Abstract
Introduction
Cardiac disease remains the largest single cause of maternal death. Whilst uncommon, left ventricular failure during pregnancy and delivery can be devastating to both mother and child. Echocardiography can play a significant role in rapidly establishing a diagnosis, guiding initial therapy and then monitoring response.
Clinical vignettes
The history, presentation and management of three cases of peri-partum left ventricular failure is examined: stress cardiomyopathy in a 34 year old with twins, left ventricular dysfunction secondary to pre-eclampsia in a 22 year old with a singleton pregnancy and a true peri-partum cardiomyopathy in a 42 year old with IVF twins. The defining risk factors, presenting characteristics and echocardiographical findings for each pathology are highlighted.
Conclusion
Echocardiography is playing an increasingly important role in the immediate assessment and management of left ventricular failure. This is especially true in the peri-partum woman, where establishing the correct therapy is both challenging and crucial due to the significant cardiovascular changes that occur around the time of delivery. To this end we believe that echocardiography should be rapidly available to guide the management of these patients by a multidisciplinary team made up of obstetricians, cardiologists, anaesthetists and intensive care physicians.
Introduction
Whilst uncommon, left ventricular failure during pregnancy and delivery can be devastating to both mother and child. Critical care clinicians may be called on to contribute to the care of these women and their management can be challenging with lack of time a major factor.
The rate of maternal death in the UK from primary cardiac disease has risen over the past 20 years. 1 It has been the leading cause of indirect maternal mortality, and the leading overall cause of maternal mortality in the UK since 2002. 2
Clinical diagnosis of cardiovascular collapse in peri-partum women mandates immediate echocardiography. Only bedside transthoracic echocardiography can provide the immediate structural and functional information required to effect immediate therapeutic manoeuvres. This skill also allows rapid reassessment to ensure progress is being made in the right direction.
Transthoracic echocardiography now plays a central role in the assessment of all causes of shock in the critical care arena and is now the modality of first choice in European consensus guidelines. 3 Its use as both a diagnostic tool and as a guide to cardiovascular therapies is well documented. 4 The MBRACE 2019 report will be focusing on both cardiac deaths and deaths after critical care which will place echocardiography at centre stage in managing the sick peri-partum mother.
Rapid recognition of the three most significant causes of de novo left ventricular failure in peri-partum women is the key to their survival. We therefore present three clinical vignettes generated from our own practice designed to signal to the reader the key differentiating clinical and echocardiographic features. We also discuss management options and present an echocardiographic algorithm intended for use at the bedside.
Clinical vignettes
Stress cardiomyopathy (SCM)

Left ventricular dysfunction secondary to pre-eclampsia

Peri-partum cardiomyopathy

Discussion and management
Maternal death is a rare but catastrophic event. Since 2002 cardiac disease has remained the single largest cause of maternal mortality, resulting in 55 deaths per 100, 000 maternities in the triennium from 2014–2016. The mortality rate from cardiac disease has remained largely unchanged despite an overall steady decrease in maternal mortality and is due to be one of the focus areas of the 2019 MBRACE report.
The three most significant causes of de novo left ventricular failure in peripartum women are stress cardiomyopathy, severe pre-eclampsia and autoimmune peri-partum cardiomyopathy in decreasing order of frequency. Autoimmune peri-partum cardiomyopathy accounts for a small number of maternal deaths: 9 out of the 153 deaths attributable to cardiac disease in the six-year period from 2009–2014. It is vital that clinicians understand its rarity, preventing over-diagnosis and potential mismanagement.
When presented with a peripartum women who has the signs and symptoms of acute left ventricular failure the primary focus should be on firstly confirming that the left ventricle has failed, secondly initiating management in a suitable critical care facility and then on locating and confirming the likely cause.
The algorithm shown here in Figure 4 gives a suggested approach based on accurate bedside echocardiography. The majority of hospitals are not equipped with a dedicated obstetric echo service. Some hospitals now have acute echo services bases either in cardiology or intensive care. It is vital that this patient group can access this service within an hour of request, however this is provided at a local level.
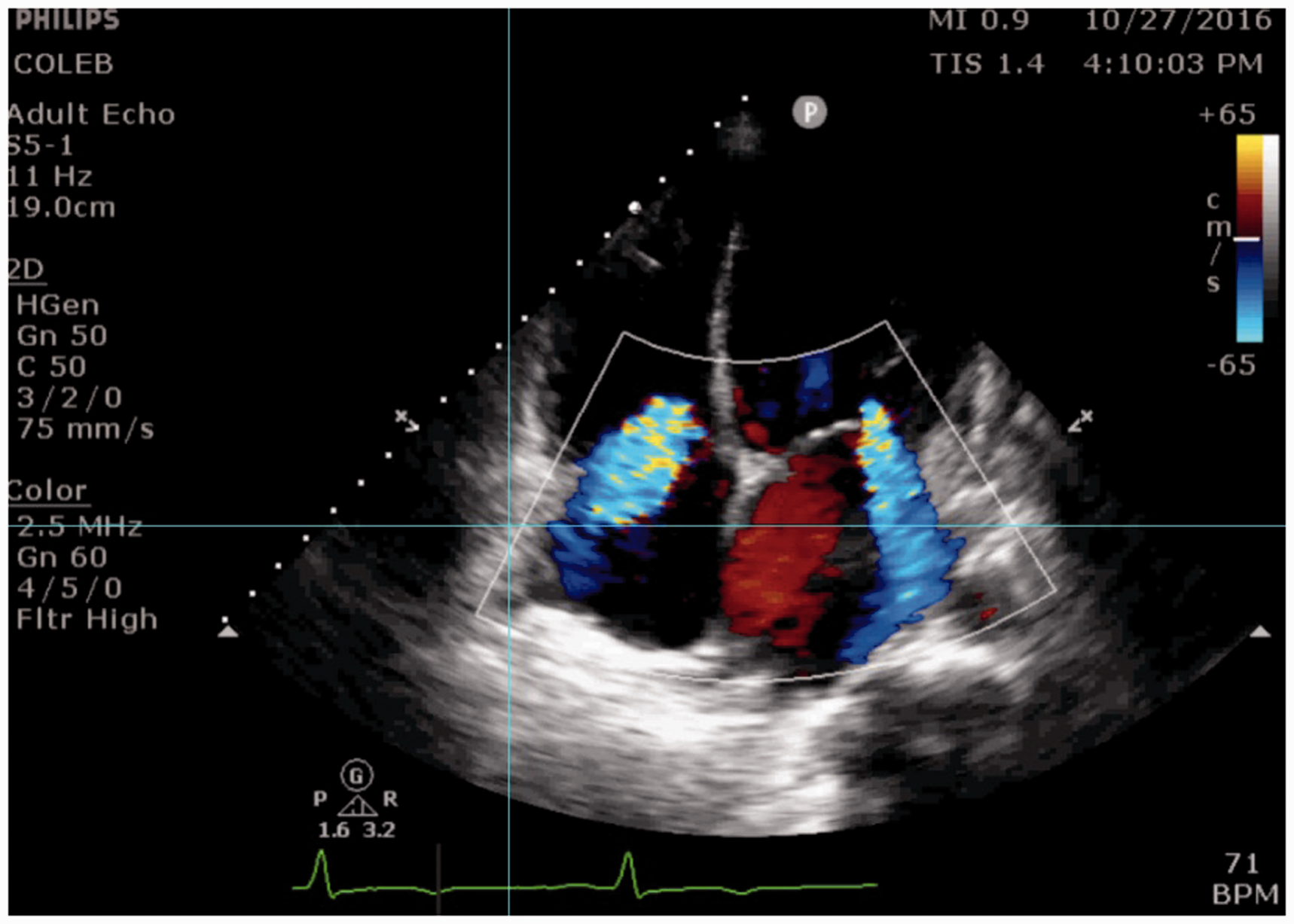
Moderate mitral and tricuspid regurgitation secondary to biventricular failure and subsequent dilatation.
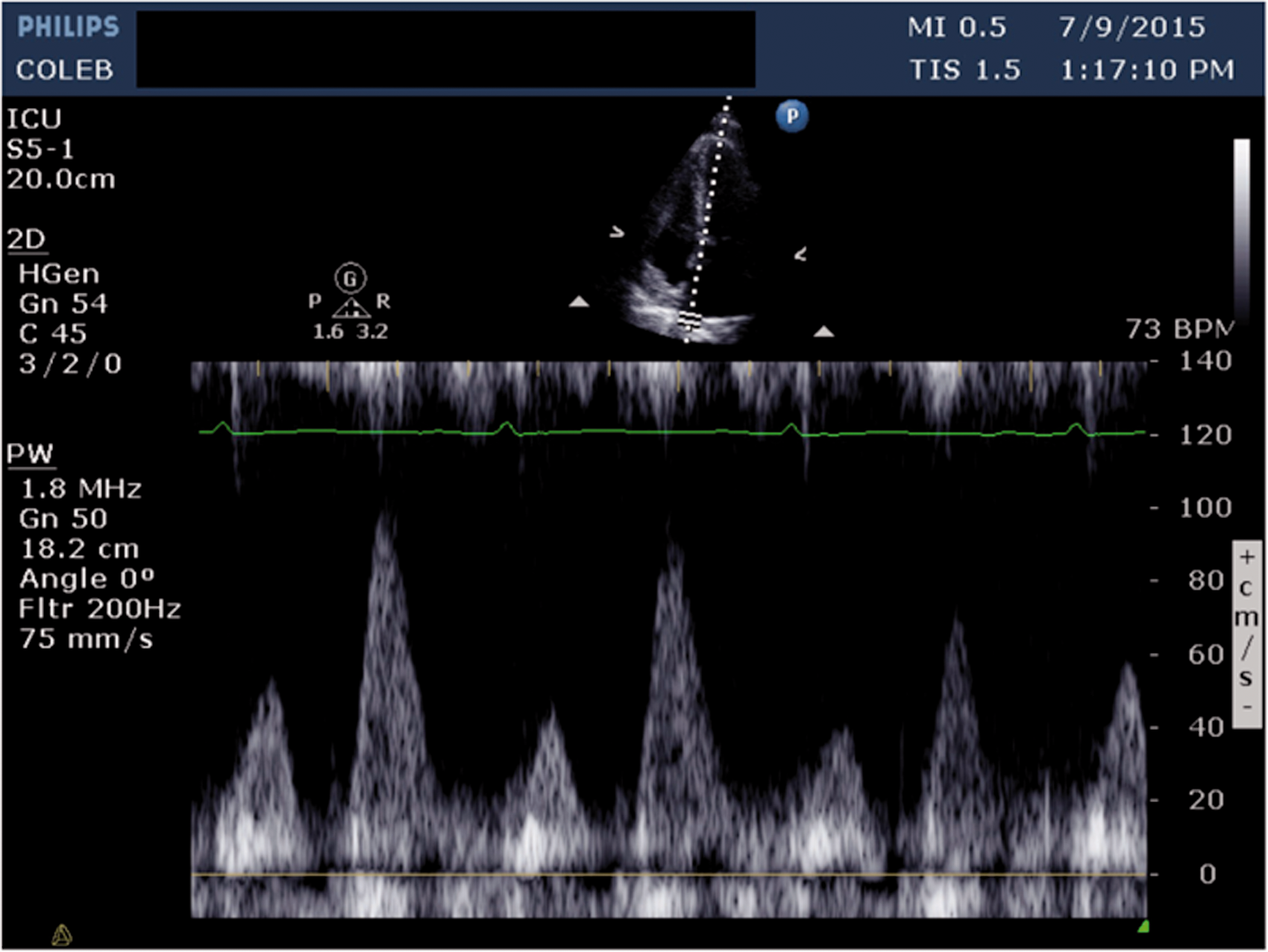
Pulsed-wave doppler demonstrating pulmonary vein systolic wave peak velocity less than the diastolic wave peak velocity (s/d ratio <1) suggestive of diastolic dysfunction grade II or III.
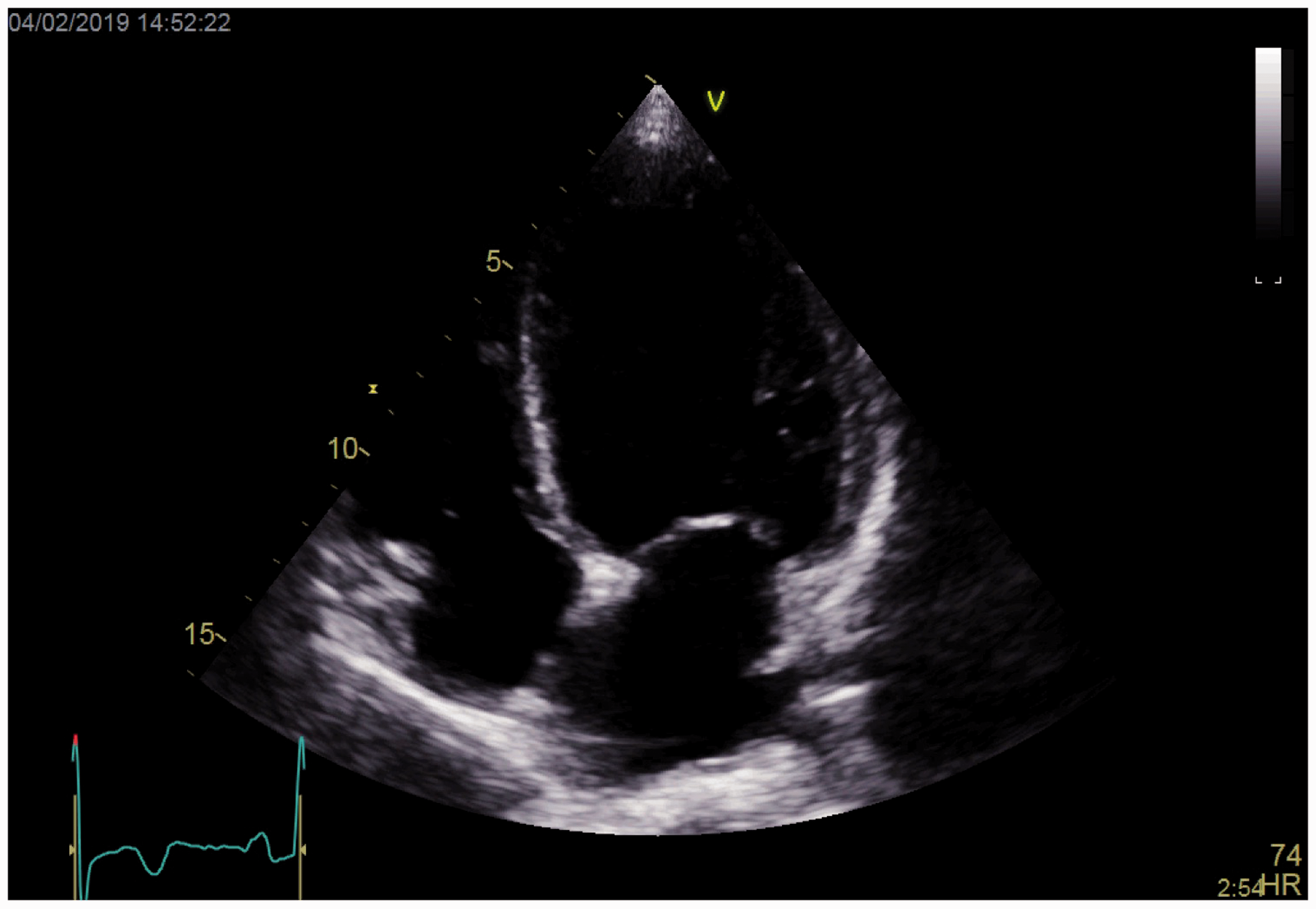
Apical-4-chamber view illustrating dilated left ventricle.
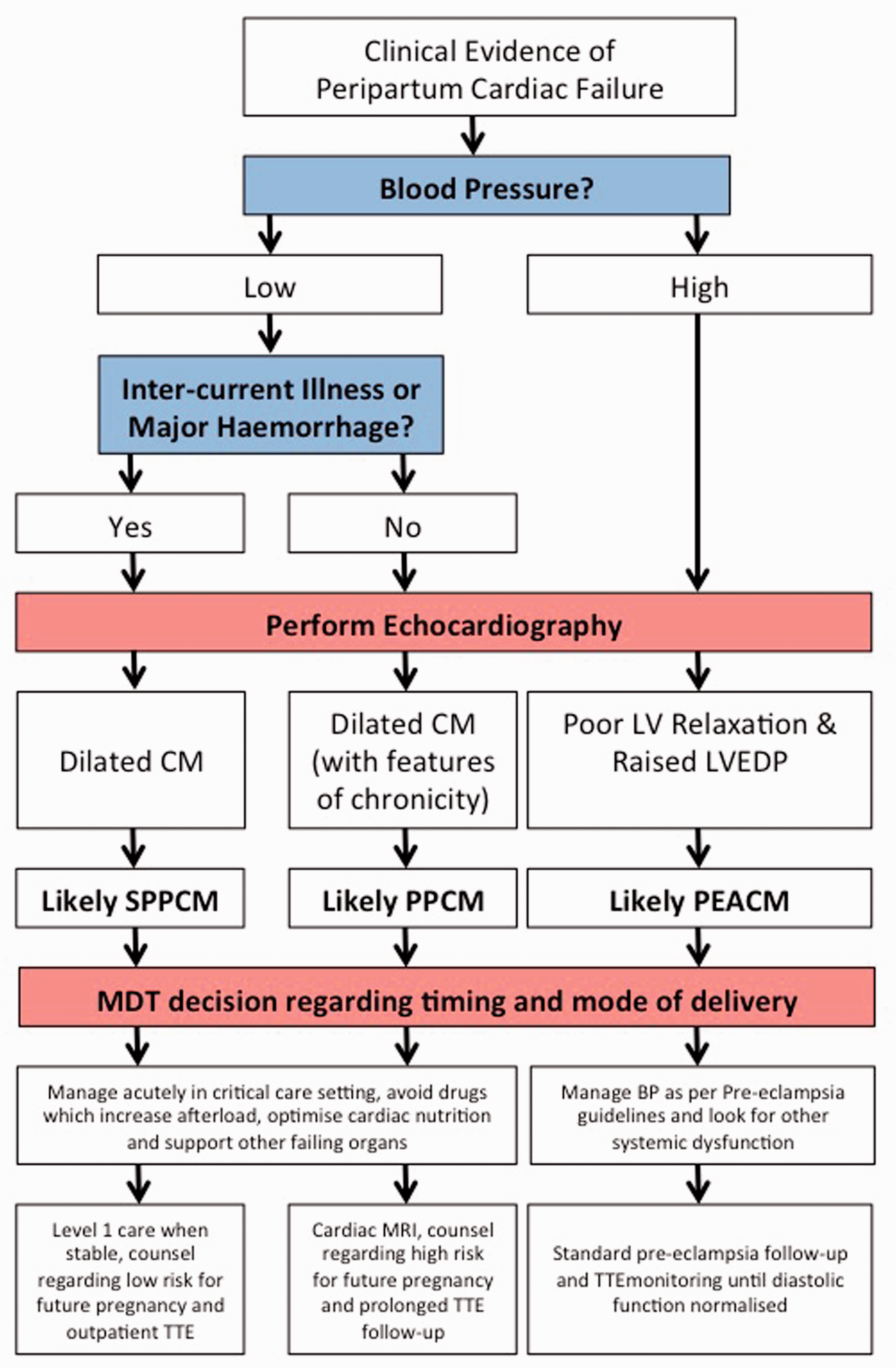
Algorithm for management of unexpected peri-partum left ventricular failure.
The key echocardiographic information can be obtained from a Level I transthoracic study or a FICE level study.32,33 The purpose of this initial assessment is to confirm left ventricular failure and rule out other causes of cardiovascular shock. In all cases of peri-partum left ventricular failure we would recommend urgent follow-up with a full Level II study orientated towards ensuring there is no unexpected background cardiac structural disease: for example an unexpected ASD, there is no co-existent valvular pathology not directly causes by the left ventricular failure itself, and that a full assessment of fluid responsiveness is undertaken.
Immediate management
General considerations
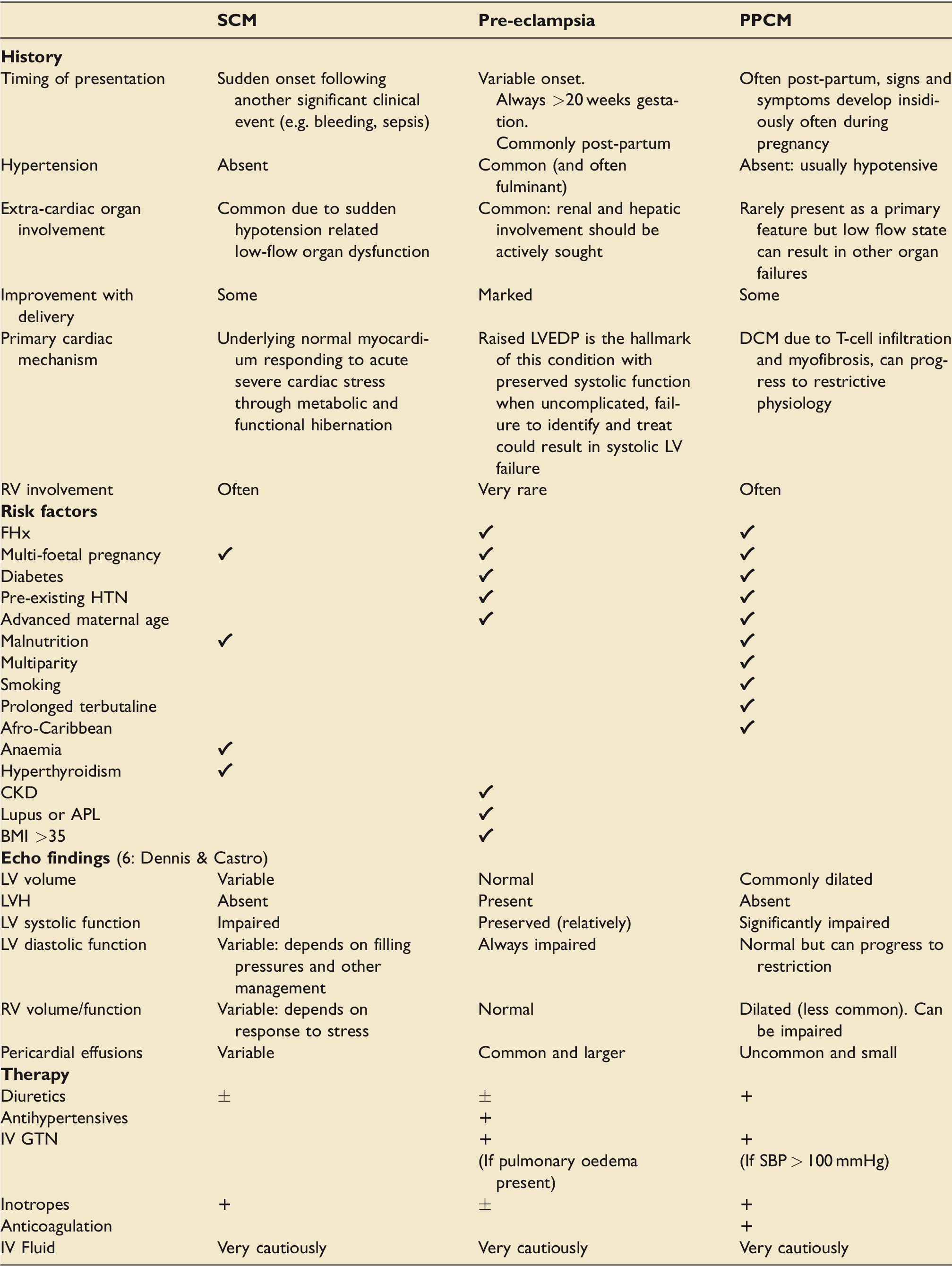
comparison of presenting features, risk factors, echocardiographical findings and therapy for PPCM, SPPCM and pre-eclampsia associated cardiac dysfunction.
Fluid balance
It is vital to remember that intravascular volume may be low in patients with left ventricular failure and as we saw in our stress cardiomyopathy clinical vignette, augmenting intravascular volume by small degrees may be crucial to the management of these patients. This should be done only after echocardiographic assessment of fluid responsiveness and in a critical care setting. Aliquots of 250mls of fluid will provide an immediately assessable response.
Echocardiography can be used to identify frank hypovolaemia by looking at the left ventricle and the IVC. Papillary muscle apposition, a left ventricular internal dimension in diastole (LVIDd) <2.2 cm/BSA and a left ventricular end diastolic area (LVEDA) <5.5 cm2/BSA are all indicative of hypovolaemia as is an IVC diameter <1.5 cm with collapse during respiration. 34 Echocardiography can also be used to predict the response to a fluid bolus by looking at the variation of both left ventricular outflow tract (LVOT) velocity time integral (VTi) and IVC diameter with the ventilatory cycle. Alternatively in a spontaneously breathing patient, LVOT VTi increment with a passive leg raise can also be used to assess fluid responsiveness.
In some cases where there is no evidence of fluid responsiveness intravenous loop diuretics may be appropriate, especially in the presence of pulmonary oedema. Their vasodilatory effect will reduce preload and their diuretic effect will reduce total body water. Judicious dosing is required to take into account the large fluid shifts around the time of delivery.
Using data generated from echocardiography, the Nagueh equation (1.24 × (E/e*) + 1.9) 35 can be used to generate an estimate of mean pulmonary capillary wedge pressure. This becomes particularly useful when considering the effect that fluid administration or removal might have on extravascular lung water in the context of predominantly diastolic heart failure, such as that seen in pre-eclampsia associated cardiomyopathy.
Vasoactive drugs
The guiding principle for the use of vasoactive drugs in the setting of a failing ventricle is that they will universally increase the workload of the ventricle and this should be the primary consideration in their use.
There will also be circumstances when a vasopressor such as Noradrenaline has to be used, for example when SCM is precipitated by sepsis or to offset the effect of sedative infusions. In these situations, repeated assessment of left ventricular function using echocardiography can be utilised to ensure that adequate cardiac output is being maintained. Maintenance of cardiac output and therefore flow as measured by the trans aortic velocity time integral is a more important but co-existent consideration to absolute measured systolic pressure.
As a general rule, reducing the afterload on the LV using vasodilatory drugs is beneficial in an effort to reduce its workload. To this end, both enteral and intravenous vasodilators such as ACE inhibitors and GTN can be used to good effect. However, this should always be done cautiously as dropping systemic blood pressure precipitously can result in a reduction in coronary perfusion pressure and thus impaired myocardial oxygen delivery.
Inotropic therapy
On occasion left ventricular function may be so severely impaired that reduction in afterload alone is not enough to compensate. When there are systemic signs of hypo-perfusion: for example, poor peripheral capillary refill, oligo-anuria, rising lactate and an echo has confirmed adequate filling with a significantly reduced cardiac output, it may be appropriate to utilize an inotrope such as Dobutamine or Enoximone. Again caution should be used and hypotension secondary to systemic vasodilation anticipated and managed cautiously. Repeated echo assessment of cardiac output and contractility can again be used to titrate the infusion rate, which should be maintained at the minimum required dose to achieve adequate cardiac output appreciating that this will come at the expense of increased demand on the failing ventricle.
Global consideration for optimum cardiac working conditions should also be given including haemoglobin level, vitamin deficiencies in particular the B complex and vitamin D, micronutrients such as selenium and electrolyte provision.
Follow on therapy and management
Once the immediate situation has been stabilized through repeated diagnostic echocardiography and provision of adequate cardiovascular support including invasive ventilation where required, longer-term therapy should be initiated. This may include ACE inhibition, rate control and other drugs aimed at managing left ventricular impairment in the medium to long term. If PPCM is thought to be the likely cause consideration can be given to steroid therapy at 24 hours from presentation. 36 This should be guided by a consultant cardiologist who can also monitor and counsel the patient following further appropriate tests in the out patient setting. Differentiation between the causes of left ventricular failure is vital at this juncture to prevent further episodes of left ventricular failure for example due to PPCM which can recur with subsequent pregnancies.
Conclusions
Echocardiography is playing an increasingly important role in the immediate assessment and management of left ventricular failure. This is especially true in the peri-partum woman, where establishing the correct therapy is both challenging and crucial due to the significant cardiovascular changes that occur around the time of delivery. To this end we believe that echocardiography should be rapidly available to guide the management of these patients by a multidisciplinary team made up of obstetricians, cardiologists, anaesthestists and intensive care physicians.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
