Abstract
Background
Leukocytosis is a rise in white blood cell (WBC) count and clinical outcomes of moderate to severe leukocytosis in trauma patients have not been described. We hypothesized that trauma patients with severe leukocytosis (SL; ≥40.0 × 109 leukocytes/L) have higher rates of in-hospital complications and mortality than those with moderate leukocytosis (ML; 25.0–39 × 109 leukocytes/L).
Methods
We performed a retrospective analysis (2010-2017) on trauma patients developing ML or SL at a single Level-I trauma center. A multivariable logistic regression analysis for risk factors were performed.
Results
From 15,807 trauma admissions, 332 (2.1%) had ML or SL. Of these, 308 (92.8%) were ML and 24 (7.2%) were SL. Patients with ML and SL reached their peak WBC count in 1 and 10 days after admission respectively (p < 0.001). SL patients suffered higher rates of in-hospital complications (p < 0.05) and mortality compared to those without ML or SL (14.5% vs. 3.3%, p < 0.001). Between ML and SL, mortality rates rose with leukocytosis severity (13.3% vs. 29.2%, p = 0.03). Among all patients with ML or SL, vasopressor use was the strongest independent risk factor for mortality (OR 12.61, p < 0.001).
Conclusion
Clinicians should be weary of the increased mortality rates and in-hospital complications in SL patients. Among patients with ML or SL, vasopressor use, rather than SL, was the strongest predictor of mortality. Patients with ML had a quicker time course to peak leukocytosis compared to SL, suggesting these two entities to be distinct in etiology and outcome, warranting future research.
Keywords
Introduction
A leukocytosis is generally defined as a leukocyte count of >11 × 109 leukocytes/L and involves a broad differential which includes malignancy, infection, severe hemorrhage, intoxication, acute hemolysis, and trauma. The release of catecholamines during periods of stress, such as severe trauma, demarginalizes white blood cells (WBCs) into the blood stream and deploys stores from the lungs, spleen, and bone marrow.1,2 This neurohumoral response leads to a reactive leukocytosis of variable severity.
In the general population, moderate leukocytosis (ML) to severe leukocytosis (SL) have been reported to have a hospital mortality rate of 32.1–61.5%.3,4 Post-injury leukocytosis has been linked with increased risk of developing multiple organ dysfunction syndrome (MODS). 5 A mean leukocytosis of 19.5 × 109 leukocytes/L have been shown to be independently associated with MODS. 6 Leukocytosis has been implicated as a predictor head injury severity, acute blood loss anemia, and hollow viscus injury in trauma patients.7–10 However, there is conflicting evidence regarding the significance of leukocytosis in stratifying injury severity and prognosis.11–13 Additionally, timing of peak leukocytosis could also hold some significance and has not been clarified in trauma patients. Schnüriger et al. found that a peak WBC count of >20 × 109 leukocytes/L within 24 hours had a 2.2-fold increased risk for a hollow viscus injury. However, the sensitivity for such an injury was only 29%, thus questioning the usefulness of leukocytosis as a sole predictor. 10 Lastly, there is no consensus regarding the WBC count cut-off to stratify the severities among leukocytosis. 14 Although a leukemoid reaction generally refers to a leukocytosis of >50 × 109 leukocytes/L, the literature also references WBC counts ranging between 25.0–50.0 × 109 leukocytes/L, creating ambiguity among these studies.4,14,15
We sought to elucidate the morbidity and mortality of varying leukocytosis severities in trauma patients. This information may help alert clinicians to prognosticate their outcomes and detect patients at risk of significant morbidity or mortality. The primary aim of this study was to examine the differences in morbidity and mortality of trauma patients with ML (25–39 × 109 leukocytes/L) and SL (≥40 × 109 leukocytes/L). We hypothesized that trauma patients with SL have higher rates of in-hospital complications and mortality compared to those with ML.
Methods
We performed a retrospective review of adult trauma patients ≥18-years-old at a single Level-I trauma center admitted from April 1st, 2010 to December 31st, 2017. The study was reviewed and approved by the Institutional Review Board (IRB). All patient data were collected in compliance with Health Insurance Portability and Accountability Act (HIPAA). Patients with a peak WBC ≥25 × 109 leukocytes/L, at any point during the index hospitalization were included in our study. Both male and female patients were included in our study. Pediatric patients as defined by ≤17-years-old and pregnant patients were excluded from the study due to their differences in physiological states. All eligible subjects underwent chart review and relevant data was collected into a secure REDCap (Research Electronic Data Capture) database. 16 Demographic information of interest included age, gender, and medical comorbidities such as congestive heart failure, end-stage renal disease, coronary artery disease, cirrhosis, atrial fibrillation, diabetes, hypertension, cerebrovascular accident, hyperlipidemia, obesity, non-hematologic malignancy, dementia, and chronic obstructive pulmonary disease (COPD) among others. Admission mechanism of injury, injury severity score (ISS), Glasgow Coma Scale (GCS), and vital signs were also collected. Laboratory studies on admission such as WBC, hemoglobin, platelets, and the number of days required until the WBC reached its peak were recorded.
A peak WBC count cutoff of 40 × 109 leukocytes/L was chosen to distinguish between ML and SL based off of previous studies that most accurately represent the trauma patient population.4,14,15 The primary outcome was mortality, comparing patients with ML and SL during index hospitalization. Mortality rate of trauma patients without ML or SL were obtained by querying our institution’s trauma registry. Additional outcomes analyzed included in-hospital complications, vasopressor requirements, and time to peak WBC count. In-hospital complications of interest included respiratory failure, pneumonia, pulmonary embolism, acute respiratory distress syndrome (ARDS), myocardial infarction, cerebrovascular accident, deep vein thrombosis, urinary tract infection, acute kidney injury (AKI), bacteremia, sepsis, rhabdomyolysis, and surgical site infection as defined by the trauma quality improvement program. 17
Variables were coded as present or absent. Outcomes of patients with ML were compared to those with SL using a Mann-Whitney-U and chi-square analysis. We used two-sided p-values and a statistical significance level of p < 0.05. Multivariable logistic regression model controlling for SL, age, ISS, and vasopressors required at any point during hospitalization was conducted to identify which factors contribute to the risk of mortality in patients with ML or SL. Variables were chosen based on known predictors for mortality in trauma patients.3,18,19 Statistical analyses were performed through IBM SPSS Statistics for Windows (Version 24, IBM Corp., Armonk, NY).
Results
Demographics of patients with ML and SL
Of the 15,807 trauma admissions during the study period, 332 (2.1%) had a ML or SL during their hospital course. Among these patients, 308 (92.8%) had ML while 24 (7.2%) had SL. There were no differences between ML and SL patients in median age (31.0 vs. 33.5 years, p = 0.76), ISS (24 vs. 29, p = 0.17), and GCS (15 vs. 15, p = 0.10). Both groups had similar vital signs and comorbidities (p > 0.05). There were also no differences in admission WBC (24.6 × 109 leukocytes/L vs. 18.3 × 109 leukocytes/L, p = 0.30) and differential counts which included neutrophils (80% vs. 71.8%, p = 0.69), lymphocytes (8.1% vs. 20.4%, p = 0.64), monocytes (4.9% vs. 6.1%, p = 0.89), eosinophils (0.2% vs. 0.3%, p = 0.93), basophils (0.0% vs 0.0%, p = 0.71) between ML and SL. Hemoglobin (13.3 g/dL vs. 13.2 g/dL, p = 0.66) and platelet counts (262 × 109 platelets/L vs. 267 × 109 platelets/L, p = 0.87) did not differ between ML and SL. Patients with SL took longer to reach their peak WBC level compared to those in the ML cohort (10 days vs. 1 day, p < 0.001). WBC peaks were different between ML and SL (28.1 × 109 leukocytes/L vs. 43.6 × 109 leukocytes/L, p < 0.001), but their differential counts did not (Table 1). ML and SL had similar proportions of blunt (83.8% vs. 87.5%, p = 0.76) and penetrating (16.2% vs 12.5%, p = 0.76) mechanisms of injury (Table 2).
Demographics and WBC characteristics of adult trauma patients with ML and SL.
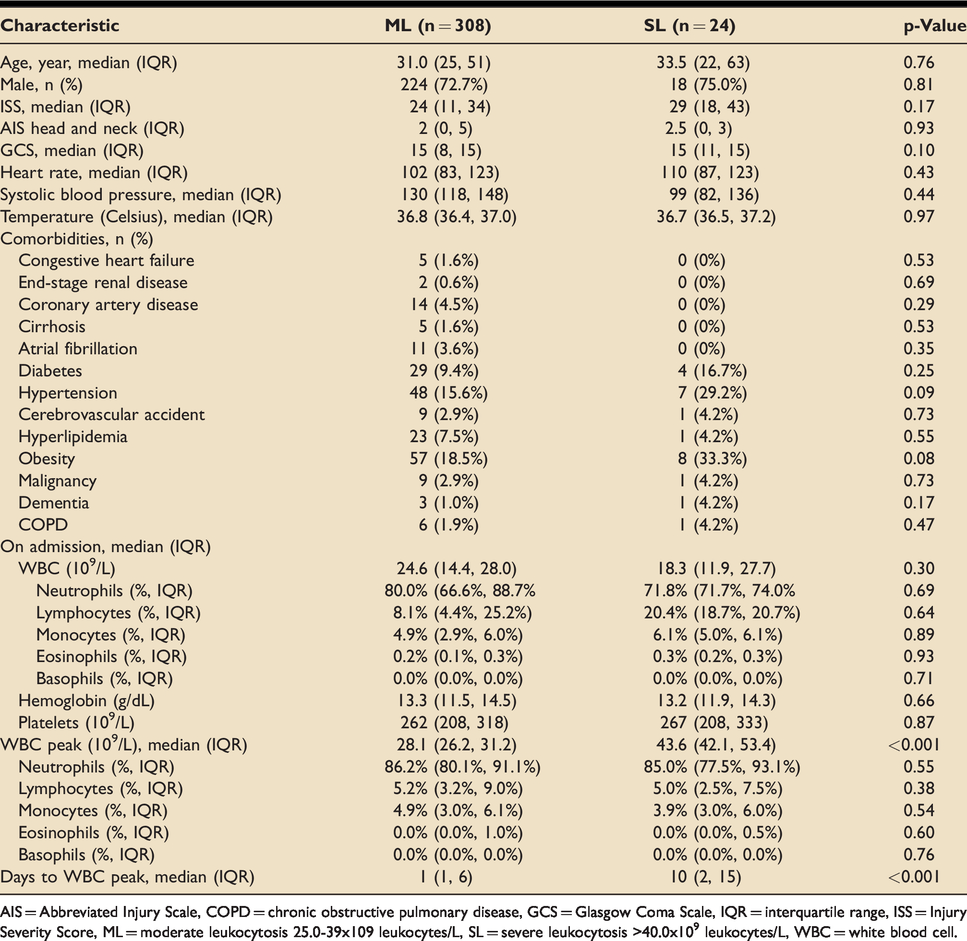
AIS = Abbreviated Injury Scale, COPD = chronic obstructive pulmonary disease, GCS = Glasgow Coma Scale, IQR = interquartile range, ISS = Injury Severity Score, ML = moderate leukocytosis 25.0-39x109 leukocytes/L, SL = severe leukocytosis>40.0x109 leukocytes/L, WBC = white blood cell.
Mechanism of injury in adult trauma patients with ML and SL.
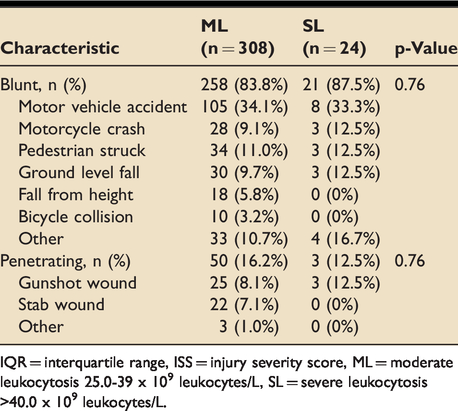
IQR = interquartile range, ISS = injury severity score, ML = moderate leukocytosis 25.0-39 x 109 leukocytes/L, SL = severe leukocytosis>40.0 x 109 leukocytes/L.
Outcomes of patients with ML and SL
ML and SL cohorts had similar rates of operative intervention (68.8% vs. 79.2%, p = 0.75), including splenectomy. There were no differences in ventilator days (1 vs. 6, p = 0.31), ICU LOS (4 vs. 7, p = 0.12), and hospital LOS (11 vs. 21, p = 0.13). The SL cohort had higher rates of therapeutic antibiotic (58.3% vs. 32.9%, p = 0.01) and vasopressor use (62.5% vs. 25.0%, p < 0.001), with more patients initiating vasopressors before peak WBC count (78.6% vs. 50.0%, p = 0.04). For those who required vasopressors, there was no difference in hospital days until WBC peak in ML and SL (2 days vs. 4 days, p = 0.91) (Table 3). Moreover, patients with SL had higher rates of ARDS (12.5% vs. 2.9%, p = 0.02), AKI (29.2% vs. 8.8%, p = 0.002), sepsis (33.3% vs. 10.1%, p = 0.001), and rhabdomyolysis (12.5% vs. 3.3%, p = 0.03). The mortality rate in patients without ML or SL was significantly lower than those with any ML or SL (3.3% vs. 14.5% p < 0.001). Among these, SL patients had a higher mortality rate than ML (29.2% vs. 13.3%, p = 0.03) (Table 4).
Clinical outcomes in adult trauma patients with ML and SL.
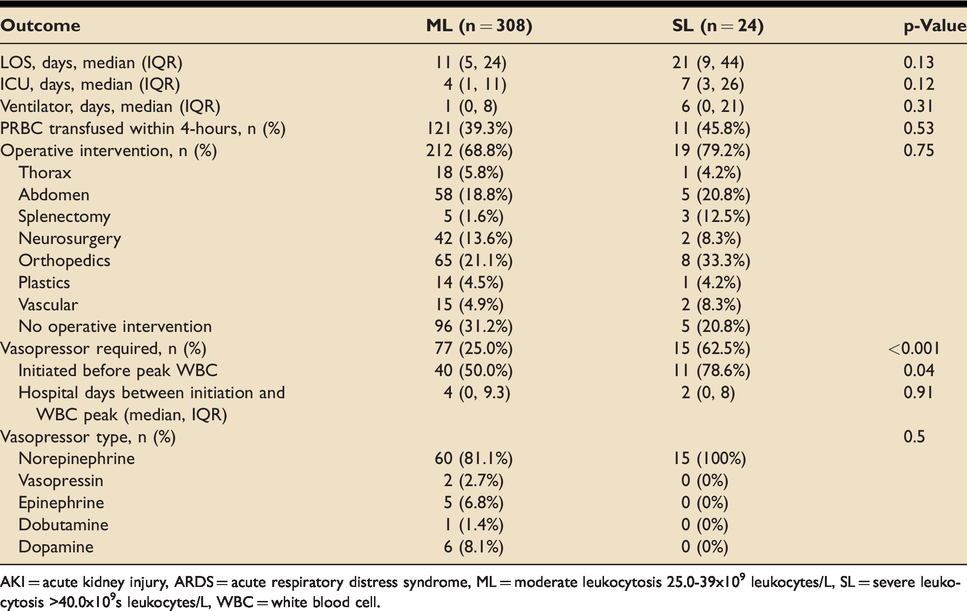
AKI = acute kidney injury, ARDS = acute respiratory distress syndrome, ML = moderate leukocytosis 25.0-39x109 leukocytes/L, SL = severe leukocytosis>40.0x109s leukocytes/L, WBC = white blood cell.
Complications and mortality in adult trauma patients with ML and SL.
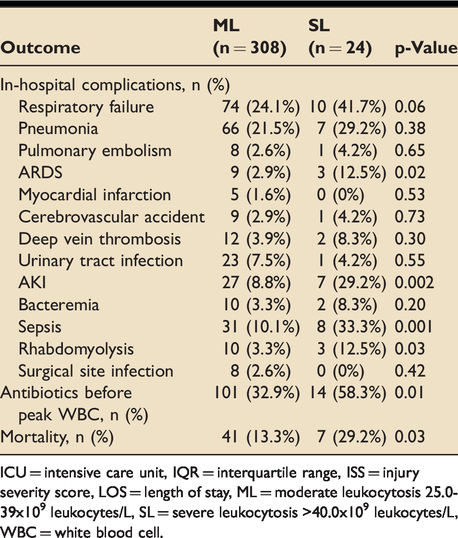
ICU = intensive care unit, IQR = interquartile range, ISS = injury severity score, LOS = length of stay, ML = moderate leukocytosis 25.0-39x109 leukocytes/L, SL = severe leukocytosis>40.0x109 leukocytes/L, WBC = white blood cell.
Multivariable logistic regression analysis for risk of mortality in all patients with ML or SL
In our multivariable logistic regression model, age (OR 1.03, CI 1.01–1.04, p = 0.001) and vasopressor requirement at any point during the hospitalization (OR 12.61, CI 5.80–27.40, p < 0.001) were independent risk factors for mortality in all patients with ML or SL. SL (OR 1.35, CI 0.45–4.01, p = 0.59) and ISS (OR 1.02, CI 1.00–1.05, p = 0.11) did not increase the risk of mortality (Table 5).
Logistic regression model predicting risk of mortality in all patients with ≥25.0–39 × 109 leukocytes/L.
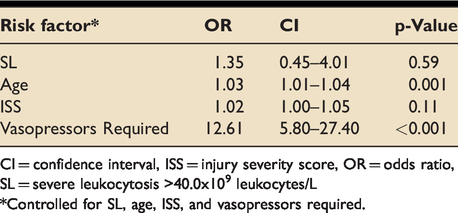
CI = confidence interval, ISS = injury severity score, OR = odds ratio, SL = severe leukocytosis>40.0x109 leukocytes/L*Controlled for SL, age, ISS, and vasopressors required.
Discussion
To our knowledge, this is the first study to investigate the associated morbidity and mortality of trauma patients with varying severities of leukocytosis. Overall, trauma patients with ML or SL had increased mortality compared to those without. Patients with SL had higher rates of mortality and in-hospital complications including ARDS, AKI, sepsis, and rhabdomyolysis, compared to those with ML. The timing of ML and SL were quite different, with ML predominantly appearing as an early post-injury phenomenon while SL took over a week to develop. Finally, we found that vasopressor use was the strongest independent risk factor for mortality in all patients with ML or SL, whereas SL alone was not a risk factor within this population.
Extreme leukocytosis has been correlated with poor clinical outcomes. Furthermore, studies have shown that WBC counts may prognosticate the severity of blunt traumatic injury.11,13 However, prior studies have only focused on the presence of milder leukocytosis and thus, outcomes regarding trauma patients with ML or SL (≥25 × 109 leukocytes/L) were previously unknown. Our study builds on existing literature by further stratifying extreme leukocytosis into ML and SL. The rate of mortality of ML and SL increase by nearly 4- and 10-fold respectively when compared to patients without ML or SL. Furthermore, patients with SL exhibited a 2.5-fold increased rate of mortality compared to ML while having no differences in mechanism of injury, comorbidities, admission vital signs, age, and ISS. While we are the first to report these findings in trauma patients with ML or SL, studies involving lower levels of leukocytosis have also exhibited similarly high mortality rates. Lawrence et al. observed that patients with ≥30 × 109 leukocytes/L in a mixed emergency department population without hematological malignancy had a 38.1% rate of mortality. 4 Among ICU patients with a leukocytosis of ≥25 × 109 leukocytes/L the mortality was 37.5%. 20 Clinicians should be wary of this dramatic increase in mortality rate among patients with ML and SL in trauma patients and vigilantly search for an opportunity to treat.
The time to peak WBC count may point to etiological differences between ML or SL, thus describing ML and SL as two different phenomena. Although our cohorts had similar demographics, the timing of peak leukocytosis development and hospital course for each group were strikingly different. WBC counts of those with ML peaked one day after admission, while those with SL peaked on the 10th day. The quicker onset of ML suggests a more acute process, likely due to an inflammatory response related to the patient’s traumatic injuries. Expression of adhesion molecules located on the surface membrane of these leukocytes are upregulated within 20 hours of tissue injury. This stimulates leukocytes to interact with endothelial receptors and lead them towards sites of injury. 21 However, when leukocytes persist and are not appropriately removed, such as during a ML or SL, dying neutrophils can release toxic cargo that can further exacerbate inflammation, tissue destruction, and autoimmunity. 22 Compared to ML, patients in our SL cohort suffered from higher rates of in-hospital complications that included ARDS, AKI, sepsis, and rhabdomyolysis. Given the delayed occurrence of peak WBC count in SL patients, we postulated that these complications may contribute to the SL etiology and thus, these patients may be more similar to the general non-trauma inpatient population who develop ML or SL. This was supported by the SL mortality rate we observed, which paralleled those described in previous non-trauma patients.23–25 In these patients, SL may be the product of infection, hemorrhage, or a pre-morbid state in addition to the systemic hyper-inflammatory response. Infection is the most common cause of SL and does align with our higher sepsis and antibiotic usage rates in this cohort. This is further supported by the fact that the WBC peak did not occur until much later in SL compared to ML patients. Interestingly, LOS and ICU days were not significantly different between the cohorts. Although there were higher rates and risk of mortality, there were no differences in comorbidities nor age in our cohort. Further investigative efforts to elucidate the causes of ML or SL onset within trauma patients are warranted. Contrary to our hypothesis, SL was not an independent risk factor for mortality according to our multivariable analysis. Instead, it was vasopressor use that contributed most significantly to this risk.
Vasopressors are a staple in managing hemodynamically unstable patients that were already volume resuscitated. The catecholamine surge during the exaggerated inflammatory response in severely ill patients may lead to leukocytosis and can be further exacerbated when vasopressors are initiated.26–29 Patients with SL had more than a 2-fold higher rate of vasopressor use compared to ML patients, with most being administered before peak WBC. Vasopressor use was the strongest independent risk factor for mortality in all patients with ML or SL – increasing the mortality risk by over 12-fold. Comparatively, previous research has shown vasopressors to increase the risk of mortality by only 2–5-fold in trauma patients.18,30–32 The more pronounced increased mortality risk from vasopressor use may be due to its interaction with concomitant traumatic injury and leukocytosis. Especially during prolonged leukocytosis, activated leukocytes may clump together with platelets in damaged vessel walls, restricting perfusion to vital organs.33,34 Meanwhile, if vasopressors were required to hemodynamically stabilize trauma patients, blood vessels would constrict to maintain intravascular volume. The combination of a platelet-leukocyte plug in the setting of vasoconstriction may further lead to tissue ischemia – especially worrisome in trauma patients with concomitant solid organ injuries. 35 Additionally, leukocytes such as neutrophils may release reactive oxygen species (ROS), an essential part of its protective ability within the immune system, but may also lead to organ failure and worsening of shock if neutrophils aren’t appropriately removed. 22 ROS interacts with proteins, membrane lipids, and nucleic acids to ultimately damage tissue and consequently exacerbate the release of pro-inflammatory agents. 33 Thus, the shock experienced by those with a ML or SL in combination with vasopressor management may be more devastating. Future research regarding the inflammatory pathways in trauma patients who experience ML or SL are warranted with hopes of identifying the specific deleterious effects of these exaggerated inflammatory response seen in this population.
There were several limitations to our study including those inherent to our retrospective methodology. Medical records may lack information to sufficiently quantify the number of patients with known inducers of leukocytosis (e.g. glucocorticoid use). 36 Conditions that have been previously known to be associated with elevated leukocytosis such as head injury, sickle cell disease, or hematologic malignancy, were included in our study to more accurately represent the demographics of trauma patients.14,37 Baseline WBC counts prior to trauma were not available as well. Our study only focused on in-hospital outcomes, as the follow-up rate in our system and the nature of trauma patients in general are not reliable enough to accurately depict 30-day or longer outcomes. Lastly, given that ML and SL only occurred in 2.1% of trauma patients, future multicenter initiatives are needed to capture a larger sample size of trauma patients to investigate this fairly rare yet lethal phenomenon.
Conclusion
This study is the first to evaluate outcomes of trauma patients with ML or SL. Severe leukocytosis is a rare occurrence as the overall incidence of any ML or SL was <2.5%. Patients with SL had a 2.5-fold increased rate of mortality compared to those with ML. In addition, SL patients took more than a week to reach their peak WBC count and had higher rates of in-hospital complications such as ARDS, AKI, sepsis, and rhabdomyolysis. Among all ML or SL patients, vasopressor use had a 12-fold increased risk of mortality – more so than what was previously reported in non-trauma patients. SL was not an independent predictor of mortality amongst patients. Clinicians caring for trauma patients with ML and SL should understand the increased rate of mortality and anticipate the increasingly complicated hospital course of SL patients. Future prospective efforts are needed to further investigate how these inflammatory pathways may contribute to a ML or SL and their poor outcomes.
Footnotes
Acknowledgements
Abstract has been presented at the 2019 Annual Meeting of the Southwestern Surgical Congress on April 14–17 in Huntington Beach, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
