Abstract
Background
Disrupted circadian rhythms can have a major effect on human physiology and healthcare outcomes, with proven increases in ICU morbidity, mortality and length of stay.
Methods
We performed a multicentre observational study to study the nocturnal lux exposure of patients in 3 intensive care units.
Results
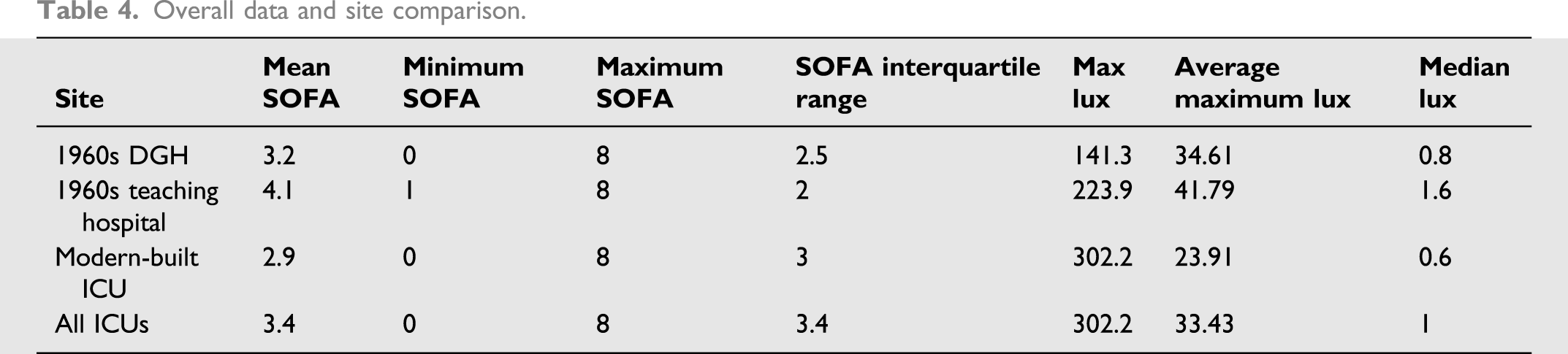
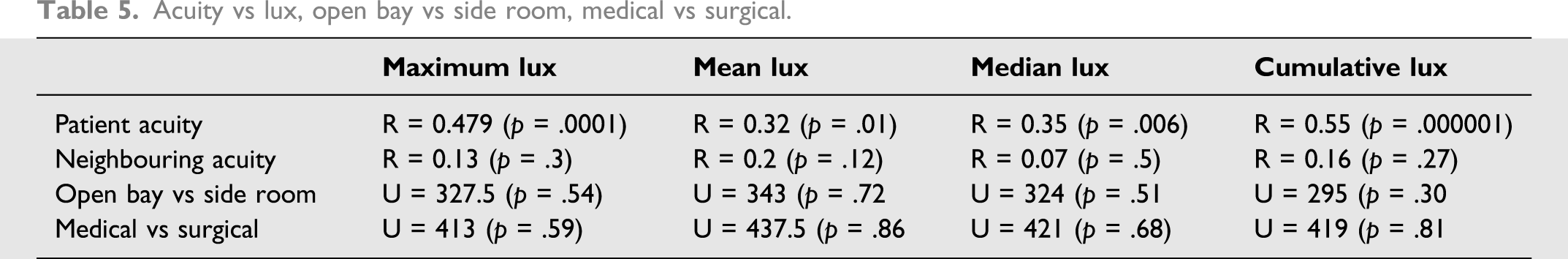
The median light intensity recorded was 1 lux over the 6-hour recording period; however, this is deceptive as it hides short periods of high lux. When looked at in shorter time segments of 30 minutes, there were significant periods of lux higher than a crude median, especially in higher acuity patients. There was a positive correlation between acuity (as estimated by SOFA score) and maximum lux (R = 0.479, p = .0001), median lux (R = 0.35, p = .006) and cumulative lux (R = 0.55, p = .000001). There was no relationship between neighbouring patient acuity and lux.
Conclusions
Clinicians should practice vigilance at night to provide optimal environmental conditions for patients to minimise potential harm.
Introduction
The circadian rhythm is a self-sustained fluctuation of various physiological processes over approximately (circa) 1 day (diem) 1 that acts as the internal ‘body clock’. Whilst the circadian rhythm is known mostly for its role in the sleep–wake cycle, it also influences the immune system, 2 gut microbiome, 3 renal function, 4 vascular health, 5 thermo-regulation, hormonal homeostasis, glucose metabolism, cell regeneration, 6 hepatic function 7 and even skin barrier function. 8 It is therefore unsurprising that a disrupted circadian rhythm will lead to organ dysfunction and consequently increases morbidity, mortality and length of ICU stay in patients with critical illness.9,10
As light is a major external influence on the circadian rhythm, exposure to artificial light may influence the normal physiological response resulting in patients in intensive care commonly having disrupted circadian rhythms. 11 Light is sensed by intrinsically photosensitive retinal ganglion cells (ipRGCs) which are independent from the image-forming rods and cones. These specialist cells utilise the photopigment melanopsin to absorb light.12-15 This information is then transmitted via the retinohypothalmic tract to the suprachiasmatic nucleus (SGN) of the anterior hypothalamus. The SGN in turn controls the amount of melatonin produced by the pineal gland.12-15 Artificial light exposure for as little as 20 min has been proven to suppress melatonin production16-19 which rapidly impacts the circadian rhythm.
Naturally occurring lux levels 30.

There are no medically recognised standards for how much light should be in ICU; however, there are industrial standards when building new ICUs. These standards recommend 20 lux as the night-time illumination within an ICU. 20
In our previous pilot single-centre study, we demonstrated a relationship between severity of illness and lux exposure. 21 The purpose of this current study was to more definitively quantify the levels of nocturnal light in an ICU and assess relationship between patient acuity, environmental acuity (neighbouring patient acuity), type of bed space (open bay or side room) and type of admission (medical or surgical) with nocturnal light exposure. To our knowledge, this study is the most comprehensive analysis of nocturnal light in an ICU that has been carried out to date.
Methods
This prospective multicentre observational study ran between April and June 2019. The project was run in conjunction with the Wessex trainee research network SPARC (South Coast Perioperative Audit and Research Collaborative). The study was registered with the Health Research Authority (IRAS 232050) and granted ethical approval via proportional review (18/LO/0258).
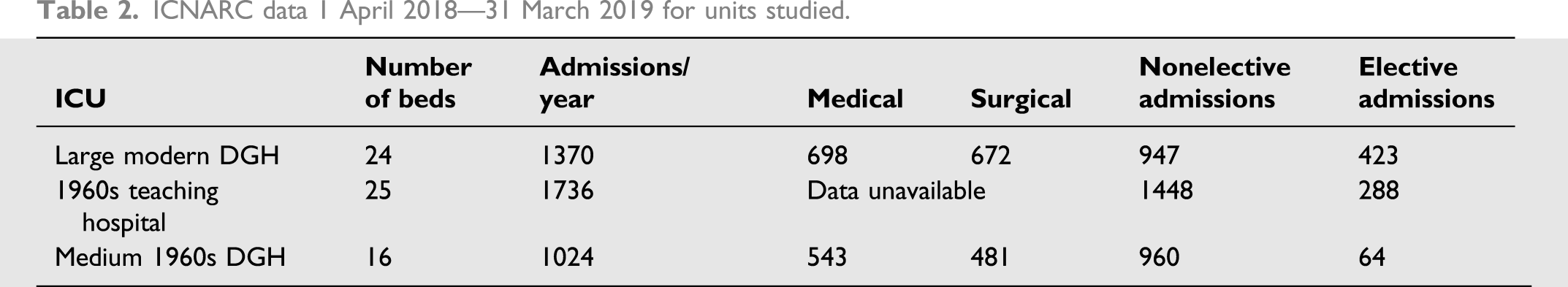
ICNARC data 1 April 2018—31 March 2019 for units studied.
Example bed representation and recording schedule.
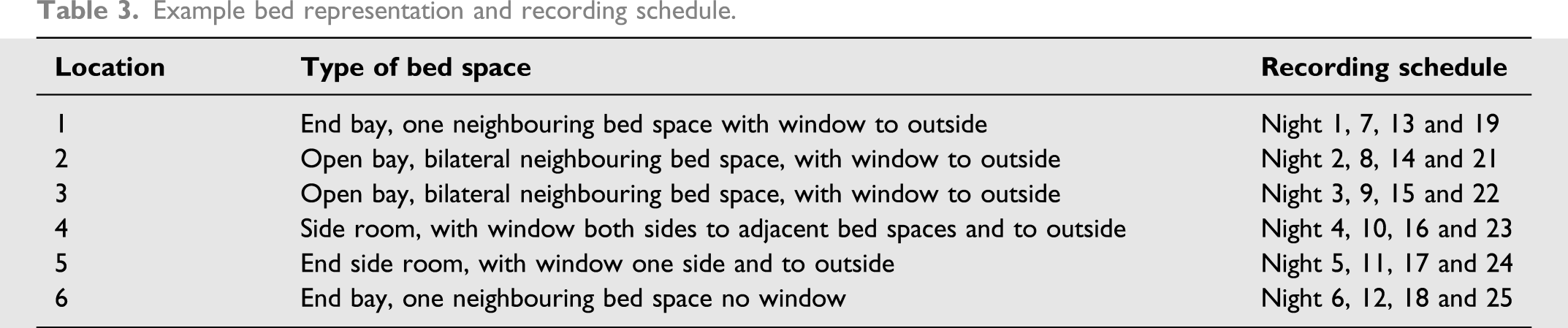
Once the bed spaces to study had been determined, a schedule was drawn up for which bed space (and its adjacent bed spaces if applicable) should be studied on which night, independent of the acuity of the admitted patients to give some randomisation to the patients studied. Participants were eligible for inclusion if they were located in one of these pre-determined bed spaces, on the night that bed space was due to be studied. If the bed space was empty on the night it was due to be studied, the next bed space on the schedule was selected.
Exclusion criteria included any patient receiving palliative care, patients at risk of cross contamination (MRSA, etc.) and bedside nursing staff not in agreement with participation for clinical reasons, of which there were zero incidents.
Digital luxometers were used (PCE-174 luxometers, externally calibrated by Exova METECH to +/−5% within 0–400 lux) to record light data using a photoreceptor which produces an electrical current proportional to lux levels via a transducer.
Light data were recorded as lux as this gives the most accurate representation of the amount of light exposure patients have and is the unit used as a standard for industrial light levels. Data were recorded from 23:00–05:00 to minimise any seasonal variation in light levels. Each ICU recorded one set of data for one bed space per night.
The luxometers were attached to a flexible extension device, to facilitate them being as close to the patient’s eyeline as possible and at the same angle they were positioned. This was in turn connected to a laptop which downloaded data directly onto a spreadsheet (Microsoft Excel) (Figure 1). Equipment set up.
The luxometers were programmed to record data every 5 s from 23:00–05:00, which led to approximately 1.3 million individual data points. The frequency of data recording was important to ensure short peaks in the light level were accurately identified.
As a marker of acuity, a single SOFA score of the patient in the predetermined bed space was calculated at the beginning of the night, as well as a single SOFA score of any adjacent patient. If there were two adjacent patients, then the sum of each patient’s SOFA was calculated to give a single numerical representation of surrounding acuity. Type of admission (medical or surgical), age of patient and type of bed space (side room or open bay) was also recorded.
Lux data was nonparametric, and therefore, Spearman’s rank coefficient was used for correlation, and the Mann–Whitney U-test was used to compare groups. A two-tailed test with a threshold p-value of .05 was used. The SOFA spread was normally distributed, and therefore, the mean SOFA was used. Because of significant variation in lux as time progresses, heat maps were used to examine light intensity throughout the recording period. In these heat maps, maximum and median lux data were split into 30-min segments to allow visual comparison of lux and time. This allowed physiologically important, but short, periods of high lux masked by crude averages to be visualised. Cumulative lux (the sum of all lux recorded throughout an individual night) was used as a measure of total light exposure throughout the night.
Results
In total, 60 nights’ data were recorded across the three ICUs, studying 49 different patients–some longer-staying patients were studied twice as they remained in the same bed space when the schedule repeated itself. Despite repeating patients, SOFA scores differed when the data were captured on subsequent nights as their clinical condition changed over time. The study resulted in 1,252,800 individual lux data points. The mean age of patients was 59 years, with a mean SOFA score of 3.4. There were 29 medical admissions and 29 surgical admissions.
Overall data and site comparison.
Acuity vs lux, open bay vs side room, medical vs surgical.
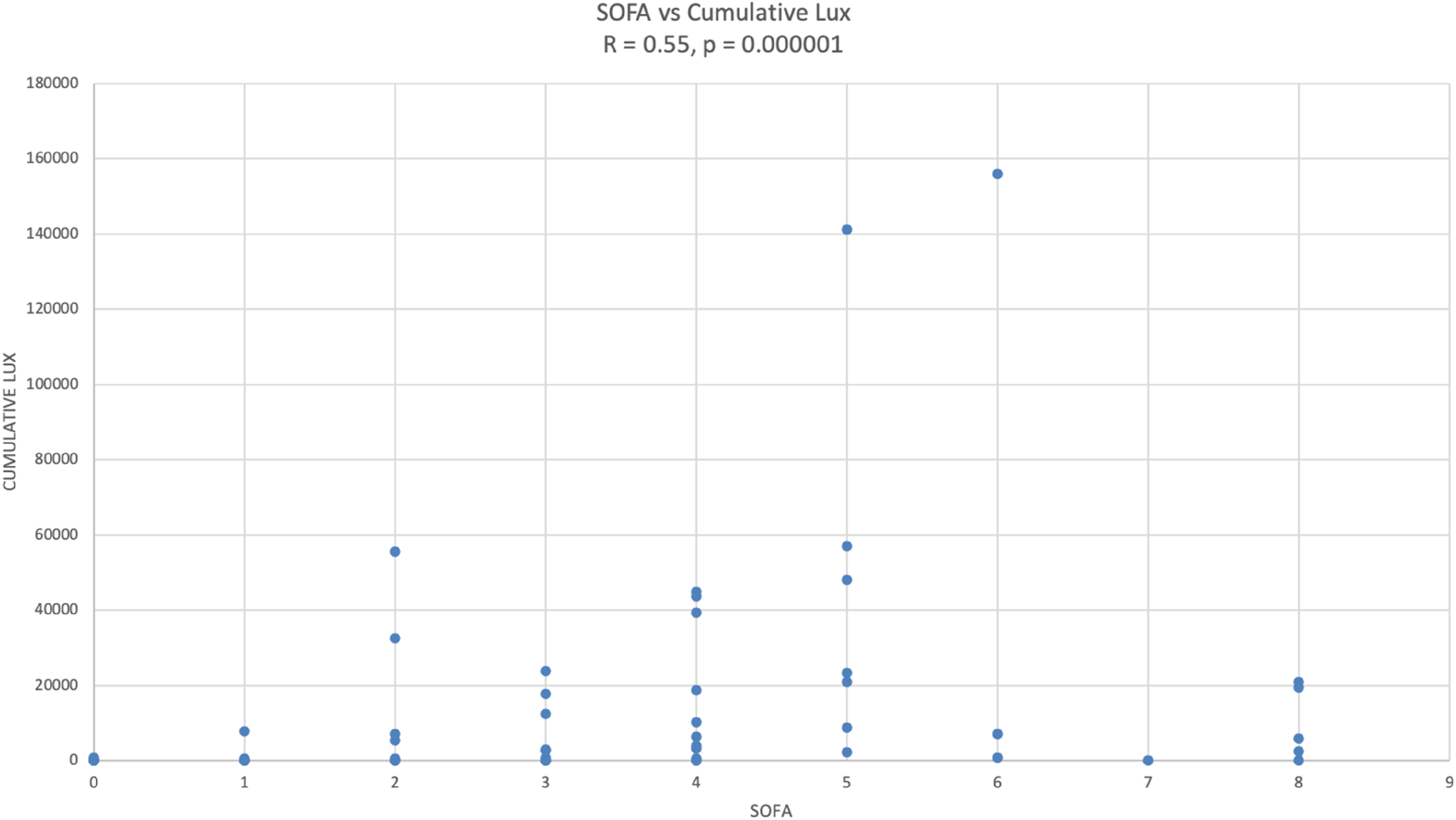
SOFA vs cumulative lux.
When split into 30-minute segments and analysed as heat maps, higher median and maximum lux levels persist for longer as the night progresses in higher acuity patients (Figure 3). There was no relationship between adjacent patient acuity and lux. Heat Map of SOFA v median and maximum lux, per 30 mins.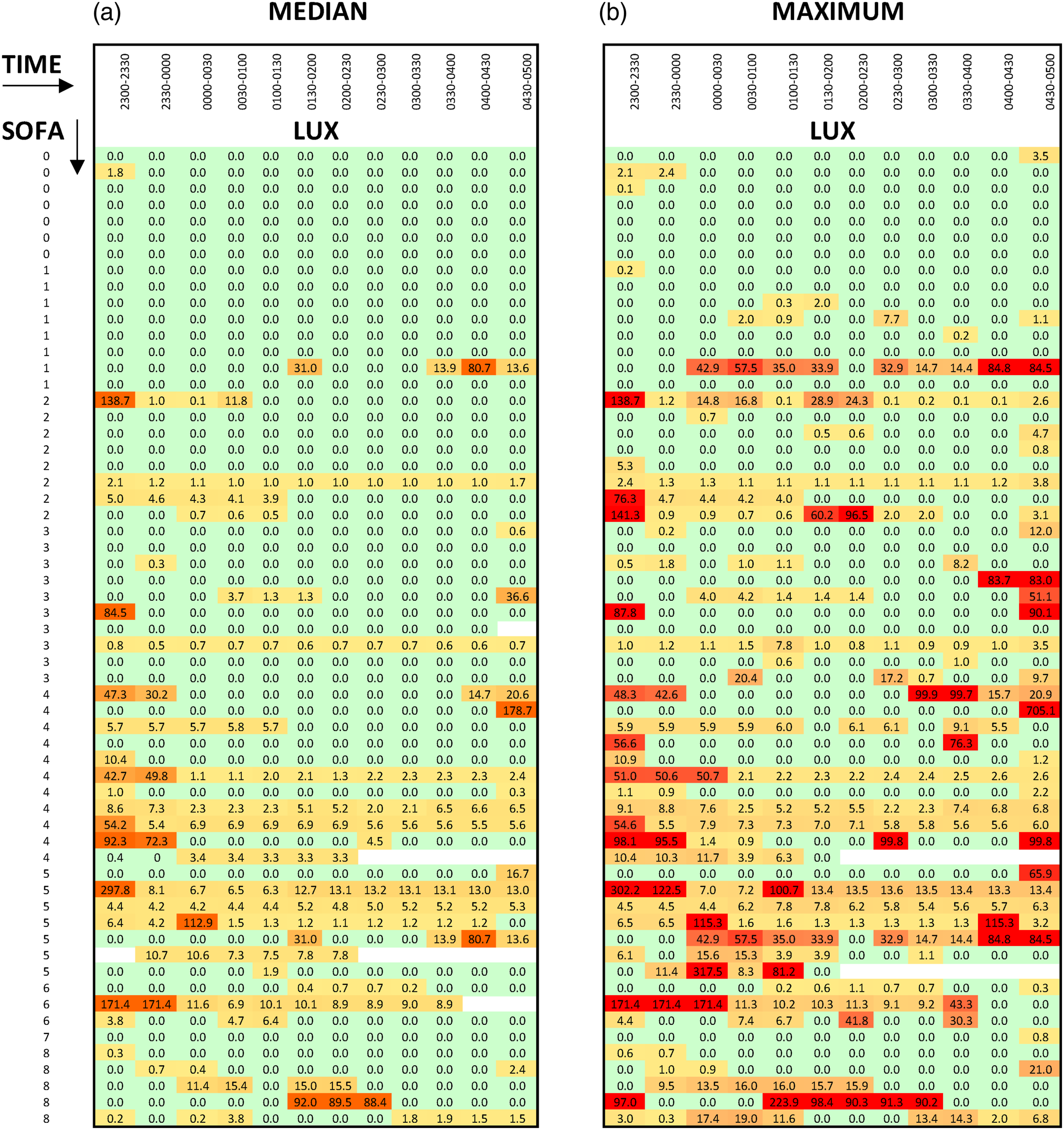
Discussion
The main observation of this prospective study of nocturnal light exposure on critical care is that there is a positive correlation between increasing SOFA score and increasing lux, which likely reflects the higher medical and nursing workload required to care for higher acuity patients. Whilst this elevated lux level may be justified as workload increases, it is also plausible that disruption in physiology through circadian disturbance could be detrimental.1-9 However, it is important to recognise that low light levels will make the delivery of care more challenging for staff and may increase errors in care. Errors due to staff working in low light environments (such as drug administration errors) need to be balanced against the physiological disturbance of circadian disruption when choosing a nocturnal light level.
There was no relationship between index patient lux exposure and neighbouring patient acuity. This is important, as it potentially impacts patient distribution within the ICU. If there was a relationship between neighbouring acuity and lux exposure, consideration may have needed to be given as to where patients are positioned in the unit in relation to other patients. It should however be recognised that this observational study was not powered to demonstrate such a difference and that results may be confounded by the individual design of the units studied.
There was also variation between the hospitals. Whilst there are many confounding factors for this variation (including acuity), it is feasible that hospital design playing a role in lux exposure and environmental light should be considered in the design of new units.
Despite lower median light exposure seen in the more modern-built ICU, current design and technology does not allow both low patient light exposure and higher light levels for clinical staff within the same bed space. Some simple measures can be employed to minimise environmental harm. Eye masks have proven to increase the perceived quality of ICU sleep22,23 and maintain normal hormonal function in ICU environments. 24 These are cheap and widely available with minimal morbidity. Exogenous melatonin is used to aid sleep in the critically unwell, and in some small studies, it has been shown to be effective in healthy patients in a simulated environment, 25 but there is conflicting evidence for its wider use. 26 Some potential areas for technological development include minimising the blue light output of medical lights and devices, 27 optimising daytime wavelength of light output 28 and even the potential development of wearable LED facemasks. 29
The device itself may have affected the results. It was made as unobtrusive as possible, with only the sensor itself visible from the end of the bed, and staff were told it was to monitor the environment, not specifically light; however, it must be acknowledged that the presence of the device may have altered staff behaviour. This likely affected the beginning of the shift the most, especially time around ‘lights out’, but over the course of a 12-h shift, it is theorised that practice returned to near normal levels.
The main limitation of this study is that it does not address patient-centred outcomes. The theorised negative impact on patients is hypothesised from previously published research. Whilst patient-centred outcomes were considered as part of this study design, the amount of confounders present and sample size required made it unviable for our work. Further, larger studies are needed which might also measure cortisol levels and delirium, and patient-centred outcomes are needed to better understand the importance of this issue and guide practice further.
Conclusion
Clinicians should be sensitive to the impact of environmental factors such as light on patients, whilst ensuring the environment is optimised for the delivery of clinical care.
In designing new hospitals, ICU lighting design should be considered and made patient centred to minimise the effect on physiology. In existing ICUs, the routine use of a combination of exogenous melatonin and eye masks may be a simple and low-morbidity management strategy for disrupted ICU circadian rhythms, but the effectiveness of this strategy has not been proven in large-scale trials.
Normalising and supporting physiology is a core principle of intensive care medicine, and although circadian rhythm dysfunction may not be as visible as other physiological processes, its effects shine brightly on patients. Intensive care interventions continue to get ever more complex and costly, but the ICU environment is often overlooked by clinicians as a barrier to wellness. It is the sickest patients that have the most to gain from normal circadian cycles that the environment may be disrupting the most. Clinicians must be cognisant to the invisible threat of disrupted circadian rhythms and always remember Airway, Breathing, Circulation, Disability…Environment.
Footnotes
Contributors – On behalf of the SPARC research network
Dr. Ben Harris, Dr. Shivan Kanani, Dr. Christopher Asplin, Dr. David Shearn, Dr. Irena Grecu, Dr. Alex Pettipher, Dr. Jatinder Pabla, Dr. Rebecca Cusack, Dr. Paul Wilson, Dr. Roger Lightfoot and Dr. Matthew Taylor.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was funded by a National Institute of Academic Anaesthesia Research Grant.
