Abstract
Purpose
To evaluate the impact of pressure-regulated volume control (PRVC/VC+) use on delivered tidal volumes in patients with acute respiratory distress syndrome (ARDS) or at risk for ARDS.
Materials and methods
Retrospective study of mechanically ventilated adult patients with severe sepsis or septic shock.
Results
A total of 272 patients were divided into patients with recognized ARDS, patients without ARDS, and patients with unrecognized ARDS. Over 90% of patients were ventilated with PRVC on admission, resulting in delivered tidal volumes significantly higher than set tidal volumes among all groups at all time points, even after ARDS recognition (p < 0.001). Tidal volumes were lower for patients with pulmonary sepsis as compared to those with a nonpulmonary origin (p < 0.001).
Conclusions
Using PRVC promotes augmented delivered tidal volumes, often in excess of 6 mL/kg ideal body weight. Correct recognition of ARDS and having pulmonary sepsis improves compliance with low-stretch protocol ventilation.
Keywords
Introduction
Acute respiratory distress syndrome (ARDS) is a major health burden, with an incidence rate of approximately 58.7 cases per 100,000 person-years, translating to more than 74,500 deaths in the United States each year.1,2 The most common risk factor is severe sepsis (70%), frequently of a pulmonary source; mortality rates have been reported as high as 50%.3–5
Mechanical ventilation protocols for patients with ARDS have changed significantly in the past two decades. The landmark ARDSNet trial demonstrated that lung protective ventilation; that is, the use of tidal volumes of 6 mL/kg or less by ideal body weight (IBW), with higher positive end-expiratory pressure (PEEP) levels and permissive hypercapnia, led to reduced mortality and increased number of ventilator-free days. 6 Subsequent reviews and meta-analyses supported this approach5,7–9 and it was incorporated into the Surviving Sepsis Campaign guidelines. 10
Multiple studies have demonstrated that the first 48 h of mechanical ventilation significantly impact outcomes of patients who present with ARDS and those who may develop it during hospitalization.4,11–14
The main ARDSNet study was done using predominantly assist control/volume control (AC/VC) ventilation. Ventilatory modifications to AC/VC have attempted to ostensibly mitigate ventilator-induced lung injury through a variety of mechanisms, which often confuse clinicians. One such mechanism, pressure-regulated volume control (PRVC, also known as volume control plus (VC+) or adaptive pressure-controlled continuous mandatory ventilation (AP-CMV)), has blended principles of pressure control and volume control ventilation; this mode uses a set tidal volume as a feedback guide to continuously adjust inspiratory pressures based on flow rates.15,16 In theory, as compared to VC ventilation, this mode is designed to minimize barotrauma and improve patient comfort. In unparalyzed patients, however, it may allow for delivery of tidal volumes which may be significantly higher than set volume when using VC; this is similar to the risk of excess tidal volume delivery in PC ventilation. There has been no rigid evaluation of the impact of the machine-driven ventilation adjustment of PRVC on the ARDSNet “low-stretch” protocol compliance and outcomes.
This study describes strategies of mechanical ventilation during the first 48 h of admission for sepsis or septic shock. The primary goal of the study was to determine whether the use of PRVC leads to higher tidal volumes than would be dictated by the ARDSNet guidelines. A secondary goal was to determine whether having a diagnosis of sepsis due to a pulmonary source had an impact on ventilator management. We hypothesized that use of PRVC led to noncompliance with ARDSNet guidelines through higher delivered tidal volumes. We also hypothesized that having a pulmonary source of sepsis led to lower set ventilator volumes.
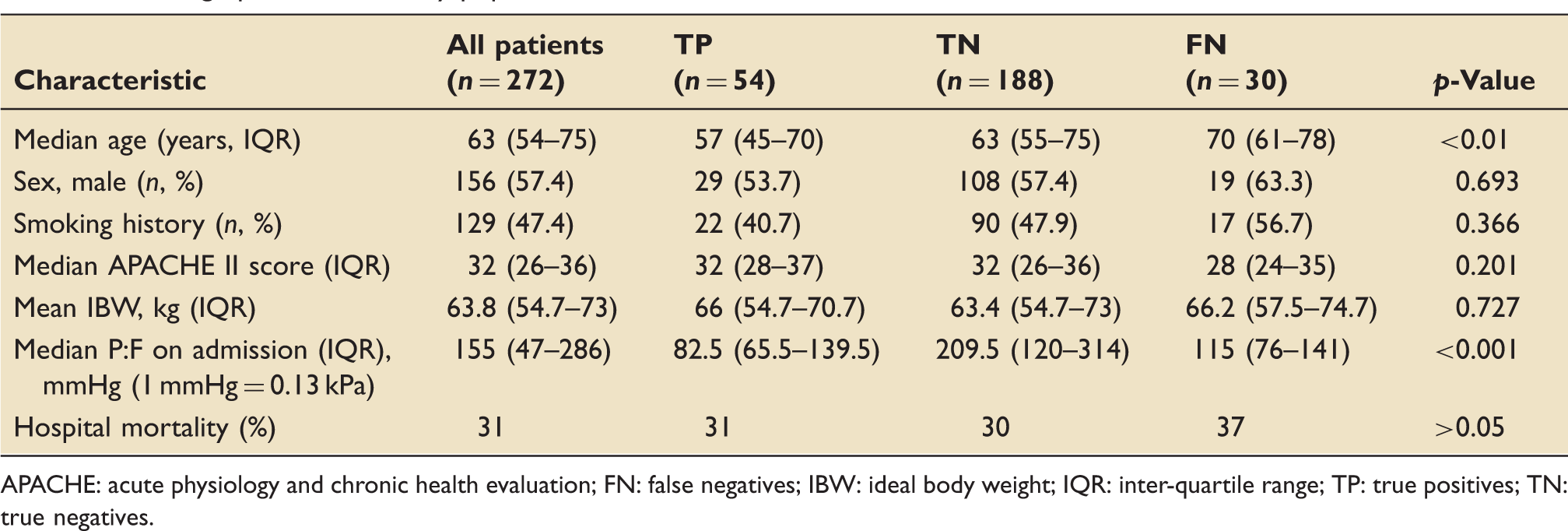
Methods
This was a retrospective review of ventilator management in mechanically ventilated adult (≥18 years old) patients admitted with severe sepsis or septic shock to Santa Barbara Cottage Hospital (SBCH) between 2012 and 2016. Sepsis was defined with Sepsis 2 terminology; that is, severe sepsis is sepsis complicated by end-organ dysfunction, and septic shock was severe sepsis with hypotension refractory to fluid resuscitation. 17 SBCH is a 483-bed community teaching hospital and Level 1 trauma center, which serves as the largest referral center between Los Angeles and the San Francisco Bay Area on California’s central coast. Using data from its actively maintained Sepsis Registry and electronic medical record system (Epic Systems, Verona, WI), data were extracted on admission date, Acute Physiology And Chronic Health Evaluation (APACHE) II score, height and weight, gender, race, smoking history, source of sepsis, and ventilator settings (mode of ventilation, set and actual tidal volumes, plateau pressures, PEEP) during the first 48 h in the ICU. In patients who were prescribed PRVC ventilation, spontaneous breaths were volume supported. Study data were collected and managed using Research Electronic Data Capture (REDCap) hosted at SBCH. 18
Clinician notes (seven experienced board-certified intensivists) and ventilator settings at intervals of every 12 h (Hour 0 = time of intubation) were reviewed in an effort to compare the accuracy of clinical diagnosis of ARDS versus a likely missed diagnosis of ARDS based on the Berlin definition (1) applied retrospectively. Ventilator management was assessed by intensivists within the first 24 h of intubation, although in some cases, initial ventilator management was done by senior internal medicine residents. The ventilators used were Hamilton G5 (Hamilton Medical, Reno, NV) and Puritan Bennett 840 (Medtronic, Minneapolis, MN). Patients with possible primary cardiogenic shock or significant cardiogenic pulmonary edema were excluded. Patients were a mixture of medical and surgical patients.
Patients were separated into groups of True Positives (TP; those patients with an accurate diagnosis of ARDS), True Negatives (TN; those patients with an accurate diagnosis of no ARDS), False Negatives (FN; those patients who likely had ARDS but were not diagnosed as such), and False Positives (FP; those patients without ARDS who were mistakenly diagnosed with ARDS). This separation was based on data on admission.
Statistical analysis
Statistical analysis was performed using the R Core Team 2016. 19 Patients were first classified into four groups: TP, TN, FN, and FP. Their demographic information, set and actual tidal volumes, PEEP, plateau pressures, and all other collected clinical information were analyzed by conducting a Kruskal–Wallis rank sum test for numerical variables and a chi-square test or a Fisher’s exact test for categorical variables. A multiple comparison test was carried out for the variables that have an overall p ≤ 0.05.
Patients were categorized as having either sepsis of pulmonary or nonpulmonary origin. Patient characteristics and clinical information were then analyzed by conducting a Student’s t test or a Wilcoxon rank sum test for numerical variables and a chi-square test or a Fisher’s exact test for categorical variables.
Results
A total of 272 intubated, mechanically ventilated patients with severe sepsis or septic shock were included in the analysis. ARDS patients, whether diagnosed during admission or retrospectively, had an interquartile PaO2/FaO2 (P:F) ≤ 200 mmHg (26.7 kPa). There was no significant difference between IBW, APACHE II scores, or gender (Table 1). The distribution in sources of sepsis (pulmonary vs. nonpulmonary) was statistically significant at p = 0.04 (Figure 1). The TP group was significantly younger (p < 0.05) than the FN group and with a significantly lower median P:F ratio; notably, the median P:F of TP patients was 82.5 mmHg (11 kPa) vs. 115 mmHg (15.3 kPa) for FN patients (p < 0.001). In-hospital mortality was 31% for the population, with no statistically significant differences between the groups, although there was a trend toward increased mortality in the FN group. There were no False Positives (i.e. those patients who were incorrectly diagnosed with ARDS).
Sources of sepsis origin. Demographics of the study population. APACHE: acute physiology and chronic health evaluation; FN: false negatives; IBW: ideal body weight; IQR: inter-quartile range; TP: true positives; TN: true negatives.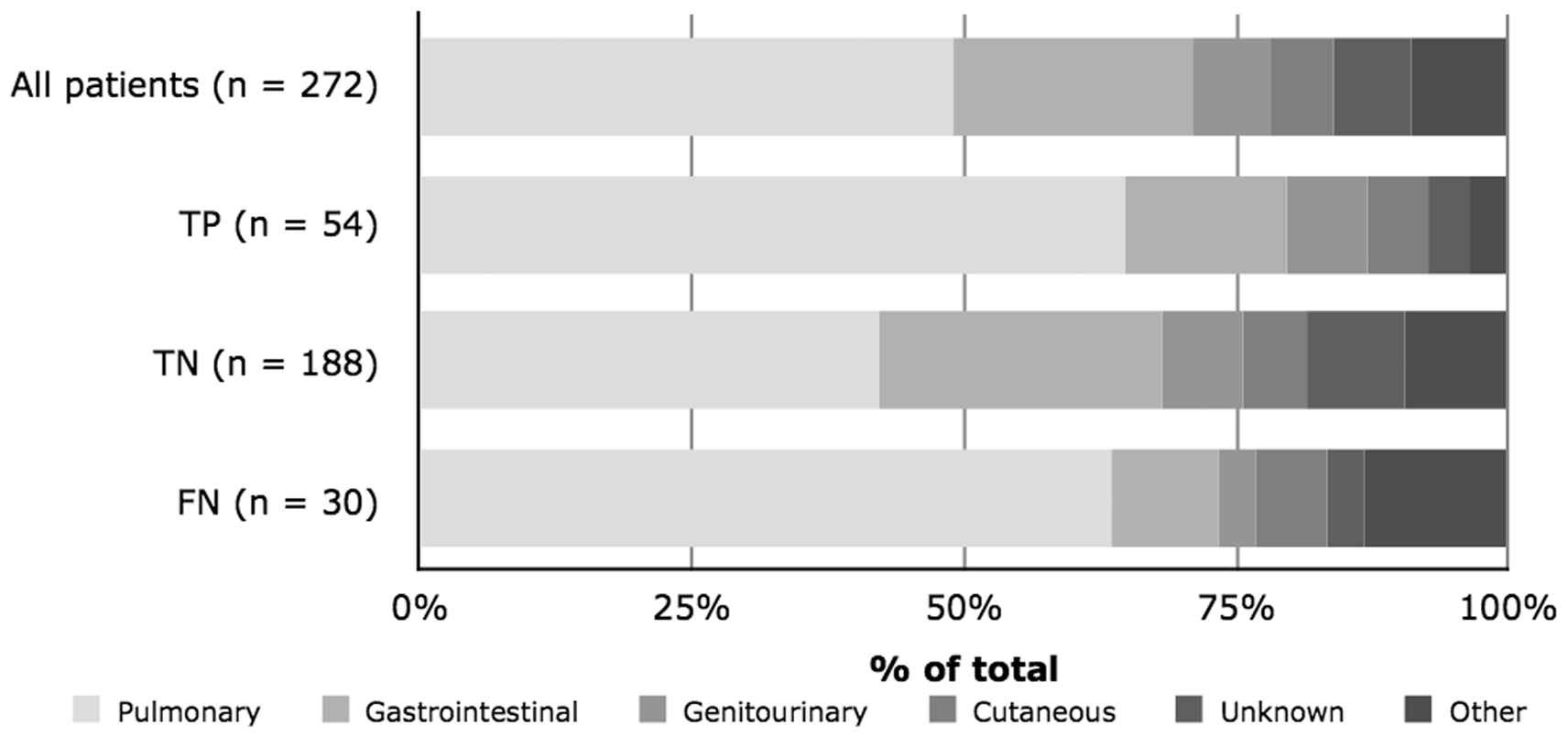
Impact of the use of PRVC on tidal volumes and airway pressure
Over 90% of patients were ventilated with PRVC ventilation on admission (Figure 2); all AC/VC ventilation was done with PRVC. Tidal volumes set at intubation (time 0), which were approximately 7.3–7.6 mL/kg of IBW, tended to be higher than at subsequent time points. Actual delivered tidal volumes were universally statistically significantly higher than set tidal volumes among all groups (Table 2). This difference in tidal volumes was, on average, approximately 0.5 mL/kg IBW. Some delivered tidal volumes were over 9 mL/kg IBW; although set and delivered tidal volumes were generally lower in the group of patients with recognized ARDS, they still frequently reached 8 mL/kg IBW.
Ventilation modes used among all groups. Comparison of set and actual tidal volumes and airway pressures at each timepoint. Vt difference reflects a difference as seen in the entire data set analysis, not difference between medians. Vt, tidal volume; mL/kg of IBW (IQR) (3-way ANOVA p-value). Pplat, plateau pressure. PEEP, positive end-expiratory pressure; cmH2O (3-way ANOVA p-value). ARDS: acute respiratory distress syndrome; FN: false negatives; IBW: ideal body weight; TP: true positives; TN: true negatives.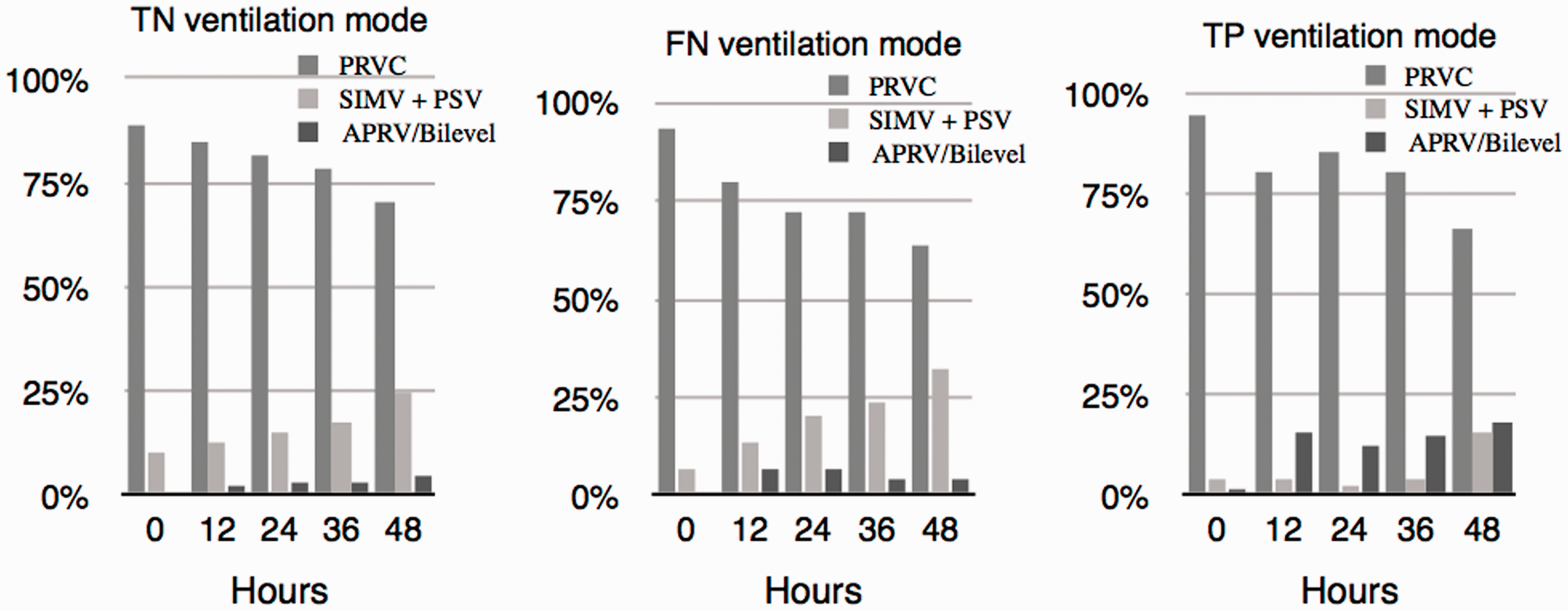
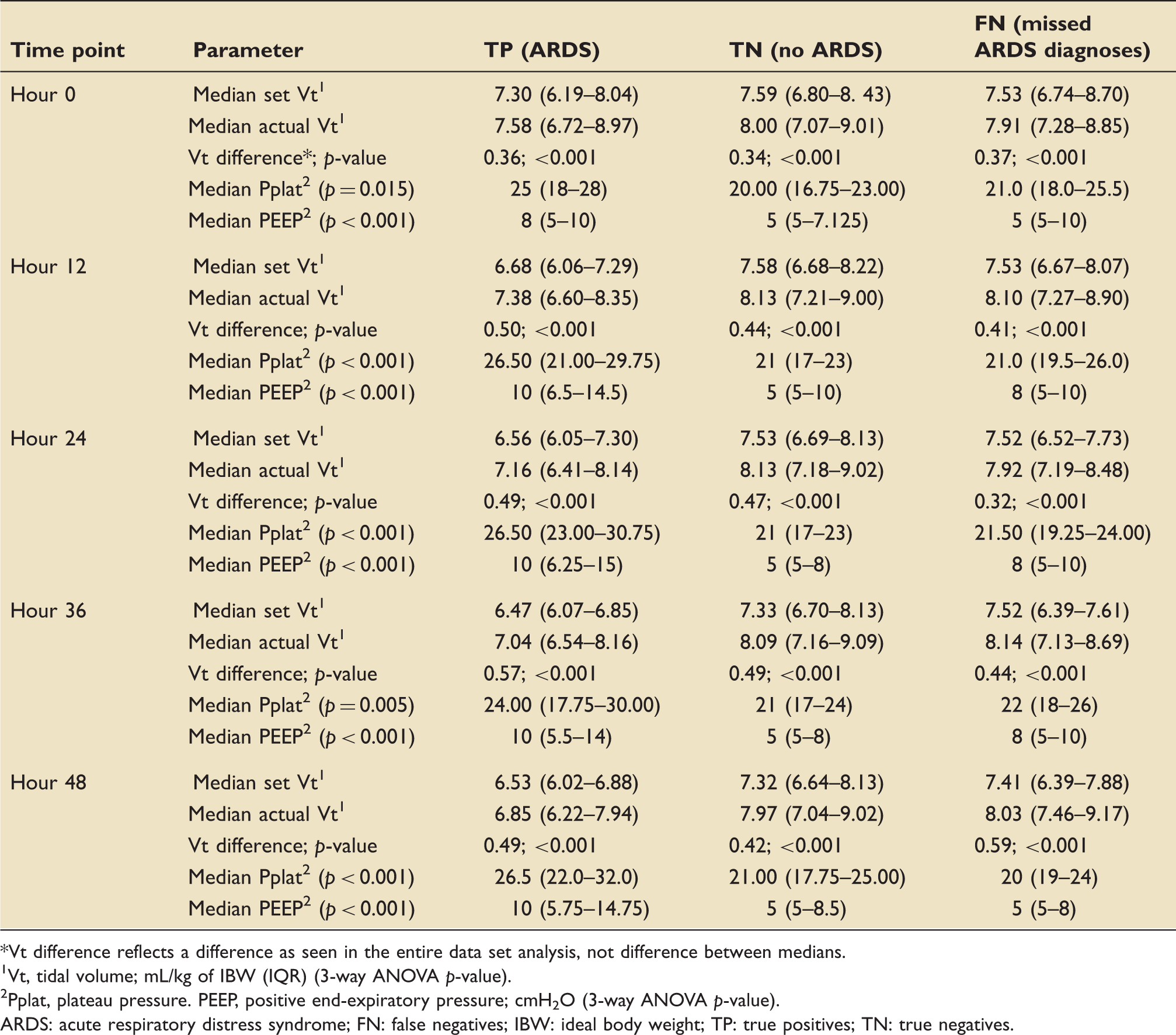
Plateau pressures were significantly higher in the ARDS group as compared to the non-ARDS (TN) group during the first 48 h, although median plateau pressures never exceeded 26.5 cmH2O even in the TP group. PEEP levels were statistically higher in the TP group as compared to the TN group at all time points, including at admission. There was no statistically significant difference between airway pressures in the TN and FN groups.
Impact of pulmonary source of sepsis on ventilator management
Pulmonary vs. nonpulmonary source of sepsis: Ventilator parameters and clinical characteristics.
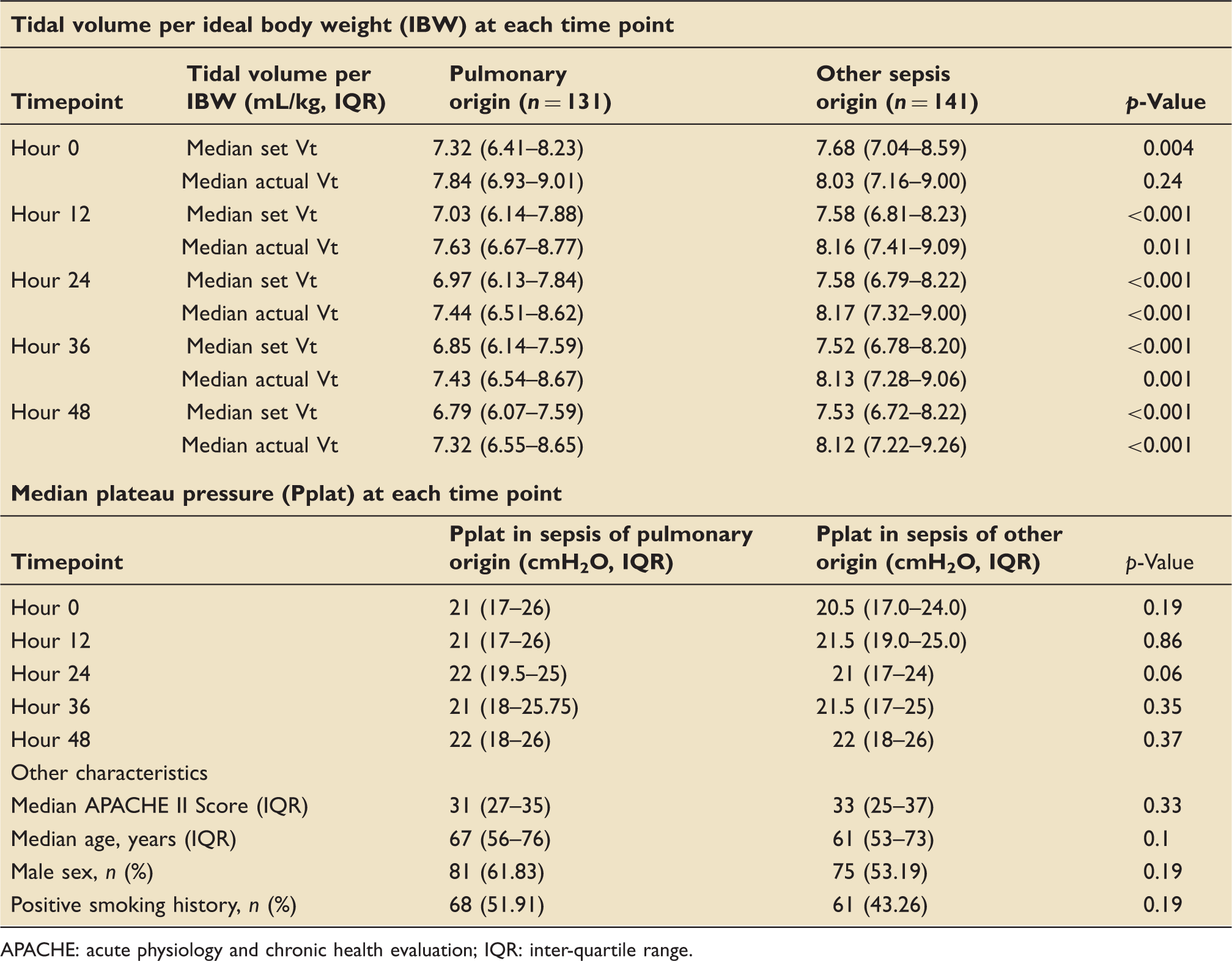
APACHE: acute physiology and chronic health evaluation; IQR: inter-quartile range.
Discussion
This study examined a real-world approach to mechanical ventilation in severe sepsis and septic shock patients with ARDS or at risk for ARDS.
The impact of PRVC on augmentation of tidal volumes has not been formally reported. In this study, 90% of patients were ventilated with PRVC on admission. We found that actual tidal volumes were statistically significantly higher than set volumes in all groups across all timepoints, suggesting that patient-driven augmentation using PRVC could impair lung-protective ventilation.
Specific data on PRVC use in ARDS is scant. One of the largest trials 20 randomized 44 patients with acute respiratory failure (P:F ≤ 300 mmHg (40 kPa), PEEP 5 cmH2O, FiO2 0.4–0.6) to PRVC or AC/VC, and suggested that peak inspiratory pressures are lower with PRVC use. Possibly, PRVC facilitates a decelerating inspiratory flow by accounting for a rise in intrathoracic pressure; in AC/VC, inspiratory flow is constant and therefore intrathoracic pressure rises potentially more than it would with PRVC. 20 Patients were randomized 8 h after admission and there was no evaluation of plateau pressures. They found no benefit of PRVC in reducing ventilator time, likelihood of failing the assigned mode of mechanical ventilation, or survival.
Initial tidal volumes were higher across the groups, with a notable decrease after 12 h. There are several potential explanations for this. First, initial ventilation parameters may have been set by less experienced trainee physicians and/or respiratory therapists before the involvement of a trained intensivist. Second, the decision may have been deliberate to compensate for an underlying acidosis. Third, the level of sedation and/or paralysis necessary to depress the patient’s respiratory drive enough to prevent spontaneous augmentation of tidal volume may not have been achieved until 12 h post-intubation. Previous work has suggested that using higher tidal volumes, even for relatively short periods of time, increases the risk of ARDS development. 4 Higher tidal volume ventilation may increase ARDS risk through increased proinflammatory cytokine production,13,14 among other reasons.
Patients with sepsis of a pulmonary origin in this study were ventilated with significantly lower set tidal volumes than sepsis of other (combined) origins, suggesting that clinicians were more likely to treat these patients closer to ARDSNet guidelines. However, actual tidal volumes were not statistically significantly different for the first 12 h, suggesting that patient-driven ventilation using PRVC may be thwarting the efforts of the clinicians to use lung-protective strategy. High tidal volumes on admission in patients without ARDS are a known independent risk factors for lung injury development,4,21 and the use of lower tidal volumes in high-risk patients, such as those with sepsis and/or pneumonia, may lower the risk of ARDS development during hospitalization. 22 Thus, a pulmonary source of sepsis has been included in several robust ARDS-predictive models, including the Lung Injury Prediction Score.8,9,23
The “right” mode of ventilation in ARDS and in patients at high risk for ARDS remains unknown. A study which compared AC, PC, and PRVC ventilation in ARDS patients showed no reduction in work of breathing in patients on PRVC; in this study, delivered tidal volumes were higher in PRVC than in VC, but lower than in PC, ventilation 24 and a recent comparison of 13 different ventilation modes highlighted that in spontaneously-breathing patients, tight control of delivered tidal volume can be challenging, particularly when those breaths are pressure-supported. 25 Protocolized mechanical ventilation in ARDS is challenging due to the great variability in compliance and extent and inhomogeneity of lung involvement between patients. Unlike ventilated healthy lungs, the same tidal volume can lead to a significant difference in lung stress and strain between patients. 26 These observations have led to suggestions that parameters other than tidal volume, such as driving pressure, should be used as targets for mechanical ventilation in ARDS. 27 Furthermore, data from a variety of clinical and preclinical studies suggest that injurious mechanical ventilation may result not only from volutrauma, but also from barotrauma (which may be more effectively limited with PC ventilation), increased work of breathing (which may be lower with VC or PRVC but can vary significantly between patients), patient-ventilator dyssynchrony (which may be better with PRVC but may be more related to the degree of sedation or paralysis than to ventilator mode) and inspiratory flow (which may be higher with PC and lower with PRVC).20,24,28,29 Thus, the choice of mechanical ventilation mode is probably one that should be individualized according to the challenges presented by each patient.
A strength of this study is the description of the largest group of ARDS patients ventilated with PRVC, with over 90% of 84 patients experiencing this ventilatory mode in the first 48 h. This study also describes a specific population of patients with very high morbidity and mortality; all patients had ARDS due to severe sepsis and septic shock, had high APACHE II scores, and generally had moderate-severe ARDS with an overall mortality of 31%. Other strengths include the collection of ventilatory data trends over the first 48 h, a three-group comparison of patients with diagnosed ARDS, missed ARDS, and no ARDS, and a comparison of patients with a pulmonary source of sepsis to those of another source.
The study is not without the weaknesses inherent to a retrospective review. The team did not collect data on adjunctive treatments (level of sedation achieved, neuromuscular blockade, corticosteroid use, prone positioning, inhaled prostaglandin use, recruitment maneuvers, etc.), which are relevant in severe ARDS (extracorporeal membrane oxygenation (ECMO) was not available at the study site and none of the patients were transferred to an ECMO-capable facility). The study also was not adequately powered and/or did not have enough data available to assess some important clinical endpoints, such as ICU length of stay and ventilator days. Additionally, this study was not able to directly compare PRVC use with other modes of ventilation. Finally, this is a single center study from a community teaching hospital and some of its findings may not be generalizable.
This study demonstrates that the use of PRVC may result in the delivery of tidal volumes, which are significantly higher than those set by the clinician, and consequently, higher than those recommended for ARDS patients. Having a pulmonary origin of sepsis may impact ventilation practices, particularly in lowering tidal volumes. Further work is needed to identify whether use of PRVC impacts duration of ventilator use and ICU stay, as well as mortality among patients with acute hypoxemic respiratory failure.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
