Abstract
Background
Right ventricular (RV) function is increasingly being recognised as an important factor influencing outcomes in ST elevation myocardial infarction (STEMI) complicated by cardiogenic shock (CS). In this study, we investigated RV echocardiographic parameters’ association with 28-day mortality in patients admitted to intensive care with STEMI complicated by CS with reduced left ventricle ejection fraction (LVEF).
Method
We performed a retrospective analysis of patients admitted to intensive care unit (ICU) in a single tertiary cardiac centre over a 34-month period with STEMI complicated by CS and LVEF < 40%. Clinical and echocardiographic data were collected and correlated with 28-day mortality.
Results
One-hundred patients were included with a mean age of 62.6 ±12.7 years and 78% were male. Mortality at 28 days was 37%. Respectively, 85%, 40% and 25% of patients required mechanical ventilation, mechanical circulatory support and renal replacement therapy. Tricuspid annulus peak systolic velocity (RV S’) was significantly higher in survivors (12 ± 3.3 v 10 ± 3.5 cm/s, p = 0.03) and was an independent predictor of mortality (odds ratio 1.2, 95% confidence interval 1.1–1.4, p = 0.04). RV S’ of 10.5 cm/s exhibited best sensitivity and specificity (64% and 65%, respectively; p = 0.02) for mortality. The Kaplan–Meier curve demonstrated 85% risk of 28-day mortality for RV S’ < 10.5 cm/s v 53% for RV S’ > 10.5 cm/s (p = 0.02).
Conclusion
RV function is associated 28-day mortality in patients admitted to ICU with STEMI complicated by CS with reduced LVEF. RV S’ predicted mortality with good sensitivity and specificity.
Introduction
Cardiogenic shock (CS) following ST elevation myocardial infarction (STEMI) is a frequent life-threatening complication. 1 CS may result in multiorgan failure requiring admission to intensive care 2 for pharmacological and often mechanical circulatory support (MCS) for cardiac output to maintain end-organ perfusion and oxygen delivery. 3
The incidence of CS complicating STEMI in the United States has been steadily increasing from 6.3% in 2003 to 10.1% in 2010. 1 Over the same period, there has been an improvement in survival, but it remains unacceptably high at 34%. 1 Also, in the United Kingdom, the incidence has risen from 7% in 2007 to 13% in 2015 with mortality up to 40%. 4 The improvement in outcome seems associated with increased early mechanical revascularisation. 1 Length of hospital stay has remained static, whilst the cost has been steadily rising. 1 End-organ dysfunction such as acute kidney injury (AKI) is part of CS definition. AKI complicating CS incidence is up to 50% and is a predictor of mortality. 5
In chronic heart failure, right ventricular (RV) function is an established predictor of outcomes.6,7 Recently, the role of the RV in acute heart failure and CS is being increasingly recognised. Patients admitted to hospital with acute decompensated heart failure and lower tricuspid annular plane systolic excursion (TAPSE) have a higher 6-month mortality. 8 Also, RV function was found to be an independent predictor of CS development and mortality in patients presenting with anterior STEMI. 9 In another study, RV function in STEMI complicated by CS was associated with higher 1-year mortality where TAPSE was adopted as a marker of RV function. 10
Little is known about the association between RV function and short-term outcomes in CS. It is plausible that RV function may influence mortality and complications in this cohort of intensive care patients. In this retrospective observational hypothesis-generating study, our objective was to investigate the association between RV parameters evaluated by echocardiography and 28-day mortality in patients admitted to intensive care unit (ICU) with STEMI complicated by CS and reduced left ventricle ejection fraction (LVEF). We also investigated the association between RV parameters and the need for mechanical ventilation, MCS and renal replacement therapy (RRT) in the same cohort.
Materials and Methods
Patients
We retrospectively reviewed consecutive patients admitted with the diagnosis of myocardial infarction complicated by cardiogenic shock to the intensive care unit (ICU) in our tertiary cardiac centre between October 2016 and September 2019. Patients were included in the study if they were more than 18 years old, admitted with a primary diagnosis of STEMI followed by primary percutaneous coronary intervention (PCI) with CS requiring ICU admission, had impaired left ventricular (LV) systolic function and underwent echocardiography within 48 hours of admission to ICU. CS was defined for the purpose of this study as a systolic arterial pressure less than 90 mmHg or a mean arterial pressure less that 65 mmHg, with or without therapy, and evidence of at least one organ hypoperfusion (e.g. delirium, acute kidney injury, and acute liver injury).11,12 LV systolic impairment was defined as LV ejection fraction (EF) less than 40%. 13 Patients were excluded from the study if they were less than 18 years old, pregnant, had non-cardiogenic cause of shock, suffered cardiac arrest with early signs of cerebral hypoxia, were considered moribund by the admitting physician (defined as imminent death with no medical therapeutic option) or patients with echocardiograms where RV and LV assessments were not possible.
Clinical information was collected about patients’ demographics, comorbidities, medical history, PCI, mechanical complications of STEMI (ventricular septal defect and moderate or more mitral valve regurgitation), clinical parameters, laboratory tests, medications, APACHE II severity score, presence of mechanical ventilation, MCS and RRT, length of stay and mortality by review of electronic case records. Clinical parameters were collected as worst documented values on Day 1 of admission to ICU. Mortality at 28 days from ICU admission was considered the primary endpoint. Need for mechanical ventilation, MCS and RRT were secondary endpoints. APACHE II score was calculated as described previously. 14 The STEMI culprit lesion was identified as recorded at the time of PCI. Data about PCI, thrombolysis in myocardial infarction (TIMI) post-PCI flow score and referral for coronary bypass surgery were collected as documented at time of PCI. The study was conducted according to Declaration of Helsinki and with local research governance approval.
Echocardiography
Echocardiography was performed by experienced accredited (British Society of Echocardiography or European Association of Cardiovascular Imaging) cardiac sonographers at the bedside in ICU using commercially available ultrasound systems with a 3.5-Mhz probe (Vivid E7 or Vivid E9, GE Health Care, Horten, Norway). Images and reports were stored in a dedicated central image storage facility. Echocardiograms including chamber quantification were conducted in accordance with the British Society of Echocardiography recommendations. 15 Chamber quantification data were collected from stored images and reports by the study team blinded to clinical information and included: LV ejection fraction by Simpson’s biplane or visual estimate at the time of the original scan, left ventricle outflow tract (LVOT) velocity time integral (VTI) by pulse-wave Doppler at 0.5–1 cm proximal to the aortic valve, LVOT diameter, right ventricular (RV) dimensions, right ventricle outflow tract (RVOT) dimensions, tricuspid annular plane systolic excursion (TAPSE) by M-mode of the lateral tricuspid annulus and tricuspid annulus peak systolic velocity (RV S’) by tissue Doppler at the lateral tricuspid annulus. 15 RV diastolic parameters were not available as they were not part of the dataset guidelines at the time of scanning. Tricuspid regurgitation maximum velocity (TR Vmax) was investigated by continuous-wave Doppler through the tricuspid valve in systole. Stroke volume (SR), cardiac output (CO) and cardiac index were calculated as LVOT VTI x π x LVOT radius, 2 SR x heart rate and CO/body surface area (BSA), respectively. Data were also collected about ventricular septal defect (VSD) and significant mitral valve regurgitation (moderate or more) as documented in the reports.
Statistical analysis
Continuous data were presented as mean and standard deviation or median and interquartile range depending on normality of distribution tested by QQ plot and Shapiro–Wilk test. Numbers and percentages were used to describe dichotomous data. Continuous data were compared by Student t and Mann–Whitney U tests for parametric and non-parametric data, respectively. Chi square tests were utilised to compare categorical data. Correlation of continuous with categorical data was performed by simple logistic regression. Results of logistic regression analysis were presented as odds ratio (OR) and 95% confidence interval (CI). Cox multivariate analysis and logistic regression were employed to investigate independent factors associated with mortality and the need for mechanical ventilation, MCS and RRT, respectively. Sensitivity and specificity were determined by the receiver-operating characteristic (ROC) curve. The Kaplan–Meier method with log-rank test was employed to compare survival between strata. The statistical analysis plan was defined prior to analysis. Missing data were not imputed. Graphpad Prism version 8.3 software package (Graphpad Software, San Diego, California, USA) for MacOS was used for statistical analysis except Cox regression multivariate analysis, which was performed using SPSS software version 26 for MacOS (IBM SPSS Inc., Chicago, Illinois, USA). A p value < 0.05 was considered significant.
Results
A total of 253 patients with a diagnosis of CS secondary to STEMI case notes were reviewed. Forty-nine patients did not have echocardiography within 48 hours, 33 patients had poor acoustic windows and 71 patients had LVEF > 40% were excluded. Therefore, 100 patients met the inclusion criteria and were included in the study (Figure 1). The mean age of the cohort was 62.6 ± 12.7 years, 78% were male and 38% suffered out of hospital cardiac arrest (OHCA). Eighty-seven patients were admitted to ICU directly following PCI and the remaining 13 patients were admitted within 48 hours. Median acute physiology and chronic health evaluation II (APACHE II) score was 17 (13–21) with a median length of ICU and hospital stay of 6 days (4–11) and 17 days (8–26), respectively. All-cause 28-day mortality was 37% (37 patients) with 32 out of 37 deaths occurring in ICU. Cardiovascular death occurred in 28 patients and 6 died due to neurological complications. Mechanical ventilation was required in 85% of cases, while MCS and RRT were initiated in 40% and 25% of patients, respectively (Supplementary Table 1). Echocardiography review revealed median LVEF 29% (20–35) with mean LVOT VTI 14.3 ± 4.6 cm, SV 47 ± 16 mL, median CO 3.83 (2.65, 4.63) l/min and cardiac index 1.93 (1.40, 2.51) l/min/m.
2
While mean RV parameters were TAPSE 16 ± 5 mm and RV S’ 11 ± 4 cm/s (Supplementary Table 2). Echocardiography data availability is summarised in Supplementary Table 3. Inclusion and exclusion flowchart.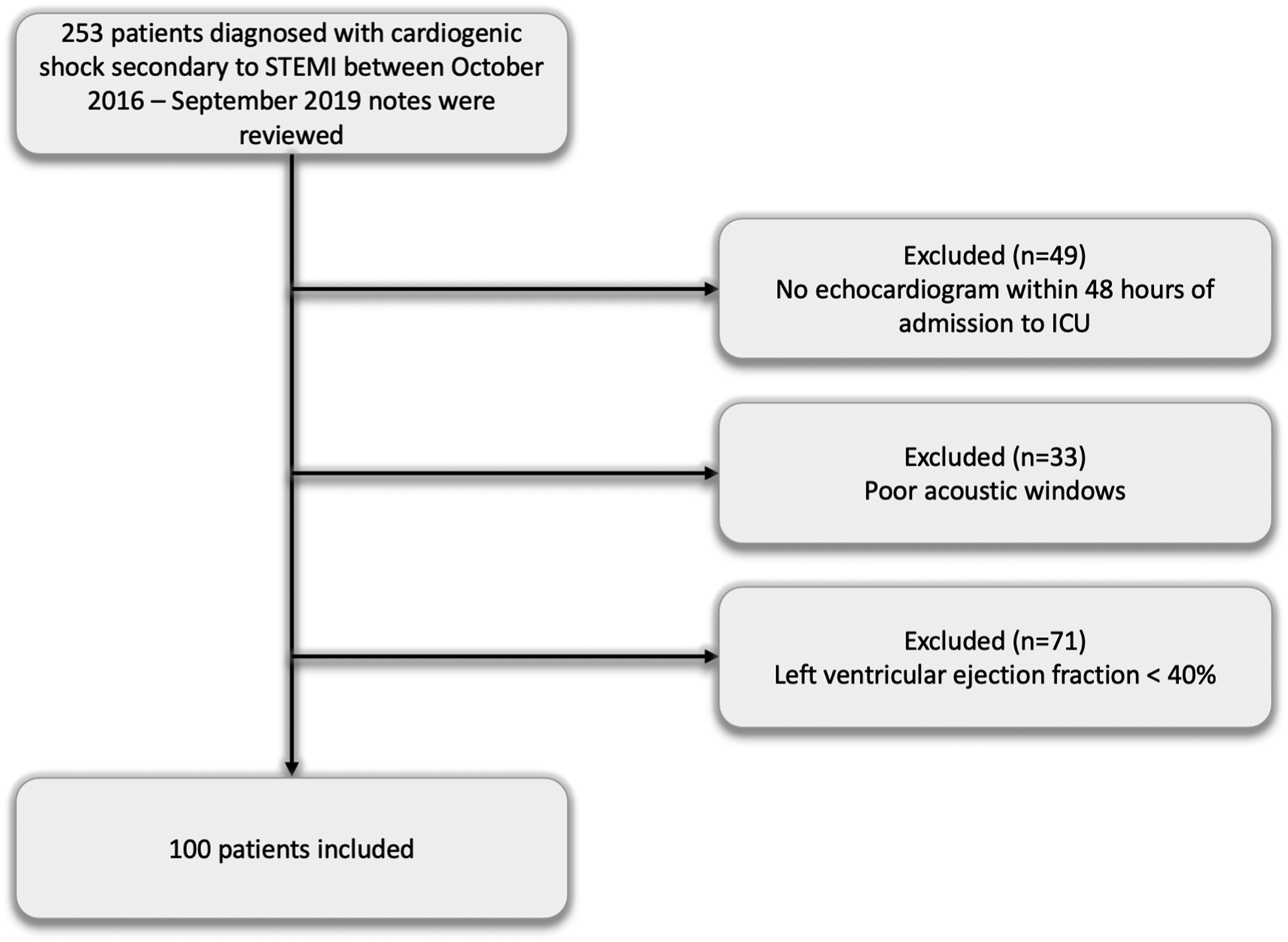
Demographics, medical history, culprit lesion and clinical parameters of 28-day survivors and non-survivors.
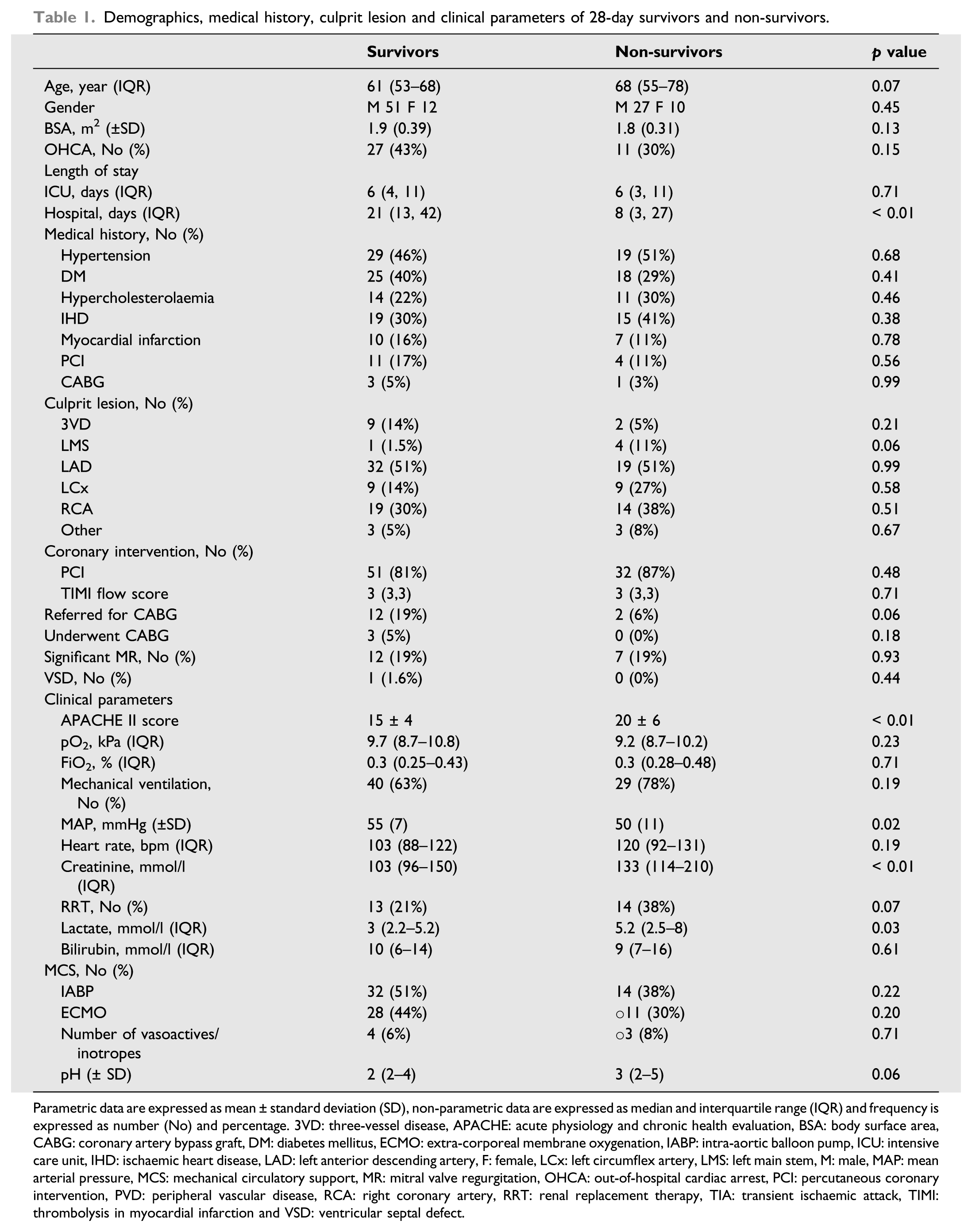
Parametric data are expressed as mean ± standard deviation (SD), non-parametric data are expressed as median and interquartile range (IQR) and frequency is expressed as number (No) and percentage. 3VD: three-vessel disease, APACHE: acute physiology and chronic health evaluation, BSA: body surface area, CABG: coronary artery bypass graft, DM: diabetes mellitus, ECMO: extra-corporeal membrane oxygenation, IABP: intra-aortic balloon pump, ICU: intensive care unit, IHD: ischaemic heart disease, LAD: left anterior descending artery, F: female, LCx: left circumflex artery, LMS: left main stem, M: male, MAP: mean arterial pressure, MCS: mechanical circulatory support, MR: mitral valve regurgitation, OHCA: out-of-hospital cardiac arrest, PCI: percutaneous coronary intervention, PVD: peripheral vascular disease, RCA: right coronary artery, RRT: renal replacement therapy, TIA: transient ischaemic attack, TIMI: thrombolysis in myocardial infarction and VSD: ventricular septal defect.
Echocardiographic parameters of 28-day survivors v non-survivors.
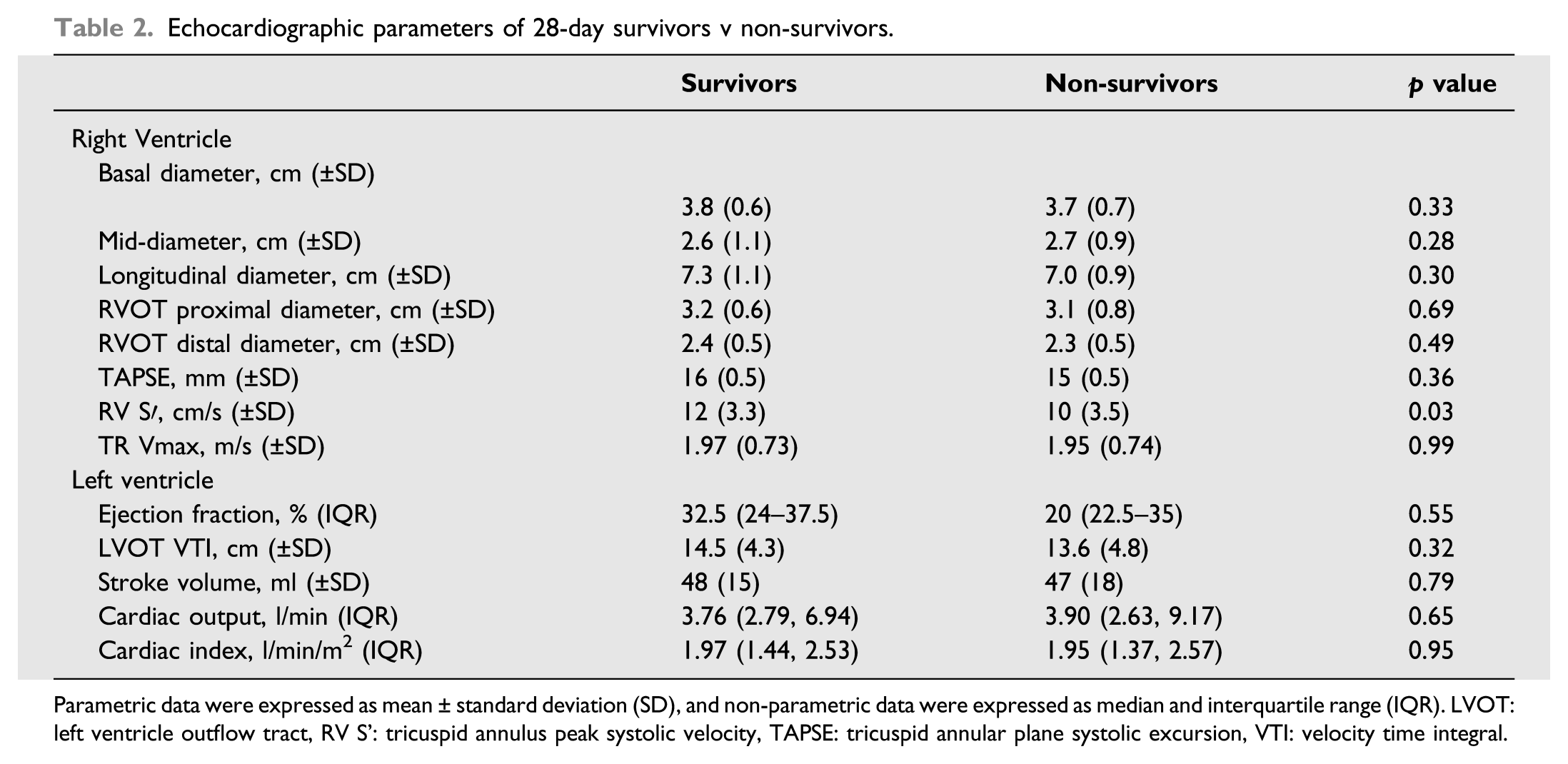
Parametric data were expressed as mean ± standard deviation (SD), and non-parametric data were expressed as median and interquartile range (IQR). LVOT: left ventricle outflow tract, RV S’: tricuspid annulus peak systolic velocity, TAPSE: tricuspid annular plane systolic excursion, VTI: velocity time integral.
Cox multivariate analysis of echocardiography and clinical parameters that are significantly different between 28-day survivors and non-survivors.
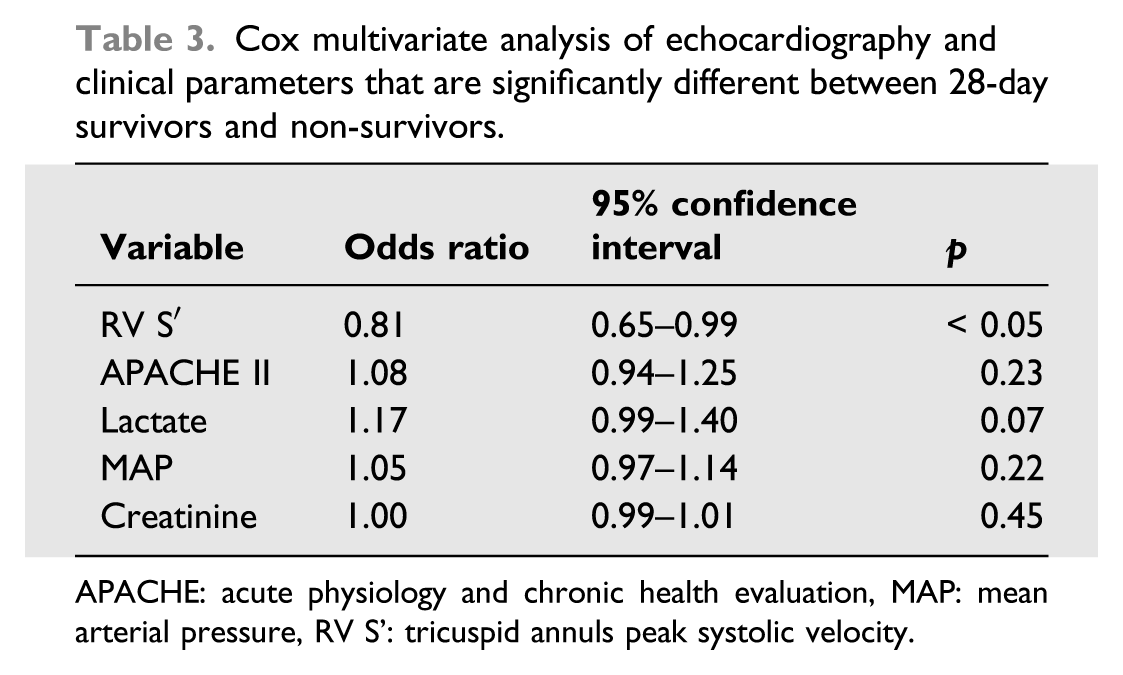
APACHE: acute physiology and chronic health evaluation, MAP: mean arterial pressure, RV S’: tricuspid annuls peak systolic velocity.
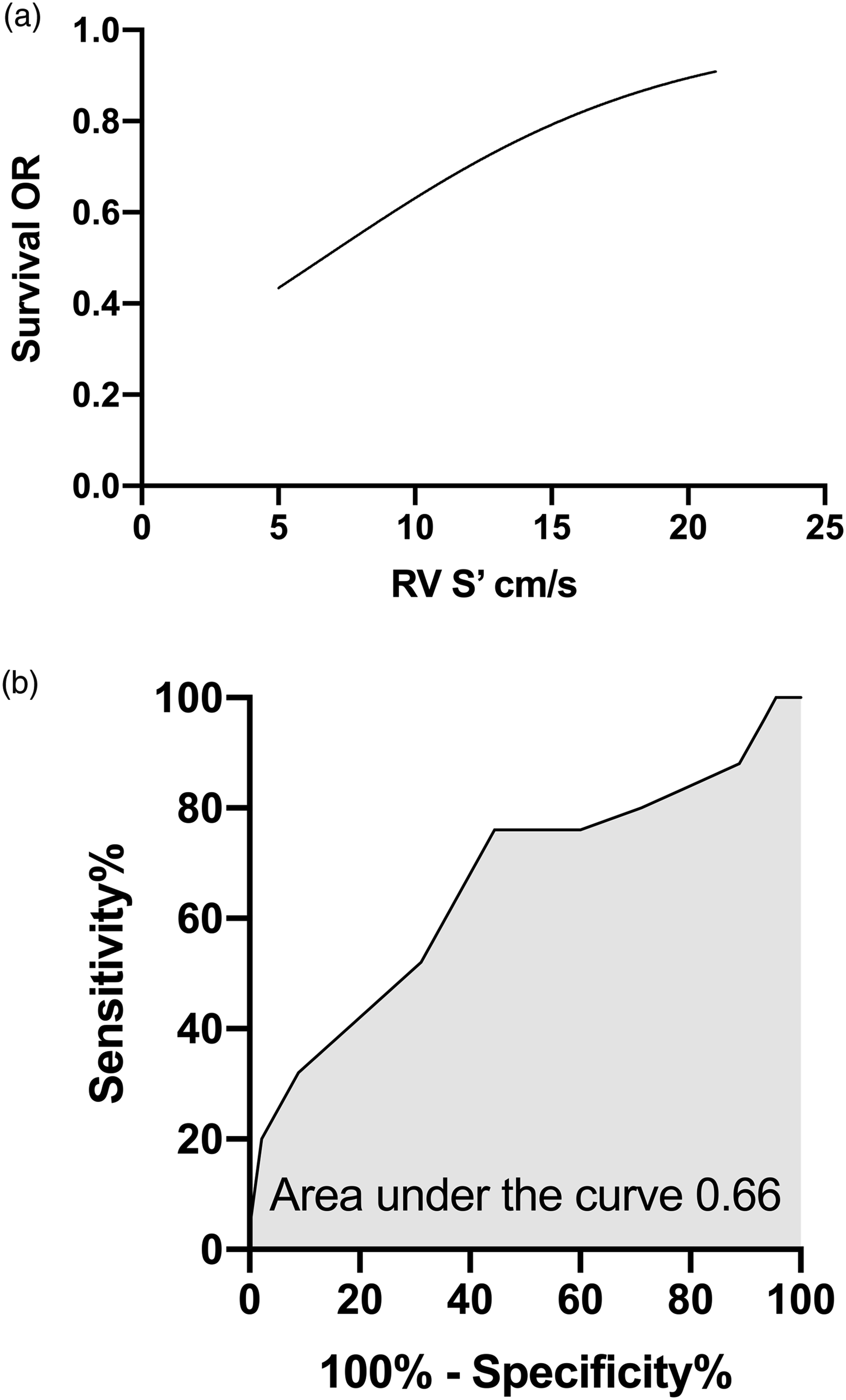
RV S′ prediction of survival. (a) Univariate logistic regression of RV S′ association with survival. (b) Receiver-operating characteristic curve (ROC) of RV S′ predicting survival. OR: odds ratio. RV S’: tricuspid annulus peak systolic velocity.
An RV S′ cut-off of 10.5 cm/s was used as the basis for Kaplan–Meier survival analysis which demonstrated that patients with RV S’ > 10.5 cm/s had better chance of 28-day survival compared to patients with RV S’ < 10.5 cm/s (85% vs 53%, p = 0.01) (Figure 3). Kaplan–Meier survival curves of 28-day mortality using RV S′ cut-off value of 10.5 cm/s. RV S’: tricuspid annulus peak systolic velocity.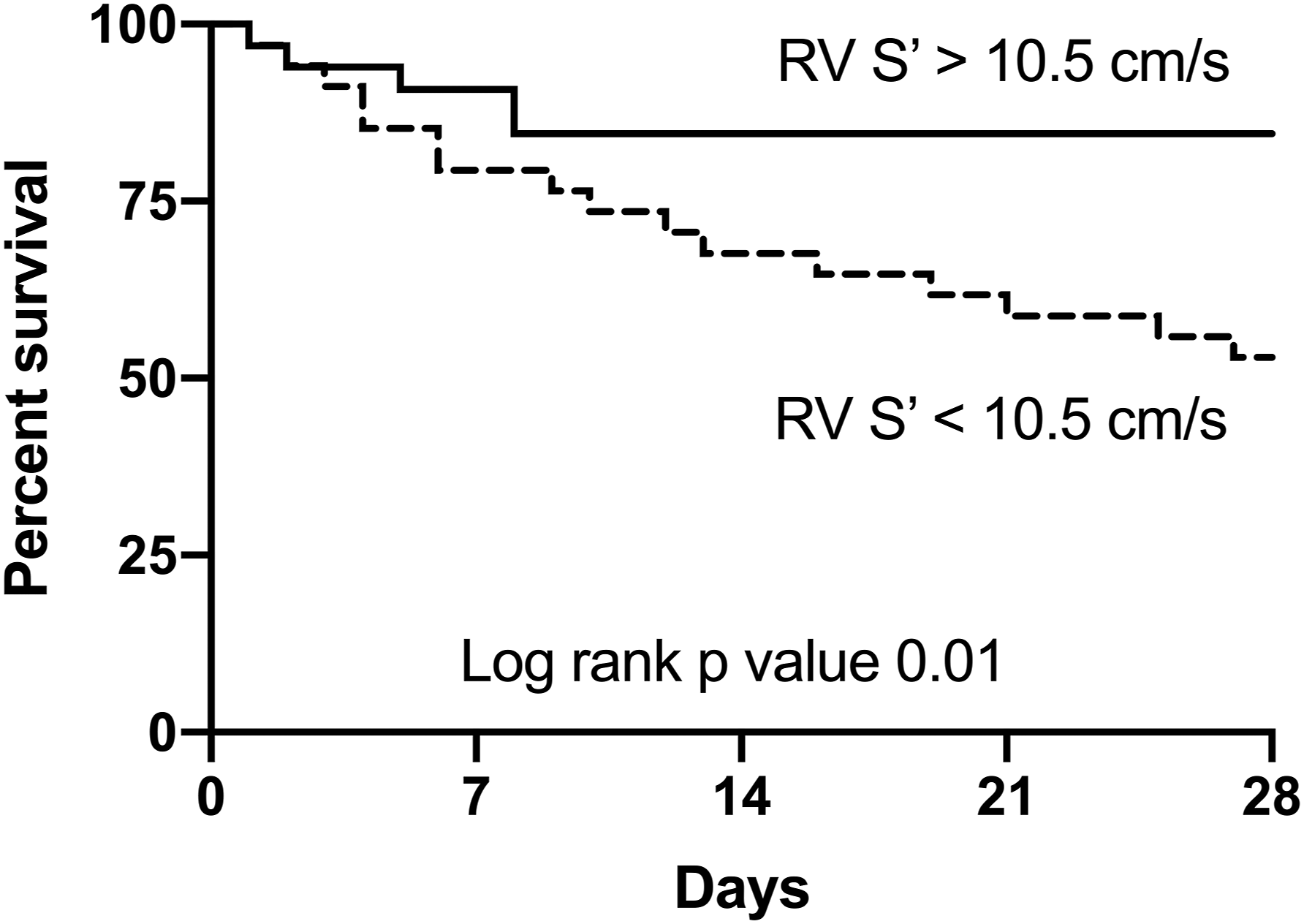
For the secondary endpoints, there was no statistically significant difference in echocardiographic parameters based on the need for mechanical ventilation (Supplementary Table 6). In patients requiring MCS, LVOT VTI (13 (10, 15) cm v 14 (12, 18) cm, p = 0.03) and SV (43 ± 16 mL v 50 ± 15 mL, p = 0.03) were significantly lower compared to those who did not (Supplementary Table 7). Other echocardiographic parameters were not significantly different. Logistic regression revealed that neither LVOT VTI nor SV was able to predict the need for MCS (LVOT VTI OR = 0.93, 95% CI 0.84–1.01, p = 0.1, SV OR = 0.97, 95% CI 0.94–1, p = 0.06). TAPSE was significantly lower in patients requiring RRT (14 ± 4 mm v 16 ± 5 mm, p = 0.03) compared to patients not needing it. TAPSE, though, was not able to predict the need for RRT in this cohort (OR = 0.41, 95% CI 0.14–1.09, p = 0.08). All other echocardiographic parameters were comparable (Supplementary Table 8).
Discussion
In this retrospective, observational, hypothesis-generating study, we investigated RV size and function parameters measured by echocardiography correlation with 28-day mortality in patients admitted to ICU with STEMI complicated by CS and reduced LVEF (< 40%). We also investigated the correlation between RV function parameters and the need for mechanical ventilation, MCS and RRT as secondary endpoints in the same cohort. Patients recruited to this study had borderline RV function by TAPSE and RV S’. Non-survivors had higher APACHE II score, creatinine and lactate; and lower MAP and RV S’. RV S’ was found to be an independent predictor of 28-day mortality in this cohort, and RV S′ of 10.5 cm/s presented the best sensitivity (64%) and specificity (62%) in prediction of mortality (area under the curve 0.66). RV S’ > 10.5 cm/s was associated with 85% survival compared to 53% survival in patients with RV S’ < 10.5 cm/s. For the secondary endpoints, TAPSE was lower in patients needing RRT, but there was no significant difference in RV parameters based on the need for mechanical ventilation or MCS.
RV ejection relies on the contraction of its longitudinal fibres and on LV contractility through the interventricular septum and inter-connection of fibres. 16 In patients presenting with STEMI complicated by CS with significantly reduced LV systolic function, RV stroke volume will depend primarily on its longitudinal fibres’ contraction to maintain cardiac output. Previous studies have shown that patients presenting with acute myocardial infarction associated with RV dysfunction had a higher incidence of developing CS.17,18 In addition, for patients presenting with STEMI complicated by CS with impaired LV and good RV function (measured by TAPSE), 1 year survival was better than those with biventricular impairment. 10 Furthermore, RV S′ was able to predict 2-year cardiovascular outcomes in patients with newly diagnosed significant LV systolic dysfunction. 19 We demonstrated that this was also the case for short-term outcome in STEMI complicated by CS with significantly impaired LV systolic function where RV S′ correlated with outcome and was able to predict 28-day mortality. This highlights the importance of RV function in this cohort and the need for increased awareness of RV status during clinical care.
The RV function variability in this cohort is probably multifactorial. LV dysfunction has been recently demonstrated to be a cause for significant reduction in RV function. 20 In our patients, LV function is significantly reduced (EF = 29%), rendering it a plausible major contributor to RV function variability. Ischaemia (acute or chronic) is another likely contributing factor. OHCA preceded presentation in 38% of this cohort which may result in variable degrees of myocardial stunning that might have an impact on the RV. The majority of patients included in this study were mechanically ventilated (85%), some due to hypoxaemia, which is known to negatively impact the RV. 21 Furthermore, positive pressure mechanical ventilation has a negative impact on the RV through changes in intra-thoracic pressure. 22 A rise in intra-thoracic pressure results in compression of pulmonary vasculature leading to increasing pulmonary vascular resistance and RV afterload. This could result in RV dysfunction and compromised stroke volume causing further embarrassment to patients already in cardiogenic shock. 23 Another contributing factor may be pre-existing disease. These contributing factors may shed some light on the reasons for the RV differences observed in this study.
RV S′ measures the tricuspid annulus peak systolic velocity, which is a marker of longitudinal function, and was recently shown along with TAPSE to correlate well with RV ejection fraction (EF) measured by thermodilution in a small mixed cohort of critically ill patients compared to other parameters such as fractional area change (FAC). 24 Also, RV S′ correlated well with RV EF measured by cardiac magnetic resonance (CMR), but RV S′ demonstrated less inter- and intra-observer variability compared to other parameters. 25 Another study examined the effect of RV function on long-term outcome of patients with newly diagnosed LV systolic dysfunction and demonstrated that RV S’ > 9.5 cm/s is associated with better outcomes (this value is close to the cut-off we found to be associated with short-term mortality). 26 RV S’ < 10 cm/s was associated with significantly compromised cardiac output (cardiac index < 2L/min/m2) by right heart catheter in patients with pulmonary hypertension 27 which is again close to the cut-off we found to be associated with 28-day mortality. This suggests that RV S′ could be used to risk-stratify patients admitted with STEMI complicated by CS and potentially play a role as a therapeutic target in the haemodynamic management in this difficult-to-manage group of patients. In addition, lower RV S′ could alert the clinician to early adoption of continuous RV function measurement via advanced cardiac output monitoring (e.g. advanced pulmonary artery catheter) as well as ‘RV protective’ strategies including optimisation of gas exchange and ventilatory pressures, pulmonary vasodilators, judicious fluid management, inotropy and MCS. 28
Guidelines advocate RV S′ of 14.1 ± 2.3 cm/s as normal and less than 9.5 cm/s as the cut-off for RV dysfunction. 29 In a cohort of sepsis patients, median RV S′ was found to range between 13 and 17 cm/s and worst outcome for 14 cm/s. 30 Other studies also demonstrated that RV S′ above the guidelines’ cut-off (12 and 15 cm/s) was associated with mortality in sepsis. 31 In our cohort, RV S′ of 10.5 cm/s which is at the lower end of the guidelines’ normal range was associated with higher risk of mortality. This suggests that in critically ill patients, echocardiographic parameters within the guidelines’ normal range do not necessarily mean adequate function. Furthermore, these guidelines were developed based on studies conducted in normal individuals and non–critically ill patients under normal loading conditions which casts a shadow on how to interpret and utilise these figures in intensive care. More research is needed to better understand the impact of critical illness and loading conditions on echocardiographic parameters.
AKI is a common complication of CS with patients often requiring RRT. RV dysfunction manifesting as lower TAPSE and RV S′ in critically ill patients were associated with developing AKI, but only TAPSE was found to be an independent predictor of AKI. 32 In another large study of more than 1800 patients admitted to ICU, those who had both RV (assessed by TAPSE) and LV dysfunction were at a higher risk of AKI and those with RV and AKI had more than a 7-fold increase in odds of mortality. Also, patients requiring RRT in cardiac and cardiothoracic ICU had a 50% prevalence of RV dysfunction. 33 In line with previous investigations, TAPSE was lower in patients requiring RRT in this study. Perhaps with a bigger sample size, TAPSE or other RV parameters (e.g. RV S′) may show stronger association with and the ability to predict the need for RRT as well as 28-day mortality.
Our investigation has several limitations inherent to its design as a retrospective, single-centre, cross-sectional study; as a result, these findings may have limited generalisability. Also, selection bias is a limitation to this study as echocardiogram quality was an inclusion criterion. The sample size may also underestimate the effect of less strong predictors of outcome. RV diastolic function parameters were not available to us, and they may play a role in this cohort of patients.
Conclusion
In this hypothesis-generating study, RV function is associated with short-term outcome in patients admitted to intensive care with STEMI complicated by CS and impaired LV systolic function. RV S′ may be a useful echocardiography-based marker for risk stratification in this cohort. Other parameters for RV function (TASPE) may have a role in the development of CS complications. Further prospective research is required to shed more light on the role of the RV in STEMI complicated by CS and which echocardiographic parameters would be suitable for risk stratification and as a potential therapeutic target in this cohort of patients.
Supplemental Material
sj-pdf-1-inc-10.1177_17511437211037927 – Supplemental Material for Right ventricular function is associated with 28-day mortality in myocardial infarction complicated by cardiogenic shock: A retrospective observational study
Supplemental Material, sj-pdf-1-inc-10.1177_17511437211037927 for Right ventricular function is associated with 28-day mortality in myocardial infarction complicated by cardiogenic shock: A retrospective observational study by Hazem Lashin, Olusegun Olusanya and Sanjeev Bhattacharyya in Journal of the Intensive Care Society
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
