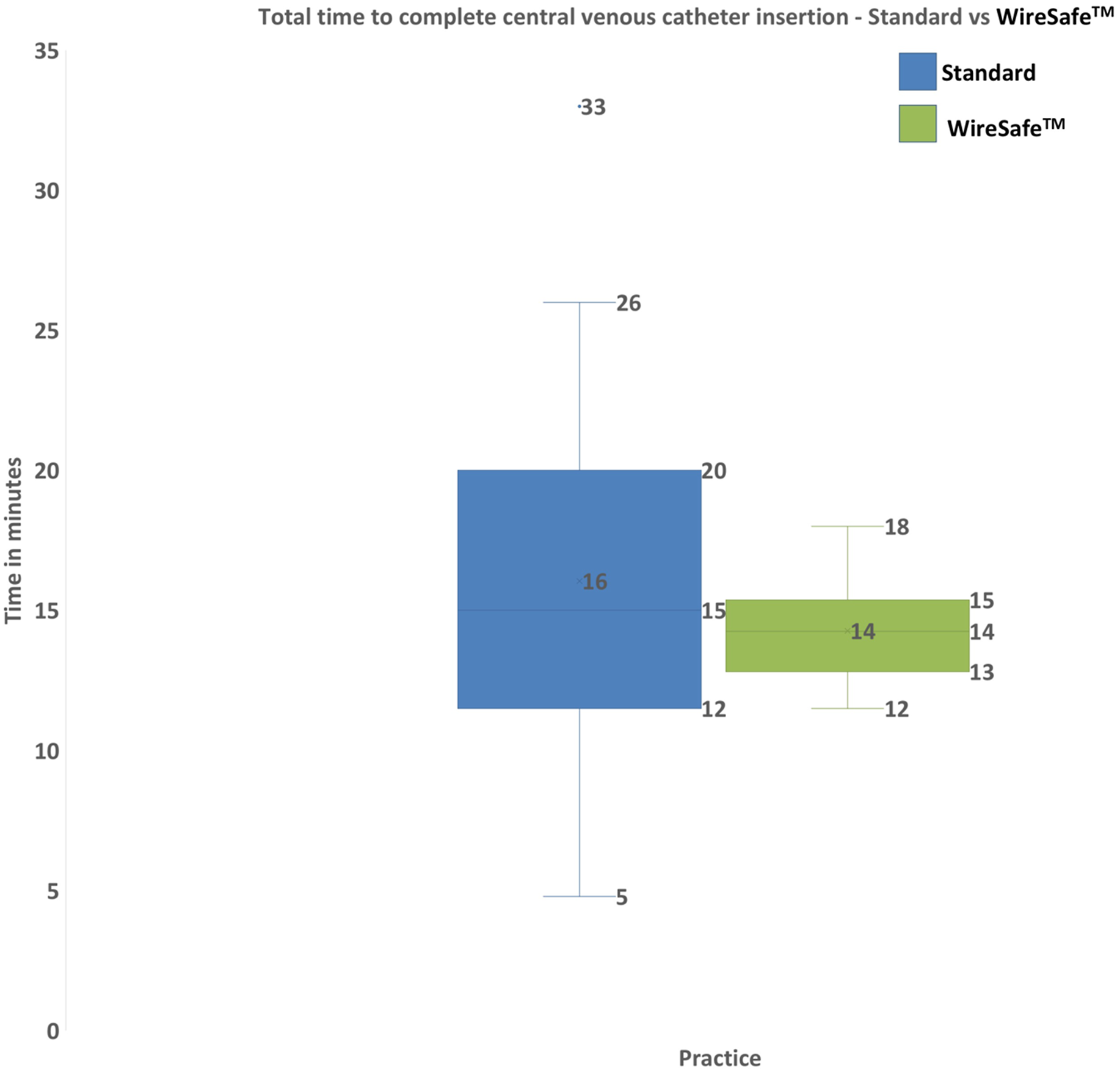
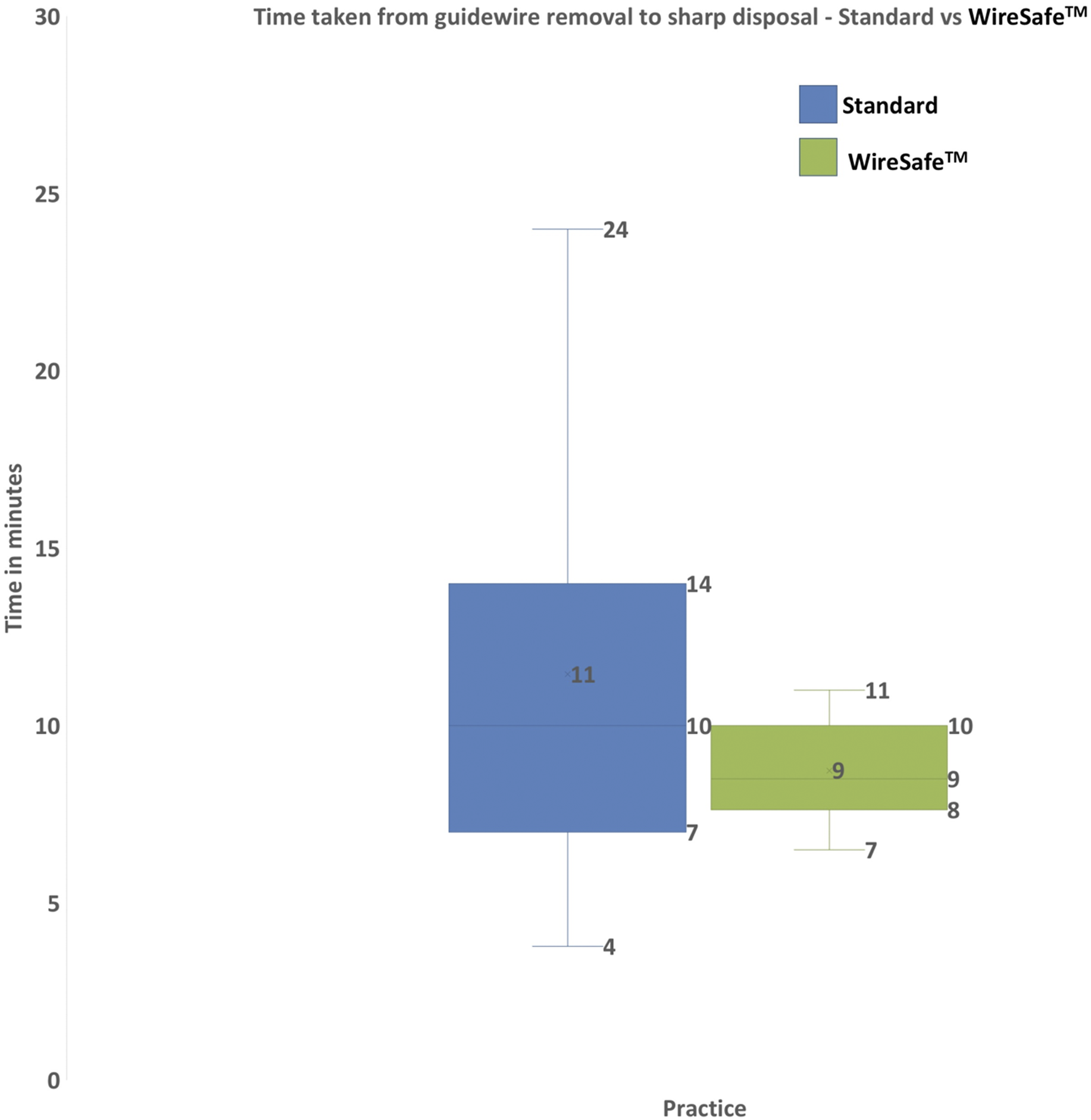
In the WireSafeTM cohort, all 16 (100%) clinicians demonstrated an appropriate and safe sharps disposal technique. In the standard group, 7 (47%) clinicians demonstrated an appropriate and safe sharps disposal technique while 8 (53%) did not. There was a statistically significant difference found between sharps disposal in the WireSafeTM versus standard group, (p = 0.0008, Fisher’s exact test). In the standard group, out of the eight procedures where sharps disposal was not deemed safe, four participants transferred open sharps to the sharps bin across the room using their hands while four participants left sharps behind them after completion of procedure.
One-year following implementation of the WireSafeTM, over 600 devices were used for central venous catheter insertion at our institution and there were no incidents of guidewire retention. Structured interview surveys were conducted with 10 anaesthetists and 10 ICU physicians of varying degrees of seniority from resident to attending. All stated that they had used the WireSafeTM technique in their clinical practice and between them had used approximately 225 WireSafeTM kits in total. The WireSafeTM equipment had been used in a variety of clinical situations including unconscious patients, awake patients and awake but agitated patients, and none of the clinicians reported any equipment-related problems whilst using the device. Half (10/20) of the clinicians surveyed felt that the WireSafeTM made the process of central venous catheter insertion easier, with the other half reporting equipoise compared to standard insertion techniques. None of the clinicians surveyed felt that the WireSafeTM made the process harder. All clinicians felt that the risk of guidewire retention was reduced by using the WireSafeTM. Most participants (16/20) stated that using the WireSafeTM was more convenient for sharps disposal at the end of the procedure, and the remaining four clinicians found no difference compared to standard practice. All 20 participants said they approved of the WireSafeTM device for central venous catheter insertion and would continue to support its use for safety and convenience benefits. Qualitative participant comments included a perceived benefit of the WireSafeTM in that it could be placed next to the surgical site (rather than on the procedure trolley which required the clinician to repeatedly twist their torso to access equipment and temporarily store sharps).
Discussion
Guidewire retention is classed as a clinical ‘never event’ in the British National Health Service and is described as a preventable error. 4 Complications of guidewire retention include cardiac dysrhythmias, blood vessel or cardiac muscle perforation 11 and can result in significant patient harm with a reported mortality of up to 20%,11,17 as well as repercussions for the clinician involved and financial implications for the hospital where the event occurred. 18 Preventative measures to date have included further education and training, the use of checklists and two person checking techniques.8,9,19,20 However, predicting and preventing an event that only occurs once in every few thousand procedures can be problematic.2,3 The complication is rare enough to not be in the forefront of the clinician’s mind and in a pressured era of waiting time targets, healthcare institutions may be reluctant to devote significant time and resources to address a problem which they perceive to be rare. Instead, a simple and low cost intervention which is easy to implement may provoke interest within the healthcare community. Devices like the WireSafeTM which are safety engineered to remove potential errors by forcing the operator to perform certain safety steps have the highest level of effectiveness in the hierarchy of intervention effectiveness in human factors terms when trying to eradicate errors out of a healthcare system. 21 By contrast, education, training, reliance on clinician vigilance and checklists are costly, of limited efficacy when addressing rare events, difficult to sustain with staff turnover and subject to ‘creeping complacency’ over time. 10 These conventional solutions to rare errors can also lead to adverse consequences in terms of distraction errors due to an increase in cognitive load for a clinician during the performance of a complex procedure.22,23 This pilot study was hence primarily designed to assess whether the introduction of the WireSafeTM device impeded existing conventional central line insertion techniques in terms of procedure duration, and investigate its impact on sharps safety. Secondarily, the device’s acceptability and perceived safety benefits to staff members were surveyed via a structured questionnaire one-year following implementation of the device.
Introduction of WireSafeTM adds a forcing function to the conventional Seldinger method of central line insertion, which could potentially prolong the duration of the procedure. However, our data did not demonstrate a statistically significant difference in total procedure time between the WireSafeTM and standard group, which demonstrates that introduction of the new forcing function into the procedure did not negatively impede its duration. There was in fact a trend towards the procedure being quicker with the device (14.2 ± 2 vs 16 ± 7 min). Whilst this study is ultimately underpowered for assessing the impact of device on the duration of the procedure, the trend is interesting and warrants further investigation. Importantly the part of the procedure directly impacted by the introduction of the device, that is guidewire removal until sharps disposal, was significantly faster in the WireSafeTM group (8.7 ± 1.4 vs 11.4 ± 5.6 min, p = 0.035), a finding which is unlikely to be explained by the procedure being performed in a different order as sharps disposal in both study groups can only occur after catheter securement and should always occur prior to clearing away the remainder of the equipment in order to prevent sharps injuries. This may suggest that in time and with clinician familiarity with the device, the WireSafeTM technique could reduce overall central venous catheter insertion time and thus provide additional benefit beyond that of safety.
NHS Employers reported that of the needlestick injuries which occurred during clinical procedures, 35% occurred after the procedure during the clearing up and disposal of sharps. 14 Our results demonstrated a significant improvement in safe sharp disposal practice among participants. The WireSafeTM gives a designated space on the trolley for sharps disposal, and following all 16 procedures observed, all clinicians placed their used sharps inside the WireSafeTM box, closed the box to seal the contents inside and transferred the sharps to the bin in the closed container during equipment clear up. As sharps were transferred inside the sealed container, this was deemed as safe practice in the study and indeed represented an upgrade in safety terms over transferring sharps to the bin in an open tray/container or on the procedure trolley, practices which were also defined as safe in the standard group. Nationally, there are substantial cost implications of needlestick injuries which could be avoided by adopting safer sharps practice, with the estimated financial burden of needle stick injuries totalling up to $591 million per year in the United States alone. 24 Should the clinician or patient contract an infectious disease due to a sharps injury, the personal cost to health is also significant.
This pilot study therefore demonstrates that central line insertion using the WireSafeTM device improves sharps safety and has no demonstrable negatives compared to the current standard of practice in terms of procedure duration and staff acceptance. Entirely positive or neutral views expressed in the structured survey of users of the device 1 year after its implementation are encouraging and confirm that the device has wide acceptance amongst staff, 100% of whom felt that the device reduced the risk of guidewire retention, and 80% of whom also felt that the device was more convenient for safe sharps disposal at the end of the procedure. Importantly, the device can also be introduced with minimal induction training.
However, there are limitations to this single centre and small sample size analysis, which made the study underpowered in terms of assessing the impact of the device on total procedure duration The study was conducted during a period when the WireSafeTM device was being introduced across our hospital and the need to ‘use up’ supplies of previous procedure equipment prior to adopting universal use of the WireSafeTM device meant that whilst participants were not necessarily given the choice to use one technique over the other, they were also not truly randomised to either group. They instead utilised the WireSafeTM device based on its availability in different clinical areas. Importantly, we are not aware of any instances of staff refusing to use the device once it was introduced into their clinical area, or vice versa of staff insisting on utilising the device whilst their clinical area was still utilising old supplies. Finally, the study was not blinded and participants were aware that they were being watched for the purposes of research, which could potentially lead to bias in participant performance.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PJY is the inventor of the WireSafeTM. The patent (WO 2017/029492 A1) is currently owned by PJY, MZAM and Venner Medical Technologies (VMT). VMT has the rights to manufacture and market the device.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Queen Elizabeth Hospital Kings Lynn.
Appendix
The steps involved in central venous catheter insertion are vessel puncture, guidewire insertion, threading the catheter over the guidewire, removing the guidewire, suturing the catheter and finally applying a dressing before sharps disposal. The WireSafeTM is a novel clinical innovation designed to ensure safe removal of the guidewire at a critical point in the procedure at which it is normally forgotten, that is after the catheter is placed over the guidewire and before the catheter is secured in place. The WireSafeTM is a locked procedure pack which contains the equipment required to complete the procedure after the guidewire is removed: a curved suture, needle holder, counter traction forceps, scissors, dressing, 20 ml syringe, filter needle and a pen, and is provided as part of the central venous catheter insertion pack. The WireSafeTM device is opened by using the guidewire as a key to unlock and open the device, and access its contents in order to complete the procedure. This ensures the clinician is always reminded to remove the guidewire at the correct point in the procedure, as they would be unable to complete the procedure without undertaking this step (Figure 1). In human factors terms this is known as a forcing function. After the procedure, the empty WireSafeTM box can be used as a safe container to transfer sharps to the bin thus reducing the risk of needlestick injury.
