Abstract
Ventilator-associated pneumonia is the most common healthcare-associated infection in mechanically ventilated patients. Despite this, accurate diagnosis of ventilator-associated pneumonia is difficult owing to the variety of criteria that exist. In this prospective national audit, we aim to quantify the existence of patients with suspected ventilator-associated pneumonia that would not be detected by our standard healthcare-associated infection screening process. Furthermore, we aim to assess the impact of tracheostomy insertion, subglottic drainage endotracheal tubes and chlorhexidine gel on ventilator-associated pneumonia rate. Of the 227 patients recruited, suspected ventilator-associated pneumonia occurred in 32 of these patients. Using the HELICS definition, 13/32 (40.6%) patients were diagnosed with ventilator-associated pneumonia (H-posVAP). Suspected ventilator-associated pneumonia rate was increased in our tracheostomy population, decreased in the subglottic drainage endotracheal tube group and unchanged in the chlorhexidine group. The diagnosis of ventilator-associated pneumonia remains a contentious issue. The formalisation of the HELICS criteria by the European CDC should allow standardised data collection throughout Europe, which will enable more consistent data collection and meaningful data comparison in the future. Our data add weight to the argument against routine oral chlorhexidine. The use of subglottic drainage endotracheal tubes in preventing ventilator-associated pneumonia is interesting and requires further investigation.
Keywords
Introduction
Ventilator-associated pneumonia (VAP) is the most common healthcare-associated infection (HAI) in patients receiving mechanical ventilation.1–4 VAPs are independently associated with an increased length of intensive care unit (ICU) stay, duration of mechanical ventilation and mortality. 3 Since VAPs have an outcome and cost implication with public, political and regulatory interest, accurate reporting of the prevalence of VAPs within our critical care environment is essential. 5 The most simplistic definition of a VAP is a new lower respiratory tract infection occurring at least 48-h following the initiation of mechanical ventilation. 6 Unfortunately, this simplistic definition is far too vague, over-sensitive and open to individual clinician interpretation.7,8
Given that many of the VAP risk factors are modifiable, there is an increasing appetite for individual units and health boards to declare their VAP rate as a quality indicator to benchmark their performance.9,10 To facilitate this, there is a requirement for a robust and standardised mechanism for allowing diagnosis and reporting of VAPs. In Scotland, HAI data are gathered by the Scottish Intensive Care Society Audit Group (SICSAG) Wardwatcher database. When considering a diagnosis of VAP, Wardwatcher utilises the Hospitals in Europe Link for Infection Control through Surveillance (HELICS) definition (Figure 1). This is a standardised and internationally recognised definition that has been adopted by the European Centre for Disease Prevention and Control (HAI-Net ICU 2017).11–13
Hospitals in Europe Linked for Infection Control through Surveillance Project (HELICS).
11

SICSAG data reveal that our VAP rate as a nation is low compared with other European countries. 14 It is believed that this is due to our focus on quality improvement and patient safety with initiatives such as the introduction of VAP prevention bundles together with robust reporting and quality assurance.15,16
It has been argued that the strict nature of the HELICS diagnostic tool, in particular the reliance on serial CXRs makes the diagnosis of VAP difficult. 17 Uncertainty, therefore, exists as to whether the HELICS definition itself underestimates our true national VAP rate. It should be noted that most units in Europe and the UK utilise the same diagnostic criteria for VAP.
VAP prevention bundles have been developed over the past decade to help minimise the rate of VAP in the ICU. 15 Although the utilisation of a VAP prevention bundle is regarded as good practice, wide variation of individual bundle elements exists. In particular, the use of sub-glottic drainage endotracheal tubes (SGTs) and chlorhexidine gel are not standardised throughout Scotland as their efficacy in preventing VAP is not widely agreed upon.
This national audit project aims to determine the rate of “suspected VAP” (S-VAP) within Scottish ICUs over a one-month period. We aim to quantify what proportion of these would have been missed by our existing diagnostic criteria. In addition, we will assess whether interventions such as chlorhexidine gel, tracheostomy or SGT have any bearing on VAP rates.
Methods
We prospectively collected non-identifiable HAI data from patients ventilated for more than 48-h, in all ICUs across Scotland. Prior to the launch of data collection, all SICSAG leads were contacted to ensure that we had local support. Local data collectors were recruited by the Scottish Intensive Care Society (SICS) Trainee Committee.
An encrypted data collection tool was devised using Microsoft Excel and disseminated to all data collectors throughout Scotland. Data collectors were all trainee medical staff who worked within each individual ICU unit and would therefore have had access to the required data as part of their daily duties. For this reason, formal national ethics approval via the Public Benefit and Privacy Panel (PBPP) was not required. All Scottish Health Board Information Governance Department, Research and Development Departments and any other relevant stake-holder within each health board were contacted, to ensure regional approval for this project.
The data collection tool was completed prospectively during the audit period. All adult ICU patients invasively ventilated for more than 48 h were included in this project. During the audit period, all eligible patients had their age, sex, admission diagnosis, time of admission and date of admission recorded. Additional HAI screening data were collected from each patient on a daily basis. This daily data consisted of the following “yes/no” questions:
– NG tube in situ? – Tracheostomy or endotracheal tube? – Subglottic endotracheal tube in place? – Chlorhexidine mouthwash prescribed regularly? – Is there a clinical suspicion of a new lower respiratory tract infection today? – Is this patient on antibiotic therapy today?
If a patient was identified to have been commenced on new antibiotic therapy to target a suspected new lower respiratory tract infection, then additional clinical information was gathered to determine whether this suspected new lower respiratory tract infection would have been positively diagnosed by the HELICS definition.
We submitted a data request to the Scottish Intensive Care Society Audit Group to obtain Wardwatcher data regarding patient outcome, length of stay, number of ventilation days and APACHE scores.
Results
We collected data from 18 ICUs within 11 health boards across Scotland over the 30-day audit period. Data collection was not obtained from the remaining Scottish ICUs (three cardiac units and one general unit) as a local data collector was not able to be recruited for these sites. During the study period, we collected data from 227 patients, totalling 1751 days of daily HAI data.
Antibiotics were commenced for new suspected lower respiratory tract infection (S-VAP) in 32 patients. Of these 32 patients, there were 13 who would have been positively diagnosed utilising the HELICS criteria (H-posVAP). We identified a sub-group of 19 patients who were started on antibiotics for a suspected chest infection but failed to meet the diagnostic HELICS criteria (H-negVAP).
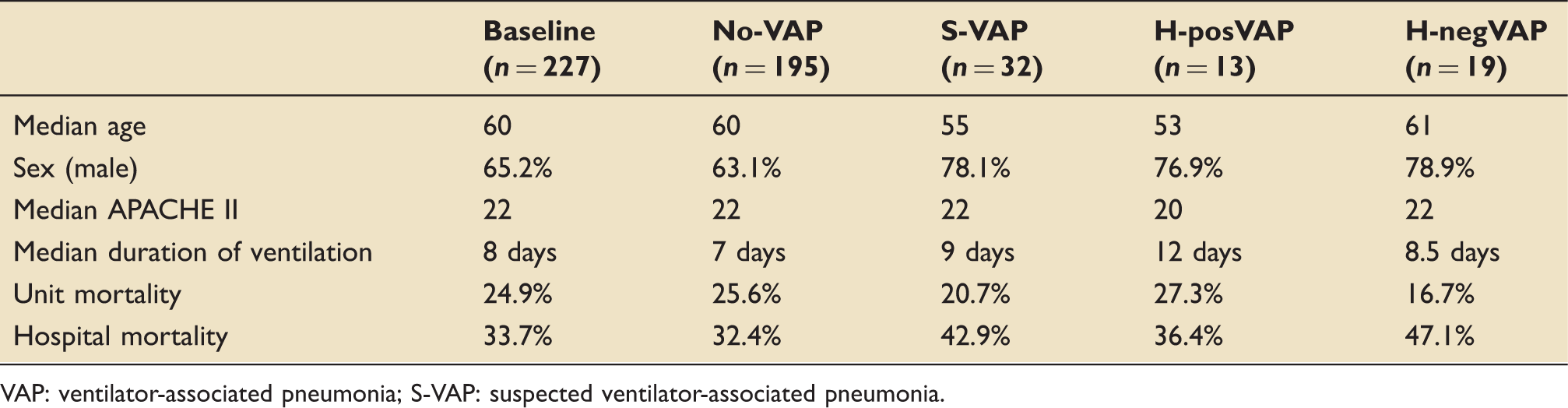
The demographics of the patient groups above (Figure 2) were all similar to our baseline in terms of age and APACHE scores. We did not collect co-morbidity data during this study. A disproportionate number of our patients who were commenced on antibiotics for suspected VAP (S-VAP) were male despite our baseline having an even sex balance (Table 1).
The duration of mechanical ventilation increased from a baseline of 8 days to 9 days in patients with a suspected ventilator-associated pneumonia (S-VAP). This increased further to 12 days in those who were positively diagnosed by the HELICS criteria (H-posVAP). Patients in whom a VAP was suspected did not demonstrate a higher unit mortality but their hospital mortality was considerably increased, particularly in the sub-group of patients who did not meet the HELICS criteria (H-negVAP).
Patient demographics.
VAP: ventilator-associated pneumonia; S-VAP: suspected ventilator-associated pneumonia.
Reason that antibiotics were commenced for patients with suspected VAP.
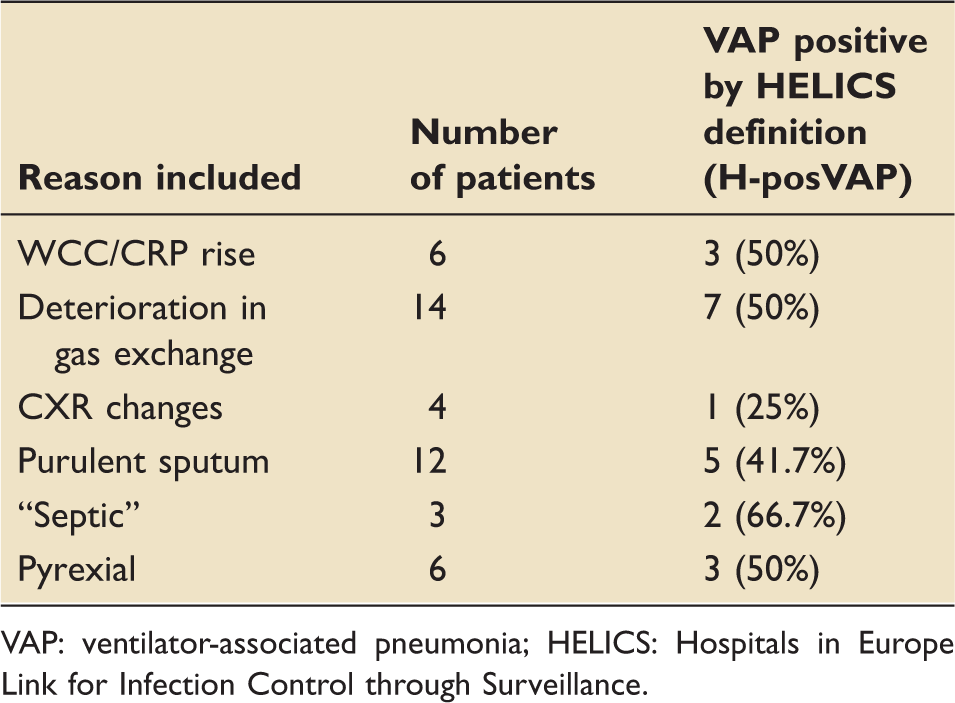
VAP: ventilator-associated pneumonia; HELICS: Hospitals in Europe Link for Infection Control through Surveillance.
Meropenem, piperacillin/tazobactam and ciprofloxacin were the most commonly utilised first line antibiotics for new suspected lower respiratory tract infections during the audit period.
Reasons that S-VAP did not meet HELICS criteria (

S-VAP: suspected ventilator-associated pneumonia; HELICS: Hospitals in Europe Link for Infection Control through Surveillance.
Subglottic tube analysis.
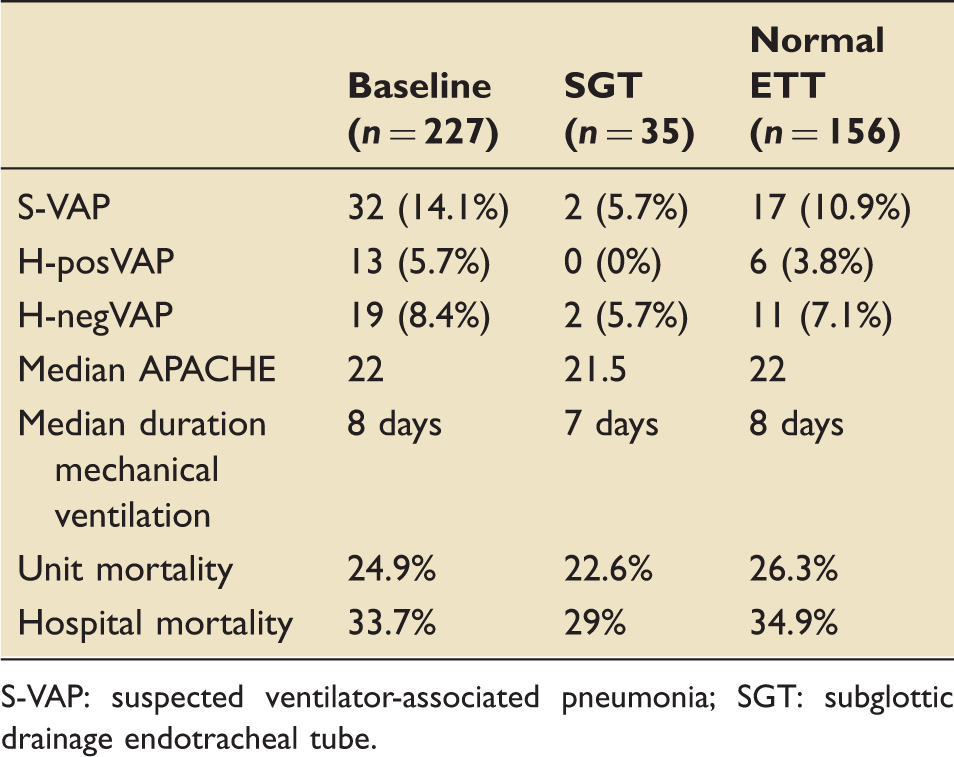
S-VAP: suspected ventilator-associated pneumonia; SGT: subglottic drainage endotracheal tube.
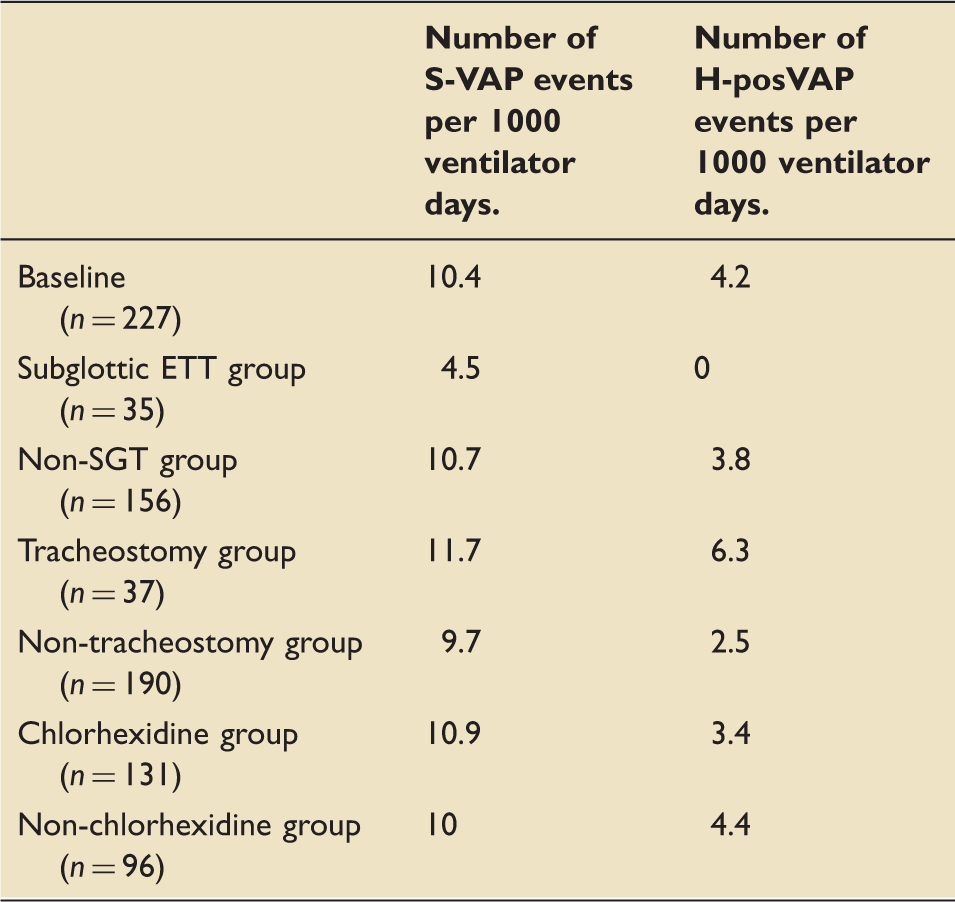
Events in each subgroup per 1000 ventilator days.
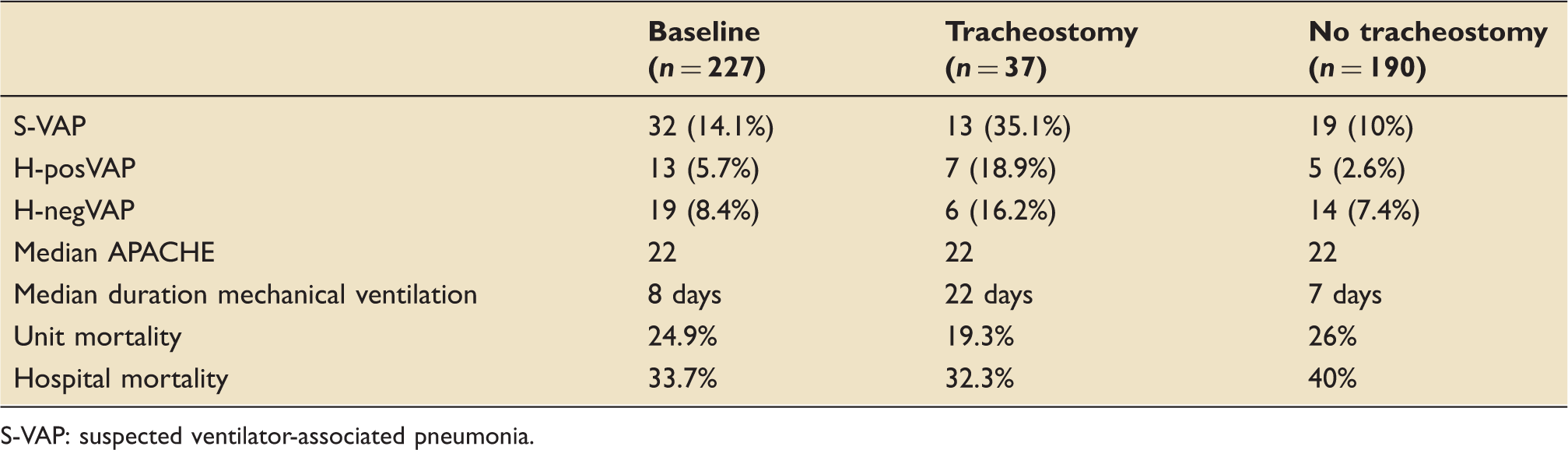
Tracheostomy analysis.
S-VAP: suspected ventilator-associated pneumonia.
Subglottic tube, chlorhexidine and tracheostomy subgroup analysis
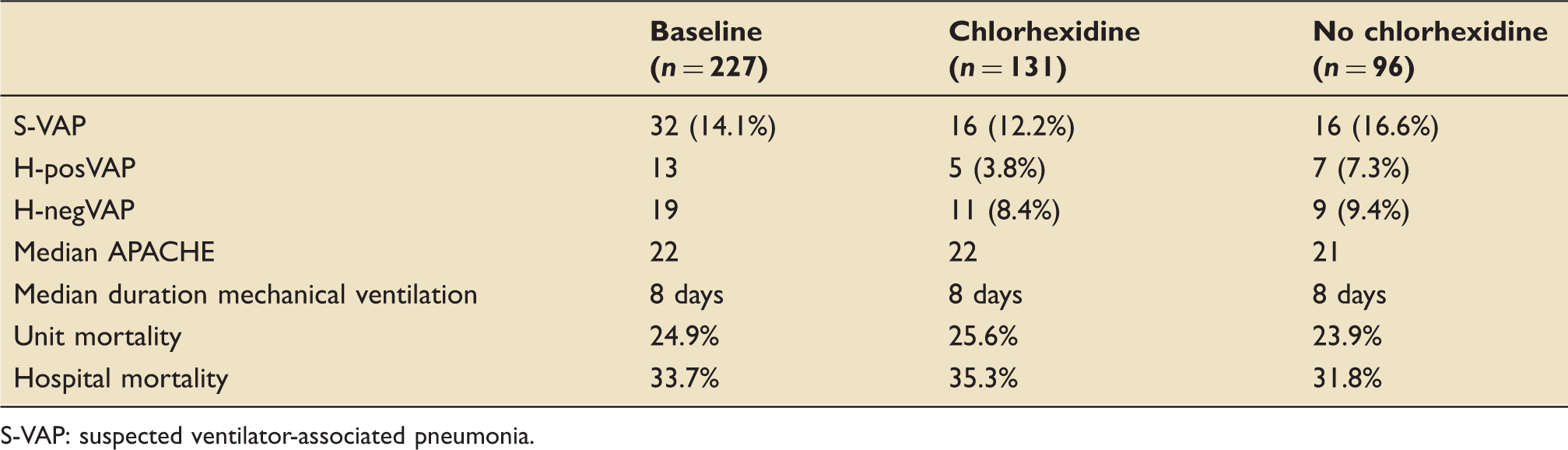
Chlorhexidine analysis.
S-VAP: suspected ventilator-associated pneumonia.
Tracheostomies were present in 37 of our 227 patients during the audit period (16.3%). S-VAP occurred in 13 of our 37 patients with tracheostomies (35.1%). In the remaining 190 patients who did not have a tracheostomy, S-VAP occurred in 19 cases (10%) which is significantly less than in the tracheostomy group (Table 6). By applying the HELICS definition, a difference between the tracheostomy and ETT group still exists with H-posVAP occurring in 7 of the 37 tracheostomy patients (18.9%) compared to 5 of the 190 ETT patients (2.6%). Number of S-VAP episodes per 1000 ventilator days was 11.7 days in the tracheostomy group compared to 9.7 days in the non-tracheostomy group.
During the audit period, 131 patients were prescribed regular chlorhexidine gel (57.7%) (Table 7). In the chlorhexidine group, S-VAP occurred in 16 patients (12.2%). In the non-chlorhexidine group, S-VAP also occurred in 16 cases (16.7%). When applying HELICS analysis, an H-posVAP occurred in five of the chlorhexidine patients (3.5%) compared to seven of the non-chlorhexidine patients (7.3%). There was no difference between the groups in terms of number of S-VAP episodes per 1000 ventilator days.
Discussion
There is no doubt that VAP is a significant healthcare burden that has significant morbidity. Despite this, the diagnosis of VAP remains a contentions issue.18–21 We have demonstrated that a sub-group of patients with suspected VAP exist, who would not have been diagnosed by the HELICS definition and therefore would not be declared by our HAI reporting tools. This is consistent with existing literature which describes a variation in the diagnosis of VAP depending upon which diagnostic tool is applied. 19 In our population, the presence of suspected VAP had little difference on unit mortality, regardless of HELICS screen. It is interesting that both the S-VAP and H-negVAP groups demonstrated a higher hospital mortality. It is difficult to attribute this rise in mortality purely to the presence of suspected VAP as hospital mortality is influenced by many other independent factors.
Our study shows the antibiotic burden of S-VAP is significant, regardless of subsequent HELICS screen. Given growing concerns regarding antibiotic resistance, it is essential that a national HAI screening tool accurately reflects the burden of antibiotic use in our ICUs. Our existing tools do not always capture this information and perhaps the burden of antibiotic usage is something that our national HAI screening processes should aspire to collect.
We were unable to identify positive microbiology in the half of our patients with S-VAP. The organisms that were isolated were those typically associated with VAPs. Those isolated with
It is no surprise that the lack of positive radiological features contributed to most patients failing to have a VAP diagnosis confirmed. It was disappointing that lack of radiological evidence contributed to most patients with suspected VAP failing to be positively diagnosed with the condition. Patients with respiratory co-morbidities require serial CXRs to confirm a diagnosis of VAP. Daily CXRs are no longer routinely performed in ICU; therefore, this might explain why a number of patients in our cohort did not meet the HELICS definition of VAP. 17 Although modern ICU practice has moved away from daily CXRs, any patient who develops a respiratory deterioration consistent with suspected VAP would still be expected to receive a CXR.
All screening tools have their limitations and there is a difficult balance to be struck between over-screening and missing potential VAPs. The variability of VAP diagnosis depending on what screening tool is utilised has been explored previously.7,19 The 2013 US Center for Disease Control National Healthcare Safety Network (CDC-NHSN) algorithm appears more adequately suited for surveillance purposes as it describes a spectrum of ventilator-associated events.22–24 Adopting this definition has potential as it may increase the proportion of patients screened since it would capture all patients who suffer a deterioration in gas exchange. However, this deterioration (or ventilator-associated condition) may not be due to an infective cause and in addition may not be a preventable complication. The merits of collecting this data are therefore questionable. Furthermore, the presence of these ventilator-associated events has been demonstrated to have a low sensitivity in diagnosing VAP. 25
The utility of VAP rates as an HAI quality indicator has been questioned, given the lack of consensus over the definition of a VAP and the discrepancy between VAP rates depending on what screening tool is used. 19 However, the pathogenesis of VAP does have modifiable elements, many of which are markers of routine high quality ICU care. 26 Therefore, the argument to continue using VAP rates as an HAI quality indicator is strong. It is therefore important that units across the UK and Europe utilise the same diagnostic tools when performing VAP screening. The adoption of the HELICS criteria by the European CDC indicates that the existing HAI screening process in Scotland is appropriate.
Randomised control trial and meta-analysis data have consistently suggested a 50% reduction in the rate of VAP development through the routine use of endotracheal tubes with subglottic suction ports (SGT).27–30 The Intensive Care Society VAP Guidelines recommend their routine use on this basis. 26 Our study, albeit with limited numbers, also demonstrates a VAP rate reduction consistent with the existing literature. On this basis, we would also recommend the routine use of SGTs. Although they have an additional cost compared with standard ETT, the benefit in terms of VAP reduction and reduced antibiotic burden would be a worthwhile use of resource.
The rate of suspected VAP in patients with tracheostomies was 11.7 per 1000 ventilator days versus 9.7 in non-tracheostomy patients. This is perhaps not a surprise given that micro-aspiration is inevitable when placing a tracheostomy. The number of these patients subsequently diagnosed with an H-posVAP was also increased in this group. There are many confounding patient factors in addition to micro-aspiration during tracheostomy insertion which may explain this. In our study, patients with tracheostomies demonstrated a notable increase in duration of mechanical ventilation, which would inevitably increase the point prevalence of VAP.
Routine oral chlorhexidine was incorporated into VAP prevention bundles as a result of large scale meta-analysis in cardiac and non-cardiac patients which demonstrated significant reduction in VAP rates. 31 Recent literature no longer seems to support this with some studies demonstrating harm in terms of increasing oral ulceration. 32 Chlorhexidine is not a benign intervention and much like latex, has been implicated in numerous cases of hypersensitivity and anaphylaxis. As such, the routine use of chlorhexidine is no longer recommended in the most recent NICE guidelines. 26 Our study, again is consistent with recent literature and demonstrates no benefit in terms of VAP reduction.
Scottish ICUs should be proud of the high standard of quality improvement work that is undertaken routinely with input from all members of the multi-disciplinary team. This work has undoubtedly led to a dramatic reduction in the rate of VAP. Our rates remain low and compare well with other developed countries. This national audit project highlights the difficulties that exist in trying to diagnose VAP. The strength of this study has been the collaborative effort of the Scottish ICU trainee cohort that has allowed a large quantity of HAI data to be gathered prospectively. The influence of chlorhexidine is consistent with existing literature and is further evidence that routine chlorhexidine should be discontinued. The effect of SGTs is interesting; however, due to the low prevalence of VAP in our patient cohort, further investigation is needed. Only by consistently gathering standardised and reliable HAI data can we continue to assess the prevalence of VAPs in our ICUs and determine what factors can help reduce this important complication of mechanical ventilation.
Categorisation of patients who were analysed during the audit period.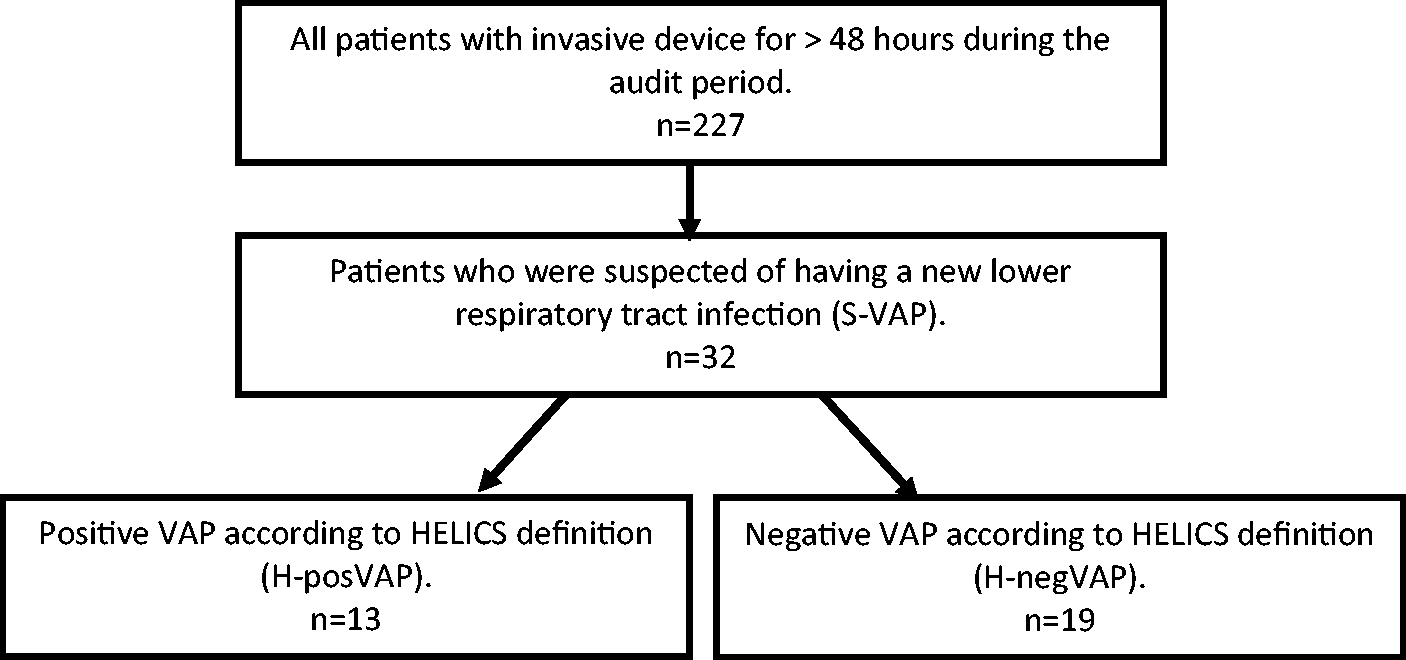
Footnotes
Acknowledgements
We would like to thank the following local data collectors for their input into this project: Stephen Cole, Dave MacPherson, Gilly Fleming, Michael Baynham, Steve Dauncey, Gemma Milne, Suleman Mulla, David Wright, Ashleigh Taylor, Chris Lochrin, Helen Jordan, Eoghan Smith, Claire McCue, Susan Chapman, Gareth Lipton, Rachael Campbell, Eleanor Waters, Neil Brain, Rhys Millington, Iain Thompson, Mina Zikry, Frances McConnachie, Richard Bauld, Aiden McIvor, Euan Kerr, Marc Vilas, Euan Chalmers, John Gardner, Maxine Bunton, Iain Belford, Martin Gannon, Phil Henderson, Thomas Dunford, Jane Hamilton, Stuart Williams, Duncan Philp.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
