Abstract
Albumin plays a key role in the critically ill patient acting as a prognostic marker and as a therapy in the form of human albumin solutions. However, the use of human albumin solution has varied over time with notable differences between health systems. Whilst its use is widely accepted for some clinical indications, its role has not always been clear in the critically ill and has been found to be harmful in some cohorts. Numerous studies have showed conflicting results and critical care clinicians have not always been guided by robust evidence. Nonetheless, at present the use of human albumin solution appears to be increasing again in the United Kingdom. Below, we discuss the latest evidence base for its use in critically ill patients.
Introduction
A stable solution of human albumin was first developed in the 1940s yet, even today, few therapeutic interventions polarise critical care physicians more.
1
Interest in albumin solution as a plasma expander has been great, and by the turn of the century was used in greater volume than any other biopharmaceutical solution available on the market.
2
However, the first prospective randomised controlled trial was not reported until 1975,
3
and after numerous small studies with conflicting results,4–6 the publication of a meta-analysis by the Cochrane Collaboration, concluded that albumin use may increase mortality in the critically ill.
4
Despite these results, and increasing competition from the synthetic colloid market,
7
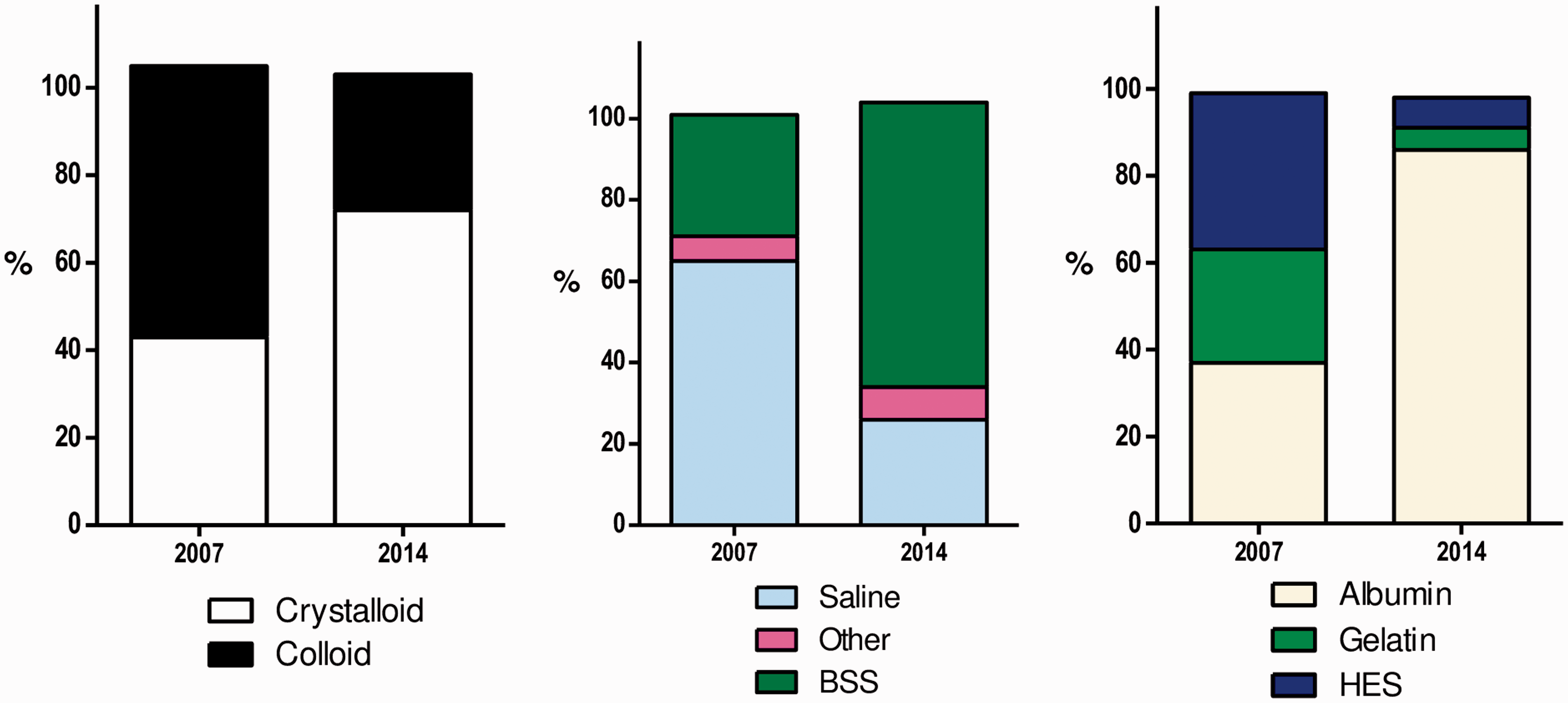
human albumin solution (HAS) remains in use in the critical care environment. Colloid use has diminished overall but HAS use has proportionally increased
8
(Figure 1).
Proportion of all fluid resuscitation episodes given in 2007 and 2014 in 84 ICUs (courtesy of Hammond et al.
8
).
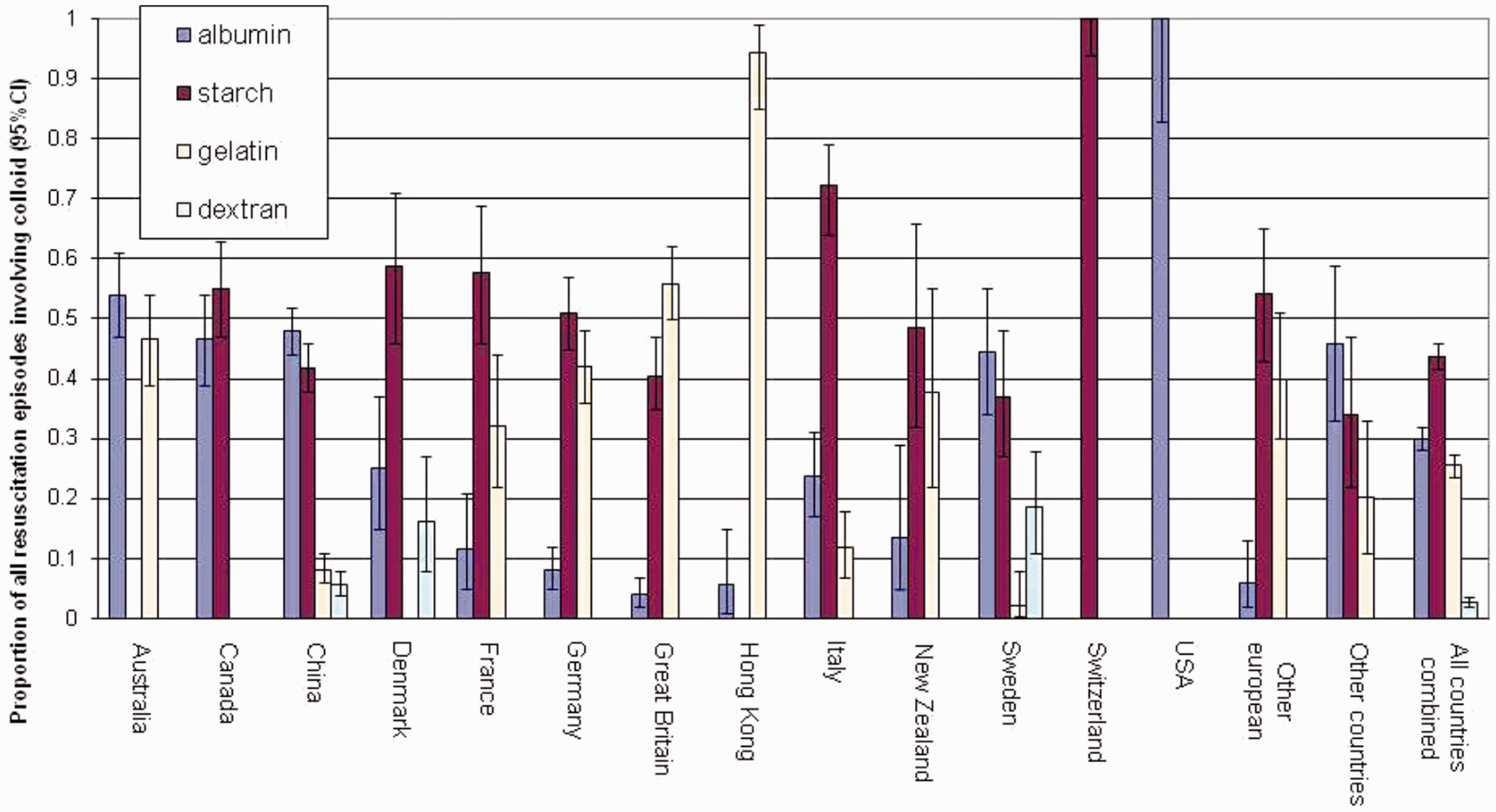
The use of albumin varies greatly between geographical locations and health systems; one trial reported no use in Switzerland, whilst in the United States it accounted for 100% of colloid used
9
(Figure 2). Despite a reliable safety profile,2,10 human albumin solution’s use is still minimal in the United Kingdom compared to other areas of the world.8,9 Acquisition cost is potentially prohibitive; a 500 mL bottle of 4.5% solution costing as much as £55.02 (Zenalb 4.5% solution for infusion 500 mL bottles (Bio Products Laboratory Ltd) NHS indicative price),
11
compared to crystalloid solutions that can be purchased for a fraction of the cost.
12
Coupled with a lack of proven superiority in the general intensive care unit (ICU) population,
13
some may find it difficult to justify the expense. And yet, international guidelines, developed and endorsed, by international critical care societies, suggest that albumin solution could be used after administration of ‘substantial amounts’ of crystalloid, when resuscitating patients.
14
The absence of specific recommendations on dose and timing likely reflects heterogeneity in the published literature.
Type of colloid used as a percentage of all colloid episodes by country (courtesy of Finfer et al.
9
).
Hypoalbuminaemia and the use of human albumin solution in acutely unwell patients has been investigated for decades. Here, we aim to briefly provide an overview of the available clinical evidence in specific conditions seen in ICU.
Hypoalbuminaemia
Hypoalbuminaemia is associated with worse outcomes across a myriad of patient cohorts, including trauma, 15 major surgery, 16 emergency medical admissions 17 and the ICU population. 18
Hypoalbuminaemia, defined as a serum albumin < 35 g/L,19,20 is common in the ICU, with a reported incidence of up to 76% in critically unwell children, 21 and 82% in certain adult groups. 22 Mechanisms for this phenomenon are complex and multifactorial, but the predominant syndrome encountered in the critical care population is one of increased capillary leak. 23 Increased renal losses (e.g. nephrotic syndrome), bleeding, protein losing enteropathy, increased catabolism, reduced hepatic synthesis and malnutrition can all potentially contribute, but are much less common. 24
A 2003 meta-analysis of almost 100 studies, including over 290,000 patients, demonstrated that as the severity of hypoalbuminaemia increased, so too did mortality, morbidity, length of stay and cost. For each 10 g/L reduction in serum albumin concentration a 137% increase in mortality was observed. The role of albumin as a therapy is less clear; recipients of human albumin solution had a reduced pooled odds ratio of morbidity, but this failed to reach statistical significance.
Dubois et al. tested the theory that supplementing serum albumin with hyperoncotic solutions daily, in patients who were hypoalbuminaemic, would result in better outcomes. In this single centre, open label, pilot study, 100 patients with serum albumin concentrations of <31 g/dL, were randomised to receive 300 mL 20% HAS on day 1, then 200 mL 20% HAS on each subsequent day, or to receive no albumin therapy. They found that mean delta SOFA score was greater in the albumin group than in the control (3.1 vs. 1.4; p = 0.03), during days 1 to 7, concluding that supplementation of albumin improves organ dysfunction, and results in less positive fluid balance, but were unable to find any difference in survival, or sustained improvement in organ dysfunction at days 14, 21 and 28. 25 However, it is worth noting that there was no statistically significant difference in absolute SOFA scores between the two groups. A subsequent large, randomised controlled trial, failed to replicate these promising results 26 – discussed further below.
Use as a resuscitation fluid
Intravenous fluid therapy is one of the most common interventions in critical care, 27 and human albumin solutions may have a role in certain syndromes and cohorts.
In 2004, the SAFE trial, of 6997 critical care patients in 16 academic centres in Australia and New Zealand, compared 4% HAS to 0.9% saline as a resuscitation fluid in a heterogeneous group of intensive care patients. Exploring all-cause mortality at 28 days, no statistically significant difference was found between the groups. Similar outcomes were observed in terms of length of stay, duration of mechanical ventilation and incidence of renal replacement therapy. Sub-group analysis of patients with trauma revealed a trend towards increased mortality (13.6% vs. 10.0% (RR 1.36; 0.99 to 1.86; p = 0.06). 13
This finding led Myburgh et al. to perform a post hoc follow-up study of 460 patients from the original SAFE trial, who had sustained a traumatic brain injury. A significantly higher mortality (33.2% vs. 20.4%), and fewer favourable neurological outcomes (47.3% vs. 60.6%) were observed at 24 months post randomisation, in the albumin group, compared to those that received saline. 28 An additional post-hoc analysis of patients who received intracranial pressure monitoring (ICP), found that those in the albumin group received more interventions to treat raised ICP, and in the group where ICP monitoring was discontinued in the first week after randomisation, there were significantly more deaths in the albumin group compared to saline (34.4% vs. 17.4). 29 The authors hypothesised this could be the result of harm associated with interventions to treat ICP, or albumin-associated coagulopathy leading to secondary haemorrhage. It is worth emphasising that these are post hoc analyses, but in the absence of large randomised controlled trials, it seems prudent to avoid the use of albumin in the brain injured patient.
The observation of a less positive fluid balance when using hyperoncotic human albumin solutions sparked interest in the critical care community and led to the concept of ‘small volume resuscitation.’ 30 This concept utilises the greater oncotic properties of hyperoncotic solutions, to maximise the proportional volume of fluid administered to remain in the intravascular compartment, and also to draw interstitial fluid intravascularly. This combined effect allows the achievement of the same haemodynamic endpoints for significantly smaller volumes of fluid administered. When using 20% HAS, patients require one quarter of the volume compared with balanced crystalloid. 31 Research has repeatedly demonstrated an association with net positive fluid balance and adverse outcomes, in both septic,25,32 and non-septic,33,34 populations, and thus the concept of ‘small volume resuscitation’ is an attractive one.
In 2018, Martensson et al. published The SWIPE trial, 35 comparing patients resuscitated with 20% albumin versus 4–5% albumin solutions, and found that at 48 h the volume of fluid administered was lower (median difference −600 mL (−800 to −400; p = <0.001)), and a lower cumulative fluid balance (mean difference −576 mL (−1033 to −119; p = 0.01) was observed in the 20% albumin group compared to the group resuscitated with 4–5% albumin. No difference in adverse events were found and, thus, the authors concluded that the findings support further exploration of ‘small volume resuscitation’ in larger randomised trials. 35 Importantly, this theory has not yet been tested against balanced crystalloid solutions in a similar manner.
In the general ICU population, HAS is a safe intravenous fluid, but without proven superiority over 0.9% saline. However, it should be avoided in those patients who have suffered a traumatic brain injury. Its role in “small volume resuscitation” appears promising but requires further large-scale research.
Use in sepsis and septic shock
Intravenous fluid therapy has been researched extensively in the treatment of sepsis and septic shock. 27 Albumin has received particular attention as a therapy in sepsis, both as a resuscitation fluid, and in the prevention of hypoalbuminaemia.
Both the Society of Critical Care Medicine (USA), and the European Society of Intensive Care Medicine, endorse the Surviving Sepsis Campaign recommendation of albumin as the colloid of choice, when resuscitating patients with sepsis and septic shock.14,36 Again, guidance on when to move from crystalloid to colloid, during resuscitation, is left to individual clinician discretion.
The SAFE Study Investigators performed an a priori, post hoc analysis of 1218 patients with severe sepsis from their original dataset. Following multivariate analysis, and after adjustment for baseline characteristics, albumin therapy was independently associated with a decreased odds ratio for death. 37 Curiously, important secondary outcome measures, including length of ICU and hospital stay, duration of mechanical ventilation, and incidence of renal replacement therapy, did not differ between the groups.
The results of the EARSS Study were presented by J Charpentier, at LIVES 2011, in Berlin, Germany. This multi-centre, open-label, randomised trial, in 29 French ICUs, looked at whether infusion of 100 mL of 20% albumin, every 8 h, for three days, would result in superior outcomes, compared to infusing 100 mL 0.9% saline, at the same intervals. Mortality did not differ significantly between the two groups (24.1% vs. 26.3%), but catecholamine requirement was significantly reduced in the albumin group. 38 It should be noted, however, that this dataset is, as yet, unpublished, and thus has not undergone the scrutiny of peer review.
Following Dubois et al.’s initial results, using 20% albumin to treat hypoalbuminemia, Caironi and colleagues performed the ALBIOS trial, a prospective, multi-centre, randomised, open-label trial, of over 1800 critically unwell adults with severe sepsis, in 100 Italian ICUs. Patients were randomised to receive either 20% albumin and crystalloid, to maintain serum albumin levels of 30 g/L or greater, or crystalloid solutions alone, for the first 28 days of their ICU stay, or until ICU discharge. Mortality at 28 days did not differ between the two groups. During the first seven days the albumin group had a significantly lower cumulative net fluid balance (median 347 mL vs. median 1220 mL; p = 0.004), and quicker resolution of shock (median days to suspension of vasopressors 3 vs. 4; p = 0.007). 26
Subsequent meta-analyses have yielded mixed results. In 2014, Patel et al. published a meta-analysis of 16 randomised trials exploring the impact of albumin on mortality in critically ill adults, and found that compared to control fluid groups, there was little difference in outcomes (RR 0.94; 95% CI 0.87–1.01; p = 0.11). 39 It is worth noting here that of the 16 trials included, all but three had fewer than 60 patients. One trial included in the meta-analysis, performed by Joachim Boldt, has since been retracted by Anesthesia & Analgesia, after Justus-Liebig Universität Giessen reported evidence of data manipulation. 40
In the same year, Wiedermann and Joannidis submitted a brief communication to the New England Journal of Medicine in response to the ALBIOS trial.
41
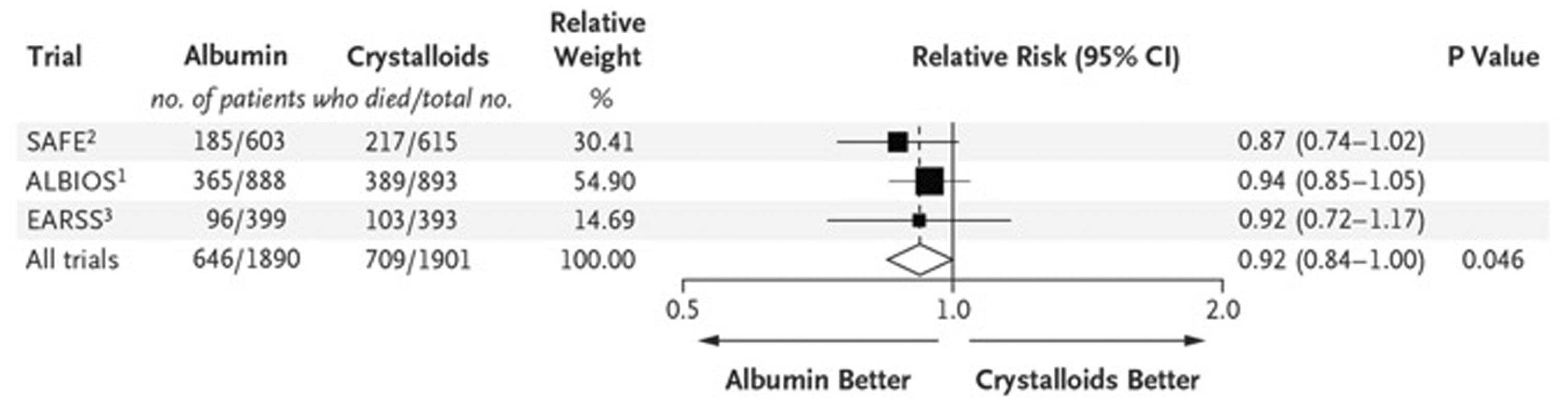
They presented their meta-analysis, including only large scale, randomised trials of albumin therapy. They concluded that, although not reaching statistical significance in the individual SAFE, ALBIOS or EARRS trials, the pooled relative risk when small trials were excluded, indicated a significant reduction in mortality with albumin use. This analysis excluded all works performed by Boldt et al., and all other trials with fewer than 60 patients included in the meta-analysis above. It is critical to note, this is not a formal meta-analysis, presented as original research. However, based on these results, a formal analysis is needed urgently (Figure 3).
Meta-analysis of mortality in large-scale randomized trials comparing albumin with crystalloids in adult patients with severe sepsis (courtesy of Wiedermann and Joannidis
41
).
Human albumin solutions have been extensively researched in sepsis, and may have benefit over other intravenous fluid therapies, but this has never been definitely proven in a large-scale RCT.
Use as an adjunct to furosemide
Diuretics are used in almost 50% of critical care admissions, with furosemide accounting for almost 95% of diuretic use. 42 The diuretic effect of furosemide is diminished in the setting of hypoalbuminaemia 43 so it would appear rational to administer both hyperoncotic albumin solution and furosemide together, in patients who are albumin deficient.
Two small prospective trials have addressed this question in the critical care population. In 2005, Martin et al. published the results of a randomised, double-blinded, placebo-controlled trial, of mechanically ventilated patients with lung injury. Patients randomised to the treatment arm received 100 mL 25% albumin, a loading dose of furosemide, followed by the initiation of a furosemide infusion. Repeated doses of albumin were administered every 8 h for three days. The control group received an identical loading dose, and infusion, of furosemide, but received 100 mL 0.9% saline every 8 h. At the end of the study period, the albumin group had a significantly greater mean net negative fluid balance (−5480 mL vs. −1490 mL; p = <0.01), and greater improvement in their oxygenation index. 44 These results should be interpreted in the context that this multi-centre trial consisted of only 40 patients across two sites.
Oczkowski et al. performed a pilot, feasibility study, to assess whether hyperoncotic albumin, in addition to furosemide, improves diuresis and enables liberation from mechanical ventilation. Unfortunately, the study did not meet its feasibility criteria, but in the limited physiological data acquired, outcomes were similar between the groups. The authors concluded that, although feasibility with their model was not proven, the design could help inform the planning of future, definitive, trials. 45
Furosemide potency is reduced in hypoalbuminaemia, but as a target for therapeutic intervention, research so far is limited.
Use in liver disease
Patients with acute, or progressive, liver disease account for more than 3% of all admissions to critical care beds in the United Kingdom. 46 Therefore, it is important that the intensivist is aware of the indications and limitations of albumin therapy in liver disease.
Paracentesis is the cornerstone of treatment of tense, symptomatic, ascites in patients with liver cirrhosis. 47 On removal of large volumes of fluid, circulatory dysfunction is prevalent in approximately 80% of cases, if adjunctive plasma expansion is not performed. 48 A meta-analysis, performed by Kwok et al., of 16 randomised trials, found that albumin use was associated with a significant reduction in post-paracentesis circulatory dysfunction (OR 0.26 95% CI 0.08–0.93), but conferred no benefit over synthetic colloids. 49 This is in contrast with numerous small studies that have shown benefit of albumin therapy over synthetic colloids in this context.50–52 This phenomenon is commonly observed in critical care research, where large analyses fail to replicate outcomes of smaller studies. The likely explanation of this is a combination of the inherent heterogeneity of the critical care population, combined with non-uniform trial design and outcome measures. 53
The British Society of Gastroenterology (BSG) recommend that large volume paracentesis (>5000 mL) be performed in one episode, and patients should receive plasma expansion after completion of the procedure, using 8 g albumin per litre of ascites removed. This equates to one 100 mL bottle of 20% human albumin solution per 3 L of ascites drained. 47
The BSG also make recommendations for its use in high risk patients with spontaneous bacterial peritonitis (SBP) and raised/rising creatinine; albumin 1.5 g/kg within the first 6 h, and 1 g/kg on day 3. 47 A paper published by Salerno et al. in 2013, presented the results of a systematic review and meta-analysis of four randomised controlled trials, with a total of 270 patients. The pooled odds ratio for a reduction in renal impairment, in patients treated with albumin, was 0.21 (0.11–0.42), and 0.34 (0.19–0.60) for a reduction in mortality. 54 Albumin’s benefit in patients with advanced liver disease and other infections, has traditionally, been less clear. In 2014, Thevenot et al. published the results of a multicentre randomised controlled trial, of 193 patients with severe liver disease, and sepsis unrelated to SBP. Patients were randomised to receive either antibiotics plus albumin, or antibiotics alone. Albumin delayed the onset of renal failure (mean time to onset, ALB: 29.0 ± 21.8 vs. 11.7 ± 9.1 days, p = 0.018), but had no effect in the incidence of established renal failure at three months, or mortality. 49
However, this is in contrast to those patients with cirrhosis and ascites who receive ‘long-term’ albumin replacement therapy. The ANSWER trial, published in 2018 by Caraceni et al., sought to compare standard medical therapy (anti-aldosterone drugs and furosemide), with standard medical therapy in combination with human albumin solution, in patients with liver cirrhosis and ascites. Participants in the albumin arm received 50 mL 20% HAS, twice per week for the first week, then once weekly for up to 18 months thereafter. Patients who received albumin therapy had a reduced risk of death of 38%, compared to those who received standard medical therapy alone (OR 0.62 CI 95% 0.40–0.95). 55
Human albumin solution is a well-established therapy in patients with liver disease. Although the benefit demonstrated in smaller studies is not replicated in larger analyses, consensus opinion favours the use of albumin in large volume paracentesis, and in spontaneous bacterial peritonitis. Its acute use in other hepatic syndromes is less clear.
Conclusions
Albumin may have an important role within certain facets of critical care medicine. It is recommended to avoid circulatory dysfunction after large volume paracentesis and is beneficial in hepatic decompensations due to SBP. Its use in sepsis and septic shock remains debated; reducing administered fluid volumes, but with inconclusive effects on mortality outcomes. Further investigation into ‘small volume resuscitation’ is urgently required. However, albumin solutions should not be administered to patients who have sustained a traumatic brain injury, and the routine correction of hypoalbuminaemia in critically ill patients is not advised.
In summary, hypoalbuminaemia is associated with worse mortality, which is not improved by replacement. HAS is useful as an intravenous therapy in aspects of hepatic failure and may still be shown to have a role in sepsis, pending a formal meta-analysis of large albumin trials. Whilst its use in the United Kingdom pales in comparison to the United States, Australia and New Zealand, its use continues to climb in proportion to other colloids. Knowledge of its uses and evidence base are paramount, allowing the critical care physician to make safe and effective, clinical and economic decisions.
Supplemental Material
sj-pdf-1-inc-10.1177_1751143720961245 - Supplemental material for Human albumin solutions in intensive care: A review
Supplemental material, sj-pdf-1-inc-10.1177_1751143720961245 for Human albumin solutions in intensive care: A review by David Melia and Benjamin Post in Journal of the Intensive Care Society
Footnotes
Authors' Note
David Melia and Benjamin Post are also affiliated with Barts Health NHS Trust, London, England.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
